Abstract
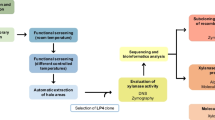
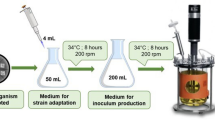
Chitin is a linear homo-polymer of N-acetyl-d-glucosamine (GlcNAc) and the second most abundant biopolymer after cellulose. Several industries rely on the bioprocesses for waste chitin recycle and hydrolysis by chitinase (EC 3.2.1.14) for potential healthcare applications through the production of its monomeric subunit, GlcNAc. In the present study, a chitinase-producing fungus (named as MFSRK-S42) was isolated from the marine water sample of North Bay of the Andaman and Nicobar Islands. It was identified as Aspergillus terreus by morphological and molecular characterization methods leveraging the internal transcribed spacer between 18S rRNA and 5.8S rRNA. Chitinase that was isolated from the fermentation broth of marine Aspergillus terreus was used to carry out biotransformation of chitineaceous wastes. Prior to the enzymatic hydrolysis step, chitins from different sources were characterized for the presence of characteristic functional groups, grain size distribution, and surface morphology. Enzymatic hydrolysis of 50 mg/ml substrate with six units of enzyme incubated for 5 days revealed 15, 36.5, 40, and 46 mg/ml GlcNAc production from ground prawn shell, chitin flakes, colloidal prawn shell, and swollen chitin respectively under standardized conditions, as determined by HPLC. In this study, 30, 73, 80, and 92% GlcNAc yields were observed from ground prawn shell, chitin flakes, colloidal prawn shell, and swollen chitin conversion respectively. The HPLC-eluted product was confirmed as GlcNAc by the presence of characteristic functional groups in FTIR and 244 Da molecular weight peak in HRMS analyses.








Similar content being viewed by others
References
Chen, J. K., Shen, G. R., & Liu, G. L. (2010). N–acetyl glucosamine: production and applications. Marine Drugs, 8(9), 2493–2516.
Cody, R. M., Davis, N. D., Lin, J., & Shaw, D. (1990). Screening microorganisms for chitin hydrolysis and production of ethanol from amino sugars. Biomass, 21(4), 285–295.
Kuk, J. H., Jung, W. J., Jo, G. H., Kim, Y. C., Kim, K. Y., & Park, R. D. (2005). Production of N-acetyl-β-D-glucosamine from chitin by Aeromonas sp. GJ-18 crude enzyme. Applied Microbiology and Biotechnology, 68(3), 384–389.
Suresh, P. V., & Anil Kumar, P. K. (2012). Enhanced degradation of α-chitin materials prepared from shrimp processing byproduct and production of N-acetyl-D-glucosamine by thermos active chitinases from soil mesophilic fungi. Biodegradation, 23(4), 597–607.
Li, H., Morimoto, K., Katagiri, N., Kimura, T., Sakka, K., Lun, S., & Ohmiya, K. (2002). A novel β-N-acetylglucosaminidase of Clostridium paraputrificum M—21 with high activity on chitobiose. Applied Microbiology and Biotechnology, 60(4), 420–427.
Gao, C., Zhang, A., Chena, K., Hao, Z., Tong, J., & Ouyanga, P. (2015). Characterization of extracellular chitinase from Chitinibacter sp. GC72 and its application in GlcNAc production from crayfish shell enzymatic degradation. Biochemical Engineering Journal, 97, 59–64.
Zhang, A., Gao, C., Wang, J., Chen, K., & Ouyang, P. (2016). An efficient enzymatic production of N-acetyl-D-glucosamine from crude chitin powders. Green Chemistry, 18(7), 2147–2154.
Sashiwa, H., Fujishima, S., Yamano, N., Kawasaki, N., Nakayama, A., Muraki, E., Hiraga, K., Oda, K., & Aiba, S. (2002). Production of N-acetyl-D-glucosamine from α-chitin by crude enzymes from Aeromonas hydrophila H-2330. Carbohydrate Research, 337(8), 761–763.
Jamialahrnadi, K., Behravan, J., Fathi Najafi, M., Tabatabaei-Yazdi, M., Shahverdi, A. R., & Faramarzi, M. A. (2011). Enzymatic production of N-acetyl-D-glucosamine from chitin using crude enzyme preparation of Aeromonas sp., PTCC1691. Biotechnology, 10(3), 292–297.
Nguyen, N. V., Young, J. K., Oh, K. T., Jung, W. K., & Park, R. D. (2008). Antifungal activity of chitinases from Trichoderma aureoviride DY-59 and Rhizopus microspores VS-9. Current Microbiology, 56(1), 28–32.
Krishnaveni, B., & Ragunathan, R. (2014). Chitinase production from marine wastes by Aspergillus terreus and its application in degradation studies. International Journal of Current Microbiology and Applied Sciences, 3(1), 76–82.
Chandrasekaran, M. (1997). Industrial enzymes from microorganisms: the Indian scenario. Journal of Marine Biotechnology, 5, 36–39.
Narayanan, K., Chopade, N., Vasanth, R. P., & Venkata, R. J. (2013). Fungal chitinase production and its application on bio-waste management. Journal of Scientific and Industrial Research, 72, 393–399.
Hsu, S. C., & Lockwood, J. L. (1975). Powdered chitin agar as a selective medium for enumeration of Actinomycetes in water and soil. Applied Microbiology, 29(3), 422–426.
Doyle, J. J., & Doyle, J. L. (1990). Isolation of plant DNA from fresh tissue. Focus, 12, 13–15.
White, T. J., Bruns, T., Lee, S., & Taylor, J. W. (1990). Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. Pp. 315–322 In: PCR protocols: a guide to methods and applications, eds. Innis, M.A., D.H. Gelfand, J. J. Sninsky, and T. J. White. Academic Press, Inc., New York.
Dey, P., Banerjee, J., & Maiti, M. K. (2011). Comparative lipid profiling of two endophytic fungal isolates—Colletotrichum sp. and Alternaria sp. having potential utilities as biodiesel feed stock. Bioresource Technology, 102(10), 5815–5823.
Tamura, K., Dudley, J., Nei, M., & Kumar, S. (2007). MEGA 4: Molecular evolutionary genetics analysis (MEGA) software version 4.0. Molecular Biology and Evolution, 24(8), 1596–1599.
De Marco, J. L., Lima, C. L. H., Desousa, M. V., & Felix, C. R. (2000). A Trichoderma harzianum chitinase destroys the cell walls of the phytopathogen Crinipellis pernicosa, the causal agent of the witches broom disease of cocoa. World Journal of Microbiology and Biotechnology, 16(4), 383–386.
Nampoothiri, K. M., Baiju, T. V., Sandhya, C., Sabu, A., Szakacs, G., & Pandey, A. (2004). Process optimization for antifungal chitinase production by Trichoderma harzianum. Process Biochemistry, 39(11), 1583–1590.
Reese, E. T., & Maguire, A. (1969). Surfactants as stimulants of enzyme production by microorganisms. Applied Microbiology, 17(2), 242–245.
Monreal, J., & Reese, E. T. (1988). The chitinase of Serratia marcescens. Canadian Journal of Microbiology, 161, 689–696.
Mudasir, R. G., Tahir, I., & Wahyni, E. T. (2008). Immobilization of dithizone onto chitin isolated from prawn sea water shells (P. merguensis) and its preliminary study for the adsorption of Cd (II) ion. Journal of Physical Science, 19, 63–78.
Lee, S. H., Kim, H. J., Sakai, E., & Daimon, M. (2003). Effect of particle size distribution of fly ash–cement system on the fluidity of cement pastes. Cement and Concrete Research, 33(5), 763–768.
Saha, P., Manna, S., Roy Chowdhury, S., Sen, R., Roy, D., & Adhikari, B. (2010). Enhancement of tensile strength of lignocellulosic jute fibers by alkali-steam treatment. Bioresource Technology, 101(9), 3182–3187.
Young, V. L., Simpson, R. M., & Ward, V. K. (2005). Characterization of an exochitinase from Epiphyas postvittana nucleopolyhedrovirus (family Baculoviridae). Journal of General Virology, 86(12), 3253–3261.
Klich, M. A. (1993). Morphological studies of Aspergillus section Versicoloes and related species. Mycologia, 85(1), 100–110.
Gardes, M., & Bruns, T. D. (1993). ITS primers with enhanced specificity for basidiomycetes: application to the identification of mycorrhiza and rusts. Molecular Ecology, 2(2), 113–118.
Velmurugan, N., Kalpana, D., Hoon, H. J., Jung, C. H., & Yang, S. L. (2011). A novel low temperature chitinase from the marine fungus Plectosphaerella sp. strain MF 1. Botanica Marina, 54, 75–81.
Chio, S., & Sangho, K. (2005). Efficient syntheses of the keto-carotenoids canthaxanthin, astaxanthin, and astacene. Journal of Organic Chemistry, 70(8), 3328–3331.
Brugnerottoa, J., Lizardib, J., Goycooleab, F. M., Argu Èelles-Monalc, W., Desbrie Áresa, J., & Rinaudoa, M. (2001). An infrared investigation in relation with chitin and chitosan characterization. Polymer, 42(8), 3569–3580.
Das, S., Sen, R., & Roy, D. (2012). Enzymatic processing of chitinaceous wastes for N-acetyl-D-glucosamine production: an example of green and efficient environmental management. Environmental Engineering and Management Journal, 11, 1849–1855.
Libowitzky, E., & Rossman, G. R. (1997). An IR absorption calibration for water in minerals. American Mineralogist, 82(11-12), 1111–1115.
Acknowledgements
The authors gratefully acknowledge the technical help received from Dr. R. Samanta and Dr. D. Gunasheelan during HPLC analyses.
Funding
SD is thankful to the National Jute Board (Govt. of India) and IIT Kharagpur for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Electronic Supplementary Material
Figure S1
(PDF 65 kb)
Rights and permissions
About this article
Cite this article
Das, S., Dey, P., Roy, D. et al. N-Acetyl-d-glucosamine Production by a Chitinase of Marine Fungal Origin: a Case Study of Potential Industrial Significance for Valorization of Waste Chitins. Appl Biochem Biotechnol 187, 407–423 (2019). https://doi.org/10.1007/s12010-018-2822-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-018-2822-3