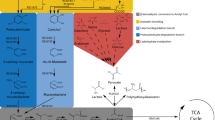
Abstract
Lignin valorization is considered an integral part for an economically viable biorefinery. However, heterogenous nature of lignin imposes a big challenge for upgrading diverse lignin-derived intermediates and subsequent downstream processing. To overcome this challenge, we proposed to explore unique convergent pathways in Rhodococus strains to funnel lignin-derived compounds into single target products. A feasible bioprocess for co-production of lipids and carotenoids from lignin by Rhodococci was developed. This process would potentially extract more values from lignin via biological upgrading.







Similar content being viewed by others
Data availability
All authors declare that all data and materials support their published claims and comply with field standards.
References
Balan, V., Chiaramonti, D., & Kumar, S. (2013). Biofuels Bioprod. Biorefin., 7(6), 732–759.
Chen, Z., & Wan, C. (2017). Biological valorization strategies for converting lignin into fuels and chemicals. Renewable and Sustainable Energy Reviews, 73, 610–621.
Xu, Z., Lei, P., Zhai, R., Wen, Z., & Jin, M. (2019). Biotechnol. Biofuels, 12(1), 32.
Martínková, L., Uhnáková, B., Pátek, M., Nešvera, J., & Křen, V. (2009). Biodegradation potential of the genus Rhodococcus. Environment International, 35(1), 162–177.
Chen, Z., & Wan, C. (2017). Co-fermentation of lignocellulose-based glucose and inhibitory compounds for lipid synthesis by Rhodococcus jostii RHA1. Process Biochemistry, 57, 159–166.
Wei, Z., Zeng, G., Huang, F., Kosa, M., Huang, D., & Ragauskas, A. J. (2015). Bioconversion of oxygen-pretreated Kraft lignin to microbial lipid with oleaginous Rhodococcus opacus DSM 1069. Green Chemistry, 17(5), 2784–2789.
Cai, C., Xu, Z., Xu, M., Cai, M., & Jin, M. (2020). ACS Sustain. Chemical Engineer, 8, 2016–2031.
Ichiyama, S., Shimokata, K., & Tsukamura, M. (1989). Carotenoid Pigments of GenusRhodococcus. Microbiology and Immunology, 33(6), 503–508.
Fiedor, J., & Burda, K. (2014). Potential Role of Carotenoids as Antioxidants in Human Health and Disease. Nutrients, 6(2), 466–488.
Mata-Gómez, L. C., Montañez, J. C., Méndez-Zavala, A. and Aguilar, C. N. (2014) Microb. Cell Fact. 13, 12, 1.
Chaudhary, R., & Dhepe, P. L. (2017). Solid base catalyzed depolymerization of lignin into low molecular weight products. Green Chemistry, 19(3), 778–788.
Chen, Z., Reznicek, W. D., & Wan, C. (2018). Deep eutectic solvent pretreatment enabling full utilization of switchgrass. Bioresource Technology, 263, 40–48.
Amara, S., Seghezzi, N., Otani, H., Diaz-Salazar, C., Liu, J. and Eltis, L. D. (2016) Sci. Rep. 6.
Vardon, D. R., Rorrer, N. A., Salvachua, D., Settle, A. E., Johnson, C. W., Menart, M. J., Cleveland, N. S., Ciesielski, P. N., Steirer, K. X., Dorgan, J. R., & Beckham, G. T. (2016). cis,cis-Muconic acid: separation and catalysis to bio-adipic acid for nylon-6,6 polymerization. Green Chemistry, 18(11), 3397–3413.
Salvachúa, D., Karp, E. M., Nimlos, C. T., Vardon, D. R., & Beckham, G. T. (2015). Towards lignin consolidated bioprocessing: simultaneous lignin depolymerization and product generation by bacteria. Green Chemistry, 17(11), 4951–4967.
Takaichi, S., Ishidsu, J.-I., Seki, T. and Fukada, S. (1990) Agric. Biol. Chem. 54, 1931-1937.
Ahn, J.-W., & Kim, K.-J. (2015). Enzyme Microb. Technol., 77, 29–37.
Chan, J. M., Bauer, S., Sorek, H., Sreekumar, S., Wang, K., & Toste, F. D. (2013). Studies on the Vanadium-Catalyzed Nonoxidative Depolymerization of Miscanthus giganteus-Derived Lignin. ACS Catalysis, 3(6), 1369–1377.
Erdocia, X., Prado, R., Fernández-Rodríguez, J., & Labidi, J. (2015). ACS Sustain. Chemical Engineer, 4, 1373–1380.
Kosa, M., & Ragauskas, A. J. (2012). Bioconversion of lignin model compounds with oleaginous Rhodococci. Applied Microbiology and Biotechnology, 93(2), 891–900.
Zhao, C., Xie, S., Pu, Y., Zhang, R., Huang, F., Ragauskas, A. J., & Yuan, J. S. (2016). Synergistic enzymatic and microbial lignin conversion. Green Chemistry, 18(5), 1306–1312.
Xie, S., Sun, Q., Pu, Y., Lin, F., Sun, S., Wang, X., Ragauskas, A. J., & Yuan, J. S. (2017). ACS Sustain. Chemical Engineer, 5, 2215–2223.
Kot, A. M., Błażejak, S., Kieliszek, M., Gientka, I., Bryś, J., Reczek, L. and Pobiega, K. (2019) World J. Microbiol. Biotechnol. 35, 157, 10.
Olson, M. L., Johnson, J., Carswell, W. F., Reyes, L. H., Senger, R. S., & Kao, K. C. (2016). Characterization of an evolved carotenoids hyper-producer ofSaccharomyces cerevisiaethrough bioreactor parameter optimization and Raman spectroscopy. Journal of Industrial Microbiology & Biotechnology, 43(10), 1355–1363.
Gao, S., Tong, Y., Zhu, L., Ge, M., Zhang, Y., Chen, D., Jiang, Y., & Yang, S. (2017). Iterative integration of multiple-copy pathway genes in Yarrowia lipolytica for heterologous β-carotene production. Metabolic Engineering, 41, 192–201.
Yamane, Y., Higashida, K., Nakashimada, Y., Kakizono, T., & Nishio, N. (1997). Influence of Oxygen and Glucose on Primary Metabolism and Astaxanthin Production by Phaffia rhodozyma in Batch and Fed-Batch Cultures: Kinetic and Stoichiometric Analysis. Applied and Environmental Microbiology, 63(11), 4471–4478.
Chávez-Cabrera, C., Flores-Bustamante, Z. R., Marsch, R., Montes, M. D. C., Sánchez, S., Cancino-Díaz, J. C. and Flores-Cotera, L. B. (2010) Appl. Microbiol. Biotechnol. 85, 1953-1960, 6.
Yang, L.-B., Zhan, X.-B., Zheng, Z.-Y., Wu, J.-R., Gao, M.-J., & Lin, C.-C. (2014). A novel osmotic pressure control fed-batch fermentation strategy for improvement of erythritol production by Yarrowia lipolytica from glycerol. Bioresource Technology, 151, 120–127.
Baral, N. R., & Shah, A. (2014). Microbial inhibitors: formation and effects on acetone-butanol-ethanol fermentation of lignocellulosic biomass. Applied Microbiology and Biotechnology, 98(22), 9151–9172.
Dias, C., Sousa, S., Caldeira, J., Reis, A., & Lopes da Silva, T. (2015). Bioresour. Technol., 189, 309–318.
Honda, H., Sugiyama, H., Saito, I., & Kobayashi, T. (1998). High cell density culture of Rhodococcus rhodochrous by pH-stat feeding and dibenzothiophene degradation. Journal of Fermentation and Bioengineering, 85(3), 334–338.
Shields-Menard, S. A., AmirSadeghi, M., Green, M., Womack, E., Sparks, D. L., Blake, J., Edelmann, M., Ding, X., Sukhbaatar, B., Hernandez, R., Donaldson, J. R., & French, T. (2017). The effects of model aromatic lignin compounds on growth and lipid accumulation of Rhodococcus rhodochrous. International Biodeterioration & Biodegradation, 121, 79–90.
Author information
Authors and Affiliations
Contributions
The authors contributed to the ideation and writing of this work.
Corresponding author
Ethics declarations
Ethics Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to Participate
The authors agreed to participate in this work.
Consent for Publication
The authors agreed to publish this work.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, Z., Wan, C. Microbial Conversion of Lignin-Based Compounds into Carotenoids by Rhodococci. Appl Biochem Biotechnol 193, 3442–3453 (2021). https://doi.org/10.1007/s12010-021-03602-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-021-03602-z