Abstract
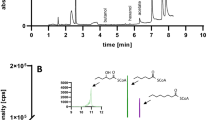
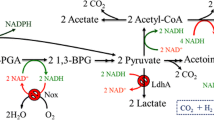
Acetogen Clostridium sp. MT1802 originally producing 336-mM acetate from inorganic carbon of CO2/CO was engineered to eliminate acetate production and sporulation using Cre-lox66/lox71-approach. The recombinant started producing 105-mM formate expressing synthetic formate dehydrogenase integrated in two copies. Formate-producing recombinant was further engineered to express synthetic formate acetyltransferase, acetolactate synthase, acetolactate decarboxylase, and alcohol dehydrogenase integrated in two copies each using Tn7 tool. The resulted recombinant started producing 102-mM 2,3-butanediol (23BD). 23BD production was confirmed in five independent single step fermentation runs 25 days long each in five repeats using syngas blend 60 % CO and 40 % H2 (v/v) (p <0.005). 23BD production was 78 % if only CO2/H2 blend was fed instead of syngas (p <0.005). 23BD from CO2/H2 blend might serve as a commercial route to mitigate global warming in proportion to CO2 fermentation scale worldwide.











Similar content being viewed by others
References
Young, K. D. (2006). The selective value of bacterial shape. Microbiology and Molecular Biology Reviews, 70(3), 660–703. doi:10.1128/MMBR.00001-06.
Berzin, V., Kiriukhin, M., & Tyurin, M. (2012). Selective production of acetone during continuous synthesis gas fermentation by engineered biocatalyst Clostridium sp. MAceT113. Letters in Applied Microbiology, 55, 149–154. doi:10.1111/j.1472-765X.2012.03272.x.
Tyurin, M., & Kiriukhin, M. (2013). Ethanol overproduction during continuous syngas fermentation due to expression of amplified ethanol biosynthesis pathway integrated using Tn7-tool and powered at the expense of eliminated acetic acid production and spore formation. Journal of Applied Microbiology. doi:10.1111/jam.12123.
Demain, A. L. (2009). Biosolutions to the energy problem. Journal of Industrial Microbiology and Biotechnology, 36(3), 319–332.
Fischer, C. R., Klein-Marcuschamer, D., & Stephanopoulos, G. (2008). Selection and optimization of microbial hosts for biofuels production. Metabolic Engieneering, 10(6), 295–304.
Köpke, M., Michalcea, C., Liew, F.-M., Tizard, J. H., Ali, M. S., Conolly, J. J., et al. (2011). 2, 3-butanediol production by acetogenic bacteria, an alternative route to chemical synthesis, using industrial waste gas. Applied and Environmental Microbiology, 77(15), 5467–5475.
Abubackar, H. N., Veiga, M. C., & Kennes, C. (2012). Biological conversion of carbon monoxide to ethanol: effect of pH, gas pressure, reducing agent and yeast extract. Bioresource Technology, 114, 518–522.
Fischer, C. R., Klein-Marcuschamer, D., & Stephanopoulos, G. (2008). Selection and optimization of microbial hosts for biofuels production. Metabolic Engineering, 10(6), 295–304.
Gaddy, J. L., Arora, D. D., Ko, C.-W., Phillips, J. R., Basu, R., Wikstrom, C. V., Clausen, E. C. (2001). Methods for increasing the production of ethanol from microbial fermentation. US Patent 7285402.
Ma, C., Wang, A., Qin, J., Li, L., Ai, X., Jiang, T., et al. (2009). Enhanced 2,3-butanediol production by Klebsiella pneumoniae SDM. Applied Microbiology and Biotechnology, 82(1), 49–57. doi:10.1007/s00253-008-1732-7.
Celińska, E., & Grayek, W. (2009). Biotechnological production of 2,3-butanediol—current state and prospects. Biotechnology Advances, 27, 715–725.
Xiu, Z. L., & Zeng, A. P. (2008). Present state and perspectives of downstream processing of biologically produced 1,3-propanediol and 2,3-butanediol. Applied Microbiology and Biotechnology, 78, 917–926.
Ji, X. J., Huang, H., & Ouyang, P. K. (2011). Microbial 2,3-butanediol production: a state-of-the-art review. Biotechnology Advances, 29, 351–364.
Oliver, J. W., Machado, I. M., Yoneda, H., & Atsumi, S. (2013). Cyanobacterial conversion of carbon dioxide to 2,3-butanediol. Proceedings of the National Academy of Sciences U S A, 110(4), 1249–1254. doi:10.1073/pnas.1213024110.
Drake, H. L. (1994). Acetogenesis. NY, London: Chapman & Hall.
Berzin, V., Kiriukhin, M., & Tyurin, M. (2012). Cre-lox66/lox71-based elimination of phosphotransacetylase or acetaldehyde dehydrogenase shifted carbon flux in acetogen rendering selective overproduction of ethanol or acetate. Applied Biochemistry and Biotechnology. doi:10.1007/s12010-012-9864-8.
Craig, N. L. (1991). Micro Review. Tn7: a target site-specific transposon. Molecular Microbiology, 5(11), 2569–2573.
Tanner, R. S., Miller, L. M., & Yang, D. (1993). Clostridium ljungdahlii sp. nov., an acetogenic species in clostridial rRNA homology group I. International Journal of Systemic Bacteriology, 43(2), 232–236.
Tyurin, M., Lynd, L. R., & Wiegel, J. (2006). Genetic delivery systems for strictly anaerobic thermophiles. In F. A. Rainey & A. Oren (Eds.), Methods in Microbiology (1st ed., Vol. 35, pp. 307–328). Amsterdam: Elsevier, Academic Press.
Berzin, V., Kiriukhin, M., & Tyurin, M. (2013). Selective n-butanol production by Clostridium sp. MTButOH1365 during continuous synthesis gas fermentation due to expression of synthetic thiolase, 3-hydroxy butyryl-CoA dehydrogenase, crotonase, butyryl-CoA dehydrogenase, butyraldehyde dehydrogenase and NAD-dependent butanol dehydrogenase. Applied Biochemistry and Biotechnology. doi:10.1007/s12010-012-0060-7.
Tyurin, MV. (1992). The method and apparatus for electric cell treatment using partial power capacitor discharge via the sample and the power tetrode. Russian Patent 2005776.
Tyurin, M., Desai, S., & Lynd, L. (2004). Electrotransformation of Clostridium thermocellum. Applied and Environmental Microbiology, 70(2), 883–890.
Tyurin, M. V., Sullivan, C. R., & Lynd, L. R. (2005). Induced oscillations play an important role in high efficiency transformation of thermophilic anaerobes. Applied and Environmental Microbiology, 71, 8069–8076.
Tyurin, M., Kiriukhin, M., & Berzin, V. (2012). Electrofusion of cells of acetogen Clostridium sp. MT351 with erm(B) or cat in the chromosome. Journal of Biotech Research, 4, 1–12.
Berzin, V., & Tyurin, M. (2012). Acetogen biocatalyst Clostridium sp. MTEtOH871 engineered with our proprietary electrotransformation technology and equipment: continuous synthesis gas fermentation for selective ethanol production. Journal of Biotech Research, 4, 54–64.
Leibig, M., Krismer, B., Kolb, M., Friede, A., Götz, F., & Bertram, R. (2008). Marker removal in staphylococci via Cre recombinase and different lox sites. Applied and Environmental Microbiology, 74(5), 1316–1323.
Shepherd, C. T., Moran, L. A. N., & Scott, M. P. (2009). Determination of transgene copy number by real-time quantitative PCR. Methods in Molecular Biology, 526, 129–134. doi:10.1007/978-1-59745-494-0_11.
Ross, S. M. (2000). Introduction to probability and statistics for engineers and scientists (2nd ed.). San Diego: Academic.
Volker, M. (2003). Energy conservation in acetogenic bacteria. Applied and Environmental Microbiology, 69(11), 6345–6353. doi:10.1128/AEM.69.11.6345-6353.2003.
Wang, A., Xu, Y., Ma, C., Gao, C., Li, L., Wang, Y., et al. (2012). Efficient 2,3-Butanediol Production from cassava powder by a crop-biomass-utilizer, Enterobacter cloacae subsp. dissolvens SDM. PLoS One, 7(7), e40442. doi:10.1371/journal.pone.0040442.
Acknowledgments
The research was supported by the funds of angel friends of MT family. Syngas Biofuels Energy, Inc. and its successors are the sole distributors of the electroporation and electrofusion equipment: www.syngasbiofuelsenergy.com.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tyurin, M., Kiriukhin, M. Synthetic 2,3-Butanediol Pathway Integrated Using Tn7-tool and Powered Via Elimination of Sporulation and Acetate Production in Acetogen Biocatalyst. Appl Biochem Biotechnol 170, 1503–1524 (2013). https://doi.org/10.1007/s12010-013-0285-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0285-0