Abstract
Background
Osseous defects reconstructed with cryopreserved structural allografts are poorly revascularized and therefore are prone to nonunion, infection, deterioration of mechanical properties, and fracture. Whether this can be mitigated by specific interventions such as intramedullary surgical revascularization has been incompletely evaluated.
Questions/purposes
We aimed to study surgical revascularization as a means to improve bone remodeling in cryopreserved allograft. Second, we questioned whether spatial histomorphometric differences occur in cortical bone areas after intramedullary surgical revascularization. Third, biomechanical properties of the graft-recipient construct in surgically revascularized allograft were compared with those of conventional allografts.
Methods
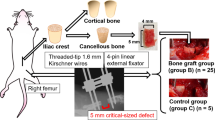
Allografts were harvested from 10 Brown Norway rats, cryopreserved, and transplanted orthotopically in a 10-mm defect in two groups of 10 Lewis rats each (major histocompatibility mismatch). In the control group, no surgical revascularization was performed, whereas in the experimental group, a saphenous arteriovenous bundle was transposed in the bone marrow cavity. Bone remodeling was measured with histomorphometry, histology, and microcomputed tomography at 16 weeks. Spatial differences were analyzed with histomorphometry. To determine biomechanical properties, load at failure and structural stiffness in bending were evaluated by the three-point bend testing. In both groups, normal values of the contralateral femur also were analyzed.
Results
Surgically revascularized allografts had increased bone remodeling (bone formation rate to bone surface ratio: 130 ± 47 µm3/µm2/year versus 44 ± 43 µm3/µm2/year, p = 0.006) and higher cortical osteocyte counts (18.6% ± 12.7% versus 3.1% ± 2.8%, p = 0.002) than nonrevascularized grafts. In nonrevascularized grafts, the bone formation rate to bone surface ratio was 35% of the contralateral normal values, whereas in surgically revascularized grafts, the bone formation rate to bone surface ratio in the grafts exceeded the contralateral values (110%). Microcomputed tomography did not show differences in bone volume between groups, however in both groups, bone volume was less in grafts compared with the contralateral femurs. Inner cortical bone formation rate to bone surface ratio was greater in surgically revascularized grafts (65 ± 30 µm3/µm2/year versus 13 ± 16 µm3/µm2/year in the control group, p = 0.012). Outer cortical bone formation rate to bone surface ratio also increased in surgically revascularized grafts (49 ± 31 µm3/µm2/year versus 19 ± 21 µm3/µm2/year, p = 0.032). No differences were found in load at failure and structural stiffness between both groups. In the control group, load at failure and structural stiffness were lower in grafts than in the contralateral femurs (p = 0.004 and p = 0.02, respectively). In the experimental group, surgically revascularized grafts also had lower load at failure and structural stiffness than the contralateral femurs (p = 0.008 and p = 0.02, respectively).
Conclusions
Surgical revascularization of large segmental allografts improved bone remodeling and viability without an adverse effect on total bone volume or bending strength and stiffness in this short-term analysis.
Clinical Relevance
Cryopreserved allografts remain largely necrotic and are associated with a high rate of complications. Surgical revascularization increases graft healing which could contribute to graft survival with time.



Similar content being viewed by others
References
Aerssens J, Boonen S, Lowet G, Dequeker J. Interspecies differences in bone composition, density, and quality: potential implications for in vivo bone research. Endocrinology. 1998;139:663–670.
Aponte-Tinao L, Farfalli GL, Ritacco LE, Ayerza MA, Muscolo DL. Intercalary femur allografts are an acceptable alternative after tumor resection. Clin Orthop Relat Res. 2012;470:728–734.
Aspenberg P, Wang JS, Jonsson K, Hagert CG. Experimental osteonecrosis of the lunate: revascularization may cause collapse. J Hand Surg Br. 1994;19:565–569.
Bullens PH, Minderhoud NM, de Waal Malefijt MC, Veth RP, Buma P, Schreuder HW. Survival of massive allografts in segmental oncological bone defect reconstructions. Int Orthop. 2009;33:757–760.
Carneiro R, Malinin T. Vascularized bone allografts: an experimental study in dogs. J Reconstr Microsurg. 1991;7:101–103.
Davy DT. Biomechanical issues in bone transplantation. Orthop Clin North Am. 1999;30:553–563.
Delloye C, Cornu O, Druez V, Barbier O. Bone allografts: what they can offer and what they cannot. J Bone Joint Surg Br. 2007;89:574–579.
Donati D, Di Bella C, Col Angeli M, Bianchi G, Mercuri M. The use of massive bone allografts in bone tumour surgery of the limb. Curr Orthop. 2005;19:393–399.
Ehrlich PJ, Lanyon LE. Mechanical strain and bone cell function: a review. Osteoporos Int. 2002;13:688–700.
Enneking WF, Campanacci DA. Retrieved human allografts: a clinicopathological study. J Bone Joint Surg Am. 2001;83:971–986.
Enneking WF, Mindell ER. Observations on massive retrieved human allografts. J Bone Joint Surg Am. 1991;73:1123–1142.
Fox EJ, Hau MA, Gebhardt MC, Hornicek FJ, Tomford WW, Mankin HJ. Long-term followup of proximal femoral allografts. Clin Orthop Relat Res. 2002;397:106–113.
Frost HM. Dynamics of bone remodeling. In: Frost HM, ed. Bone Biodynamics. Boston, MA: Little, Brown and Company; 1964:315–333.
Gouin F, Passuti N, Verriele V, Delecrin J, Bainvel JV. Histological features of large bone allografts. J Bone Joint Surg Br. 1996;78:38–41.
Hori Y, Tamai S, Okuda H, Sakamoto H, Takita T, Masuhara K. Blood vessel transplantation to bone. J Hand Surg Am. 1979;4:23–33.
Kasashima T, Minami A, Kato H, Kaneda K. Experimental study of vascularized bone grafts: hypertrophy of the grafted bone. J Reconstr Microsurg. 2000;16:121–128.
Kumta S, Yip K, Roy N, Lee SK, Leung PC. Revascularisation of bone allografts following vascular bundle implantation: an experimental study in rats. Arch Orthop Trauma Surg. 1996;115:206–210.
Mankin HJ, Doppelt S, Tomford W. Clinical experience with allograft implantation: the first ten years. Clin Orthop Relat Res. 1983;174:69–86.
Mankin HJ, Hornicek FJ, Raskin KA. Infection in massive bone allografts. Clin Orthop Relat Res. 2005;432:210–216.
McBroom RJ, Cheal EJ, Hayes WC. Strength reductions from metastatic cortical defects in long bones. J Orthop Res. 1988;6:369–378.
Mills LA, Simpson AH. In vivo models of bone repair. J Bone Joint Surg Br. 2012;94:865–874.
Pelker RR, McKay J Jr, Troiano N, Panjabi MM, Friedlaender GE. Allograft incorporation: a biomechanical evaluation in a rat model. J Orthop Res. 1989;7:585–589.
Stevenson S, Emery SE, Goldberg VM. Factors affecting bone graft incorporation. Clin Orthop Relat Res. 1996; 324:66–74.
Stevenson S, Horowitz M. The response to bone allografts. J Bone Joint Surg Am. 1992;74:939–950.
Tamai S. Experimental vascularized bone transplantations. Microsurgery. 1995;16:179–185.
Wheeler DL, Enneking WF. Allograft bone decreases in strength in vivo over time. Clin Orthop Relat Res. 2005;435:36–42.
Willems WF, Kremer T, Friedrich P, Bishop AT. Surgical revascularization induces angiogenesis in orthotopic bone allograft. Clin Orthop Relat Res. 2012;470:2496–2502.
Author information
Authors and Affiliations
Corresponding author
Additional information
This research was funded by the Musculoskeletal Transplant Foundation. (WFW, ATB)
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research editors and board members are on file with the publication and can be viewed on request.
Each author certifies that his or her institution approved the animal protocol for this investigation and that all investigations were conducted in conformity with ethical principles of research.
Clinical Orthopaedics and Related Research neither advocates nor endorses the use of any treatment, drug, or device. Readers are encouraged to always seek additional information, including FDA-approval status, of any drug or device prior to clinical use.
About this article
Cite this article
Willems, W.F., Kremer, T., Friedrich, P. et al. Surgical Revascularization in Structural Orthotopic Bone Allograft Increases Bone Remodeling. Clin Orthop Relat Res 472, 2870–2877 (2014). https://doi.org/10.1007/s11999-014-3658-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-014-3658-y