Abstract
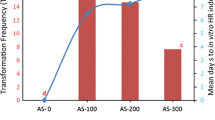
An optimized protocol for induction and establishment of Agrobacterium rhizogenes-mediated hairy root cultures of Plumbago zeylanica L. was developed through selection of suitable explant type and the bacterial strain. The infection of internodal explants from an in vivo plant and leaves of in vitro origin with the A4 strain resulted in the emergence of hairy roots at a transformation frequency of 86.33 and 42.33 %, respectively. Independent transformed root somaclones (rhizoclones) capable of sustained growth were maintained under a low illumination in auxin-free agar-solidified Murashige and Skoog (MS) medium through subcultures at periodic intervals. The presence of pRi T L -DNA rolB or rolC genes and pRi T R -DNA mas2 gene in the transformed rhizoclone genome was ascertained by PCR amplification. Concentrations and type of carbon source, auxin and media strength were optimized for root biomass growth. Five independent rhizoclones each from A4- and LBA9402-transformed root lines were studied for their plumbagin accumulation at different growth phases, using HPLC analysis. The potential for plumbagin biosynthesis was expressed in all the tested rhizoclones, although distinct inter-clonal variations were noted. It was evident that maturation of hairy roots was more important for plumbagin accumulation; slow-growing and early-maturing rhizoclones accumulated more plumbagin compared to fast-growing and late-maturing rhizoclones. A4-induced rhizoclone HRA2B5 was identified as the most superior clone with a higher plumbagin yield potential in comparison with other tested hairy root clones, in vitro-grown non-transformed roots and in vivo roots of naturally occurring P. zeylanica.










Similar content being viewed by others
References
The Wealth of India (1989). A dictionary of Indian raw materials and industrial products (Vol.2, pp. 163-164). New Delhi, India: CSIR.
Kirtikar, K. R., & Basu, B. D. (1993). Indian medicinal plants (2). Dehradun: Shiva Publishers.
Gebre-Mariam, T., Neubert, R., Schmidt, P. C., Wutzler, P., & Schmidtke, M. (2006). Journal of Ethnopharmacology, 104, 182–187.
Wang, Y. C., & Huang, T. L. (2005). Journal of Chromatography A, 1094, 99–104.
Simonsen, H. T., Nordskjold, J. B., Smitt, U. W., Nyman, U., Palpu, P., Joshi, P., & Varughese, G. (2001). Journal of Ethnopharmacology, 74, 195–204.
Dai, Y., & Hou, L. F. (2005). Pharmaceutical Biology, 43(3), 243–248.
Kavimani, S., Ilango, R., Madheswaran, M., Jayakar, B., Guupta, M., & Majumdar, U. K. (1996). Indian Journal of Pharmaceutical Science, 58(5), 194–196.
Kumar, R., Kumar, S., Patra, A., & Jayalakshmi, S. (2009). International Journal of Pharmacology and Pharmaceutical Science, 1(1), 171–175.
Ram, A. (1996). Indian Journal of Pharmacology, 28, 161–166.
Bopaiah, C. P., & Pradhan, N. (2001). Phytotherapy Research, 15, 153–156.
Itogawa, M., Takeya, K., & Furukawa, H. (1991). Planta Medica, 57, 317–319.
de Paiva, S. R., Figueiredo, M. R., Aragao, T. V., & Kaplan, M. A. C. (2003). Memórias do Instituto Oswaldo Cruz, 98, 959–961.
Bhargava, S. K. (1984). Indian Journal of Experimental Biology, 22, 153–156.
Srinivasan, L., Mathew, N., & Muthuswamy, K. (2009). Parasitology Research, 105, 1179–1182.
Bermejo-Besco´s, P., Martı´n-Arago´n, S., Jime´nez-Aliaga, K. L., Ortega, A., Molina, M. T., Buxaderas, E., Orellana, G., & Csa´ky¨, A. G. (2010). Biochemical and Biophysical Research Communications, 400, 169–174.
Hsu, Y. L., Cho, C. Y., Kuo, P. L., Huang, Y. T., & Lin, C. C. (2006). Journal of Pharmacology and Experimental Therapeutics, 318, 484–494.
Sandur, S. K., Ichikawa, H., Sethi, G., Ahn, K. S., & Aggarwal, B. B. (2006). Journal of Biological Chemistry, 281, 17023–17033.
Kitanov, G. M., & Pashankov, P. P. (1994). Pharmazie, 49, 642–646.
Komaraiah, P., Kavi Kishor, P. B., & Ramakrishna, S. V. (2001). Biotechnology Letters, 23, 1269–1272.
Panichayupakaranant, P., & Tewtrakul, S. (2002). Electronic Journal of Biotechnology, 5(3), 228–232.
Kim, Y. J., Weathers, P. J., & Wyslouzil, B. E. (2002). Biotechnology and Bioengineering, 80(4), 454–464.
Kim, Y. J., Wyslouzil, B. E., & Weathers, P. J. (2002). In Vitro Cellular and Developmental Biology-Plant, 38, 1–10.
Nayak, P. (2013). In vitro propagation and plumbagin production from Agrobacterium-transformed hairy root cultures of Plumbago zeylanica L.—an important medicinal plant species. Ph.D. Thesis, Bhubaneswar (Odisha), India: Utkal University.
Guillon, S., Trémouillaux-Guiller, J., Pati, P. K., Rideau, M., & Gantet, P. (2006). Current Opinion in Plant Biology, 9, 341–346.
Guillon, S., Tremouillaux-Guiller, J., Pati, P. K., Rideau, M., & Gantet, P. (2006). Trends in Biotechnology, 24, 403–409.
Georgiev, M. I., Pavlov, A. I., & Bley, T. (2007). Applied Microbiology and Biotechnology, 74, 1175–1185.
Veena, V., & Taylor, C. G. (2007). In Vitro Cellular and Developmental Biology-Plant, 43, 383–403.
Nilsson, O., & Olsson, O. (1997). Physiologia Plantarum, 100, 463–473.
Aoki, S., & Syono, K. (1999). Plant and Cell Physiology, 40, 252–256.
Bulgakov, V. P. (2008). Biotechnology Advances, 26, 318–324.
Chandra, S. (2012). Biotechnology Letters, 34, 407–415.
Shkryl, Y. N., Veremeichik, G. N., Bulgakov, V. P., Tchernoded, G. K., Mischenko, N. P., Fedoreyev, S. A., & Zhuravlev, Y. N. (2008). Biotechnology and Bioengineering, 100, 118–125.
Kiselev, K. V., Dubrovina, A. S., & Bulgakov, V. P. (2009). Applied Microbiology and Biotechnology, 82(4), 647–655.
Majumdar, S., Garai, S., & Jha, S. (2011). Plant Cell Reports, 30, 941–954.
Ahlawat, S., Saxena, P., Ram, M., Alam, P., Nafis, T., Mohd, A., & Abdin, M. Z. (2012). African Journal of Biotechnology, 11(35), 8684–8691.
Swain, S. S., Rout, K. K., & Chand, P. K. (2012). Applied Biochemistry and Biotechnology, 168, 487–503.
Georgiev, M. I., Agostini, E., Ludwig-Müller, J., & Xu, J. (2012). Trends in Biotechnology, 30(10), 528–537.
Gangopadhyay, M., Sircar, D., Mitra, A., & Bhattacharya, S. (2008). Biologia Plantarum, 52(3), 533–537.
Gangopadhyay, M., Dewanjee, S., Chakraborty, D., & Bhattacharya, S. (2011). Industrial Crops Products, 33, 445–450.
Yogananth, N., & Jothi Basu, M. (2009). Global Journal of Biotechnology and Biochemistry, 4(1), 66–69.
Verma, P. C., Singh, D., Rahman, L., Gupta, M. M., & Banerjee, S. (2002). Journal of Plant Physiology, 159, 547–552.
Sivanesan, I., & Jeong, B. R. (2009). African Journal of Biotechnology, 8(20), 5294–5300.
Sakamoto, S., Putalun, W., Pongkitwitoon, B., Juengwatanatrakul, T., Shoyama, Y., Tanaka, H., & Morimoto, S. (2012). Plant Cell Reports, 31, 103–110.
Murashige, T., & Skoog, F. (1962). Physiologia Plantarum, 15, 473–497.
Nayak, P., Thirunavoukkarasu, M., & Chand, P. K. (2011). Plant Science Research, 33(1&2), 44–51.
Doyle, J. J., & Doyle, J. L. (1990). Focus, 12, 13–15.
Slightom, J. L., Durandtardif, M., Jouanin, L., & Tepfer, D. (1986). Journal of Biological Chemistry, 261, 108–121.
Jouanin, L. (1984). Plasmid, 12, 91–102.
Soudek, P., Podlipna, R., Marsik, P., & Vanek, T. (2005). Biologia Plantarum, 49(4), 487–492.
Pal, A., Swain, S. S., Mukherjee, A. K., & Chand, P. K. (2013). Food Technology and Biotechnology, 51(1), 26–35.
Chaudhuri, K. N., Ghosh, B., Tepfer, D., & Jha, S. (2005). Plant Cell Reports, 24, 25–35.
Verma, P. C., Rahman, L. U., Nagi, A. S., Jain, D. C., Khanuja, S. P. S., & Banerjee, S. (2007). Plant Biotechnology Reports, 1, 169–174.
Swain, S. S., Sahu, L., Barik, D. P., & Chand, P. K. (2010). Scientia Horticulturae, 125, 461–468.
Swain, S. S., Sahu, L., Pal, A., Barik, D., Pradhan, C., & Chand, P. K. (2012). World Journal of Microbiology and Biotechnology, 28, 729–739.
Tiwari, R. K., Trivedi, M., Guang, Z. C., Guo, G. Q., & Zheng, G. C. (2007). Plant Cell Reports, 26, 199–210.
Garg, S. (2001). Biotechnological approaches for genetic improvement and conservation of Coleus forskolii. Ph.D. Thesis, Lucknow, India: Lucknow University.
Kuzma, L., Rozalskib, M., Walenckac, E., Rozalskac, B., & Wysokinska, H. (2007). Phytomedicine, 14, 31–35.
Farkya, S., & Bisaria, V. S. (2008). Journal of Bioscience and Bioengineering, 105(2), 140–146.
Tepfer, D. (1984). Cell, 37, 959–967.
Kamada, H., Okamura, N., Satake, M., Harada, H., & Shimomura, K. (1986). Plant Cell Reports, 5, 239–242.
Fu, C. X., De Zhao, X., Xue, X. F., Jin, Z. P., & Ma, F. S. (2005). Process Biochemistry, 40, 3789–3794.
Palazón, J., Cusidó, R. M., Roig, C., & Piñol, M. T. (1998). Plant Cell Reports, 17, 384–390.
Palazón, J., Cusidó, R. M., Gonzalo, J., Bonfill, M., Morales, S., & Piñol, M. T. (1998). J Plant Physiology, 153, 712–718.
Bulgakov, V. P., Khodakovskaya, M. V., Labetskaya, N. V., Tchernoded, G. K., & Zhuravlev, Y. N. (1998). Phytochemistry, 49, 1929–1934.
Bonhomme, V., Laurain Mattar, D., & Fliniaux, M. A. (2000). Journal of Natural Products, 63, 1249–1252.
Hu, Z.-B., & Du, M. (2006). Journal of Integrative Plant Biology, 48(2), 121–127.
Oksman-Caldentey, K. M., Sevón, N., Vanhala, L., & Hiltunen, R. (1994). Plant Cell Tissue and Organ Culture, 38, 263–272.
Jung, K. H., Kwak, S. S., Kim, S. W., Lee, H., Choi, C. Y., & Lin, J. R. (1992). Biotechnology Letters, 14(8), 695–700.
Young-Am, C., Yu, H., Song, J., Chun, H., & Park, S. (2000). Biotechnology Letters, 22, 1527–1530.
Liu, C. Z., Wang, Y. C., Ouyang, F., Ye, H. C., & Li, G. F. (1997). Biotechnology Letters, 19(9), 927–929.
Jacob, A., & Malpathak, N. (2004). Current Science, 87(10), 1442–1447.
Uozumi, N., Kohketsu, K., Kondo, O., Honda, H., & Kobayashi, T. (1991). Journal of Fermentation Bioengineering, 72, 457–460.
Avigad, G. (1982). Sucrose and other disaccharides. In F. A. Loewus & W. Tanner (Eds.), Plant carbohydrates (Vol. 13A, pp. 217–271). Berlin: Springer-Verlag.
Borkowska, B., & Szczebra, J. (1991). Journal of Experimental Botany, 42, 911–915.
Sim, S. J., Chang, H. N., Liu, J. R., & Jung, K. H. (1994). Journal of Fermentation Bioengineering, 78, 229–234.
Xu, H., Park, J. H., Kim, Y. K., Park, N., Lee, S. Y., & Park, S. U. (2009). Journal of Medicinal Plant Research, 3(11), 978–981.
Drewes, F. E., & Staden, J. V. (1995). Plant Growth Regulation, 17, 27–31.
Shen, W. H., Petit, A., Guern, J., & Tempe, J. (1988). Proceedings of National Academy of Sciences USA, 58, 3417–3421.
Deno, H., Yamagata, H., Emoto, T., Yoshioka, T., Yamada, Y., & Fujita, Y. (1987). Journal of Plant Physiology, 131, 315–323.
Rodriguez-Talou, J., & Giulielti, A. M. (1995). Biotechnology Letters, 17, 1337–1342.
Croes, A. F., Van der Berg, A. J. R., Bosveld, M., Breteler, H., & Wullems, G. J. (1989). Planta, 179, 43–50.
Batra, J., Dutta, A., Singh, D., Kumar, S., & Sen, J. (2004). Plant Cell Reports, 23, 148–154.
Acknowledgments
The technical support provided by the Directors of the Institute of Minerals and Materials Technology (CSIR), Bhubaneswar, India, and of the Indian Institute of Technology, Kanpur, India, is gratefully acknowledged. One of the authors (P. Nayak) acknowledges the Council of Scientific and Industrial Research (CSIR), Government of India, for financial support in form of a Senior Research Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nayak, P., Sharma, M., Behera, S.N. et al. High-Performance Liquid Chromatographic Quantification of Plumbagin from Transformed Rhizoclones of Plumbago zeylanica L.: Inter-Clonal Variation in Biomass Growth and Plumbagin Production. Appl Biochem Biotechnol 175, 1745–1770 (2015). https://doi.org/10.1007/s12010-014-1392-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-1392-2