Abstract
Purpose of review
While care for patients with movement disorders has traditionally focused on motor symptoms, there is increasing evidence that optimal care for these disorders is more complex both in terms of the spectrum of symptoms experienced by patients (e.g., pain, depression) and the multidimensional needs of patients and their families. Palliative care is an approach to the care of patients and families affected by serious illnesses that seeks to relieve suffering by addressing complex medical symptoms, psychosocial issues, spiritual well-being, and goals of care. While traditionally associated with cancer and hospice, more recent work in palliative care has focused on integrating a palliative care approach from the time of diagnosis for patients with chronic illnesses, including movement disorders.
Recent findings
Studies of patients with movement disorders and their family caregivers suggest that these patients have significant unmet needs under current models of care, including underrecognition and treatment of non-motor symptoms, inadequate psychosocial support, and suboptimal end-of-life care. We describe how a palliative care approach can empower clinicians, patients, and families to reduce common sources of suffering and optimize quality of life. This field recognizes the importance of primary palliative care (palliative skills useful for any clinician caring for persons caring for serious illness) as a foundation of the palliative care approach and complementary to specialist palliative care. In this article, we will focus on primary palliative care skills for movement disorder specialists including providing a diagnosis and prognosis with compassion, discussing goals of care, complex symptom management, caregiver support, spiritual and emotional well-being, and referral to hospice and specialist palliative care.
Summary
A palliative care approach complements other efforts in movement disorder care. Research is needed to evaluate and develop therapeutic interventions and models of care applying a palliative care approach.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
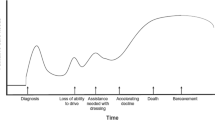
The World Health Organization defines palliative care as “an approach that improves the quality of life (QOL) of patients and families facing life-threatening illness, through the prevention and relief of suffering by means of early identification and impeccable assessment and treatment of pain and other problems, physical, psychosocial and spiritual” [1]. Although originating in the hospice movement for cancer, there are several developments within the field of palliative care relevant to patients and families affected by movement disorders [2••, 3, 4••]. First, palliative approaches are indicated for non-cancer illnesses which are incurable, progressive, or associated with high symptom burden such as movement disorders [5]. Second, palliative care can be provided at any time during an illness, and evidence from other conditions suggests that early palliative care improves patient QOL, reduces health care costs, and may prolong life [6•, 7]. This palliative care approach begins at the time of diagnosis and is integrated into future visits based on ongoing assessment of patient and caregiver needs, rather than estimated prognosis or waiting for “the right time.” Third, palliative care can be integrated with standard care based on the needs and goals of patients and their disease trajectory (Fig. 1). Finally, palliative care may be conceptualized in terms of primary palliative care (skills relevant to all clinicians) and secondary or tertiary palliative care (complex or advanced care provided by specially trained teams or providers) [8, 9]. In this manuscript, we focus on primary palliative skills for movement disorder specialists (Table 1). Our manuscript focuses on Parkinson’s disease (PD) and Huntington’s disease (HD) as the palliative needs of these populations are better described in the literature, but this approach is highly relevant to other movement disorders [4••, 10•, 11••].
Relevance of palliative care to movement disorders and current gaps
Palliative models of care address several important issues facing patients and families affected by movement disorders, including advance care planning, caregiver support, and intensive management of distressing medical and psychiatric symptoms. Unfortunately, the majority of movement disorder patients do not receive palliative services under current models of care even when approaching the end of life [12]. Hospital deaths for individuals with movement disorders are high despite the fact that, when asked, most patients express a preference to die at home [13, 14]. Many non-motor symptoms, including pain and depression, are underrecognized and undertreated in current models of care [15] and other issues, such as caregiver support and spiritual well-being, are rarely addressed [16, 17].
In addition to general misperceptions about palliative care, there are additional barriers which may contribute to underutilization of palliative care in movement disorders. In the case of PD, physicians commonly tell patients that they will die with PD and not from it, despite the fact that age-adjusted mortality is significantly higher in PD patients, that leading causes of death in PD are related to complications of PD (e.g., falls, pneumonia), and that the Centers for Disease Control and Prevention lists PD as the 14th leading cause of death in the USA [18, 19]. Patients also report that their physicians underestimate the emotional impact of receiving a PD diagnosis and desire more support around this event [20•, 21]. Even for HD, which is widely recognized as a fatal illness that merits palliative and other multidisciplinary services, there are disparities in terms of who receives this care and hospice services are underutilized [4••, 22]. Finally, many clinicians are uncomfortable with addressing these issues, possibly due to inadequate training and high moral distress in confronting palliative care and end-of-life issues [8, 23, 24].
Despite these limitations, there is cause for encouragement. Preliminary studies suggest palliative care approaches improve symptom burden, improve patient QOL, reduce caregiver distress, and reduce hospitalizations [25,26,27,28]. Use of the physician orders for life-sustaining treatment (POLST) form, a specific form of advance directive, may also reduce hospital deaths [29•]. Patients and caregivers report being open to palliative approaches and having high satisfaction with these interventions, and report preferences for earlier advance care discussions [20•, 30•].
Providing a diagnosis and prognosis with compassion
There are few events as memorable or traumatic to patients and their families as receiving their diagnosis [21]. However, the physician can improve the experience by responding empathetically to the patient’s emotional reaction, sharing information and realistic goals, listening to the patient’s hopes and expectations, and making a follow-up plan. The SPIKES mnemonic can be used to guide these conversations (setting up the interview; assessing patient’s perception; obtaining patient’s invitation; giving knowledge; addressing emotions; strategy and summary) [31] as can the VitalTalk website or smartphone application [32]. Similarly, the mnemonic NURSE can be used to address patient emotions (name the emotion; understand the emotion; respect the emotion; support the emotion; explore the emotion) [33••]. Physicians should provide adequate time for these conversations and rapid follow-up to prevent feelings of confusion and abandonment. Regarding prognosis, it is important to assess how much patients and caregivers want to know and to reassess their desire for information at future visits as it may shift over time. Holloway et al. formulated an approach to answering the questions of “How long?” and “How well?” when addressing patient survival in advanced neurologic disease, providing guidance on tailoring this approach to individual patients and reducing common biases [34]. Regarding both diagnosis and prognosis, studies support an open approach and not withholding information unless specifically requested by the patient [35].
Setting and updating goals of care
Goals of care discussions are conversations in which patients, families, and clinicians work together to develop guidelines for current and future care. In this conversation, patients and family members share their values, hopes, and fears, and clinicians provide information regarding the diagnosis, prognosis, and guidance on available resources. Advance care planning is an important part of these discussions but should not be the sole focus. Other important issues to address include what activities or events are most important to maintain QOL and how the family would like to navigate ongoing or increasing levels of patient dependence. We find the following questions helpful in better understanding patients’ and families’ values: How do you define QOL? What do you currently enjoy and look forward to? What about your illness is most troubling to you? What are you most afraid of?
One of the medical team’s primary responsibilities in these discussions is to prioritize safety and possibly revise goals of independence if there are adverse safety implications. Both PD and HD are associated with motor vehicle accidents [36, 37], and a recent longitudinal study [38] of driving safety in PD demonstrated significant increase in driving safety errors after 2 years. While state laws vary, we recommend patients either stop driving or get a specialty driving evaluation. In some cases, hiding keys or selling vehicles may be justified for patient and community safety. Firearms and other potentially dangerous items should be secured or removed from the house, particularly in patients with psychotic symptoms. Verbal, physical, and/or sexual threats or abuse may be directed at either the caregiver or patient and may be more common in PD than previously believed [39]. If suspected or confirmed, adult protective services should be contacted, as such activity strongly suggests a need for placement, even if contrary to goals of care. Financial risk may arise through scams targeting persons with impaired cognition or through intentional spending by patients with poor judgment or compulsive behavior [40, 41]. The use of cash allowances or low-limit credit cards may minimize risks while maintaining a patient’s sense of independence. Wandering behaviors in those with dementia place patients at risk for adverse outcomes and may necessitate 24/7 supervision or placement [42]. Identification bracelets or necklaces are also highly recommended. Finally, falls are a significant cause of morbidity and mortality for patients as well as caregiver distress [43]. More than 50% of individuals with HD are recurrent fallers, experiencing two or more falls per year [44]; and in a recent prospective study [45] of newly diagnosed PD, 79.7% participants fell over the 54 months, and the majority (89.3%) were recurrent fallers. Home safety evaluations and medical alert bracelets or necklaces may mitigate some of this risk, particularly in situations where the primary caregiver is also frail. Mobility restrictions (e.g., use of wheelchair or walking only with assistance) may also be needed to prevent falls and related complications. It is often helpful for the clinician to play the role of the bad guy to alleviate family strain and blame.
While goals of care conversations may not be opportune at the time of diagnosis, they should be initiated early, due to the possibility of progressive cognitive impairment impacting patients’ abilities to set goals for themselves. In these conversations, the clinician should be mindful of the patient’s capacity, being careful not to overemphasize the primacy of patient desires when they appear to lack capacity, nor to discount the desires of patients with the capacity to provide input despite mild to moderate dementia [46•, 47]. Capacity is best assessed in relation to specific issues rather than globally (e.g., one may have capacity to decide general goals of care but not complex financial decisions) and can be assessed by remembering four “Cs” (a patient’s ability to understand their condition, their choices, the consequences of choices, and whether decisions are consistent over time) [48].
While studies have not yet been completed to show the benefits of goals of care discussions in movement disorder populations, the Care Coordination [49] for Health Promotion and Activities in PD (CHAPS) is a program within several of the Veterans Affairs Medical Centers which proactively addresses both motor and non-motor health problems in veterans with PD and includes tools to enhance patient self-management and care planning. Advance care directives and long-term planning are addressed and a randomized controlled trial involving a comparison between arm group receiving the nurse-led CHAPS Program intervention and a control arm receiving care as usual is underway. The work has evolved from earlier health services research that identified gaps in care and will likely show efficacy in improving performance in quality measures and in the secondary outcomes of patient health-related quality of life, self-efficacy, and perceptions of care quality.
Advance care planning
Advance care planning requires conversation(s) about patient preferences for care if the patient is unable to communicate. Medicare Part B pays for advance care planning discussions between patients and health care providers [50]. There are many types of advance directives, but most contain two key elements: (1) identifying a health care surrogate and (2) creating a living will outlining care preferences at the end of life. Organ and tissue donation preferences as well as brain donation for research may be discussed and provide a means of alleviating suffering by bringing meaning to the illness. The most important element is identifying a health care surrogate to be a voice for the patient if the patient is unable to speak for himself. Without an identified surrogate, state law governs who is legally responsible for making medical decisions and may defer to persons the patient would not want involved. As surrogates guide health care providers on what patients would want if they could speak for themselves, patients should discuss their wishes with their surrogate and advise them of any changes. A living will outlines preferences for the type and intensity of care provided at the end of life. Such a document typically includes preferences about cardio-pulmonary resuscitation (CPR), mechanical ventilation, and artificial nutrition, but may also include instructions for symptom management and religious rituals.
Individuals often confuse advance care planning with estate planning and frequently complete these forms with a lawyer, which can result in lengthy and confusing documents that do not match patients’ goals or medical practices. In most cases, advance directives may simply be witnessed and signed by two adults and do not need to be notarized or prepared by an attorney. Individuals may download a free copy of their state’s advance directive form at http://www.caringinfo.org.
A copy of the advance directive should be shared with the health care surrogate, other family members, and the health care provider and a copy placed on the refrigerator where emergency medical technicians can easily find it. The advance directive should be filed in electronic medical records and patients who are “do not resuscitate” may include this on medical bracelets to avoid unnecessary interventions outside of the home. Several advance directive applications using smartphone technology allow individuals to carry their wishes with them at all times.
Advance care planning is a process that may take place over several visits and should be revisited annually and with changes in functional status. In one cross-sectional study in PD [51], 94% of those surveyed wanted prognosis and treatment information early in the disease course, while much fewer wanted early discussions about end-of-life care planning (27%) or end-of-life care options such as hospice (21%). It is important to emphasize that there are no right or wrong answers, and that patients can change their minds at a later date should their values or circumstances change. In a survey of patients with HD [52], the majority of interviewees expressed some kind of wish regarding end of life, probably more than they had revealed to their physician, but were sometimes hesitant to discuss it. Activities that can complement discussions with the provider include http://theconversationproject.org/, http://www.emmisolutions.com/, or http://www.gowish.org/.
Complex symptom management
A thorough review of the management of non-motor symptoms goes beyond the scope of this article. Rather, we will discuss potential differences in a palliative care approach and refer to relevant reviews for other details. As QOL is heavily driven by non-motor issues across movement disorders and these disorders are frequently not mentioned by patients or recognized by providers, we recommend taking an active assessment of these issues as a routine part of care including pain, mood, psychotic and behavioral symptoms, sleep and energy, communication, autonomic symptoms, swallowing, and nutrition [15, 53, 54].
Pain is a common symptom in many movement disorders [53, 55]. A palliative care approach to pain includes understanding not only the qualities or the pain, but also its functional, psychological, and social impact [56]. Clinicians should talk with patients about their goals for pain management, emphasizing that functional improvement may be more attainable than complete pain elimination and that risks and side effects of medications must be weighed against function [57]. A proactive approach should be taken to avoid preventable pain (e.g., keeping joints mobilized, good skin care in bed- or chair-bound individuals) and treatment should be directed at specific causes when possible. Opioids may be used judiciously as there is some evidence to support their efficacy in this population [58•] (see https://www.cdc.gov/drugoverdose/prescribing/guideline.html for best practices).
It is important to differentiate mood disorders from other difficult emotions (see below) that may contribute to, or be mistaken for, mood disorders. It is equally important to assess mood disorders as potential drivers of end-of-life goals of care, including physician-assisted dying. Sleep disorders and fatigue are common and may impact both patient and caregiver well-being [59, 60]. Management requires identifying and treating contributing factors such as depression, specific sleep disturbances, anemia, pain, and medications [61]. Non-pharmacological approaches could include exercise, changes in nutrition, energy conservation strategies, mindfulness training, cognitive behavioral therapy, or acupuncture [61, 62].
Many movement disorders lead to dysphagia, weight loss, anorexia, and changes in appetite. These symptoms may be important indicators of progression of the illness and may signal that patients are nearing the end of life [63•]. Modifications to diet should be made within the context of an individual’s goals of care. For example, patients may forego pureed diets or thickened liquids and choose to continue eating their favorite foods if their goals place a higher value on the pleasures of eating than maximizing safety. Feeding tubes are rarely indicated in these conditions and evidence does not support their use to prevent aspiration or prolong survival [64, 65]. A high rate of complications secondary to percutaneous endoscopic gastrostomy (PEG) tube placement has been described in neurodegenerative disease likely to be due to multiple factors including the inherent morbidity and poor conditioning associated with neurodegenerative disease, poor baseline nutritional status, ongoing risk factors for aspiration associated with neurodegenerative disease, and long in-patient stay with consequent increased susceptibility to infection but also the opportunity for improved ascertainment of complications [66]. Enteral feeding may be considered when prolonged and difficult mealtimes reduce QOL or situations in which maintaining hydration and nutrition despite dysphagia is consistent with goals of care, particularly in situations where dysphagia is advanced out of proportion to other symptoms such as dementia.
Caregiver assessment and support
Patients with movement disorders often develop long-term disabilities and rely on family members to meet their daily care needs. Family and other informal unpaid caregivers represent an increasing yet infrequently recognized investment of human resources who, in addition to helping with activities of daily living, commonly perform medical or nursing tasks [67].
Substantial loss of function and symptom burden can occur relatively early in diseases such as HD, resulting in years of marked morbidity [10•, 68]. As a result, caregivers frequently suffer from sleeplessness, fatigue, anxiety, depression, and impaired immunological responses [69]. In a study comparing caregiver mood and QOL for those caring for patients with progressive neurological illnesses, caregivers of patients with HD experienced among the highest levels of anxiety, depression, and confusion as well as the greatest decrement in social relationships compared to caregivers of other illnesses [70].
Caregiver burden is defined as the extent to which caregivers perceive adverse effects to their emotional, social, financial, physical, and spiritual functioning [71]. Risk factors for caregiver burden include female sex, low educational attainment, residing with the care recipient, depression, social isolation, financial stress, higher number of hours spent caregiving, and lack of choice in being a caregiver [72]. Compassion fatigue reduces the capacity for compassion as a consequence of being exhausted from absorbing the suffering of others and may affect 40–80% of caregivers [73]. Distressed caregivers often develop negative attitudes toward the disease, relationship problems with the patient and other family members arise, and they often experience complex bereavement [74].
Attending to the health and well-being of the family caregiver is not a standard part of movement disorder care and may be facilitated through interdisciplinary clinics including social work and counseling resources [75]. However, clinicians can perform a brief assessment of caregiver well-being without a team. In addition to detecting caregiver issues early and making appropriate referrals, even brief inquiries directed to the caregiver are deeply appreciated and let the caregiver know they are not alone in this struggle [30•]. Caregiver assessment may be an informal addition to the clinical interview, asking questions about caregiver well-being and support, or utilizing one of the many formal assessment tools [67, 71, 76,77,78,79]. The National Consensus Development Conference for Caregiver Assessment recommends the following approach: (1) identify primary and additional caregivers, (2) incorporate needs and preferences of both the patient and caregiver in all care planning, (3) improve caregivers’ understandings of their roles and teach them the skills necessary to carry out the tasks of caregiving, and (4) recognize the need for periodic reassessment of care outcomes for the patient and caregiver [72, 80].
Protective factors against caregiver burden include resilience, perceived competence, self-efficacy emotional regulation, social support, optimism, and emphasis on the positive aspects of care [74]. Some of these protective characteristics can be trained and enhanced through targeted interventions. Available support for caregivers include educational materials, home health care or skilled nursing support, respite care, advocacy organizations, and local or online support groups [4••]. Specialty palliative care, including outpatient clinics and home palliative care, may provide more intensive caregiver support [4••]. The involvement of an interdisciplinary team for PD, for example, reduces caregiver strain and aggression between patient and caregiver [68], and earlier involvement helps adapt services to meet the changing needs of the patient [81].
Addressing common social, emotional, and spiritual issues
The palliative care model goes beyond the medical model of psychiatric illness to embrace the many social, emotional, and spiritual issues that may arise in the course of illness. Neurodegenerative movement disorders slowly rob patients of basic abilities to work, performing activities of daily living and purposeful tasks, and being independent, resulting in a continuum of loss. These changes impact one’s sense of identity and social networks through the eventual inability to work, limitations of leisurely activities, and loss of mobility. The unpredictability of PD symptoms and ability to participate in activities is a common source of distress and frustration [82]. Social activity is critical for maintenance of cognitive ability [83] and social isolation adversely affects QOL, physical and mental health, and mortality especially in PD [84, 85]. Like exercise, social activity should be assessed and promoted through awareness, community organizations, and social work referrals, if needed.
It is important to distinguish common but difficult emotional reactions to chronic illness from psychiatric illness, provide appropriate support, and not further isolate patients and families who may already experience shame around these feelings. Common difficult emotions include anticipatory grief (sense of loss), guilt, frustration, demoralization, anger, and worries and fears about the future including financial issues and future health events such as death or dementia [17, 20•, 30•, 86,87,88,89]. These emotions are frequently driven by spiritual or existential issues including hopelessness, meaninglessness, loneliness, death anxiety, and an inability to find sources of love, peace, comfort, transcendence, or connection [90]. The search for meaning is one of the central coping mechanisms when facing progressive illness and at the end of life, and may buffer against depression, hopelessness, and desire for hastened death among terminally ill patients [91,92,93]. Spirituality, religion, and culture also may provide meaning, and these factors may guide patient decision-making during illness and at the end of life. Chaplains, spiritual counselors, or community religious leaders may provide emotional and spiritual support, particularly for patients adverse to the idea of psychotherapy [94]. Standardized tools are available to assess and identify religious or spiritual/existential background, preferences, and beliefs [90, 95, 96]. Interventions that enhance meaning and build resilience include traditional psychotherapy, mindfulness-based approaches, gratitude journals, narrative-based approaches (e.g., “dignity therapy”), and family therapy [97,98,99,100].
Referring to hospice or specialty palliative care services
Hospice care refers specifically to end-of-life palliative care. In the USA, the Medicare hospice benefit is available to patients certified by two physicians to have a prognosis of 6 months or less. Hospice is fully covered by Medicare and Medicaid coverage varies by state. Medication costs are included for conditions related to the terminal illness [101]. We recommend three questions to make appropriate hospice referrals and discuss hospice with patients. First, would the patient and family benefit from the services available from hospice? Hospice care is most commonly delivered in the home or current place of residence (e.g., assisted living) and includes intensive home monitoring, visiting nurses, social workers, dieticians, chaplains, and bereavement support for the family. Other services, including physical therapy, massage, and volunteer services, may vary between hospices. Second, are the patient’s and family’s goals of care aligned with hospice? A helpful question is, “If you were to develop a serious pneumonia, would you want to go to a hospital for intravenous fluids and antibiotics?” If the answer is no and the goals of care are comfort, hospice may be appropriate. If the answer is yes, one may consider home palliative care or other home services. Finally, does the patient qualify for hospice? Table 2 provides current Medicare eligibility guidelines relevant to movement disorders. These guidelines are not strict criteria, and patients demonstrating other end-of-life signs, including an accelerating disease course, loss of interest in eating or other activities, or loss of will/desire to live, may be appropriate for hospice. As with other aspects of palliative care, we recommend initiating hospice early to allow patients and families to receive the full benefit of these services, as waiting for “the right time” frequently means either no hospice or hospice stays of days to weeks.
A growing number of centers now offer specialized outpatient palliative care for patients with movement disorders [11••]. Referrals may be made to transfer care for patients with advanced disease or for assistance with complex medical symptoms such as pain, caregiver distress, psychosocial support, spiritual well-being, or complex goals of care discussions.
Physician-assisted death
Patients may request physician-assisted death (PAD) because of unmet psychosocial, physical, or spiritual needs [91, 102]. PAD is now legal in several states (OR, VT, WA, CA, CO, DC, and MT). PAD laws permit terminally ill patients with adequate mental capacity to request a prescription for a lethal dose of medication that the patient may ingest at a future date but may also elect not to take it [103]. Physicians practicing in states with legalized PAD who do not support this practice may opt out. For patients who contemplate PAD, we recommend referral to palliative care to achieve optimal symptom palliation, including physical and existential sources of suffering [103].
Conclusions and suggestions for future research
Palliative care issues are common across a wide range of movement disorders and a palliative care approach may improve the QOL of patients and families. Although interest in this area is growing, this field is still in its infancy and there is significant work ahead to ensure a palliative care approach is widely available. Barriers to palliative care include minimal training and exposure for the majority of neurology residents, movement disorder fellows and practicing clinicians, concerns over reimbursement for time-intensive and team-based services, and a paucity of evidence-based studies to justify and guide models of care and therapeutic interventions. High-priority research items to address these barriers include (1) validation of needs assessment tools to guide participant selection, (2) validation of outcome measures, (3) evaluation of patient and caregiver interventions, and (4) dissemination and implementation efforts.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
World Health Organization. WHO definition of palliative care [Online]. 2016. http://www.who.int/cancer/palliative/definition/en/ Accessed August 28, 2016.
•• Oliver DJ, Borasio GD, Caraceni A, de Visser M, Grisold W, Lorenzl S et al. A consensus review on the development of palliative care for patients with chronic and progressive neurological disease. European journal of neurology. 2016;23(1):30–8. doi:https://doi.org/10.1111/ene.12889. Important position statement and review of the role of palliative care in neurology.
Miyasaki JM, Kluger B. Palliative care for Parkinson’s disease: has the time come? Curr Neurol Neurosci Rep. 2015;15(5):542. https://doi.org/10.1007/s11910-015-0542-4.
•• Tarolli CG, Chesire AM, Biglan KM. Palliative care in Huntington disease: personal reflections and a review of the literature. Tremor Other Hyperkinet Mov (N Y). 2017;7:454. doi:https://doi.org/10.7916/D88057C7. Excellent review of palliative care issues in Huntington’s disease.
Boersma I, Miyasaki J, Kutner J, Kluger B. Palliative care and neurology: time for a paradigm shift. Neurology. 2014;83(6):561–7. https://doi.org/10.1212/WNL.0000000000000674.
•• Higginson IJ, Mc Crone P, Hart SR, Burman R, Silber E, Edmonds PM. Is short-term palliative care cost-effective in multiple sclerosis? A randomized phase II trial. J Pain Symptom Manage. 2009;38(6):816–26. https://doi.org/10.1016/j.jpainsymman.2009.07.002. One of the few randomized controlled trials of palliative care approaches for neurologic disease.
Temel JS, Greer JA, Muzikansky A, Gallagher ER, Admane S, Jackson VA, et al. Early palliative care for patients with metastatic non-small-cell lung cancer. N Engl J Med. 2010;363(8):733–42. https://doi.org/10.1056/NEJMoa1000678.
Creutzfeldt CJ, Gooley T, Walker M. Are neurology residents prepared to deal with dying patients? Arch Neurol. 2009;66(11):1427–8. https://doi.org/10.1001/archneurol.2009.241.
Quill TE, Abernethy AP. Generalist plus specialist palliative care—creating a more sustainable model. The New England journal of medicine. 2013;368(13):1173–5. https://doi.org/10.1056/NEJMp1215620.
•• Wiblin L, Lee M, Burn D. Palliative care and its emerging role in multiple system atrophy and progressive supranuclear palsy. Parkinsonism Relat Disord. 2017;34:7–14. https://doi.org/10.1016/j.parkreldis.2016.10.013. Good overview of palliative care as applied to other parkinsonian conditions.
•• Kluger BM, Fox S, Timmons S, Katz M, Galifianakis NB, Subramanian I et al. Palliative care and Parkinson’s disease: meeting summary and recommendations for clinical research. Parkinsonism Relat Disord. 2017;37:19–26. https://doi.org/10.1016/j.parkreldis.2017.01.008. Sumary of findings from an international working group on the current practice, state of evidence and research gaps.
Moens K, Houttekier D, Van den Block L, Harding R, Morin L, Marchetti S, et al. Place of death of people living with Parkinson’s disease: a population-level study in 11 countries. BMC Palliat Care. 2015;14:28. https://doi.org/10.1186/s12904-015-0021-3.
Snell K, Pennington S, Lee M, Walker R. The place of death in Parkinson’s disease. Age Ageing. 2009;38(5):617–9. https://doi.org/10.1093/ageing/afp123.
Gomes B, Calanzani N, Gysels M, Hall S, Higginson IJ. Heterogeneity and changes in preferences for dying at home: a systematic review. BMC Palliat Care. 2013;12:7. https://doi.org/10.1186/1472-684X-12-7.
Shulman LM, Taback RL, Rabinstein AA, Weiner WJ. Non-recognition of depression and other non-motor symptoms in Parkinson’s disease. Parkinsonism & related disorders. 2002;8(3):193–7.
Kessler S. Forgotten person in the Huntington disease family. Am J Med Genet. 1993;48(3):145–50. https://doi.org/10.1002/ajmg.1320480306.
Goy ER, Carter JH, Ganzini L. Needs and experiences of caregivers for family members dying with Parkinson disease. J Palliat Care. 2008;24(2):69–75.
Fall PA, Saleh A, Fredrickson M, Olsson JE, Granerus AK. Survival time, mortality, and cause of death in elderly patients with Parkinson’s disease: a 9-year follow-up. Mov Disord. 2003;18(11):1312–6. https://doi.org/10.1002/mds.10537.
Murphy S, Kochanek K. Deaths: preliminary data for 2010. National Vital Statistics Reports. 2012;60:1–52.
•• Boersma I, Jones J, Carter J, Bekelman D, Miyasaki J, Kutner J et al. Parkinson disease patients’ perspectives on palliative care needs: what are they telling us? Neurol Clin Pract. 2016;6(3):209–19. https://doi.org/10.1212/CPJ.0000000000000233. Qualitative study of the experiences of PD patients and how they may inform palliative and movement disorders care.
Phillips LJ. Dropping the bomb: the experience of being diagnosed with Parkinson’s disease. Geriatr Nurs. 2006;27(6):362–9. https://doi.org/10.1016/j.gerinurse.2006.10.012.
Klager J, Duckett A, Sandler S, Moskowitz C. Huntington’s disease: a caring approach to the end of life. Care Manag J. 2008;9(2):75–81.
Manu E, Marks A, Berkman CS, Mullan P, Montagnini M, Vitale CA. Self-perceived competence among medical residents in skills needed to care for patients with advanced dementia versus metastatic cancer. J Cancer Educ. 2012;27(3):515–20. https://doi.org/10.1007/s13187-012-0351-2.
Oliver D, Maguirre S, Hardiman O, Bede P. “Palliative care and end of life care” in neurodegenerative disorders: a clinical guide. 2nd ed. New York: Springer International Publishing; 2016.
Miyasaki JM, Long J, Mancini D, Moro E, Fox SH, Lang AE, et al. Palliative care for advanced Parkinson disease: an interdisciplinary clinic and new scale, the ESAS-PD. Parkinsonism & related disorders. 2012;18(Suppl 3):S6–9. https://doi.org/10.1016/j.parkreldis.2012.06.013.
Veronese S, Gallo G, Valle A, Cugno C, Chio A, Calvo A et al. Specialist palliative care improves the quality of life in advanced neurodegenerative disorders: NE-PAL, a pilot randomised controlled study. BMJ Support Palliat Care. 2015. https://doi.org/10.1136/bmjspcare-2014-000788.
Garden G, Green S, Pieniak S, Gladman J. The Bromhead Care Home Service: the impact of a service for care home residents with dementia on hospital admission and dying in preferred place of care. Clin Med (Lond). 2016;16(2):114–8. https://doi.org/10.7861/clinmedicine.16-2-114.
Singer AE, Goebel JR, Kim YS, Dy SM, Ahluwalia SC, Clifford M, et al. Populations and interventions for palliative and end-of-life care: a systematic review. J Palliat Med. 2016;19(9):995–1008. https://doi.org/10.1089/jpm.2015.0367.
•• Tuck KK, Zive DM, Schmidt TA, Carter J, Nutt J, Fromme EK. Life-sustaining treatment orders, location of death and co-morbid conditions in decedents with Parkinson’s disease. Parkinsonism Relat Disord. 2015;21(10):1205–9. https://doi.org/10.1016/j.parkreldis.2015.08.021. Important study on the influence of advance directives in PD.
•• Boersma I, Jones J, Coughlan C, Carter J, Bekelman D, Miyasaki J, et al. Palliative care and Parkinson’s disease: caregiver perspectives. J Palliat Med. 2017; https://doi.org/10.1089/jpm.2016.0325. Qualitative study of PD caregivers’ experiences that may inform movement disorders and palliative care
Baile WF, Buckman R, Lenzi R, Glober G, Beale EA, Kudelka AP. SPIKES—a six-step protocol for delivering bad news: application to the patient with cancer. Oncologist. 2000;5(4):302–11.
VITALtalk. VITALtalk. 2014. http://www.vitaltalk.org. March 18,2014.
•• Creutzfeldt CJ, Robinson MT, Holloway RG. Neurologists as primary palliative care providers: communication and practice approaches. Neurol Clin Pract. 2016;6(1):40–8. https://doi.org/10.1212/CPJ.0000000000000213. Excellent overview of primary palliative care skills relevant to movement disorders clinicians.
Holloway RG, Gramling R, Kelly AG. Estimating and communicating prognosis in advanced neurologic disease. Neurology. 2013;80(8):764–72. https://doi.org/10.1212/WNL.0b013e318282509c.
Hancock K, Clayton JM, Parker SM, der Wal S, Butow PN, Carrick S, et al. Truth-telling in discussing prognosis in advanced life-limiting illnesses: a systematic review. Palliat Med. 2007;21(6):507–17. https://doi.org/10.1177/0269216307080823.
Beglinger LJ, Prest L, Mills JA, Paulsen JS, Smith MM, Gonzalez-Alegre P, et al. Clinical predictors of driving status in Huntington’s disease. Mov Disord. 2012;27(9):1146–52. https://doi.org/10.1002/mds.25101.
Classen S, DP MC, Shechtman O, Awadzi KD, Lanford DN, Okun MS, et al. Useful field of view as a reliable screening measure of driving performance in people with Parkinson’s disease: results of a pilot study. Traffic Inj Prev. 2009;10(6):593–8. https://doi.org/10.1080/15389580903179901.
Uc EY, Rizzo M, O’Shea AMJ, Anderson SW, Dawson JD. Longitudinal decline of driving safety in Parkinson disease. Neurology. 2017; https://doi.org/10.1212/WNL.0000000000004629.
Bruno V, Mancini D, Ghoche R, Arshinoff R, Miyasaki JM. High prevalence of physical and sexual aggression to caregivers in advanced Parkinson’s disease. Experience in the palliative care program. Parkinsonism Relat Disord. 2016;24:141–2. https://doi.org/10.1016/j.parkreldis.2016.01.010.
Verbaan D, van Rooden SM, Visser M, Marinus J, Emre M, van Hilten JJ. Psychotic and compulsive symptoms in Parkinson’s disease. Mov Disord. 2009;24(5):738–44. https://doi.org/10.1002/mds.22453.
Han SD, Boyle PA, James BD, Yu L, Bennett DA. Mild cognitive impairment and susceptibility to scams in old age. J Alzheimers Dis. 2015;49(3):845–51. https://doi.org/10.3233/JAD-150442.
Futrell M, Melillo KD, Remington R, Butcher HK. Evidence-based practice guideline: wandering. J Gerontol Nurs. 2014;40(11):16–23. https://doi.org/10.3928/00989134-20140911-01.
Schrag A, Hovris A, Morley D, Quinn N, Jahanshahi M. Caregiver-burden in parkinson’s disease is closely associated with psychiatric symptoms, falls, and disability. Parkinsonism Relat Disord. 2006;12(1):35–41. https://doi.org/10.1016/j.parkreldis.2005.06.011.
Busse ME, Wiles CM, Rosser AE. Mobility and falls in people with Huntington’s disease. J Neurol Neurosurg Psychiatry. 2009;80(1):88–90. https://doi.org/10.1136/jnnp.2008.147793.
Lord S, Galna B, Yarnall AJ, Morris R, Coleman S, Burn D et al. Natural history of falls in an incident cohort of Parkinson’s disease: early evolution, risk and protective features. J Neurol. 2017;264(11):2268–76. https://doi.org/10.1007/s00415-017-8620-y.
•• Abu Snineh M, Camicioli R, Miyasaki JM. Decisional capacity for advanced care directives in Parkinson’s disease with cognitive concerns. Parkinsonism Relat Disord. 2017;39:77–9. https://doi.org/10.1016/j.parkreldis.2017.03.006. Important reminder to assess capacity when making advance care plans.
Dening KH, Jones L, Sampson EL. Advance care planning for people with dementia: a review. Int Psychogeriatr. 2011;23(10):1535–51. https://doi.org/10.1017/S1041610211001608.
Tunzi M. Can the patient decide? Evaluating patient capacity in practice. Am Fam Physician. 2001;64(2):299–306.
Connor K, Cheng E, Siebens HC, Lee ML, Mittman BS, Ganz DA, et al. Study protocol of “CHAPS”: a randomized controlled trial protocol of Care Coordination for Health Promotion and Activities in Parkinson’s Disease to improve the quality of care for individuals with Parkinson’s disease. BMC Neurol. 2015;15:258. https://doi.org/10.1186/s12883-015-0506-y.
Jones CA, Acevedo J, Bull J, Kamal AH. Top 10 Tips for using advance care planning codes in palliative medicine and beyond. J Palliat Med. 2016;19(12):1249–53. https://doi.org/10.1089/jpm.2016.0202.
Tuck KK, Brod L, Nutt J, Fromme EK. Preferences of patients with Parkinson’s disease for communication about advanced care planning. Am J Hosp Palliat Care. 2015;32(1):68–77. https://doi.org/10.1177/1049909113504241.
Booij SJ, Rodig V, Engberts DP, Tibben A, Roos RA. Euthanasia and advance directives in Huntington’s disease: qualitative analysis of interviews with patients. J Huntingtons Dis. 2013;2(3):323–30. https://doi.org/10.3233/JHD-130060.
Kuyper DJ, Parra V, Aerts S, Okun MS, Kluger BM. Nonmotor manifestations of dystonia: a systematic review. Mov Disord. 2011;26(7):1206–17. https://doi.org/10.1002/mds.23709.
Bernal-Pacheco O, Limotai N, Go CL, Fernandez HH. Nonmotor manifestations in Parkinson disease. Neurologist. 2012;18(1):1–16. https://doi.org/10.1097/NRL.0b013e31823d7abb.
Ha AD, Jankovic J. Pain in Parkinson’s disease. Mov Disord. 2012;27(4):485–91. https://doi.org/10.1002/mds.23959.
Roditi D, Robinson ME. The role of psychological interventions in the management of patients with chronic pain. Psychol Res Behav Manag. 2011;4:41–9. https://doi.org/10.2147/PRBM.S15375.
Gatchel RJ. Clinical Essentials of Pain Management. Washington, DC, US: American Psychological Association; 2005.
•• Trenkwalder C, Chaudhuri KR, Martinez-Martin P, Rascol O, Ehret R, Valis M et al. Prolonged-release oxycodone-naloxone for treatment of severe pain in patients with Parkinson’s disease (PANDA): a double-blind, randomised, placebo-controlled trial. Lancet Neurol. 2015;14(12):1161–70. https://doi.org/10.1016/S1474-4422(15)00243-4. Positive clinical trial on use of opiates for PD pain.
Herlofson K, Ongre SO, Enger LK, Tysnes OB, Larsen JP. Fatigue in early Parkinson’s disease. Minor inconvenience or major distress? Eur J Neurol. 2012;19(7):963–8. https://doi.org/10.1111/j.1468-1331.2012.03663.x.
Schapira AHV, Chaudhuri KR, Jenner P. Non-motor features of Parkinson disease. Nat Rev. Neurosci. 2017; https://doi.org/10.1038/nrn.2017.62.
Herlofson K, Kluger BM. Fatigue in Parkinson’s disease. J Neurol Sci. 2017;374:38–41. https://doi.org/10.1016/j.jns.2016.12.061.
Kluger BM, Rakowski D, Christian M, Cedar D, Wong B, Crawford J et al. Randomized, controlled trial of acupuncture for fatigue in Parkinson’s disease. Mov Disord. 2016;31(7):1027–32. https://doi.org/10.1002/mds.26597.
•• Goy ER, Bohlig A, Carter J, Ganzini L. Identifying predictors of hospice eligibility in patients with Parkinson disease. Am J Hosp Palliat Care. 2015;32(1):29–33. https://doi.org/10.1177/1049909113502119. Only study to date to look specifically at predictors of hospice in PD.
Cervo FA, Bryan L, Farber S. To PEG or not to PEG: a review of evidence for placing feeding tubes in advanced dementia and the decision-making process. Geriatrics. 2006;61(6):30–5.
Stavroulakis T, McDermott CJ. Enteral feeding in neurological disorders. Pract Neurol. 2016;16(5):352–61. https://doi.org/10.1136/practneurol-2016-001408.
Sarkar P, Cole A, Scolding NJ, Rice CM. Percutaneous endoscopic gastrostomy tube insertion in neurodegenerative disease: a retrospective study and literature review. Clin Endosc. 2017;50(3):270–8. https://doi.org/10.5946/ce.2016.106.
Anderson EW, White KM. “It has changed my life”: an exploration of caregiver experiences in serious illness. The American journal of hospice & palliative care. 2017:1049909117701895. doi:https://doi.org/10.1177/1049909117701895.
Fallon M, Foley P. Rising to the challenge of palliative care for non-malignant disease. Palliat Med. 2012;26(2):99–100. https://doi.org/10.1177/0269216312437281.
Marziali E, Donahue P. Caring for others: Internet video-conferencing group intervention for family caregivers of older adults with neurodegenerative disease. Gerontologist. 2006;46(3):398–403.
MP MC, Firth L, O’Connor E. A comparison of mood and quality of life among people with progressive neurological illnesses and their caregivers. J Clin Psychol Med Settings. 2009;16(4):355–62. https://doi.org/10.1007/s10880-009-9168-5.
Zarit SH, Todd PA, Zarit JM. Subjective burden of husbands and wives as caregivers: a longitudinal study. Gerontologist. 1986;26(3):260–6.
Adelman RD, Tmanova LL, Delgado D, Dion S, Lachs MS. Caregiver burden: a clinical review. JAMA. 2014;311(10):1052–60. https://doi.org/10.1001/jama.2014.304.
Sabo BM. Compassion fatigue and nursing work: can we accurately capture the consequences of caring work? Int J Nurs Pract. 2006;12(3):136–42. https://doi.org/10.1111/j.1440-172X.2006.00562.x.
Palacio C, Krikorian A, Limonero JT. The influence of psychological factors on the burden of caregivers of patients with advanced cancer: resiliency and caregiver burden. Palliative & supportive care. 2017:1–9. https://doi.org/10.1017/S1478951517000268.
Collins LG, Swartz K. Caregiver care. Am Fam Physician. 2011;83(11):1309–17.
Novak M, Guest C. Application of a multidimensional caregiver burden inventory. Gerontologist. 1989;29(6):798–803.
Zhong M, Evans A, Peppard R, Velakoulis D. Validity and reliability of the PDCB: a tool for the assessment of caregiver burden in Parkinson’s disease. Int Psychogeriatr. 2013;25(9):1437–41. https://doi.org/10.1017/S1041610213000586.
Tebb SS, Berg-Weger M, Rubio DM. The Caregiver Well-Being Scale: developing a short-form rapid assessment instrument. Health Soc Work. 2013;38(4):222–30.
Stull DE. The multidimensional caregiver strain index (MCSI). Its measurement and structure. Journal of Clinical Geropsychology. 1996;2(3):175–96.
Alliance FC. Caregiver assessment: principles, guidelines and strategies for change. .1.
Lanoix M. Palliative care and Parkinson’s disease: managing the chronic-palliative interface. Chronic Illn. 2009;5(1):46–55. https://doi.org/10.1177/1742395309102819.
Murdock C, Cousins W, Kernohan WG. “Running water won’t freeze”: how people with advanced Parkinson’s disease experience occupation. Palliative & supportive Care. 2015;13(5):1363–72. https://doi.org/10.1017/S1478951514001357.
Bassuk SS, Glass TA, Berkman LF. Social disengagement and incident cognitive decline in community-dwelling elderly persons. Ann Intern Med. 1999;131(3):165–73.
Holt-Lunstad J, Smith TB, Layton JB. Social relationships and mortality risk: a meta-analytic review. PLoS Med. 2010;7(7):e1000316. https://doi.org/10.1371/journal.pmed.1000316.
Forsaa EB, Larsen JP, Wentzel-Larsen T, Herlofson K, Alves G. Predictors and course of health-related quality of life in Parkinson’s disease. Mov Disord. 2008;23(10):1420–7. https://doi.org/10.1002/mds.22121.
Carter JH, Lyons KS, Lindauer A, Malcom J. Pre-death grief in Parkinson’s caregivers: a pilot survey-based study. Parkinsonism Relat Disord. 2012;18 Suppl 3:S15–8. https://doi.org/10.1016/j.parkreldis.2012.06.015.
Hall K, Sumrall M, Thelen G, Kluger BM. Parkinson’s Disease Foundation sponsored “Palliative C, Parkinson’s Disease” Patient Advisory C. Palliative care for Parkinson’s disease: suggestions from a council of patient and carepartners. NPJ Parkinsons Dis. 2017;3:16. https://doi.org/10.1038/s41531-017-0016-2.
Hall K. Window of opportunity: living with the reality of Parkinson’s and the threat of dementia. Colorado: North Slope Publications; 2014.
Cook DG, Hall KW, Hall LL, Schmidt TA, Sumrall MA, Tuck KK. The rest of the iceberg. Neurology. 2017;89(22):e254–6.
Anandarajah G, Hight E. Spirituality and medical practice: using the HOPE questions as a practical tool for spiritual assessment. Am Fam Physician. 2001;63(1):81–9.
Breitbart W, Rosenfeld B, Pessin H, Kaim M, Funesti-Esch J, Galietta M, et al. Depression, hopelessness, and desire for hastened death in terminally ill patients with cancer. JAMA. 2000;284(22):2907–11.
Fegg MJ, Kogler M, Abright C, Hensler M, Lorenzl S. Meaning in life in patients with progressive supranuclear palsy. Am J Hosp Palliat Care. 2014;31(5):543–7. https://doi.org/10.1177/1049909113492411.
Folkman S, Greer S. Promoting psychological well-being in the face of serious illness: when theory, research and practice inform each other. Psychooncology. 2000;9(1):11–9.
Richardson P. Spirituality, religion and palliative care. Ann Palliat Med. 2014;3(3):150–9. https://doi.org/10.3978/j.issn.2224-5820.2014.07.05.
Borneman T, Ferrell B, Puchalski CM. Evaluation of the FICA tool for spiritual assessment. J Pain Symptom Manage. 2010;40(2):163–73. https://doi.org/10.1016/j.jpainsymman.2009.12.019.
Maugans TA. The SPIRITual history. Arch Fam Med. 1996;5(1):11–6.
Guerrero-Torrelles M, Monforte-Royo C, Rodriguez-Prat A, Porta-Sales J, Balaguer A. Understanding meaning in life interventions in patients with advanced disease: a systematic review and realist synthesis. Palliat Med. 2017;31(9):798–813. https://doi.org/10.1177/0269216316685235.
Chochinov HM, Hack T, Hassard T, Kristjanson LJ, Mc Clement S, Harlos M. Dignity therapy: a novel psychotherapeutic intervention for patients near the end of life. J Clin Oncol. 2005;23(24):5520–5. https://doi.org/10.1200/JCO.2005.08.391.
Carr A. The evidence base for family therapy and systemic interventions for child-focused problems. Journal of Family Therapy. 2014;36(2):107–57.
Gotink RA, Chu P, Busschbach JJ, Benson H, Fricchione GL, Hunink MG. Standardised mindfulness-based interventions in healthcare: an overview of systematic reviews and meta-analyses of RCTs. PLoS One. 2015;10(4):e0124344. https://doi.org/10.1371/journal.pone.0124344.
Kelley AS, Morrison RS. Palliative care for the seriously ill. N Engl J Med. 2015;373(8):747–55. https://doi.org/10.1056/NEJMra1404684.
Tolle SW, Tilden VR, Drach LL, Fromme EK, Perrin NA, Hedberg K. Characteristics and proportion of dying Oregonians who personally consider physician-assisted suicide. J Clin Ethics. 2004;15(2):111–8.
Bernat JL, MP MQ. Physician-assisted death in chronic neurologic diseases. Neurology. 2017;88(16):1488–9. https://doi.org/10.1212/WNL.0000000000003832.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors. All reported studies/experiments with human or animal subjects performed by the authors have been previously published and complied with all applicable ethical standards (including the Helsinki declaration and its amendments, institutional/national research committee standards, and international/national/institutional guidelines).
Additional information
This article is part of the Topical Collection on Movement Disorders
Rights and permissions
About this article
Cite this article
Vaughan, C.L., Kluger, B.M. Palliative Care for Movement Disorders. Curr Treat Options Neurol 20, 2 (2018). https://doi.org/10.1007/s11940-018-0487-4
Published:
DOI: https://doi.org/10.1007/s11940-018-0487-4