Abstract
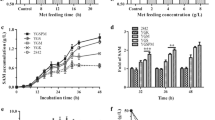
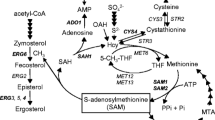
S-Adenosyl-l-methionine (SAM) plays important roles in trans-methylation, trans-sulfuration, and polyamine synthesis in all living cells, and it is also an effective cure for liver disease, depressive syndromes, and osteoarthritis. The increased demands of SAM in pharmaceuticals industry have aroused lots of attempts to improve its production. In this study, a multiple-copy integrative plasmid pYMIKP-SAM2 was introduced into the chromosome of wild-type Saccharomyces cerevisiae strain ZJU001 to construct the recombined strain R1-ZJU001. Further studies showed that the recombinant yeast exhibited higher enzymatic activity of methionine adenosyltransferase and improved its SAM biosynthesis. With a three-phase fed-batch strategy in 15-liter bench-top fermentor, 8.81 g/L SAM was achieved after 52 h cultivation of R1-ZJU001, about 27.1 % increase over its parent strain ZJU001, whereas the SAM content was also improved from 64.6 mg/g DCW to 91.0 mg/g DCW. Our results shall provide insights into the metabolic engineering of SAM pathway in yeast for improved productivity of SAM and subsequent industrial applications.





Similar content being viewed by others
References
Giulidori, P., Galli-Kienle, M., Catto, E., & Stramentinoli, G. (1984). Transmethylation, trans-sulfuration, and aminopropylation reactions of S-adenosyl-l-methionine in vivo. Journal of Biological Chemistry, 259, 4205–4211.
Winkler, W. C., Nahvi, A., Sudarsan, N., Barrick, J. E., & Breaker, R. R. (2003). An mRNA structure that controls gene expression by binding S-adenosylmethionine. Nature Structural Biology, 10, 701–707.
Lieber, C. S. (1999). Role of S-adenosyl-l-methionine in the treatment of liver diseases. Journal of Hepatology, 30, 1155–1159.
Cimino, M., Vantini, G., Algeri, S., Curatola, G., Pezzoli, C., & Stramentinoli, G. (1984). Age-related modification of dopaminergic and beta-adrenergic receptor system: restoration to normal activity by modifying membrane fluidity with S-adenosyl-l-methionine. Life Sciences, 34, 2029–2039.
Barcelo, H. A., Wiemeyer, J. C., Sagasta, C. L., Macias, M., & Barreira, J. C. (1990). Experimental osteoarthritis and its course when treated with S-adenosyl-l-methionine. Revista Clínica Española, 187, 74–78.
Shiozaki, S., Shimizu, S., & Yamada, H. (1986). Production of S-adenosyl-l-methionine by Saccharomyces sake. Journal of Biotechnology, 4, 345–354.
Lee, S. W., Park, B. S., Choi, E. S., & Oh, M. K. (2010). Overexpression of ethionine resistance gene for maximaized production of S-adenosylmethionine in Saccharomyces cerevisiae sake kyokai No.6. Korean Journal of Chemical Engineering, 27, 587–589.
Thomas, D., & Surdin-Kerjan, Y. (1991). The synthesis of the two S-adenosyl- methionine synthetases is differently regulated in Saccharomyces cereviasiae. Molecular and General Genetics, 226, 224–232.
Cantoni, G. L. (1951). Methylation of nicotinamide with a soluble enzyme system from rat liver. Journal of Biological Chemistry, 189, 203–216.
Markham, G. D., Hafner, E. W., Tabor, C. W., & Tabor, H. (1980). S-adenosyl-L-methionine synthetase from Escherichia coli. Journal of Biological Chemistry, 19(255), 9082–9092.
He, J. Y., Deng, J. J., Zheng, Y. H., & Gu, J. (2006). A synergistic effect on the production of S-adenosyl-L-methionine in Pichia pastoris by knocking in of the S-adenosyl-L-methionine synthase and knocking out of cystathionine-beta-synthase. Journal of Biotechnology, 4(126), 519–527.
Hu, X. Q., Chu, J., Zhang, S. L., Zhuang, Y. P., Wang, Y. H., Zhu, S., Zhu, Z. G., & Yuan, Z. Y. (2007). A novel feeding strategy during the production phase for enhancing the enzymatic synthesis of S-adenosyl-L-methionine by methylotrophic Pichia pastoris. Enzyme and Microbial Technology, 40, 669–674.
Zhang, J. G., Wang, X. D., Zheng, Y., Fang, G. C., & Wei, D. Z. (2008). Enhancing yield of S-adenosylmethionine in Pichia pastoris by controlling NH4 + concentration. Bioprocess and Biosystems Engineering, 2(31), 63–67.
Hu, H., Qian, J. C., Chu, J., Wang, Y. H., Zhuang, Y. P., & Zhang, S. L. (2009). Optimization of L-methionine feeding strategy for improving S-adenosyl-L-methionine production by methionine adenosyltransferase overexpressed Pichia pastoris. Applied Microbiology and Biotechnology, 83, 1105–1114.
Cao, X. T., Yang, M. H., & Xia, Y. (2012). Strain improvement for enhanced production of S-adenosyl-L-methionine in Saccharomyces cerevisiae based on ethionine-resistance and SAM synthetase activity. Annals of Microbiology, 62, 1395–1402.
Huang, Y., Gou, X., Hu, H., Xu, Q., Lu, Y., & Cheng, J. (2012). Enhanced S-adenosyl-L-methionine production in Saccharomyces cerevisiae by spaceflight culture, overexpressing methionine adenosyltransferase and optimizing cultivation. Journal of Applied Microbiology, 112, 683–694.
Chan, S. Y., & Appling, D. R. (2003). Regulation of S-adensylmethionine levels in Saccharomyces cerevisiae. Journal of Biological Chemistry, 278, 43051–43059.
Liu, H., Lin, J. P., Wu, J. P., & Cen, P. L. (2002). Production of S-adenosyl-L-methionine using Saccharomyces cerevisiae by bioconversion of L-methionine. Chemical Reaction Engineering and Technology (China), 4(18), 310–314.
Wang, Y., Shi, W. L., Liu, X. Y., Shen, Y., Bao, X. M., Bai, F. W., & Qu, Y. B. (2004). Establishment of a xylose metabolic pathway in an industrial strain of Saccharomyces cerevisiae. Biotechnology Letters, 11(26), 885–900.
Acknowledgments
We thank Prof. Xiaoming Bao (State Key Laboratory of Microbial Technology, Shandong University, China) for the donation of Plasmid pYMIKP and her critical review of this manuscript. This work was financially supported by the National Natural Science Foundation of China (21176214, 21276226) and the National High Technology Research and Development Program of China (2012AA022105A, 2011AA02A114).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, W., Shi, F., Hang, B. et al. The Improvement of SAM Accumulation by Integrating the Endogenous Methionine Adenosyltransferase Gene SAM2 in Genome of the Industrial Saccharomyces cerevisiae Strain. Appl Biochem Biotechnol 178, 1263–1272 (2016). https://doi.org/10.1007/s12010-015-1943-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1943-1