Abstract
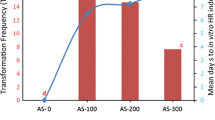
A single-chain variable fragment antibody (scFv) against plumbagin (PL) accumulated the PL production in the hairy roots of Plumbago zeylanica. Recombinant Agrobacterium rhizogenes (ATCC 15834) containing an scFv gene against PL (PL-scFv) were obtained through triparental mating and transformed into P. zeylanica to induce PL-scFv protein in the hairy roots. Up to 40 μg recombinant PL-scFv were expressed per milligram of soluble protein in transgenic P. zeylanica hairy root cultures. The mean PL content obtained from transgenic hairy roots (12.24 μg/100 mg dry weight) exhibited 2.2 times higher than those obtained from wild-type (5.48 μg/100 mg dry weight). The high correlation between the PL-scFv expression level and PL content of the recombinant plants suggested that the PL biosynthesis pathway had been modulated by the expression of PL-scFv protein in the hairy roots of P. zeylanica.




Similar content being viewed by others
References
Artsaenko O, Peisker M, Zurnieden U, Fiedler U, Weiler EW, Muntz K, Conrad U (1995) Expression of a single-chain Fv antibody against abscisic acid creates a wilty phenotype in transgenic tobacco. Plant J 8:745–750
Bermejo-Bescós P, Martín-Aragón S, Jiménez-Aliaga KL, Ortega A, Molina MT, Buxaderas E, Orellana G, Csákÿ AG (2010) In vitro antiamyloidogenic properties of 1, 4-naphthoquinones. Biochem Biophys Res Commun 400:169–174
Bhargava SK (1984) Effects of plumbagin on reproductive function of male dog. Indian J Exp Biol 22:153–156
Bradford MM (1976) A rapid and sensitive for the quantitation of microgram quantitites of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
de Paiva SR, Figueiredo MR, Aragao TV, Kaplan MAC et al (2003) Antimicrobial activity in vitro of plumbagin isolated from Plumbago species. Mem Inst Oswaldo Cruz 98:959–961
Eto J, Suzuki Y, Ohkawa H, Yamaguchi I (2003) Anti-herbicide single-chain antibody expression confers herbicide tolerance in transgenic plants. FEBS Lett 550:179–184
Fiedler U, Philips J, Artsaenko O, Conrad U (1997) Optimization of scFv antibody production in transgenic plants. Immunotechnology 3:205–216
Gangopadhyay M, Sircar D, Mitra A, Bhattacharya S (2008) Hairy root culture of Plumbago indica as a potential source for plumbagin. Biol Plant 52:533–537
Hiatt AC, Cafferkey R, Bowdish K (1989) Production of antibodies in transgenic plants. Nature 342:76–78
Harmsen MM, Langedijk AC, Vantuinen E, Geerse RH, Raue HA, Maat J (1993) Effect of a pmr 1 disruption and different signal sequences on the intracellular processing and secretion of Cyamopsis tetragonoloba alpha-galactosidase by Saccharomyces cerevisiae. Gene 125:115–123
Hsu YL, Cho CY, Kuo PL, Huang YT, Lin CC (2006) Plumbagin (5-hydroxy-2-methyl-1, 4-naphthoquinone) induces apoptosis and cell cycle arrest in A549 cells through p53 accumulation via c-Jun NH2-terminal kinase-mediated phosphorylation at serine 15 in vitro and in vivo. J Pharmacol Exp Ther 318:484–494
Itogawa M, Takeya K, Furukawa H (1991) Cardiotonic action of plumbagin on guinea-pig papillary muscle. Planta Med 57:317–319
Kitanov GM, Pashankov PP (1994) Quantitative investigation on the dynamics of plumbagin in Plumbago europea L. roots and herb by HPLC. Pharmazie 49:642–646
Komaraiah P, Kavi Kishor PB, Ramakrishna SV (2001) Production of plumbagin from cell suspension cultures of Plumbago rosea L. Biotechnol Lett 23:1269–1272
Komaraiah P, Naga Amrutha RP, Kavi Kishor B, Ramakrishna SV (2002) Elicitor enhanced production of plumbagin in suspension cultures of Plumbago rosea L. Enzyme Microb Technol 31:634–639
Putalun W, Udomsin O, Yusakul G, Juengwatanatrakul T, Sakamoto S, Tanaka H (2010) Enhanced plumbagin production from in vitro cultures of Drosera burmanii using elicitation. Biotechnol Lett 32:721–724
Putalun W, Taura F, Qing W, Matsushita H, Tanaka H, Shoyama Y (2003) Anti-solasodine glycoside single-chain Fv antibody stimulates biosynthesis of solasodine glycoside in plants. Plant Cell Rep 22:344–349
Sakamoto S, Putalun W, Tsuchihashi R, Morimoto S, Kinjo J, Tanaka H (2008) Development of an enzyme-linked immunosorbent assay (ELISA) using highly-specific monoclonal antibodies against plumbagin. Anal Chim Acta 607:100–105
Sakamoto S, Taura F, Putalun W, Pongkitwitoon B, Tsuchihashi R, Morimoto S, Kinjo J, Shoyama Y, Tanaka H (2009) Construction and expression of specificity-improved single-chain variable fragments against the bioactive naphthoquinone, plumbagin. Biol Pharm Bull 32:434–439
Sandur SK, Ichikawa H, Sethi G, Ahn KS, Aggarwal BB (2006) Plumbagin (5-hydroxy-2-methyl-1, 4-naphthoquinone) suppresses NF-kappaB activation and NF-kappaB-regulated gene products through modulation of p65 and IkappaBalpha kinase activation, leading to potentiation of apoptosis induced by cytokine and chemotherapeutic agents. J Biol Chem 281:17023–17033
Srinivasan L, Mathew N, Muthuswamy K (2009) In vitro antifilarial activity of glutathione S-transferase inhibitors. Parasitol Res 105:1179–1182
Suzuki Y, Mizuno T, Urakami E, Yamaguchi I, Asami T (2008) Immunomodulation of bioactive gibberellin confers gibberellin-deficient phenotypes in plants. Plant Biotechnol J 6:355–367
Taussig R, Carlson M (1983) Nucleotide sequence of the yeast SUC2 gene for invertase. Nucl Acids Res 11:1943–1954
Verma PC, Singh D, Rahman LU, Gupta MM, Banerjee S (2002) In vitro-studies in Plumbago zeylanica: rapid micropropagation and establishment of higher plumbagin yielding hairy root cultures. J Plant Physiol 159:547–552
Acknowledgments
We appreciate for the technical supports from the Research Support Center, Graduate School of Medical Sciences, Kyushu University. This work was funded by the Research Fellowship of the Japan Society for the Promotion of Science for Young Scientists. The research in this paper was also supported, in part, by a Grant in Aid from the Japan Society for the Promotion of Science Asian CORE Program; the Ministry of Education, Culture, Sports, Science, and Technology of Japan; and the National Center for Genetic and Biotechnology (BIOTEC), Thailand.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Q. Zhao.
Rights and permissions
About this article
Cite this article
Sakamoto, S., Putalun, W., Pongkitwitoon, B. et al. Modulation of plumbagin production in Plumbago zeylanica using a single-chain variable fragment antibody against plumbagin. Plant Cell Rep 31, 103–110 (2012). https://doi.org/10.1007/s00299-011-1143-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-011-1143-6