Abstract
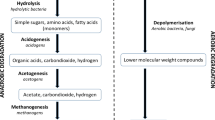
Batch methane fermentation was conducted using seed sludge collected from six methane fermentation facilities. Swine feces were centrifuged and autoclaved, followed by its use as a substrate for methanogenesis. This “swine feces supernatant medium” facilitates the cultivation of the microbes of the seed sludge, sampling of the digested liquid using a syringe, and subculturing of the digested liquid in a subsequent medium using a syringe. Through 15 subcultures, digested liquids with high and low methane production potential were obtained, which were named “H-DS” and “L-DS,” respectively. On the day 10 of cultivation, chemical oxygen demand (COD) of H-DS significantly decreased by 31% and that of L-DS did not differ significantly compared with that on the day 0 of cultivation. Acetic acid concentration of H-DS (1009 mg/L) was significantly lower than that of L-DS (2686 mg/L). These chemical characteristics indicate that organics decomposition in L-DS was not successful and suggest that H-DS has high relative abundance of bacteria decomposing organic matter and methanogen utilizing acetic acid compared with those in L-DS. Microbial community analysis revealed that Shannon index of H-DS was significantly higher than that of L-DS, and the relative abundance of acetogenic bacteria (e.g., Syntrophomonas) and acetic acid-utilizing methanogen (Methanosarcina) in H-DS was significantly higher than that in L-DS. Thus, the high methane production potential of H-DS might be attributable to the smooth flow from acetogenesis to methanogenesis step in the methane fermentation, compared with the case of L-DS.





Similar content being viewed by others
References
Batstone, D. J., Keller, J., Angelidaki, I., Kalyuzhnyi, S. V., Pavlostathis, S. G., Rozzi, A., Sanders, W. T. M., Siegrist, H., & Vavilin, V. A. (2002). The IWA anaerobic digestion model no 1 (ADM1). Water Science and Technology, 45(10), 65–73.
Beaty, P. S., & McInerney, M. J. (1989). Effects of organic acid anions on the growth and metabolism of Syntrophomonas wolfei in pure culture and in defined consortia. Applied and Environmental Microbiology, 55(4), 977–983.
Boone, D. R., & Mah, R. A. (2015). Methanosarcina. In W. B. Whitman, F. Rainey, P. Kämpfer, M. Trujillo, J. Chun, P. DeVos, B. Hedlund, & S. Dedysh (Eds.), In bergey’s manual of systematics of archaea and bacteria. https://doi.org/10.1002/9781118960608.gbm00519.
Bosshard, P. P. (2015). Turicibacter. In W. B. Whitman, F. Rainey, P. Kämpfer, M. Trujillo, J. Chun, P. DeVos, B. Hedlund, & S. Dedysh (Eds.), In bergey’s manual of systematics of archaea and bacteria. https://doi.org/10.1002/9781118960608.gbm00766.
Cai, S., & Dong, X. (2010). Cellulosilyticum ruminicola gen. nov., sp. nov., isolated from the rumen of yak, and reclassification of Clostridium lentocellum as Cellulosilyticum lentocellum comb. nov. International Journal of Systematic and Evolutionary Microbiology, 60(4), 845–849.
Caporaso, J. G., Kuczynski, J., Stombaugh, J., Bittinger, K., Bushman, F. D., Costello, E. K., Fierer, N., Peña, A. G., Goodrich, J. K., Gordon, J. I., Huttley, G. A., Kelley, S. T., Knights, D., Koenig, J. E., Ley, R. E., Lozupone, C. A., McDonald, D., Muegge, B. D., Pirrung, M., Reeder, J., Sevinsky, J. R., Turnbaugh, P. J., Walters, W. A., Widmann, J., Yatsunenko, T., Zaneveld, J., & Knight, R. (2010). QIIME allows analysis of high-throughput community sequencing data. Nature Methods, 7(5), 335–336.
Cerrillo, M., Viñas, M., & Bonmatí, A. (2016). Overcoming organic and nitrogen overload in thermophilic anaerobic digestion of pig slurry by coupling a microbial electrolysis cell. Bioresource Technology, 216, 362–372.
Cibis, K. G., Gneipel, A., & König, H. (2016). Isolation of acetic, propionic and butyric acid-forming bacteria from biogas plants. Journal of Biotechnology, 220, 51–63.
Edgar, R. C. (2010). Search and clustering orders of magnitude faster than BLAST. Bioinformatics, 26(19), 2460–2461.
Edgar, R. C., Haas, B. J., Clemente, J. C., Quince, C., & Knight, R. (2011). UCHIME improves sensitivity and speed of chimera detection. Bioinformatics, 27(16), 2194–2200.
Gerritsen, J., Fuentes, S., Grievink, W., Niftrik, L. V., Tindall, B. J., Timmerman, H. M., Rijkers, G. T., & Smidt, H. (2014). Characterization of Romboutsia ilealis gen. nov., sp. nov., isolated from the gastro-intestinal tract of a rat, and proposal for the reclassification of five closely related members of the genus Clostridium into the genera Romboutsia gen. nov., Intestinibacter gen. nov., Terrisporobacter gen. nov. and Asaccharospora gen. nov. International Journal of Systematic and Evolutionary Microbiology, 64(Pt 5), 1600–1616.
Grabowski, A., Tindall, B. J., Bardin, V., Blanchet, D., & Jeanthon, C. (2005). Petrimonas sulfuriphila gen. nov., sp. nov., a mesophilic fermentative bacterium isolated from a biodegraded oil reservoir. International Journal of Systematic and Evolutionary Microbiology, 55(3), 1113–1121.
Guo, L., Li, X. M., Bo, X., Yang, Q., Zeng, G. M., Liao, D. X., & Liu, J. J. (2008). Impacts of sterilization, microwave and ultrasonication pretreatment on hydrogen producing using waste sludge. Bioresource Technology, 99(9), 3651–3658.
Hahnke, S., Langer, T., Koeck, D. E., & Klocke, M. (2016). Description of Proteiniphilum saccharofermentans sp. nov., Petrimonas mucosa sp. nov. and Fermentimonas caenicola gen. nov., sp. nov., isolated from mesophilic laboratory-scale biogas reactors, and emended description of the genus Proteiniphilum. International Journal of Systematic and Evolutionary Microbiology, 66, 1466–1475.
Horino, H., Fujita, T., & Tonouchi, A. (2014). Description of Anaerobacterium chartisolvens gen. nov., sp. nov., an obligately anaerobic bacterium from Clostridium rRNA cluster III isolated from soil of a Japanese rice field, and reclassification of Bacteroides cellulosolvens Murray et al. 1984 as Pseudobacteroides cellulosolvens gen. nov., comb. nov. International Journal of Systematic and Evolutionary Microbiology, 64(Pt 4), 1296–1303.
Ikeda, S., Watanabe, K. N., Minamisawa, K., & Ytow, N. (2004). Evaluation of soil DNA from arable land in Japan using a modified direct-extraction method. Microbes and Environments, 19, 301–309.
Japan Sewage Works Association. (1997). Japanese standard testing methods for sewage. Japan Sewage Works Association (in Japanese).
Jarrell, K. F., Saulnier, M., & Ley, A. (1987). Inhibition of methanogenesis in pure cultures by ammonia, fatty acids, and heavy metals, and protection against heavy metal toxicity by sewage sludge. Canadian Journal of Microbiology, 33(6), 551–554.
Jiménez, J., Guardia-Puebla, Y., Romero-Romero, O., Cisneros-Ortiz, M. E., Guerra, G., Morgan-Sagastume, J. M., & Noyola, A. (2014). Methanogenic activity optimization using the response surface methodology, during the anaerobic co-digestion of agriculture and industrial wastes. Microbial community diversity. Biomass Bioenergy., 71, 84–97.
Kim, E., Lee, J., Han, G., & Hwang, S. (2018). Comprehensive analysis of microbial communities in full-scale mesophilic and thermophilic anaerobic digesters treating food waste-recycling wastewater. Bioresource Technology, 259, 442–450.
Lechner, U. (2015). Sedimentibacter. In W. B. Whitman, F. Rainey, P. Kämpfer, M. Trujillo, J. Chun, P. DeVos, B. Hedlund, & S. Dedysh (Eds.), In bergey’s manual of systematics of archaea and bacteria. https://doi.org/10.1002/9781118960608.gbm00718.
Li, J., Jha, A. K., & Bajracharya, T. R. (2014). Dry anaerobic co-digestion of cow dung with pig manure for methane production. Applied Biochemistry and Biotechnology, 173(6), 1537–1552.
Li, D., Liu, S., Mi, L., Li, Z., Yuan, Y., Yan, Z., & Liu, X. (2015). Effects of feedstock ratio and organic loading rate on the anaerobic mesophilic co-digestion of rice straw and pig manure. Bioresource Technology, 187, 120–127.
Magoc, T., & Salzberg, S. L. (2011). FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics, 27(21), 2957–2963.
Meng, X., Zhang, Y., Sui, Q., Zhang, J., Wang, R., Yu, D., Wang, Y., & Wei, Y. (2018). Biochemical conversion and microbial community in response to ternary pH buffer system during anaerobic digestion of swine manure. Energies, 11(11), 2991.
Morotomi, M., Nagai, F., & Watanabe, Y. (2012). Description of Christensenella minuta gen. nov., sp. nov., isolated from human faeces, which forms a distinct branch in the order Clostridiales, and proposal of Christensenellaceae fam. nov. International Journal of Systematic and Evolutionary Microbiology, 62(1), 144–149.
Mumtaz, T., Abd-Aziz, S., Yee, P. L., Yunus, W. M. Z. W., Shirai, Y., & Hassan, M. A. (2010). Synthesis, characterization and structural properties of intracellular copolyester poly(3-hydroxybutyrate-co-3-hydroxyvalerate) produced by Comamonas sp. EB172 from renewable resource. International Journal of Polymer Analysis and Characterization, 15(6), 329–340.
Niu, Q., Kubota, K., Qiao, W., Jing, Z., Zhang, Y., & Yu-You, L. (2015). Effect of ammonia inhibition on microbial community dynamic and process functional resilience in mesophilic methane fermentation of chicken manure. Journal of Chemical Technology and Biotechnology, 90, 2161–2169.
Oz, N. A., Ince, O., Turker, G., & Ince, B. K. (2012). Effect of seed sludge microbial community and activity on the performance of anaerobic reactors during the start-up period. World Journal of Microbiology and Biotechnology, 28(2), 637–647.
Peng, X., Zhang, Z., Luo, W., & Jia, X. (2013). Biodegradation of tetrabromobisphenol a by a novel Comamonas sp. strain, JXS-2-02, isolated from anaerobic sludge. Bioresource Technology, 128, 173–179.
Rainey, F. A., Hollen, B. J., & Small, A. M. (2015). Clostridium. In W. B. Whitman, F. Rainey, P. Kämpfer, M. Trujillo, J. Chun, P. DeVos, B. Hedlund, & S. Dedysh (Eds.), In bergey’s manual of systematics of Archaea and Bacteria. https://doi.org/10.1002/9781118960608.gbm00619.
Ren, J., Yuan, X., Li, J., Ma, X., Zhao, Y., Zhu, W., Wang, X., & Cui, Z. (2014). Performance and microbial community dynamics in a two-phase anaerobic co-digestion system using cassava dregs and pig manure. Bioresource Technology, 155, 342–351.
Schulz, H., & Eder, B. (2002). Biogas Praxis (Japanese translation edition by Y. Ukita). Ohmsha: Tokyo (in Japanese).
Sekiguchi, Y. (2015). Syntrophomonas. In W. B. Whitman, F. Rainey, P. Kämpfer, M. Trujillo, J. Chun, P. DeVos, B. Hedlund, & S. Dedysh (Eds.), bergey’s manual of systematics of archaea and bacteria. https://doi.org/10.1002/9781118960608.gbm00682.
Shah, H. N., & Hookey, J. V. (2015). Tissierella. In W. B. Whitman, F. Rainey, P. Kämpfer, M. Trujillo, J. Chun, P. DeVos, B. Hedlund, & S. Dedysh (Eds.), In bergey’s manual of systematics of archaea and bacteria. https://doi.org/10.1002/9781118960608.gbm00721.
Shen, P., Fei, H., Shuquan, S., Junya, Z., Zhineng, C., Junfang, L., Jiayi, G., Bin, F., & Bo, W. (2014). Using pig manure to promote fermentation of sugarcane molasses alcohol wastewater and its effects on microbial community structure. Bioresource Technology, 155, 323–329.
Shiratori-Takano, H., & Ueda, K. (2017). Lutispora. In W. B. Whitman, F. Rainey, P. Kämpfer, M. Trujillo, J. Chun, P. DeVos, B. Hedlund, & S. Dedysh (Eds.), In bergey’s manual of systematics of Archaea and Bacteria. https://doi.org/10.1002/9781118960608.gbm01393.
Takahashi, S., Tomita, J., Nishioka, K., Hisada, T., & Nishijima, M. (2014). Development of a prokaryotic universal primer for simultaneous analysis of Bacteria and Archaea using next-generation sequencing. PLoS One, 9(8), e105592.
Tang, Y.-Q., Shigematsu, T., Morimura, S., & Kida, K. (2015). Dynamics of the microbial community during continuous methane fermentation in continuously stirred tank reactors. Journal of Bioscience and Bioengineering, 119, 375–383.
Ueki, A., Ohtaki, Y., Kaku, N., & Ueki, K. (2016). Descriptions of Anaerotaenia torta gen. nov., sp. nov. and Anaerocolumna cellulosilytica gen. nov., sp. nov. isolated from a methanogenic reactor of cattle waste and reclassification of Clostridium aminovalericum, Clostridium jejuense and Clostridium xylanovorans as Anaerocolumna species. International Journal of Systematic and Evolutionary Microbiology, 66(8), 2936–2943.
Vrieze, J. D., Hennebel, T., Boon, N., & Verstraete, W. (2012). Methanosarcina: The rediscovered methanogen for heavy duty biomethanation. Bioresource Technology, 112, 1–9.
Wang, M., Zhou, J., Yuan, Y. X., Dai, Y. M., Li, D., Li, Z. D., Liu, X. F., Zhang, X. Y., & Yan, Z. Y. (2017). Methane production characteristics and microbial community dynamics of mono-digestion and co-digestion using corn stalk and pig manure. International Journal of Hydrogen Energy, 42(8), 4893–4901.
Yabe, S., Sakai, Y., Abe, K., & Yokota, A. (2017). Diversity of Ktedonobacteria with Actinomycetes-like morphology in terrestrial environments. Microbes and Environments, 32, 61–70.
Yang, Z., Wang, W., He, Y., Zhang, R., & Liu, G. (2018). Effect of ammonia on methane production, methanogenesis pathway, microbial community and reactor performance under mesophilic and thermophilic conditions. Renewable Energy, 125, 915–925.
Yokoyama, M. T., Spence, C., Hengemuehle, S. M., Whitehead, T. R., von Bernuth, R., & Cotta, M. (2016). Sodium tetraborate decahydrate treatment reduces hydrogen sulfide and the sulfate-reducing Bacteria population of swine manure. Journal of Environmental Quality, 45(6), 1838–1846.
Yoon, S. H., Ha, S. M., Kwon, S., Lim, J., Kim, Y., Seo, H., & Chun, J. (2017). Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. International Journal of Systematic and Evolutionary Microbiology, 67(5), 1613–1617.
Yutin, N., & Galperin, M. Y. (2013). A genomic update on clostridial phylogeny: gram-negative spore formers and other misplaced clostridia. Environmental Microbiology, 15(10), 2631–2641.
Zakaria, M. R., Abd-Aziz, S., Ariffin, H., Rahman, N. A. A., & Lai, P. (2008). Comamonas sp. EB172 isolated from digester treating palm oil mill effluent as potential polyhydroxyalkanoate (PHA) producer. African Journal of Biotechnology, 7, 4118–4121.
Zhao, H., Yang, D., Woese, C. R., & Bryant, M. P. (1993). Assignment of fatty acid-β-oxidizing syntrophic bacteria to Syntrophomonadaceae fam. nov. on the basis of 16S rRNA sequence analyses. International Journal of Systematic and Evolutionary Microbiology, 43(2), 278–286.
Zhao, Y., Yuan, X., Wen, B., Wang, X., Zhu, W., & Cui, Z. (2017). Methane potential and microbial community dynamics in anaerobic digestion of silage and dry cornstalks: a substrate exchange study. Applied Biochemistry and Biotechnology, 181(1), 91–111.
Zhou, J., Zhang, R., Liu, F., Yong, X., Wu, X., Zheng, T., Jiang, M., & Jia, H. (2016). Biogas production and microbial community shift through neutral pH control during the anaerobic digestion of pig manure. Bioresource Technology, 217, 44–49.
Zhou, S., Nikolausz, M., Zhang, J., Riya, S., Terada, A., & Hosomi, M. (2016). Variation of the microbial community in thermophilic anaerobic digestion of pig manure mixed with different ratios of rice straw. Journal of Bioscience and Bioengineering, 122(3), 334–340.
Acknowledgments
We thank Mitsui E&S Environment Engineering Co., Ltd., and Misaki Youton Corp. for providing the digested liquids, and Shoji Ookutsu (Kagoshima University) for providing the swine feces.
Funding
This research was supported by Research grant for advanced research, United Graduate School of Agricultural Sciences, Kagoshima University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(PDF 1592 kb)
Rights and permissions
About this article
Cite this article
Nakamura, Y., Ishibashi, M., Kamitani, Y. et al. Microbial Community Analysis of Digested Liquids Exhibiting Different Methane Production Potential in Methane Fermentation of Swine Feces. Appl Biochem Biotechnol 191, 1140–1154 (2020). https://doi.org/10.1007/s12010-020-03228-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-020-03228-7