Abstract
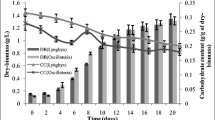
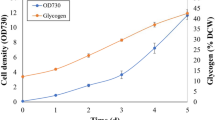
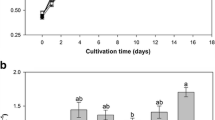
Cyanobacteria as biofertilizers are benefit to reduce the use of chemical fertilizers and reestablish the ecological system in soil. In general, several strains of cyanobacteria were involved in the biofertilizers. The co-cultivation of cyanobacteria were characterized on growth profile, production of polysaccharides and extracellular proteins, nitrogenase activity, and photosynthetic activity for three selected N2-fixing cyanobacteria, Anabaena cylindrica (B1611 and F243) and Nostoc sp. (F280). After eight-day culture, the highest dry weights were obtained in F280 pure culture and co-cultivation of B1611 and F280. Higher production of extracellular proteins and cell-bonding polysaccharides (CPS) were observed in co-cultivations compared with pure culture. The highest released polysaccharides (RPS) contents were obtained in pure culture of F280 and co-cultivation of F280 and F243. Galactose and glucose were major components of CPS and RPS in all samples. Trehalose was a specific component of RPS in F280 pure culture. Based on the monosaccharide contents of CPS and RPS, F280 was the dominant species in the related treatments of co-cultivation. The nitrogenase activities in all treatments exhibited a sharp rise at the late stage while a significant decrease existed when three cyanobacteria strains were mixed. Photosynthetic activities for all treatments were determined with rapid light curve, and the related parameters were estimated.






Similar content being viewed by others
References
Zhang, X., Davidson, E. A., Mauzerall, D. L., Searchinger, T. D., Dumas, P., & Shen, Y. (2015). Managing nitrogen for sustainable development. Nature, 528, 51–59.
Liu, J., Chu, Y., Cao, X., Zhao, Y., Xie, H., & Xue, S. (2015). Rapid transesterification of micro-amount of lipids from microalgae via a micro-mixer reactor. Biotechnology for Biofuels, 8, 229.
Li, D. J., Wang, L., Zhao, Q. Y., Wei, W., & Sun, Y. H. (2015). Improving high carbon dioxide tolerance and carbon dioxide fixation capability of Chlorella sp by adaptive laboratory evolution. Bioresource Technology, 185, 269–275.
Wang, L., Xue, C., Wang, L., Zhao, Q., Wei, W., & Sun, Y. (2016). Strain improvement of Chlorella sp. for phenol biodegradation by adaptive laboratory evolution. Bioresource Technology, 205, 264–268.
Gupta, V., Ratha, S. K., Sood, A., Chaudhary, V., & Prasanna, R. (2013). New insights into the biodiversity and applications of cyanobacteria (blue-green algae)—prospects and challenges. Algal Research, 2, 79–97.
Agarwal, A. (1979). Blue-green-algae to fertilize Indian rice paddies. Nature, 279, 181.
Bhardwaj, D., Ansari, M. W., Sahoo, R. K., & Tuteja, N. (2014). Biofertilizers function as key player in sustainable agriculture by improving soil fertility, plant tolerance and crop productivity. Microbial Cell Factory, 13, 66.
Pereira, I., Ortega, R., Barrientos, L., Moya, M., Reyes, G., & Kramm, V. (2009). Development of a biofertilizer based on filamentous nitrogen-fixing cyanobacteria for rice crops in Chile. Journal of Applied Phycology, 21, 135–144.
Gupta, A. B., & Shukla, A. C. (1969). Effect of algal extracts of Phormidium species on growth and development of rice seedlings. Hydrobiologia, 34, 77–84.
Shukla, A. C., & Gupta, A. B. (1967). Influence of algal growth-promoting substances on growth yield and protein contents of rice plants. Nature, 213, 744.
Gauri, S. S., Mandal, S. M., & Pati, B. R. (2012). Impact of Azotobacter exopolysaccharides on sustainable agriculture. Applied Microbiology Biotechnology, 95, 331–338.
Kumar, M., Prasanna, R., Bidyarani, N., Babu, S., Mishra, B. K., Kumar, A., Adak, A., Jauhari, S., Yadav, K., Singh, R., & Saxena, A. K. (2013). Evaluating the plant growth promoting ability of thermotolerant bacteria and cyanobacteria and their interactions with seed spice crops. Scientia Horticulturae, 164, 94–101.
Prasanna, R., Joshi, M., Rana, A., Shivay, Y. S., & Nain, L. (2012). Influence of co-inoculation of bacteria-cyanobacteria on crop yield and C-N sequestration in soil under rice crop. World Journal of Microbiology & Biotechnology, 28, 1223–1235.
Rana, A., Joshi, M., Prasanna, R., Shivay, Y. S., & Nain, L. (2012). Biofortification of wheat through inoculation of plant growth promoting rhizobacteria and cyanobacteria. European Journal of Soil Biology, 50, 118–126.
Karthikeyan, N., Prasanna, R., Nain, L., & Kaushik, B. D. (2007). Evaluating the potential of plant growth promoting cyanobacteria as inoculants for wheat. European Journal of Soil Biology, 43, 23–30.
Nain, L., Rana, A., Joshi, M., Jadhav, S. D., Kumar, D., Shivay, Y. S., Paul, S., & Prasanna, R. (2010). Evaluation of synergistic effects of bacterial and cyanobacterial strains as biofertilizers for wheat. Plant and Soil, 331, 217–230.
Jittawuttipoka, T., Planchon, M., Spalla, O., Benzerara, K., Guyot, F., Cassier-Chauvat, C., & Chauvat, F. (2013). Multidisciplinary evidences that Synechocystis PCC6803 exopolysaccharides operate in cell sedimentation and protection against salt and metal stresses. PloS One, 8, e55564.
Singh, S., & Das, S. (2011). Screening, production, optimization and characterization of cyanobacterial polysaccharide. World Journal of Microbiology & Biotechnology, 27, 1971–1980.
Tiwari, O. N., Khangembam, R., Shamjetshabam, M., Sharma, A. S., Oinam, G., & Brand, J. J. (2015). Characterization and optimization of bioflocculant exopolysaccharide production by cyanobacteria Nostoc sp BTA97 and Anabaena sp BTA990 in culture conditions. Applied Biochemistry and Biotechnology, 176, 1950–1963.
Rippka, R., Deruelles, J., Waterbury, J. B., Herdman, M., & Stanier, R. Y. (1979). Generic assignments, strain histories and properties of pure cultures of cyanobacteria. Journal of General Microbiology, 111, 1–61.
Kratz, F., Müller, C., Körber, N., Umanskaya, N., Hannig, M., & Ziegler, C. (2013). Characterization of protein films on dental materials: bicinchoninic acid assay (BCA) studies on loosely and firmly adsorbed protein layers. Physica Status Solidi A, 210, 964–967.
Dubois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A., & Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 28, 350–356.
Berntzon, L., Erasmie, S., Celepli, N., Eriksson, J., Rasmussen, U., & Bergman, B. (2013). BMAA inhibits nitrogen fixation in the cyanobacterium Nostoc sp. PCC 7120. Marine Drugs, 11, 3091–3108.
Ralph, P. J., & Gademann, R. (2005). Rapid light curves: a powerful tool to assess photosynthetic activity. Aquatic Botany, 82, 222–237.
Ritchie, R. J. (2008). Fitting light saturation curves measured using modulated fluorometry. Photosynthesis Research, 96, 201–215.
Acknowledgments
This study was supported by the Shanghai Municipal Agricultural Commission Program (2014, No. 5–7).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Xue, C., Wang, L., Wu, T. et al. Characterization of Co-Cultivation of Cyanobacteria on Growth, Productions of Polysaccharides and Extracellular Proteins, Nitrogenase Activity, and Photosynthetic Activity. Appl Biochem Biotechnol 181, 340–349 (2017). https://doi.org/10.1007/s12010-016-2215-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-016-2215-4