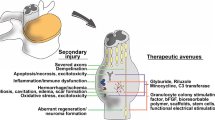
Abstract
Purpose of Review
This review provides guidance for acute spinal cord injury (SCI) management through an analytical assessment of the most recent evidence on therapies available for treating SCI, including newer therapies under investigation. We present an approach to the SCI patient starting at presentation to acute rehabilitation and prognostication, with additional emphasis on the pediatric population when evidence is available.
Recent Findings
Further studies since the Surgical Timing in Acute Spinal Cord Injury Study (STASCIS) demonstrated a potential functional outcome benefit with ultra-early surgical intervention ≤ 8 h post-SCI. Subsequent analysis of the National Acute Spinal Cord Injury Study (NASCIS) II and NASCIS III trials have demonstrated potentially serious complications from intravenous methylprednisolone with limited benefit. Newer therapies actively being studied have demonstrated limited or no benefit in preclinical and clinical trials with insufficient evidence to support use in acute SCI treatment.
Summary
Care for SCI patients requires a multi-disciplinary team. Immediate evaluation and management are focused on preventing additional injury and restoring perfusion to the affected cord. Rapid assessment and intervention involve focused neurological examination, targeted imaging, and surgical intervention when indicated. There are currently no evidence-based recommendations for pathomechanistically targeted therapies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Annually, there are approximately 54 new spinal cord injuries (SCI) per one million people in the USA, nearly 17,900 new cases per year based on 2020 estimates. Roughly 296,000 people live with SCI in the USA [1]. The morbidity associated with SCI has a substantial impact on the healthcare system. Lifetime care of traumatic SCI (tSCI) patients ranges from $1,600,000 to $4,800,000 [1]. Given the staggering impact of SCI on society, proper care of these patients is paramount. Immediate evaluation and management are focused on preventing further injury to the spinal cord, which can be caused by spine instability, spinal cord compression, systemic hypotension, vascular compromise, and elevated intrathecal pressure.
Initial Evaluation
Examination
Early recognition is imperative to avoid additional damage, especially during transport. Patients are at risk for airway compromise, respiratory failure, and circulatory failure. Initial evaluation begins with assessing the airway, breathing, and circulation (ABCs) [2••]. Other components of the examination include the patient’s Glasgow Coma Scale, pupil size and reactivity, extremity movement, and SCI level based on motor and sensory deficits. Additional signs should be evaluated, including loss of rectal tone, hyperreflexia/areflexia, and sacral exam (anal reflex and/or bulbocavernosus reflex) [2••, 3••]. Typically, reflex arcs at or below the level of the injury are impaired; however, more distal reflexes such as the anal and bulbocavernosus reflex may remain intact [4].
Imaging
Patients presenting with suspected acute tSCI require imaging to evaluate the extent of osseus and spinal cord parenchymal injury, to evaluate for ongoing mechanisms of injury or compression, and to determine spinal stability. Extent of injury provides prognosis while ongoing compression or instability informs treatment. Initial spinal computed tomography (CT) scan should be rapidly obtained to evaluate for fracture. In patients with neurologic deficit, emergent magnetic resonance imaging (MRI) is indicated to evaluate for disc herniation, epidural hematoma, spinal cord contusion or injury, and ligamentous injury. CT angiogram can evaluate for vertebral artery injury in the cervical spine or aorto-iliac injury in the thoracolumbar spine. In the subacute setting, dynamic flexion and extension imaging can evaluate for instability. Historically, dynamic imaging consisted of radiographs; however, flexion extension CT and MRI are now often utilized [5].
Validated clinical radiographic scoring systems may predict the need for operative fixation and arthrodesis, such as the modern Subaxial Cervical Spine Injury Classification (SLIC), Thoracolumbar Injury Classification and Severity Scale (TLICS), and AOSpine, including the pioneering White and Panjabi score. Historically, fracture types were used to predict stability. The White and Panjabi model of the spine based on three structural columns (anterior vertebral body, posterior vertebral body, and facet complex) posited when two or three columns were injured, the fracture was unstable [6, 7]. The modern classification systems use CT, MRI, and clinical data to better predict fracture stability and need for surgery. The SLIC system gives points for fracture morphology, integrity of disc-ligamentous complex, and patient neurologic status on a scale from 0 to 10; non-operative treatment is recommended for scores < 4, and operative treatment is recommended for scores ≥ 5 [8]. Since inter-rater reliability can be variable, new classifications systems continue to be published.
Non-traumatic SCI presenting acutely may necessitate a similar work-up; however, early MRI may be indicated to detect spinal tumor, epidural abscess, spontaneous spinal cord hemorrhage, spine syrinx, acute demyelination, or other non-traumatic etiologies. Conventional angiogram or CT angiogram may be indicated to evaluate for arteriovenous malformation (AVM) or arteriovenous fistula. A minor trauma with neurologic deficit out of proportion to the injury may suggest one of these etiologies but can be associated with Chiari malformation as well. Pediatric patients are also susceptible to spinal cord injury without radiologic abnormality (SCIWORA), which can be seen in up to a quarter of pediatric patients presenting with SCI; therefore, careful evaluation is critical [9]. Notably, the radiological modality classically refers to CT imaging.
Etiology
There are several distinct causes of SCI. Most share clinical characteristics; however, a thorough evaluation and better understanding of etiology are required to guide possible further therapies, prevent complications, and define prognosis for recovery.
Trauma
TSCI represents a significant cause of neurological morbidity in the USA [1, 10]. The most common cause of tSCI is motor vehicle accidents, accounting for more than one third of adult cases, followed by falls, the most common cause in elderly patients. Violence and sports-related injuries are also common, especially in children [1, 10, 11]. Most injuries related to trauma affect the cervical cord. The predominant mechanism of injury is direct impact due to mechanical forces associated with cord compression, fracture/displacement of the vertebrae, and/or cord laceration/transection. Secondary mechanisms of injury include hypoxia, hypoperfusion, inflammation, swelling, microhemorrhages, and thrombosis among others [12–15].
Vascular
Although rare, vascular causes of SCI include ischemic and hemorrhagic infarctions. The most common cause of ischemic injury is related to surgical procedures involving the thoracic or abdominal aorta [16]. Risk factors include peri-procedural systemic hypotension, aortic cross-clamping, and increased spinal canal pressure [17]. Other surgical procedures have also been associated with spinal cord infarction.
Other causes include stenosis or occlusion of the arteries supplying the spinal cord (anterior spinal artery, artery of Adamkiewicz (thoracic spine), and posterior spinal arteries). Aortic dissection can also cause infarction, especially in adults. Fibrocartilaginous embolism (FCE) is a rare cause of spinal cord infarction. It is thought that FCE results from embolized fibrocartilaginous material from intervertebral discs into the spinal vascular system after minor head/neck injury causing occlusion and ischemia [18].
Vascular malformations can also cause SCI. The most common types include dural arteriovenous fistula and intramedullary spinal AVM. Injury is typically secondary to the mass effect of the malformation or by ischemia/hemorrhage into the cord. The clinical presentation could be progressive rather than sudden or acute.
Spinal epidural hematoma is typically seen in tSCI but has also been documented in patients with coagulopathy or those receiving anti-thrombotic therapy [12, 19]. The main clinical features include acute severe back pain followed by weakness/paralysis.
Compression
Compressive SCI can be seen secondary to benign and malignant tumors. Injury commonly results from external compression, caused by metastases to the extradural space or intramedullary growth. Clinical presentation is typically progressive weakness below the level of the lesion, associated with sensory symptoms and/or bladder dysfunction, and aching back pain (which may occur prior to neurological symptoms). Compression can occur in the thoracic (60%), lumbar (25%), and cervical spine (15%) [12].
Inflammatory/Infectious
A bacterial infection, such as epidural abscess, is rare but constitutes a neurosurgical emergency. Myelitis occurs with bacterial meningitis, especially streptococcal meningitis. A virus may selectively involve anterior horn cells, such as poliovirus, enterovirus (E71, D68), and West Nile virus. Recent outbreaks of acute flaccid myelitis (AFM) causing a polio-like presentation occur with enterovirus D68. Polio causes asymmetric lower extremity weakness, whereas AFM usually involves the upper extremities.
Acute transverse myelitis/myelopathy (ATM) is an immune-mediated disorder. Inflammatory conditions, such as neuromyeltis optica (aquaporin-4) or myelin oligodendrocyte antibody associated disease (MOG), have myelitis and optic neuritis [20, 21].
Management
Immediate Management and Stabilization
Anatomical Reduction and Surgical Management
External immobilization is indicated for initial management of a suspected spine injury in the pre-hospital setting. After diagnosis of spine instability, rigid external cervical orthosis and spinal precautions (limiting movement) are used to prevent further injury. For infants with large heads, a backboard with occipital recess or thoracic padding is needed to prevent neck flexion while supine [5]. Patients with a fracture that is causing spinal cord compression from spinal canal narrowing may benefit from acute (< 24 h) or hyper-acute (< 12 h) reduction or surgical decompression as recent studies have demonstrated potentially improved functional outcomes [22••, 23].
In 2012, the multicenter, international, prospective cohort Surgical Timing in Acute Spinal Cord Injury Study (STASCIS) found surgical decompression within 24 h was safe and associated with improved American Spinal Injury Association Impairment Scale (AIS), a standardized tool for predicting ambulation after SCI, of at least 2 grades at 6 months after injury [24, 25]. Subsequent studies suggest that early surgery within 8 h may provide additional benefit [26, 27, 28••].
In the trial, surgical decompression was performed either through closed reduction with traction or open surgical reduction. Closed reduction is performed by cranial fixation with controlled distraction to re-align the spine, decompressing the spinal cord, and preventing continued injury. Pre-reduction MRI can evaluate for disc herniation that may require operative treatment. If a misaligned spine is not able to be reduced with traction, then operative reduction is indicated [25]. Results from the Spinal Cord Injury–Prospective Observational European Multicenter (SCI-POEM) study are expected to be published in late 2022 and will assess outcomes with hyper-acute surgical intervention (< 12 h) [29].
Cervical traction begins by application of a halo or tongs. Halo is preferred for children because it allows multiple skull pins to be applied at a lower pressure per pin, as opposed to the two-pin fixation of tongs. In addition, halo allows for fixation to a vest for long-term external fixation, if needed. For jumped or perched facets, a patient is placed in slight flexion with distraction. Increased weight is applied with serial radiographs until the distracted facet is realigned or the adjacent disc spaces appear over-distracted. Care must be used to not over-distract the spine, which may cause further injury. A disc could herniate during traction, causing acute worsening of injury. Frequent neurologic examinations during traction are critical. Once the spine is reduced, the patient may remain in traction or placed in a halo vest until surgery. Most dislocations occur because of significant disc or ligamentous disruption, which will not heal with time. Significant disc or ligamentous injury requires internal fixation and arthrodesis to achieve spine stability.
Most patients with spinal cord injury have spine fracture or disruption of disc or ligaments. Spine fracture without disc or ligament injury may heal with external immobilization for 6 to 12 weeks. External fixation devices for cervical fractures include halo vest and rigid external cervical orthosis. For fractures that span the cervical thoracic junction, a thoraco-lumbar sacral orthosis (TLSO) or cervical thoracic orthosis (CTO) is indicated.
Internal fixation may be indicated for unstable fracture or fractures requiring operative decompression. Internal fixation provides stability until solid arthrodesis between unstable vertebrae. Surgical treatment may be anterior, posterior, or both, depending on the extent of the injury, need for anterior decompression, and ability to reduce the fracture dislocation. Internal fixation may be augmented by external orthosis until the bone is healed. Guidelines exist for surgical indication to fuse traumatic injuries; however, the extent of fixation and arthrodesis is typically patient and surgeon dependent with goal to provide adequate fixation to prevent dislocation during the bone healing process [30, 31]. Bone healing is induced by compression and immobilization. Fracture segments that move or are not held in compression during the healing process may form a fibrous union called pseudoarthrosis that may not be stable long-term. Surgical options for rigid fixation include anterior vertebral body screws held to a buttress plate, anterior interbody cage to provide structural stability, and posterior pedicle screw, pars screw, or lateral mass screw fixation between vertebra connected by posterior rod construct.
Cervical vertebral fractures predict potential vertebral artery injury [32]. CT angiogram is often used to detect dissection or intraluminal thrombus. Treatment is directed towards prevention of progression of intraluminal dissection and prevention of thromboembolism, often using a single antiplatelet agent. A technique to increase perfusion of the spinal cord is to place a lumbar intrathecal drain to lower intrathecal pressure to augment perfusion of the spinal cord. This technique was pioneered by vascular surgeons when treating aortic aneurysms that could block spinal radicular arteries, decreasing perfusion to the spinal cord. Preclinical studies in pigs and clinical human studies show promising results that this technique may decrease the extent of injury to the spinal cord; however, the technique is still experimental and not standard of care at this time [33, 34]. As with post-traumatic brain injury monitoring, intraspinal pressure monitoring and spinal cord autoregulation are currently being studied in SCI and targeted perfusion therapy may be on the horizon [35–38].
Spinal Shock
Acutely after SCI, a transient phenomenon referred to as spinal shock can occur. It is characterized by sudden loss of spinal reflexes and muscle tone below the level of injury [4]. The underlying pathophysiological mechanism in spinal shock is poorly understood. The onset is immediately after or hours after injury and can be varied in presentation depending on the acuity of injury [4]. Spinal shock is considered to be multi-phasic with variable presentation and resolution [39]. The presentation also depends on the level of injury and severity (complete vs incomplete). While the sacral reflexes may be intact with high cervical lesions, it is important to not confuse the presentation with sacral sparing as over time these reflex arcs may be impacted [40]. Spinal shock is initially characterized by areflexia/hyporeflexia within the first 24 h. Within 1 to 3 days after the injury, initial return of reflexes can be seen [39]. Further return of reflexes is typically a gradual process and often begins with delayed plantar response and can be followed by the cutaneous reflexes (bulbocavernosus, anal, and cremaster) [40]. Over the next few weeks, deep tendon reflexes may return and can follow the sequence of ankle, Babinski sign (replacing delayed plantar reflex), and patellar [39, 40]. The final phase of recovery includes return of the bladder reflex typically with the emergence of spasticity/hyperreflexia in severe cases [39]. The pattern of reflex recovery appears to be cutaneous polysynaptic reflexes followed by monosynaptic reflexes [40].
ICU Management
Respiratory Management
Failure to maintain a patent airway and breathe effectively is common following SCI. The diaphragm is controlled by C3–C5, and intercostal muscles may also be affected. Patients may have a weak cough and difficulty controlling secretions. Many also have concomitant brain injuries, which can impair the ability to protect the airway with loss of central control of breathing. Additional pulmonary injuries including pneumothorax and direct laryngeal, tracheal, or vocal cord injuries may be present. Trauma is a known trigger for the development of acute respiratory distress syndrome (ARDS) [41].
Adults and children with high cervical lesions at or above C5 are likely to require immediate intubation due to respiratory arrest and should be considered for early intubation [2••, 42]. Unlike adults, children are more likely to experience progression of symptoms over the first few days due to the acute inflammatory response and development of edema; therefore, early intubation is preferred [2••]. If patients are not immediately intubated, close monitoring of their respiratory status is needed with both pulse oximetry and end tidal CO2 monitoring. Noninvasive ventilation is often not optimal due to the inability to protect the airway and control secretions.
Best practice for securing the airway has not been well studied in pediatric patients. In adults, it has been suggested that awake fiberoptic intubation may be ideal if intubation is not emergent [43]. This is not an option for most children too young to cooperate; sedation and analgesia are required to ensure safety. Selection of medications for intubation should be carefully considered given the risk of neurogenic shock. Avoidance of medications that may exacerbate vasoplegia and hypotension is important. Adrenal insufficiency has been reported in this population and must be considered when intubating [44]. Manipulation of the airway may result in vagal response leading to bradycardia, which may be exacerbated by hypoxia [45, 46]. Atropine may be required. It is important to ensure adequate intravascular volume. It is suggested to have intravenous fluids with a push/pull set up or rapid infuser as well as vasoactive infusions at the bedside for hypotension. It is advisable to have the most experienced provider in airway management attempt intubation first. Care must be taken to avoid movement of the cervical spine with inline manual stabilization by a second provider to ensure that the primary provider can focus solely on management of the airway. A head tilt maneuver should not be used. Video laryngoscopy should be considered to minimize movement; however, it should also be recognized that in some settings, this has prolonged time to intubation [2••, 6, 47]. Risks and benefits must be weighed based on the patient’s immediate clinical situation and availability of equipment. Many patients will require long-term mechanical ventilation [48]. Optimal timing of tracheostomy is not known but may be necessary for some patients [49]. Early tracheostomy may have advantages of minimizing continuous sedative and analgesic infusions and may facilitates earlier mobilization and rehabilitation. Long-term, diaphragmatic pacing may be an option to minimize or eliminate the need for ventilation for select patients [50, 51].
Hemodynamic Management
Neurogenic shock, a distinct phenomenon from spinal shock, is common following SCI and is characterized by profound vasoplegia and hypotension and often bradycardia. Adult guidelines for blood pressure (BP) management recommend maintaining goal MAPs > 85–90 for 1 week following injury, which is associated with improved neurological outcomes [52–54]. There are no specific pediatric recommendations for BP control, though it is reasonable to target MAPs equivalent to adult goals for age. In older children, targeting MAPs 85–90 is likely appropriate. Recommendations suggest ensuring BP is above the 5th percentile for age; however, targeting higher blood pressures may help to avoid intermittent measurements below goal [2••]. Cardiac arrhythmias, cardiac arrest, and EKG abnormalities have been reported and close monitoring is imperative [55].
No agent has single proven to be superior in targeting BP. Agents with both alpha- and beta-adrenergic effects such as norepinephrine may be ideal to help with both peripheral vasoconstriction and bradycardia. One study in adult patients showed that norepinephrine was comparable to phenylephrine to maintain blood pressures but norepinephrine showed better spinal cord perfusion and oxygenation (Table 1) [2, 9, 45, 47, 89, 90].
Future directions may be individualized BP goals based on optimal spinal perfusion pressures using lumbar catheters and pressure reactivity index (a surrogate measure of cerebrovascular autoregulation) [56]. Ongoing studies are evaluating optimization of spinal cord perfusion and oxygenation using infrared spectroscopy [57].
Pain
Managing pain can be very challenging in SCI. It is best to start with medications that do not impair respiratory drive such as acetaminophen and nonsteroidal anti-inflammatory medications, if the general clinical condition allows. For musculoskeletal pain, narcotics can be considered; however, caution must be used in patients without a definitive airway as they can cause respiratory depression and hypoventilation, which may be more profound if weakness is present. Medications targeting neuropathic pain should be used if there are characteristic features including burning, electric-like sensations, and allodynia [58]. Gabapentinoids, selective serotonin reuptake inhibitors, tricyclic antidepressants, and lidocaine patches can be considered [59]. Side effect profile must be considered when initiating these medications, including potential cardiac and behavioral complications. Partnering with a multi-disciplinary team experienced in pain management in this population is important.
Autonomic Dysreflexia
Autonomic dysreflexia (AD) is a life-threatening condition that can occur in individuals with SCI at the T6 level or above [60•]. With parasympathetic input cut-off, sympathetic activation is unopposed resulting in elevated systolic blood pressure with baroreflex-mediated bradycardia [61]. Patients present with sudden onset, severe headache with flushing and sweating above the level of injury and pale, cool skin with piloerection below the level of the lesion [62]. AD can lead to seizures, stroke, and intracerebral hemorrhage [63]. The most common cause is an overdistended bladder. A popular pneumonic to remember triggers is “6 Bs”: bladder (urinary tract infection, urinary retention, nephrolithiasis, blocked catheters), bowel (constipation, impaction), back passage (hemorrhoids and fissures), boils (skin damage), bones (fractures), and babies (pregnancy, sexual intercourse, breast feeding) [64]. The first step in managing AD is to eliminate noxious stimuli, focusing on known triggers. The patient should be positioned upright to promote an orthostatic drop in BP and any constrictors, such as tight clothing and dressing should be loosened [65]. If the patient remains hypertensive despite these measures, treat with fast-acting anti-hypertensives (Table 1). If the episode is lasting greater than 30 min, the patient should be moved to the ICU for aggressive management [66].
Bowel and Bladder
Bowel Management Depending on the level of injury, individuals will exhibit either upper or lower motor neuron bowel patterns. Upper motor neuron (UMN) injuries result in hyperreflexic bowels and spastic anal sphincters, leading to constipation and stool retention. Lower motor neuron (LMN) injuries result in areflexic bowels with an atonic external anal sphincter, leading to a mix of constipation and incontinence. Bowels do not always declare themselves immediately if the injury is in the low thoracic or lumbar regions. A bowel regimen should be started for regular daily timed bowel movements. An UMN program can include digital stimulation to trigger the retrocolic reflux to induce colonic emptying along with oral and rectal medications. Manual evacuation may be necessary for LMN programs [67].
Bladder Management Long-term bladder management varies depending on the level of injury. Individuals start in the ICU with a foley catheter to prevent excessive pressure in the bladder and kidneys and to allow for close monitoring of their volume status. Once an individual is stable, the patient should be transitioned to an intermittent catheterization program to protect from urinary tract infections and to facilitate normal stretch and contraction of the bladder [68].
Immobility
Venous Thromboembolism (VTE) Prevention As paralysis leads to significant venous stasis, the risk of VTE is elevated after SCI and further elevated with comorbid polytrauma with major fractures, traumatic brain injury, and the use of central venous catheters [69, 70]. The risk of VTE in pre-pubescent children is very low; thus, consensus is to treat them with mechanical prophylaxis. Chemoprophylaxis is used for children post-puberty and adults for 8 weeks, a time which is sufficient for the body to adapt to its new patterns [71].
Pressure Wounds Pressure wounds are common in SCI due to immobility, lack of sensation, and atrophy over bony prominences. The best prevention techniques are frequent repositioning and pressure relief measures. The standard of care is repositioning a supine individual every 2 h. More frequent repositioning or offloading is recommended when seated, at least every 30 min for a minimum of 30 s [72].
Therapies Studied in Clinical Trials (Summarized in Table 1)
Steroids
Intravenous methylprednisolone (MPS) for neuroprotection and reduction of secondary injury remains controversial in tSCI. In prior studies, MPS initiated within hours of injury had been associated with short term motor score improvement; however, long-term benefits were not demonstrated after 6 or more months [73]. Subsequent structured analysis from NASCIS I and NASCIS II studies failed to demonstrate clinically significant improvement. Three randomized trials and one prospective observational study failed to show effect on motor function recovery at 6 weeks, 6 months, or 12 months [74–76].
Safety studies remain controversial. A Cochrane review of NASCIS II showed a potential increase in the risk for wound infection and gastrointestinal hemorrhage [76]. Subsequent analysis associates the use of high-dose steroids with harmful side effects including death [5, 12, 75]. NASCIS III compared 24-h versus 48-h MPS infusion trended towards a higher incidence of severe pneumonia in patients receiving 48-h infusion and a potential increase in severe sepsis, although this was not statistically significant [74, 77]. The impact on mortality remains unclear; however, increased risk for major complications in the MPS treatment group was noted [78]. Although steroids are not recommended in tSCI, they are used first-line for inflammatory etiologies.
GM1 Ganglioside
Gangliosides (GM-1) have been studied as a therapeutic option for SCI. Early animal studies demonstrated a positive response to therapy, with GM-1 therapy helping to enhance mitochondrial activity thought to protect against neuronal injury and degeneration. GM-1 is thought to be important for regrowth and regeneration of axons, which may help to repair and replace damaged tissue following SCI and may help restore function. An early 1990 study demonstrated improvement in motor score from baseline to 1-year follow-up [79]. However, a larger follow-up study failed to report a difference in proportion of patients achieving neurologic improvement [80]. Research for GM-1 therapy in neuroinflammatory disorders is still ongoing.
Riluzole
Recent studies suggest riluzole, a glutamatergic modulator, may be neuroprotective through sodium channel blockade and glutamate uptake modulation [30, 79, 81]. A systematic review identified three clinical studies evaluating potential for use in humans. Major findings included significant motor improvement with treatment but with associated risks, including elevated liver enzymes and bilirubin levels [82]. Unfortunately, a recent study terminated early due to recruitment issues and slow patient enrollment rates [54] (ClinicalTrials.gov: NCT01597518); however, analysis of the data collected is now underway.
Granulocyte Colony-stimulating Factor
Experimental animal studies hypothesize that granulocyte colony-stimulating factor (G-CSF) suppresses neuronal apoptosis [83]. Phase I/IIa studies compared two different doses to MPS. Intravenous administration minimizes risk of elevated white blood cell counts and potential splenic rupture when compared to subcutaneous administration. Patients receiving 10 mcg/kg/day doses observed higher American Spinal Injury Association Impairment Scale (AIS) motor scores; however, this finding was not statistically significant [82]. A recent phase III clinical trial was conducted to evaluate the effect of intravenous G-CSF 400 mcg/m2/day given daily for 5 days. A change in AIS motor score was not observed at 3, 6, or 12 months [82].
Minocycline
Preclinical animal studies of minocycline suggest neuroprotective effects. In murine models of SCI, minocycline is thought to be anti-inflammatory through reduction of caspase-3 activation, in turn reducing neuronal apoptosis and improving recovery early after SCI [84]. In human studies with SCI, minocycline administration at various timepoints within the first 24 h of injury did not affect functional recovery. A phase II clinical trial failed to show improvement in motor recovery after 1 year [82].
Prognostication
Early neurological examination is of utmost importance for prognostication with regard to ambulation, bowel/bladder function, and other self-care activities. The AIS classifies the preservation of motor function and sensation, with grade A being the most severe and D being the mildest (Fig. 1) [24, 85]. Preservation of sacral sensation indicates that an individual’s AIS grade may improve by one level. Individuals with lower AIS grades and those under 50 years old have significantly better chances of motor function restoration. MRI of the spinal cord can provide highly valuable information. The length of intramedullary hemorrhage, canal diameter at maximal spinal cord compression, and spinal cord swelling often correlate with the severity of impairment [86]. Another ancillary testing modality that may be useful includes neurophysiological studies such as somatosensory-evoked potentials (SSEPs) and transcranial magnetic stimulation (TMS). SSEPs assess the integrity of posterior column function, whereas TMS directly tests the motor pathways. The decision to use one or another depends on the clinical spinal syndrome.
American Spinal Injury Association (ASIA) Form [84].
Early Rehabilitation Rehabilitation plays a significant role in recovery after SCI, starting immediately after acute stabilization. Early upright mobilization should be initiated gradually using a tilt table. Orthostatic hypotension can be managed with abdominal binders, compression stockings, and premedication with drugs such as midodrine [87]. Respiratory therapists can begin to slowly minimize vent settings, while speech therapists work with individuals on breath control techniques. Physical and occupational therapists begin passive and active range of motion exercises and when appropriate strengthening exercises. Orthotic devices should be implemented immediately to allow for neutral positioning of limbs to prevent contracture formation. Although efficacy was demonstrated in a small case series of three individuals, epidural electrical stimulation in conjunction with aggressive targeted therapy was able to restore a trunk and leg motor function after complete sensorimotor paralysis in a select group of patients [88••]. Novel and innovative therapies such as these are on the horizon and provide hope for the future of SCI treatment.
Conclusion
Although rare, SCI is a potentially devastating neurological emergency requiring rapid identification, assessment, and management. Given the potential of severe functional deficits or even death without proper care, management of SCI remains a subject of utmost importance in neurocritical care. Given the paucity of high-quality interventional studies in SCI, much of the management of SCI is driven by experience and expert consensus. Further research in early interventions, pathomechanistically targeted therapeutics, and acute rehabilitation is needed to facilitate development of robust evidence-based recommendations and guidelines.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Bellon K, Kolakowsky-Hayner SA, Chen D, McDowell S, Bitterman B, Klaas SJ. Evidence-based practice in primary prevention of spinal cord injury. Top Spinal Cord Inj Rehabil. 2013;19:25–30. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23678282
•• Chen JW, Meurer WJ, Dangayach NS, LaRovere KL, Tran DS. Emergency neurological life support: traumatic spine injury. Neurocrit Care. 2020;32:Suppl 11. Available from: https://doi.org/10.1007/s12028-019-00810-8. Current Emergency Neurological Life Support tSCI recommendations by the Neurocritical Care Society.
•• Shank CD, Walters BC, Hadley MN. Current topics in the management of acute traumatic spinal cord injury. Neurocrit Care. 2019;30:261–71. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29651626. In-depth overview of current topics in acute tSCI management.
Atkinson PP, Atkinson JL. Spinal shock. Mayo Clin Proc. 1996;71:384–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/8637263
Rozzelle CJ, Aarabi B, Dhall SS, Gelb DE, Hurlbert RJ, Ryken TC, et al. Management of pediatric cervical spine and spinal cord injuries. Neurosurgery. 2013;72 Suppl 2:205–26. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23417192
Panjabi MM. Clinical spinal instability and low back pain. J Electromyogr Kinesiol. 2003;13:371–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12832167
White A, Panjabi MM. Physical properties and functional biomechanics of the spine. Clin Biomech Spine. 2nd ed. Philadelphia: Lippincott Williams & Wilkens. 1990;22.
Patel AA, Dailey A, Brodke DS, Daubs M, Anderson PA, Hurlbert RJ, et al. Subaxial cervical spine trauma classification: the Subaxial Injury Classification system and case examples. Neurosurg Focus. 2008;25:E8. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18980482
Goldsmith TL, Barker DE, Strodel WE. Prolonged labetalol infusion for management of severe hypertension and tachycardia in a critically ill trauma patient. DICP. 1990;24:235–8. Available from: https://doi.org/10.1177/106002809002400303
Devivo MJ. Epidemiology of traumatic spinal cord injury: trends and future implications. Spinal Cord. 2012;50:365–72. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22270188
Jones TM, Anderson PA, Noonan KJ. Pediatric cervical spine trauma. J Am Acad Orthop Surg. 2011;19:600–11. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21980025
Ropper AE, Ropper AH. Acute spinal cord compression. N Engl J Med. 2017;376:1358–69. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28379788
Sekhon LH, Fehlings MG. Epidemiology, demographics, and pathophysiology of acute spinal cord injury. Spine (Phila Pa 1976). 2001;26:S2–12. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11805601
Rabinstein AA. Traumatic spinal cord injury. Continuum (Minneap Minn). 2018;24:551–66. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29613899
Stein DM, Sheth KN. Management of acute spinal cord injury. Continuum (Minneap Minn). 2015;21:159–87. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25651224
Cheshire WP, Santos CC, Massey EW, Howard JF. Spinal cord infarction: etiology and outcome. Neurology. 1996;47:321–30. Available from: http://www.ncbi.nlm.nih.gov/pubmed/8757000
Maniar HS, Sundt TM, Prasad SM, Chu CM, Camillo CJ, Moon MR, et al. Delayed paraplegia after thoracic and thoracoabdominal aneurysm repair: a continuing risk. Ann Thorac Surg. 2003;75:113–9; discussions 119–20. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12537202
AbdelRazek MA, Mowla A, Farooq S, Silvestri N, Sawyer R, Wolfe G. Fibrocartilaginous embolism: a comprehensive review of an under-studied cause of spinal cord infarction and proposed diagnostic criteria. J Spinal Cord Med. 2016;39:146–54. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26833287
Thiele RH, Hage ZA, Surdell DL, Ondra SL, Batjer HH, Bendok BR. Spontaneous spinal epidural hematoma of unknown etiology: case report and literature review. Neurocrit Care. 2008;9:242–6. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18373224
Bravar G, Luchesa Smith A, Siddiqui A, Lim M. Acute myelopathy in childhood. Children. 2021;8:1055. Available from: https://www.mdpi.com/2227-9067/8/11/1055
Absoud M, Greenberg BM, Lim M, Lotze T, Thomas T, Deiva K. Pediatric transverse myelitis. Neurology. 2016;87:S46–52. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27572861
•• Hsieh YL, Tay J, Hsu SH, Chen WT, Fang YD, Liew CQ, et al. Early versus late surgical decompression for traumatic spinal cord injury on neurological recovery: a systematic review and meta-analysis. J Neurotrauma. 2021;38:2927–36. Available from: http://www.ncbi.nlm.nih.gov/pubmed/34314253. Systematic review looking at early versus late decompression after tSCI.
Ter Wengel PV, Martin E, De Witt Hamer PC, Feller RE, van Oortmerssen JAE, van der Gaag NA, et al. Impact of Early (<24 h) Surgical decompression on neurological recovery in thoracic spinal cord injury: a meta-analysis. J Neurotrauma. 2019;36:2609–17. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30816058
Goodwin-Wilson C, Watkins M, Gardner-Elahi C. Developing evidence-based process maps for spinal cord injury rehabilitation. Spinal Cord. 2010;48:122–7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19687799
Fehlings MG, Vaccaro A, Wilson JR, Singh A, W Cadotte D, Harrop JS, et al. Early versus delayed decompression for traumatic cervical spinal cord injury: results of the Surgical Timing in Acute Spinal Cord Injury Study (STASCIS). PLoS One. 2012;7:e32037. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22384132
Back T, Hoehn M, Mies G, Busch E, Schmitz B, Kohno K, et al. Penumbral tissue alkalosis in focal cerebral ischemia: relationship to energy metabolism, blood flow, and steady potential. Ann Neurol. 2000;47:485–92. Available from: http://www.ncbi.nlm.nih.gov/pubmed/10762160
Ingvar DH, Lassen NA. Methods for cerebral blood flow measurements in man. Br J Anaesth. 1965;37:216–24. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0007091217535643
•• Badhiwala JH, Wilson JR, Witiw CD, Harrop JS, Vaccaro AR, Aarabi B, et al. The influence of timing of surgical decompression for acute spinal cord injury: a pooled analysis of individual patient data. Lancet Neurol. 2021;20:117–26. Available from: http://www.ncbi.nlm.nih.gov/pubmed/33357514. Pivitol study of demonstrating the relationship between early surgical deompression and improved motor score.
van Middendorp JJ, Barbagallo G, Schuetz M, Hosman AJF. Design and rationale of a Prospective, Observational European Multicenter study on the efficacy of acute surgical decompression after traumatic spinal cord injury: the SCI-POEM study. Spinal Cord. 2012;50:686–94. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22508536
Nagoshi N, Nakashima H, Fehlings MG. Riluzole as a neuroprotective drug for spinal cord injury: from bench to bedside. Molecules. 2015;20:7775–89. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25939067
Dauleac C, Mottolese C, Beuriat P-A, Szathmari A, Di Rocco F. Superiority of thoracolumbar injury classification and severity score (TLICS) over AOSpine thoracolumbar spine injury classification for the surgical management decision of traumatic spine injury in the pediatric population. Eur Spine J. 2021;30:3036–42. Available from: http://www.ncbi.nlm.nih.gov/pubmed/33475841
Merrill S, Clifton W, Valero-Moreno F, Damon A, Rahmathulla G. Vertebral artery injury with coinciding unstable cervical spine trauma: mechanisms, evidence-based management, and treatment options. Cureus. 2020;12:e7225. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32274283
Strohm TA, John S, Hussain MS. Cerebrospinal fluid drainage and blood pressure elevation to treat acute spinal cord infarct. Surg Neurol Int. 2018;9:195. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30294499
Martirosyan NL, Kalani MYS, Bichard WD, Baaj AA, Gonzalez LF, Preul MC, et al. Cerebrospinal fluid drainage and induced hypertension improve spinal cord perfusion after acute spinal cord injury in pigs. Neurosurgery. 2015;76:461–8; discussion 468–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25621979
Werndle MC, Saadoun S, Phang I, Czosnyka M, Varsos G V, Czosnyka ZH, et al. Monitoring of spinal cord perfusion pressure in acute spinal cord injury: initial findings of the injured spinal cord pressure evaluation study*. Crit Care Med. 2014;42:646–55. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24231762
Tykocki T, Poniatowski Ł, Czyż M, Koziara M, Wynne-Jones G. Intraspinal pressure monitoring and extensive duroplasty in the acute phase of traumatic spinal cord injury: a systematic review. World Neurosurg. 2017;105:145–52. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28578120
Hogg FRA, Gallagher MJ, Chen S, Zoumprouli A, Papadopoulos MC, Saadoun S. Predictors of intraspinal pressure and optimal cord perfusion pressure after traumatic spinal cord injury. Neurocrit Care. 2019;30:421–8. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30328047
Hogg FRA, Kearney S, Zoumprouli A, Papadopoulos MC, Saadoun S. Acute spinal cord injury: correlations and causal relations between intraspinal pressure, spinal cord perfusion pressure, lactate-to-pyruvate ratio, and limb power. Neurocrit Care. 2021;34:121–9. Available from: https://link.springer.com/10.1007/s12028-020-00988-2
Ditunno JF, Little JW, Tessler A, Burns AS. Spinal shock revisited: a four-phase model. Spinal Cord. 2004;42:383–95. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15037862
Ko H-Y. Revisit spinal shock: pattern of reflex evolution during spinal shock. Korean J neurotrauma. 2018;14:47–54. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30402418
Veeravagu A, Jiang B, Rincon F, Maltenfort M, Jallo J, Ratliff JK. Acute respiratory distress syndrome and acute lung injury in patients with vertebral column fracture(s) and spinal cord injury: a nationwide inpatient sample study. Spinal Cord. 2013;51:461–5. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23478670
Velmahos GC, Toutouzas K, Chan L, Tillou A, Rhee P, Murray J, et al. Intubation after cervical spinal cord injury: to be done selectively or routinely? Am Surg. 2003;69:891–4. Available from: http://www.ncbi.nlm.nih.gov/pubmed/14570369
Grassner L, Wutte C, Klein B, Mach O, Riesner S, Panzer S, et al. Early decompression (< 8 h) after traumatic cervical spinal cord injury improves functional outcome as assessed by spinal cord independence measure after one year. J Neurotrauma. 2016;33:1658–66. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27050499
Pastrana EA, Saavedra FM, Murray G, Estronza S, Rolston JD, Rodriguez-Vega G. Acute adrenal insufficiency in cervical spinal cord injury. World Neurosurg. 2012;77:561–3. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22120347
Krassioukov A V, Karlsson A-K, Wecht JM, Wuermser L-A, Mathias CJ, Marino RJ, et al. Assessment of autonomic dysfunction following spinal cord injury: rationale for additions to International Standards for Neurological Assessment. J Rehabil Res Dev. 2007;44:103–12. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17551864
Mathias CJ. Bradycardia and cardiac arrest during tracheal suction--mechanisms in tetraplegic patients. Eur J Intensive Care Med. 1976;2:147–56. Available from: http://www.ncbi.nlm.nih.gov/pubmed/793846
Altaf F, Griesdale DE, Belanger L, Ritchie L, Markez J, Ailon T, et al. The differential effects of norepinephrine and dopamine on cerebrospinal fluid pressure and spinal cord perfusion pressure after acute human spinal cord injury. Spinal Cord. 2017;55:33–8. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27271117
Claret Teruel G, Trenchs Sáinz de la Maza V, Palomeque Rico A. Pediatric acute spinal cord injury. An Pediatr (Barc). 2006;65:162–5. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16948980
Foran SJ, Taran S, Singh JM, Kutsogiannis DJ, McCredie V. Timing of tracheostomy in acute traumatic spinal cord injury: a systematic review and meta-analysis. J Trauma Acute Care Surg. 2022;92:223–31. Available from: http://www.ncbi.nlm.nih.gov/pubmed/34508010
Posluszny JA, Onders R, Kerwin AJ, Weinstein MS, Stein DM, Knight J, et al. Multicenter review of diaphragm pacing in spinal cord injury: successful not only in weaning from ventilators but also in bridging to independent respiration. J Trauma Acute Care Surg. 2014;76:303–9; discussion 309–10. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24458038
Onders RP, Elmo M, Kaplan C, Schilz R, Katirji B, Tinkoff G. Long-term experience with diaphragm pacing for traumatic spinal cord injury: early implantation should be considered. Surgery. 2018;164:705–11. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30195400
Levi L, Wolf A, Belzberg H. Hemodynamic parameters in patients with acute cervical cord trauma: description, intervention, and prediction of outcome. Neurosurgery. 1993;33:1007–16; discussion 1016–7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/8133985
Vale FL, Burns J, Jackson AB, Hadley MN. Combined medical and surgical treatment after acute spinal cord injury: results of a prospective pilot study to assess the merits of aggressive medical resuscitation and blood pressure management. J Neurosurg. 1997;87:239–46. Available from: http://www.ncbi.nlm.nih.gov/pubmed/9254087
Walters BC, Hadley MN, Hurlbert RJ, Aarabi B, Dhall SS, Gelb DE, et al. Guidelines for the management of acute cervical spine and spinal cord injuries. Neurosurgery. 2013;60:82–91. Available from: https://academic.oup.com/neurosurgery/article/60/CN_suppl_1/82/2595462
Grossman RG, Frankowski RF, Burau KD, Toups EG, Crommett JW, Johnson MM, et al. Incidence and severity of acute complications after spinal cord injury. J Neurosurg Spine. 2012;17:119–28. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22985378
Chen S, Smielewski P, Czosnyka M, Papadopoulos MC, Saadoun S. Continuous monitoring and visualization of optimum spinal cord perfusion pressure in patients with acute cord injury. J Neurotrauma. 2017;34:2941–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28351230
Shadgan B, Macnab A, Fong A, Manouchehri N, So K, Shortt K, et al. Optical assessment of spinal cord tissue oxygenation using a miniaturized near infrared spectroscopy sensor. J Neurotrauma. 2019;36:3034–43. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31044642
Colloca L, Ludman T, Bouhassira D, Baron R, Dickenson AH, Yarnitsky D, et al. Neuropathic pain. Nat Rev Dis Prim. 2017;3:17002. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28205574
Walker SM. Neuropathic pain in children: steps towards improved recognition and management. EBioMedicine . 2020;62:103124. Available from: http://www.ncbi.nlm.nih.gov/pubmed/33248373
• Lakra C, Swayne O, Christofi G, Desai M. Autonomic dysreflexia in spinal cord injury. Pract Neurol. 2021;21:532–8. Available from: http://www.ncbi.nlm.nih.gov/pubmed/34353860. Thorough overview of AD management.
Karlsson AK. Autonomic dysreflexia. Spinal Cord. 1999;37:383–91. Available from: http://www.ncbi.nlm.nih.gov/pubmed/10432257
Eldahan KC, Rabchevsky AG. Autonomic dysreflexia after spinal cord injury: systemic pathophysiology and methods of management. Auton Neurosci. 2018;209:59–70. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28506502
Wan D, Krassioukov A V. Life-threatening outcomes associated with autonomic dysreflexia: a clinical review. J Spinal Cord Med. 2014;37:2–10. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24090418
Allen KJ, Leslie SW. Autonomic dysreflexia . StatPearls. 2021. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29494041
Consortium for Spinal Cord Medicine. Acute management of autonomic dysreflexia: individuals with spinal cord injury presenting to health-care facilities. J Spinal Cord Med. 2002;25 Suppl 1:S67–88. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12051242
Flack CK, Mellon MJ. Current management strategies for autonomic dysreflexia. Curr Bladder Dysfunct Rep. 2018;13:224–9. Available from: http://link.springer.com/10.1007/s11884-018-0488-x
Qi Z, Middleton JW, Malcolm A. Bowel dysfunction in spinal cord injury. Curr Gastroenterol Rep. 2018;20:47. Available from: http://link.springer.com/10.1007/s11894-018-0655-4
Eswara JR, Castellan M, González R, Mendieta N, Cendron M. The urological management of children with spinal cord injury. World J Urol. 2018;36:1593–601. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30105455
Thompson AJ, McSwain SD, Webb SA, Stroud MA, Streck CJ. Venous thromboembolism prophylaxis in the pediatric trauma population. J Pediatr Surg. 2013;48:1413–21. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23845640
Jones T, Ugalde V, Franks P, Zhou H, White RH. Venous thromboembolism after spinal cord injury: incidence, time course, and associated risk factors in 16,240 adults and children. Arch Phys Med Rehabil. 2005;86:2240–7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16344018
Consortium for Spinal Cord Medicine. Prevention of venous thromboembolism in individuals with spinal cord injury: clinical practice guidelines for health care providers, 3rd ed.: Consortium for Spinal Cord Medicine. Top Spinal Cord Inj Rehabil. 2016;22:209–40. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29339863
Groah SL, Schladen M, Pineda CG, Hsieh C-HJ. Prevention of pressure ulcers among people with spinal cord injury: a systematic review. PMR. 2015;7:613–36. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25529614
Evaniew N, Belley-Côté EP, Fallah N, Noonan VK, Rivers CS, Dvorak MF. Methylprednisolone for the treatment of patients with acute spinal cord injuries: a systematic review and meta-analysis. J Neurotrauma. 2016;33:468–81. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26529320
Fehlings MG, Wilson JR, Harrop JS, Kwon BK, Tetreault LA, Arnold PM, et al. Efficacy and safety of methylprednisolone sodium succinate in acute spinal cord injury: a systematic review. Glob spine J. 2017;7:116S-137S. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29164020
Sayer FT, Kronvall E, Nilsson OG. Methylprednisolone treatment in acute spinal cord injury: the myth challenged through a structured analysis of published literature. Spine J. 2006;6:335–43. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16651231
Bracken MB. Steroids for acute spinal cord injury. Cochrane Database Syst Rev . 2012; Available from: https://doi.wiley.com/10.1002/14651858.CD001046.pub2
Bracken MB, Shepard MJ, Holford TR, Leo-Summers L, Aldrich EF, Fazl M, et al. Administration of methylprednisolone for 24 or 48 hours or tirilazad mesylate for 48 hours in the treatment of acute spinal cord injury. Results of the Third National Acute Spinal Cord Injury Randomized Controlled Trial. National Acute Spinal Cord Injury. JAMA. 1997;277:1597–604. Available from: http://www.ncbi.nlm.nih.gov/pubmed/9168289
Chikuda H, Yasunaga H, Takeshita K, Horiguchi H, Kawaguchi H, Ohe K, et al. Mortality and morbidity after high-dose methylprednisolone treatment in patients with acute cervical spinal cord injury: a propensity-matched analysis using a nationwide administrative database. Emerg Med J. 2014;31:201–6. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23449889
Geisler FH, Dorsey FC, Coleman WP. GM1 gangliosides in the treatment of spinal cord injury: report of preliminary data analysis. Acta Neurobiol Exp (Wars). 1990;50:515–21. Available from: http://www.ncbi.nlm.nih.gov/pubmed/2130666
Geisler FH, Coleman WP, Grieco G, Poonian D, Sygen Study Group. The Sygen multicenter acute spinal cord injury study. Spine (Phila Pa 1976). 2001;26:S87–98. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11805614
Meshkini A, Salehpour F, Aghazadeh J, Mirzaei F, Naseri Alavi SA. Riluzole can improve sensory and motor function in patients with acute spinal cord injury. Asian J Neurosurg. 13:656–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30283521
Srinivas S, Wali AR, Pham MH. Efficacy of riluzole in the treatment of spinal cord injury: a systematic review of the literature. Neurosurg Focus. 2019;46:E6. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30835675
Gibson CL, Jones NC, Prior MJW, Bath PMW, Murphy SP. G-CSF suppresses edema formation and reduces interleukin-1beta expression after cerebral ischemia in mice. J Neuropathol Exp Neurol. 2005;64:763–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16141785
Festoff BW, Ameenuddin S, Arnold PM, Wong A, Santacruz KS, Citron BA. Minocycline neuroprotects, reduces microgliosis, and inhibits caspase protease expression early after spinal cord injury. J Neurochem . 2006;97:1314–26. Available from: https://onlinelibrary.wiley.com/10.1111/j.1471-4159.2006.03799.x
American Spinal Injury Association. International Standards for Neurological Classification of Spinal Cord Injury, revised 2019. Richmond, VA.
Talbott JF, Whetstone WD, Readdy WJ, Ferguson AR, Bresnahan JC, Saigal R, et al. The Brain and Spinal Injury Center score: a novel, simple, and reproducible method for assessing the severity of acute cervical spinal cord injury with axial T2-weighted MRI findings. J Neurosurg Spine. 2015;23:495–504. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26161519
Krassioukov A, Eng JJ, Warburton DE, Teasell R, Spinal Cord Injury Rehabilitation Evidence Research Team. A systematic review of the management of orthostatic hypotension after spinal cord injury. Arch Phys Med Rehabil. 2009;90:876–85. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19406310
•• Rowald A, Komi S, Demesmaeker R, Baaklini E, Hernandez-Charpak SD, Paoles E, et al. Activity-dependent spinal cord neuromodulation rapidly restores trunk and leg motor functions after complete paralysis. Nat Med. 2022;28:260–71. Available from: http://www.ncbi.nlm.nih.gov/pubmed/35132264. Landmark study demonstrating restoration of trunk and leg in patients with complete sensorimotor paralysis.
Casha S, Zygun D, McGowan MD, Bains I, Yong VW, Hurlbert RJ. Results of a phase II placebo-controlled randomized trial of minocycline in acute spinal cord injury. Brain. 2012;135:1224–36. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22505632
Kucher K, Johns D, Maier D, Abel R, Badke A, Baron H, et al. First-in-man intrathecal application of neurite growth-promoting anti-Nogo-A antibodies in acute spinal cord injury. Neurorehabil Neural Repair. 2018;32:578–89. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29869587
Author information
Authors and Affiliations
Contributions
AXT, JJR, DD-W, SPT, JCE, DFB, and JAC performed the literature search, analysis, drafted, and critically revised the work.
Corresponding author
Ethics declarations
Conflict of Interest
The authors did not receive support from any organization for the submitted work. The authors have no relevant financial or non-financial interests to disclose. Open access of this publication was supported by the Texas Children’s Hospital Young Investigator Endowment.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Topical Collection on Critical Care Neurology
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Thomas, A.X., Riviello, J.J., Davila-Williams, D. et al. Pharmacologic and Acute Management of Spinal Cord Injury in Adults and Children. Curr Treat Options Neurol 24, 285–304 (2022). https://doi.org/10.1007/s11940-022-00720-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11940-022-00720-9