Abstract
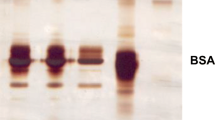
The chemical modification of developed ethyl cellulose-based membrane was carried out to make it suitable for bioseparation. The different reagents were used for the modification of membrane to couple protein A (PA) to study the purification of immunoglobulin G (IgG) from blood. The chemical modification was carried out using relatively simple and mild reaction conditions. The attenuated total reflectance Fourier transform infrared analysis of chemically modified membrane showed new peak at 1,596.06 and 1,716.49 cm−1. The scanning electron microscopy of PA-coupled membrane, which was used for IgG purification showed open pores and 950 ± 21.5 LMH (L m−2 h−1) operational flux at 0.5-bar out pressure. The flux of unmodified membrane was 1,746 ± 18.5 LMH at 0.5-bar out pressure. The equilibrium adsorption concentration (318.5 ± 5.9 μg cm−2) was obtained at 3 h. The adsorption character of PA-coupled membrane was consistent with the Langmuir adsorption model and the non-specific binding was 67.08 ± 1.3 μg cm−2. The sodium dodecyl sulfate polyacrylamide gel electrophoresis analysis showed similar purification pattern for purified IgG from human serum and commercial preparation of IgG. All the results have suggested a high potential of PA-coupled ethyl cellulose-based membrane for large-scale purification of IgG.





Similar content being viewed by others
References
Castilho, L. R., Anspach, F. B., & Deckwer, W.-D. (2002). Journal of Membrane Science, 207, 253–264.
Barroso, T., Temtem, M., Hussain, A., Aguiar-Ricardo, A., & Roque, A. C. A. (2010). Journal of Membrane Science, 348, 224–230.
Bresolin, I. T. L., Fioritti, R. R., & Bueno, S. M. A. (2011). Process Biochemistry, 46, 2277–2285.
Wongchuphan, R., Tey, B. T., Tan, W. S., Subramanian, S. K., Taip, F. S., & Ling, T. C. (2011). Process Biochemistry, 46, 101–107.
Suck, K., Walter, J., Menzel, F., Tappe, A., Kasper, C., Naumannb, C., Zeidler, R., & Scheper, T. (2006). Journal of Biotechnology, 121, 361–367.
Liu, S., Zeng, J., Tao, D., & Zhang, L. (2010). Cellulose, 17, 1159–1169.
Gemeiner, P., Polakovič, M., Mislovičová, D., & Štefuca, V. J. (1998). Chromatography B, 715, 245–271.
Boeden, H.-F., Pommerening, K., Becker, M., Rupprich, C., Holtzhauer, M., Loth, F., & Müller, R. (1991). Journal of Chromatography. A, 552, 389–414.
Hari, P. R., Paul, W., & Sharma, C. P. (2000). Journal of Biomedical Materials Research, 50, 110–113.
Chandel, A. K., Rao, L. V., Narasu, M. L., & Singh, O. V. (2008). Enzyme and Microbial Technology, 42, 199–207.
Malakian, A., Golebiowska, M., & Bellefeuille, J. (1993). American Laboratory, 25, 40P–40V.
Hou, K. C., Zaniewski, R., & Roy, S. (1991). Biotechnology and Applied Biochemistry, 13, 257–268.
Jia, L., Yang, L., Zou, H., Zhang, Y., Zhao, J., Fan, C., & Sha, L. (1999). Biomedical Chromatography, 13, 472–477.
Yang, L., & Chen, P. (2002). Journal of Membrane Science, 205, 141–153.
Charcosset, C. (1998). Journal of Chemical Technology and Biotechnology, 71, 95–110.
Castilho, L. R., Deckwer, W.-D., & Anspach, F. B. (2000). Journal of Membrane Science, 172, 269–277.
Adikane, H. V., & Thakar, D. M. (2010). Applied Biochemistry and Biotechnology, 160, 1130–1145.
Wei, X., Yunjun, L., Xuejing, S., Jingru, L., Yin, Y., Yong, L., & Yongbin, J. (2008). Journal of Membrane Science, 325, 592–598.
Shi, P., Zuo, Y., Zou, Q., Shen, J., Zhang, L., Li, Y., & Morsi, Y. S. (2009). International Journal of Pharmaceutics, 375, 67–74.
Larsson, M., Hjärtstam, J., Berndtsson, J., Stading, M., & Larsson, A. (2010). European Journal of Pharmaceutics and Biopharmaceutics, 76, 428–432.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Journal of Biological Chemistry, 193, 265–275.
Laemmli, U. K. (1970). Nature, 227, 680–685.
Ho, Y.-S., Chiu, W.-T., & Wang, C.-C. (2005). Bioresource Technology, 96, 1285–1291.
Porath, J., & Axén, R. (1976). In K. Mosbach (Ed.), Methods in enzymology, vol, 44: immobilized enzymes (pp. 19–45). New York: Academic.
Fang, F., Yu, L., Binyuan, Z., & Ke'ao, H. (2005). J Wuhan Univ Technol Mater Sci, 20, 20–23.
Park, D., Haam, S., Jang, K., Ahn, I., & Kim, W. (2005). Process Biochemistry, 40, 53–61.
Beeskow, T. C., Kroner, K. H., & Anspach, F. B. (1997). Journal of Colloid and Interface Science, 196, 278–291.
Boussif, O., Lezoualc'h, F., Zanta, M. A., Mergny, M. D., Scherman, D., Demeneix, B., & Behr, J.-P. (1995). Proceedings of the National Academy of Sciences of the United States of America, 92, 7297–7301.
Ukeda, H., Ishii, T., Sawamura, M., & Kusunose, H. (1995). Analytica Chimica Acta, 308, 261–268.
Zeng, X., & Ruckenstein, E. (1996). Journal of Membrane Science, 117, 271–278.
Sugiura, T., Imagawa, H., & Kondo, T. (2000). Journal of Chromatography B, 742, 327–334.
Randy, G. L., & Sung, W. K. (1974). Journal of Biomedical Materials Research, 8, 251–259.
Acknowledgment
We thank DBT, New Delhi, India, for giving the financial support to carry out the work (BT/PR2937/PID/06/147/2002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Adikane, H.V., Iyer, G.J. Chemical Modification of Ethyl Cellulose-Based Highly Porous Membrane for the Purification of Immunoglobulin G. Appl Biochem Biotechnol 169, 1026–1038 (2013). https://doi.org/10.1007/s12010-012-0085-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-0085-y