Abstract
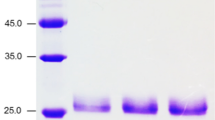
Xylans are major hemicellulose components of plant cell wall which can be hydrolyzed by xylanolytic enzymes. Three forms of endo-β-1,4-xylanases (XynSW1, XynSW2A, and XynSW2B) produced by thermotolerant Streptomyces sp. SWU10 have been reported. In the present study, we described the expression and characterization of the fourth xylanase enzyme from this bacteria, termed XynSW3. The gene containing 726 bp was cloned and expressed in Escherichia coli. The recombinant enzyme (rXynSW3) was purified from cell-free extract to homogeneity using Ni-affinity column chromatography. The apparent molecular mass of rXynSW3 was 48 kDa. Amino acid sequence analysis revealed that it belonged to a xylanase of glycoside hydrolase family 11. The optimum pH and temperature for enzyme activity were 5.5–6.5 and 50 °C, respectively. The enzyme was stable up to 40 °C and in wide pH ranges (pH 0.6–10.3). Xylan without arabinosyl side chain is the most preferable substrate for the enzyme. By using birch wood xylan as substrate, rXynSW3 produced several oligosaccharides in the initial stage of hydrolysis, and their levels increased with time, demonstrating that the enzyme is an endo-acting enzyme. The major products were xylobiose, triose, and tetraose. The rXynSW3 can be applied in several industries such as food, textile, and biofuel industries, and waste treatment.




Similar content being viewed by others
References
Collins, T., Gerday, C., & Feller, G. (2005). FEMS Microbiology Review, 29, 3–23.
Wilkie, K. C. B. (1979). Advances in Carbohydrate Chemistry and Biochemistry, 36, 215–264.
Kormelink, F. J. M., & Voragen, A. G. J. (1993). Applied Microbiology and Biotechnology, 38, 688–695.
Hector, R. E., Qureshi, N., Hughes, S. R., & Cotta, M. A. (2008). Applied Microbiology and Biotechnology, 80, 675–684.
Beg, Q. A., Kapoor, M., Mahajan, G., & Hoondal, S. (2001). Applied Microbiology and Biotechnology, 56, 326–338.
Kuhad, R. C., & Singh, A. (1993). Critical Reviews in Biotechnology, 13, 151–172.
Collins, T., De Vos, D., Hoyoux, A., Savvides, S. N., Gerday, C., Van Beeumen, J., et al. (2005). Journal of Molecular Biology, 354, 425–435.
Biely, P. (1985). Trends in Biotechnology, 3, 286–290.
Biely, P., Vrsanská, M., & Claeyssens, M. (1991). European Journal of Biochemistry, 200, 157–163.
Vrancken, K., Van Mellaert, L., & Anné, J. (2010). Methods in Molecular Biology, 668, 97–107.
Deesukon, W., Nishimura, Y., Harada, N., Sakamoto, T., & Sukhumsirichart, W. (2011). Process Biochemistry, 46, 2255–2262.
Deesukon, W., Nishimura, Y., Sakamoto, T., & Sukhumsirichart, W. (2013). Molecular Biotechnology, 54, 37–46.
Somogyi, M. (1952). Journal of Biological Chemistry, 195, 19–23.
Li, N., Yang, P., Wang, Y., Luo, H., Meng, K., Wu, N., et al. (2008). Journal of Microbiology and Biotechnology, 18, 410–416.
Altschul, S. F., Madden, T. L., Schaffer, A. A., Zhang, J., Zhang, Z., Miller, W., et al. (1997). Nucleic Acids Research, 25, 3389–3402.
Bendtsen, J. D., Nielsen, H., von Heijne, G., & Brunak, S. (2004). Journal of Molecular Biology, 340, 783–795.
Laemmli, U. K. (1970). Nature, 227, 680–685.
Blumenkrantz, N., & Asboe-Hansen, G. (1973). Analytical Biochemistry, 54, 484–489.
Bentley, S. D., Chater, K. F., Cerdeno-Tarraga, A. M., Challis, G. L., Thomson, N. R., James, K. D., et al. (2002). Nature, 417, 141–147.
Shareck, F., Roy, C., Yaguchi, M., Morosoli, R., & Kluepfel, D. (1991). Gene, 107, 75–82.
Huang, J., Wang, G., & Xiao, L. (2006). Bioresource Technology, 97, 802–808.
Mao, L., Meng, P., Zhou, C., Ma, L., Zhang, G., & Ma, Y. (2012). World Journal of Microbiology and Biotechnology, 28, 777–784.
Qu, W., & Shao, W. (2011). Biotechnology Letters, 33, 1407–1416.
Li, N., Shi, P., Yang, P., Wang, Y., Luo, H., Bai, Y., et al. (2009). Applied Biochemistry and Biotechnology, 159, 521–531.
Kui, H., Luo, H., Shi, P., Bai, Y., Yuan, T., Wang, Y., et al. (2010). Applied Biochemistry and Biotechnology, 162, 953–965.
Liu, W., Shi, P., Chen, Q., Yang, P., Wang, G., Wang, Y., et al. (2010). Applied Biochemistry and Biotechnology, 162, 1–12.
Luo, H., Wang, Y., Li, J., Wang, H., Yang, J., Yang, Y., et al. (2009). Enzyme and Microbial Technology, 45, 126–133.
Decelle, B., Tsang, A., & Storms, R. K. (2004). Current Genetics, 46, 166–175.
Biely, P., Vrsanská, M., Tenkanen, M., & Kluepfel, D. (1997). Journal of Biotechnology, 57, 151–166.
Vardakou, M., Katapodis, P., Samiotaki, M., Kekos, D., Panayotou, G., & Christakopoulos, P. (2003). International Journal of Biological Macromolecules, 33, 129–134.
Pollet, A. (2010). Ph.D. Thesis, Katholieke Universiteit Leuven, Leuven.
Prade, R. A. (1996). Biotechnology and Genetic Engineering Reviews, 13, 101–131.
Acknowledgments
This work was supported by grants from the Royal Golden Jubilee Ph.D. Program of Thailand Research Fund (PHD/0257/2549), Srinakharinwirot University (2557), and JSPS/NRCT (ACP program). The authors would like to thank Dr. Alfredo Villarroel for proofreading the article.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Sukhumsirichart, W., Deesukon, W., Kawakami, T. et al. Expression and Characterization of Recombinant GH11 Xylanase from Thermotolerant Streptomyces sp. SWU10. Appl Biochem Biotechnol 172, 436–446 (2014). https://doi.org/10.1007/s12010-013-0508-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0508-4