Abstract
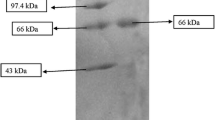
This study illustrates the biophysicochemical properties of an alkaline protease, BAP (Beauveria sp. alkaline protease) from Beauveria sp. MTCC 5184. This protease exhibited maximum activity at 50 °C, pH 9.0, and stability in a broad pH range, in the presence of organic solvents, denaturants, as well as detergents. Wash performance studies revealed that BAP was able to remove blood clots/stains from blood-soaked cloth. Peptide mass fingerprinting results demonstrated partial homology of BAP with subtilisin-like proteinase. BAP showed catalytic activity against natural as well as synthetic substrates. Active site characterization of BAP confirmed the involvement of serine, tryptophan, and aspartic acid in catalytic activity. Detailed kinetic and thermodynamic studies of BAP demonstrated that the activation energy (Ea) for casein hydrolysis was 82.55 kJ/M, the specificity constant (Kcat/K m), and the values of ∆G (change in Gibbs free energy) decreased with increase in temperature, whereas ∆H (change in enthalapy) and ∆S (change in entropy) were constant. The results of the present study indicate that BAP has potential for applications as detergent additive, in peptide synthesis, and in basic research.



Similar content being viewed by others
References
Shankar, S., Rao, M., & Laxman, R. S. (2011). Process Biochemistry, 46, 579–585.
Rao, M. B., Tanksale, A. M., Ghatge, M. S., & Deshpande, V. V. (1998). Microbiology and Molecular Biology Reviews, 62, 597–635.
Gupta, R., Beg, Q. K., & Lorenz, P. (2002). Applied Microbiology and Biotechnology, 59, 15–32.
Karbalaei-Heidari, H. R., Shahbazi, M., & Absalan, G. (2013). Applied Biochemistry and Biotechnology, 170, 573–586.
Paul, T., Das, A., Mandal, A., Halder, S. K., DasMohapatra, P. K., Pati, B. R., & Mondal, K. C. (2013). Waste Biomass Valor. doi:10.1007/s12649-013-9265-4.
Zambre, V. P., Nilegaonkar, S. S., & Kanekar, P. P. (2013). IIOAB Letter. doi:10.5195/iioablett.2013.19.
Makhdum, Z., Rehman, H., Larik, J. M., Bux, P., & Hameed, A. (2013). Journal of Applied Animal Research, 41, 218–222.
Boyce, A., & Walsh, G. (2012). Chemosphere, 88, 211–218.
More, S. V., Khandelwal, H. B., Joseph, M. A., & Laxman, R. S. (2013). Journal of Natural Fibres, 10, 98–111.
Shankar, S., More, S. V., & Laxman, R. S. (2010). Kathmandu University Journal of Science Engineering & Technology, 6, 60–69.
Wang, S. L., Yang, C. H., Liang, T. W., & Yen, Y. H. (2008). Bioresource Technology, 99, 3700–3707.
Ghorbel, B., Kamoun, A. S., & Nasri, M. (2003). Enzyme and Microbial Technology, 32, 513–518.
Gupta, A., & Khare, S. K. (2009). Critical Reviews in Biotechnology, 29, 44–54.
Vijayaraghavan, P., Vijayan, A., Arun, A., Jenisha, J. K., & Vincent, S. G. P. (2012). SpringerPlus, 1, 76–84.
Patel, G. K., Kawale, A. A., & Sharma, A. K. (2012). Plant Physiology & Biochemistry, 52, 104–111.
Rathinaraj, K., Sakhare, P. Z., Sachindra, N. M., & Mahendrakar, N. S. (2010). Food Bioprocess Technology, 3, 783–788.
Erlacher, A., Sousa, F., Schroeder, M., Jus, S., Kokol, V., Cavaco-Paulo, A., & Guebitz, G. M. (2006). Biotechnology Letter, 28, 703–710.
Pena-Montes, C., González, A., Castro-Ochoa, D., & Farres, A. (2008). Applied Microbiology & Biotechnology, 78, 603–612.
Laxman, R. S., Shankar, S., More, S. V., Khandelwal, H. B., Narasimhan, C. B. K., Palanivel, S. & Balaram, P. (2013). Euro. Patent. EP 2531596. US. Patent. US 20130095523
Laxman, R. S., Sonawane, A. P., More, S. V., Rao, B. S., Rele, M. V., Jogdand, V. V., Deshpande, V. V., & Rao, M. B. (2005). Process Biochemistry, 40, 3152–3158.
Bradford, M. M. (1976). Analytical Biochemistry, 72, 248–254.
Laemmli, U. K. (1970). Nature, 227, 680–685.
Shashidhara, K. S., & Gaikwad, S. M. (2009). International Journal of Biological Macromolecule, 44, 112–115.
Barberis, S., Quiroga, E., Morcelle, S., Priolo, N., & Luco, J. M. (2006). Journal of Molecular Catalysis B: Enzymatic, 38, 95–103.
Gupta, A., & Khare, S. K. (2007). Enzyme & Microbial Technology, 42, 11–16.
Ogino, H., Watanabe, F., Yamada, M., Nakagawa, S., Hirose, T., Noguchi, A., Yasuda, M., & Ishikawa, H. (1999). Journal of Bioscience & Bioengineering, 87, 61–68.
Barberis, S., Quiroga, E., Arribére, M. C., & Priolo, N. (2002). Journal of Molecular Catalysis B: Enzymatic, 17, 39–47.
Gupta, A., Roy, I., Khare, S. K., & Gupta, M. N. (2005). Journal of Chromatography. A, 1069, 155–161.
Ruiz, D. M., & De Castro, R. E. (2007). Journal of Industrial Microbiology & Biotechnology, 34, 111–115.
Hajji, M., Kanoun, S., Nasri, M., & Gharsallah, N. (2007). Process Biochemistry, 42, 791–797.
Esposito, T. S., Marcuschi, M., Amatal, I. P. G., Carvalho, L. B., & Bezerra, R. S. (2010). Journal of Agricultural Food Chemistry, 58, 6433–6439.
Phadatare, S. U., Srinivasan, M. C., & Deshpande, V. V. (1993). Enzyme & Microbial Technology, 15, 72–76.
Tanksale, A., Manish, C. P., Rao, M., & Deshpande, V. (2001). Biotechnology Letter, 23, 51–54.
Sindhu, R., Suprabha, G. N., & Shashidhar, S. (2009). African Journal of Microbiological Research, 3, 515–522.
Pappin, D. J. C., Hojrup, P., & Bleasby, A. J. (1993). Current Biology, 3, 327–332.
Sutar, I. I., Srinivasan, M. C., & Vartak, H. G. (1991). Biotechnology Letter, 13, 119–124.
Tsuchiya, K., Arai, T., Seki, K., & Kimura, T. (1987). Agricultural BioChemistry, 51, 2959–2965.
Bidochka, M. J., & Khachatourians, G. G. (1987). Applied & Environmental Biology, 53, 1679–1684.
Pol, D., Menon, V., & Rao, M. (2012). Extremophiles, 16, 135–146.
Singh, R. S., Saini, G. K., & Kennedy, J. F. (2010). Carbohydrate Polymer, 81, 252–259.
Marin, E., Sanchez, L., Perez, M. D., Puyol, P., & Calvo, M. (2003). Journal of Food Science, 68, 89–93.
Gote, M. M., Khan, M. I., & Khire, J. M. (2007). Enzyme & Microbial Technology, 40, 1312–1320.
Phadtare, S., Rao, M., & Deshpande, V. (1997). Archive of Microbiology, 166, 414–417.
Chellappan, S., Jasmin, C., Basheer, S. M., Kishore, A., Elyas, K. K., Bhat, S. G., & Chandrasekaran, M. (2011). Journal of Industrial Microbiology & Biotechnology, 38, 743–752.
Savitha, S., Sadhasivam, S., Swaminathan, K., & Lin, F. H. (2011). Journal of the Taiwan Institute of Chemical Engineers, 42, 298–304.
Papagianni, M., & Sergelidis, D. (2014). Applied Biochemistry & Biotechnology, 172, 3926–3938.
Cavello, I. A., Hours, R. A., Rojas, N. L., & Cavalitto, S. F. (2013). Process Biochemistry, 48, 972–978.
Acknowledgments
Shiv Shankar is grateful to the Council of Scientific and Industrial Research (CSIR) for financial assistance. Authors acknowledge the financial support from NMITLI project funded by CSIR, Govt. of India, and Dr. Mahesh J Kulkarni for analysis of samples in MALDI.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Shankar, S., Laxman, R.S. Biophysicochemical Characterization of an Alkaline Protease from Beauveria sp. MTCC 5184 with Multiple Applications. Appl Biochem Biotechnol 175, 589–602 (2015). https://doi.org/10.1007/s12010-014-1314-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-1314-3