Abstract
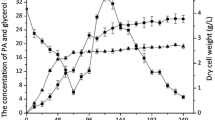
Comparative metabolomics was carried out to investigate the metabolic differences of Penicillium chrysogenum in the pilot and industrial fermentations that resulted from the scale-up. By principal component analysis, the early stages of two fermentation processes were clearly distinguished, whereas the middle and final stages were clustered together. It indicated that the different metabolisms of cells in the pilot and industrial fermentations mainly existed during the early stage. Furthermore, the levels of polyamines, polyols, glycolysis, and tricarboxylic acid cycle intermediates, which changed more dramatically during the pilot process, were all higher in the pilot than in the industrial fermentation during the early stage. This indicated that the fermentation conditions of the early stage should be the focus of process management which is aimed at increasing penicillin production. Additionally, the comparative accumulations of the precursors of penicillin (valine, cysteine, and lysine) revealed that penicillin biosynthesis in the industrial process was more affected during the middle stage of fermentation. These findings provide new insights to further regulate the industrial process and improve the production of penicillin. More generally, this study attempts to address the scarcity of studies that contrast the metabolic outcomes between commercial- and pilot-scale conditions.








Similar content being viewed by others
References
Fleming, A. (1929). On the antibacterial action of cultures of a Penicillium, with special reference to their use in the isolation of B. influenza. British Journal of Experimental Pathology, 10, 226–236.
Nielsen, J. (1997) Physiological engineering aspects of Penicillium chrysogenum. World Scientific, Singapore. 139 p.
Nasution, U., van Gulik, W. M., Ras, C., Proell, A., & Heijnen, J. J. (2008). A metabolome study of the steady-state relation between central metabolism, amino acid biosynthesis and penicillin production in Penicillium chrysogenum. Metabolic Engineering, 10, 10–23.
Gunnarsson, N., Eliasson, A., & Nielsen, J. (2004). Control of fluxes towards antibiotics and the role of primary metabolism in production of antibiotics. Advances in Biochemical Engineering/Biotechnology, 88, 137–178.
Christensen, B., Thykaer, J., & Nielsen, J. (2000). Metabolic characterization of high- and low-yielding strains of Penicillium chrysogenum. Applied Microbiology and Biotechnology, 54, 212–217.
Charles, M. (1985). Fermentation scale-up: problems and possibilities. Trends in Biotechnology, 3, 134–139.
Yang, H., & Allen, D. G. (1999). Model-based scale-up strategy for mycelial fermentation processes. Canadian Journal of Chemical Engineering, 77, 844–854.
Junker, B. H., Hesse, M., Burgess, B., Masurekar, P., Connors, N., & Seeley, A. (2004). Early phase process scale-up challenges for fungal and filamentous bacterial cultures. Applied Biochemistry and Biotechnology, 119, 241–277.
Smith, J. J., Lilly, M. D., & Fox, R. I. (1990). 1990. Biotechnology and Bioengineering, 35, 1011–1023.
Jüsten, P., Paul, G. C., Nienow, A. W., & Thomas, C. R. (1998). Dependence of Penicillium chrysogenum growth, morphology, vacuolation, and productivity in fed-batch fermentations on impeller type and agitation intensity. Biotechnology and Bioengineering, 59, 762–775.
Schmidt, F. R. (2005). Optimization and scale up of industrial fermentation processes. Applied Microbiology and Biotechnology, 68, 425–435.
Makagiansar, H. Y., Ayazi Shamlou, P., Thomas, C. R., & Lilly, M. D. (1993). The influence of mechanical forces on the morphology and penicillin production of Penicillium chrysogenum. Bioprocess and Biosystems Engineering, 9, 83–90.
Nielsen, J., & Krabben, P. (1995). Hyphal growth and fragmentation of Penicillium chrysogenum in submerged cultures. Biotechnology and Bioengineering, 46, 588–598.
Paul, G. C., & Thomas, C. R. (1996). A structured model for hyphal differentiation and penicillin production using Penicillium chrysogenum. Biotechnology and Bioengineering, 51, 558–572.
Rokem, J. S., Lantz, A. E., & Nielsen, J. (2007). Systems biology of antibiotic production by microorganisms. Natural Product Reports, 24, 1262–1287.
Cao, Y. X., Qiao, B., Lu, H., Chen, Y., & Yuan, Y. J. (2011). Comparison of the secondary metabolites in Penicillium chrysogenum between pilot and industrial penicillin G fermentations. Applied Microbiology and Biotechnology, 89, 1193–1202.
van Gulik, W. M., Antoniewicz, M. R., De Laat, W. T. A. M., Vinke, J. L., & Heijnen, J. J. (2001). Energetics of growth and penicillin production in a high-producing strain of Penicillium chrysogenum. Biotechnology and Bioengineering, 72, 185–193.
Cheng, J. S., Ding, M. Z., Tian, H. C., & Yuan, Y. J. (2009). Inoculation density-dependent responses and pathway shifts in Saccharomyces cerevisiae. Proteomics, 9, 4704–4713.
Han, P. P., & Yuan, Y. J. (2009). Lipidomic analysis reveals activation of phospholipid signaling in mechanotransduction of Taxus cuspidata cells in response to shear stress. The FASEB Journal, 23, 623–630.
Li, B. Z., Cheng, J. S., Qiao, B., & Yuan, Y. J. (2010). Genome-wide transcriptional analysis of Saccharomyces cerevisiae during industrial bioethanol fermentation. Journal of Industrial Microbiology & Biotechnology, 37, 43–55.
Fiehn, O., Kopka, J., Dormann, P., Altmann, T., Trethewey, R. N., & Willmitzer, L. (2000). Metabolite profiling for plant functional genomics. Nature Biotechnology, 18, 1157–1161.
Christensen, L. H., Mandrup, G., Nielsen, J., & Villadsen, J. (1994). A robust liquid chromatographic method for measurement of medium components during penicillin fermentation. Analytica Chimica Acta, 296, 51–62.
Ding, M. Z., Li, B. Z., Cheng, J. S., & Yuan, Y. J. (2010). Metabolome analysis of differential responses of diploid and haploid yeast to ethanol stress. OMICS., 14, 553–561.
Ding, M. Z., Cheng, J. S., Xiao, W. H., Qiao, B., & Yuan, Y. J. (2009). Comparative metabolomic analysis on industrial continuous and batch ethanol fermentation processes by GC–TOF/MS. Metabolomics, 5, 229–238.
Nasution, U., van Gulik, W. M., Kleijn, R. J., van Winden, W. A., Proell, A., & Heijnen, J. J. (2006). Measurement of intracellular metabolites of primary metabolism and adenine nucleotides in chemostat cultivated Penicillium chrysogenum. Biotechnology and Bioengineering, 94, 159–166.
Jorgensen, H., Nielsen, J., Villadsen, J., & Mollgaard, H. (1995). Metabolic flux distributions in Penicillium chrysogenum during fed-batch cultivations. Biotechnology and Bioengineering, 46, 117–131.
van Gulik, W. M., de Laat, W. T., Vinke, J. L., & Heijnen, J. J. (2000). Application of metabolic flux analysis for the identification of metabolic bottlenecks in the biosynthesis of penicillin-G. Biotechnology and Bioengineering, 68, 602–618.
Pirt, S. J., & Righelato, R. C. (1967). Effect of growth rate on the synthesis of penicillin by Penicillium chrysogenum in batch and chemostat cultures. Applied Microbiology, 15, 1284–1290.
Ding, M. Z., Tian, H. C., Cheng, J. S., & Yuan, Y. J. (2009). Inoculum size-dependent interactive regulation of metabolism and stress response of Saccharomyces cerevisiae revealed by comparative metabolomics. Journal of Biotechnology, 144, 279–286.
Ding, M. Z., Zhou, X., & Yuan, Y. J. (2010). Metabolome profiling reveals adaptive evolution of Saccharomyces cerevisiae during repeated vacuum fermentations. Metabolomics, 6, 42–55.
Henriksen, C. M., Christensen, L. H., Nielsen, J., & Villadsen, J. (1996). Growth energetics and metabolic fluxes in continuous cultures of Penicillium chrysogenum. Journal of Biotechnology, 45, 149–164.
Jennings, D. H. (1984). Polyol metabolism in fungi. Advances in Microbial Physiology, 25, 149–193.
Adler, L., Pedersen, A., & Tunblad-Johansson, I. (1982). Polyol accumulation by two filamentous fungi grown at different concentrations of NaCl. Plant Physiology, 56, 139–142.
Taber, W. A., & Tertzakian, G. (1965). Sequential primary and secondary shunt metabolism in Penicillium chrysogenum. Applied and Environmental Microbiology, 13, 590–594.
Mitsui, K., Igarashi, K., Kakegawa, T., & Hirose, S. (1984). Preferential stimulation of the in vivo synthesis of a protein by polyamines in Penicillium chrysogenum: purification and properties of the specific protein. Biochemistry, 23, 2679–2683.
Tabor, C. W., & Tabor, H. (1985). Polyamines in microorganisms. Microbiological Reviews, 49, 81–99.
Sekowska, A., Bertin, P., & Danchin, A. (1998). Characterization of polyamine synthesis pathway in Bacillus subtilis 168. Molecular Microbiology, 29, 851–858.
Flores, H. E., & Galston, A. W. (1982). Polyamines and plant stress: activation of putrescine biosynthesis by osmotic shock. Science, 217, 1259–1261.
Tabor, C. W., & Tabor, H. (1976). 1,4-Diaminobutane (putrescine), spermidine, and spermine. Annual Review of Biochemistry, 45, 285–306.
Feuerstein, B. G., Pattabiraman, N., & Marton, L. J. (1986). Spermine–DNA interactions: a theoretical study. Proceedings of the National Academy of Sciences of the United States of America, 83, 5948–5952.
Yuki, M., Grukhin, V., Lee, C. S., & Haworth, I. S. (1996). Spermine binding to GC-rich DNA: experimental and theoretical studies. Archives of Biochemistry and Biophysics, 325, 39–46.
Park, M. H., Joe, Y. A., Kang, K. R., Lee, Y. B., & Wolf, E. C. (1996). The polyamine-derived amino acid hypusine: its post-translational formation in eIF-5A and its role in cell proliferation. Amino Acids, 10, 109–121.
Acknowledgments
The authors are grateful for the financial support from the National High-Tech R&D Program (863 Program: 2012AA021204), the National Basic Research Program of China (973 Program: 2011CBA00802), and the National Natural Science Foundation of China (Key Program: 20736006, Major International Joint Research Project: 21020102040).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ding, MZ., Lu, H., Cheng, JS. et al. Comparative Metabolomic Study of Penicillium chrysogenum During Pilot and Industrial Penicillin Fermentations. Appl Biochem Biotechnol 168, 1223–1238 (2012). https://doi.org/10.1007/s12010-012-9852-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-9852-z