Abstract
Purpose of review
There are numerous health benefits to exercise, including improved cardiovascular, metabolic, and mental health. Despite these many benefits, children with cardiomyopathy have been traditionally restricted from engaging in exercise or participating in organized sports. The purpose of this review is to summarize the current literature regarding the utility of cardiopulmonary exercise testing, discuss the safety and benefits of exercise and sports participation in children with cardiomyopathy, and to identify research gaps in the field.
Recent findings
We highlight relevant literature discussing exercise and sports participation in children with hypertrophic cardiomyopathy, dilated cardiomyopathy, anthracycline-induced cardiomyopathy, and those with ventricular assist device support. We discuss recent literature describing methods for risk stratification.
Summary
The current literature globally favors promoting exercise and sports participation after appropriate risk stratification. Additionally, we offer guidance for how to safely promote exercise in children with cardiomyopathy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Opinion statement
Pediatric cardiomyopathies are a broad and heterogeneous group of diseases. Each type of cardiomyopathy presents itself with unique challenges, and generally, there are limited treatment options for children with cardiomyopathy. In this review paper, we globally portend that exercise is safe for most children with cardiomyopathy, and exercise prescriptions remain an underutilized therapy. Given the multiple physical and psychological benefits of exercise, we would highly encourage children with cardiomyopathy to engage in exercise and recreational sports. However, each patient deserves an individualized approach when recommending exercise or allowing for sports participation.
For example, children with hypertrophic cardiomyopathy (HCM) have a unique pathophysiologic substrate, and therefore, an appropriate risk assessment accounts for HCM-specific risk factors, which are outlined in our review paper. In this review, we summarized the cardiovascular and mental health benefits of exercise as well, and we believe that all children with HCM should engage in at least light or moderate exercise, depending on their risk profile. Multi-disciplinary cardiac rehabilitation programs that include cardiologists, exercise physiologists, and psychologists should be the standard of care for children with HCM. In children with dilated cardiomyopathy (DCM), we highlighted that exercise is safe for most, and structured exercise programs may increase the 6-min walk distance. This is an important finding, as the 6-min walk distance is associated with transplant free survival. We also presented literature, demonstrating that exercise is safe in certain patients with DCM. For cancer survivors with anthracycline-induced cardiomyopathy, our review explains that exercise is safe and may in fact improve cancer relapse rates. In general, we encourage exercise and sports participation to improve quality of life and mental health in children with cardiomyopathy. We would take an individualized approach when counseling patients.
Introduction
Despite the well-recognized health benefits associated with physical activity [1,2,3,4,5,6,7,8,9,10,11,12,13,14], the field of pediatric cardiology has traditionally been cautious when recommending exercise to children with cardiomyopathy (CM) due to concern for sudden cardiac death (SCD) and provoking disease progression [15,16,17]. However, recent data among adults has demonstrated that exercise can be safe and offer health benefits for certain patients with CM [18], which in turn has led to a less restrictive approach to exercise and sports participation. While childhood CM has distinct differences, there is an increasing interest in understanding and promoting safe and effective forms of exercise in the pediatric CM population. The purpose of this review is to summarize the current literature regarding the utility of cardiopulmonary exercise testing (CPET), the safety and benefits of exercise and sports participation in children with CM, and to identify research gaps which we hope will prompt future investigation.
Importance of sports participation and exercise
Cardiovascular health
Childhood obesity continues to rise and remains a major issue in America, in part due to increasing sedentary lifestyles. One in three American children are overweight or obese, with associated comorbidities including diabetes mellitus, hypertension, and nonalcoholic fatty liver disease [18, 19]. Children with CM are not spared from this epidemic; in fact, they may be at even a higher risk than the general population, with studies demonstrating decreased exercise capacity and poor cardiovascular risk profiles in children with hypertrophic cardiomyopathy (HCM) [20, 21]. However, participation in aerobic exercise has been shown to reduce the incidence and severity of diabetes, obesity, and hypertension, many co-morbidities that affect long term cardiac health [22]. Therefore, in children with non-modifiable cardiovascular risk factors, optimizing their cardiovascular risk profile through physical activity may be an important way to improve outcomes.
Mental health
Quality of life (QOL) is higher in those who exercise, and participation in organized sports improves psychological well-being in children. Obese children who enrolled in an after-school exercise program showed improvement in QOL and depression [8], and for patients with depression, exercise prescriptions can improve symptoms [6, 11]. This is particularly relevant for children with CM, who have impaired QOL in comparison to peers, and high rates of anxiety and depression [14, 23]. Although certain patients with CM are at an increased risk of SCD, it is worth noting that other mental health issues such as suicide account for 11% of deaths in youth aged 15–24 [24, 25] emphasizing the importance of identifying interventions which positively impact mental health and to avoid undue activity restriction in all children.
Exercise and sports participation by form of cardiomyopathy
Hypertrophic cardiomyopathy
HCM is the second most common form of pediatric CM, with an estimated incidence of 1 in 500 [26,27,28]. HCM is defined by a left ventricular (LV) wall thickness > 15 mm, or a septal thickness Z-score > 2 without any pressure or volume-loading components [29]. Pathologic examination of the myocardium in HCM reveals myocardial disarray, often due to genetic mutations of sarcomere formation, RASopathies or neurodegenerative disorders, usually inherited in an autosomal dominant fashion [30, 31]. Patients with HCM are at an increased risk for SCD compared to the general population and may have physiologic limitations to exercise performance [25, 32, 33]. As a result, a thoughtful approach to these patients and sports participation is crucial.
Current guidelines regarding exercise
Historically, exercise has been considered unsafe for patients with HCM, due to the perception that physical activity increased the risk of SCD. In 2009, Maron et al. described HCM as the most common cause of SCD in athletes [34]. However, more recent studies have challenged this finding with Finocchiaro et al. showing that among a cohort including 3,684 cases, HCM constituted only 6% of SCD [35], and Weissler-Snir et al. reporting more deaths among patients with HCM occurring during sleep or at rest (30% and 34%, respectively) than during activity (17% during moderate/vigorous exercise and 17% during light exercise) [32]. It was in this developing context that Saberi et al. performed a trial of moderate intensity exercise in patients with HCM and ultimately demonstrated that this type of exercise training was safe and improved peak oxygen consumption (VO2) in patients with HCM [36].
These studies have contributed to a shift in the approach of exercise counseling for patients with HCM. In 2020, the American Heart Association (AHA)/American College of Cardiology (ACC) published an updated set of guidelines which introduced new class I recommendations regarding the safety of physical activity, stating that “for most patients with HCM, mild-to-moderate intensity recreational exercise is beneficial to improve cardiorespiratory fitness, physical functioning, and quality of life, and for their overall health in keeping with physical activity guidelines for the general population” [29]. Although exercise guidelines were limited to mild-to-moderate intensity, more recent data demonstrated no difference in SCD risk between those with HCM who did vigorous exercise versus moderate exercise or were sedentary [37•]. Another class I recommendation states “for athletes with HCM, a comprehensive evaluation and shared discussion of potential risks of sports participation by an expert provider is recommended.” These guidelines also suggest that participation in low-intensity competitive sports for patients with HCM is reasonable (class IIa). Yet, despite these revised recommendations, most adults with HCM remain less active than unaffected individuals [38].
Exercise for children with hypertrophic cardiomyopathy
Studies of CPET in children with HCM reveal reduced peak VO2, low peak physical working capacity, O2 pulse, peak heart rate (HR), and high rates of abnormal Ve/VCO2 slopes [21, 39, 40]. The reasons for abnormal exercise performance and reduced levels of physical activity levels [41•] are likely multi-factorial, including physiologic limitations, patient/parent concerns, and provider influence. While studies in adults with HCM demonstrate low levels of physical activity [38], this has not been thoroughly studied in children with HCM.
Physiologic limitations of exercise performance
Certain phenotypes of HCM are characterized by significant LV hypertrophy and LV outflow tract (LVOT) obstruction which reduces the LV cavity, and in turn, the stroke volume [42], which is an important determinant of exercise performance [43, 44]. Diastolic function likely also plays a role in exercise performance in children with HCM, with echocardiographic markers of LV diastolic dysfunction by left atrial (LA) size and LA index shown to correlate with peak VO2 on CPET [45, 46]. Chronotropic incompetence (either intrinsic or in the setting of beta blockade) has also been observed in patients with HCM, which further reduces peak exercise capacity [39, 47]. Finally, physiologic or anatomic limitations in exercise performance contribute to physical deconditioning, muscle weakness, and restriction from physical activity which perpetuates abnormal exercise performance.
Patient perceptions of physical limitations
Patients with HCM face many psychological issues related to exercise, including fear of SCD, and peer pressure to engage with activity beyond their abilities. A study among teenagers with HCM utilizing qualitative methodology to identify perceptions regarding exercise [48•] identified several factors which influence decisions to be active including: self‐awareness, ownership and autonomy, feeling restricted, support from parents, and support from their cardiologist. This work highlights the barriers and motivators to activity which likely differ for children with HCM compared to healthy peers and emphasizes the necessity of participatory study design in developing effective activity related interventions.
Provider influence on exercise and sports participation
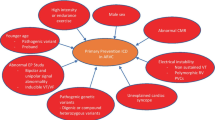
Providers who care for children with HCM play a critical role in exercise and sports counseling. Teenagers with HCM report entrusting their cardiologist for guidance with physical activity [48•]; however, there is wide variability in how exercise counseling is performed. This is likely due to a deficiency of pediatric literature, shifting guidelines, provider discomfort or lack of knowledge in safe counseling, and fear of litigation. A study published in 2022 of pediatric heart failure and electrophysiology specialists revealed differences in counseling patterns with heart failure specialist being more likely to restrict exercise than electrophysiologists [16]. The heterogeneity in guidance demonstrates a need to develop a more unified and evidence-based approach to activity in these patients. In Fig. 1, we provide a brief list of findings that may warrant caution with competitive sports participation.
There are numerous health benefits of exercise. For patients with cardiomyopathy, there are various factors that increase the risk of sudden cardiac death, and a risk–benefit discussion should occur on a case-by-case basis. (VO2, oxygen consumption; SCD, sudden cardiac death; BP, blood pressure; LGE, late gadolinium enhancement; cMRI, cardiac magnetic resonance imaging; CPET, cardiopulmonary exercise testing. Ventricular assist device icon adapted from Mehra et al. [97]). Image created with BioRender.com.
Risk stratification
Patients with HCM are at risk for SCD given their unique physiologic substrate. Prior to routine exercise or organized sports participation, a complete risk assessment should take place. There are well-established risk factors for SCD in adult patients with HCM [29]. By contrast, children with HCM have a different risk profile for SCD [49], and applying adult risk models may not be appropriate to estimate SCD in children [50]. Recently, the primacy and HCM risk-kids model have been developed to specifically measure pediatric risk [50, 51]. While neither of these models include data from CPET in their algorithms, this may be due to a lack of consistent practice, and we contend that incorporating exercise related data, along with personal and family history, and imaging data will allow for a more accurate understanding of patient specific risk.
The initial risk stratification should include a personal and family history of syncope or SCD, echocardiogram, and cardiac magnetic resonance imaging (MRI) with contrast to detect late gadolinium enhancement (LGE). With echocardiographic evaluation, it is important to measure LA size, LV function, LV and mitral valve morphology, an LVOT gradient, and coronary artery anatomy. CPET with consideration of stress echocardiography to measure an inducible LVOT gradient is recommended as well [29], being mindful that outflow tract obstruction can also occur in athletes or otherwise healthy individuals [52]. Various studies in children have demonstrated prognostic utility with CPET. In a single center retrospective cohort study, ectopy (atrial or ventricular) during surveillance CPET was associated with cardiac death, cardiac transplantation, or the need for an implantable cardiac defibrillator (ICD) [21]. In a large multi-center study of 724 children with HCM, an abnormal CPET, defined as: an abnormal blood pressure (BP) response (inability to augment systolic BP by 20 mmHg or a drop in systolic BP with exercise), ischemia, or complex ventricular ectopy (frequent single PVCs, or consecutive PVCs), was associated with a lower 5-year transplant-free survival. A subgroup analysis in this study revealed that ischemia during CPET was most predictive of 5-year transplant-free survival in this cohort. Other studies have shown that an abnormal BP response was associated with a higher risk of all-cause mortality when compared to those with normal CPET [41•], though it is worth remembering that accurately measuring BPs during exercise can be challenging. Metabolic measurements have also been shown to associate with clinical outcomes. A multi-center analysis performed in Italian hospitals including 66 children with HCM found an association with reduced percent predicted peak VO2 (< 60% of predicted) and circulatory power (defined as peak VO2 in mL/kg/min x systolic BP) with the composite outcome including: SCD (SCD events, aborted SCD, ICD shock, or sustained ventricular tachycardia), death from heart failure, cardiac transplantation, progression to NYHA class III–IV caused by end-stage disease with or without LV ejection fraction < 50%, hospitalization due to heart failure, and septal reduction therapy for heart failure symptoms [40].
With the increased use of genetic testing, questions have arisen as to how to best counsel patients who test positive for pathologic genes associated with HCM, without manifesting any clinical findings, a group known as “genotype-positive/phenotype-negative.” While patients who are genotype-positive are at risk for disease progression, exercise should still be promoted. In a study of 187 adults, split into groups of HCM-patients and genotype-positive/phenotype-negative, vigorous exercise was not associated with an increased incidence of ventricular arrhythmias for those in the genotype-positive/phenotype-negative group [53]. Furthermore, in a retrospective analysis of pediatric patients with genotype-positive HCM, Burstein et al. revealed no association of genetic mutations with adverse cardiac events [54]. These studies overall suggest that genotype-positivity in isolation is not an indication to restrict from exercise. In general, patients who are genotype-positive, regardless of phenotype, should be monitored for clinical progression. At this current time, there is little evidence to suggest that pediatric patients who are genotype-positive/phenotype-negative are at an elevated risk of sudden cardiac events with exercise, although additional caution should be taken in those with multiple pathogenic genes [55].
Implantable cardiac defibrillators
Some patients with HCM have an ICD due to an elevated risk of SCD or a prior event. Evidence reveals that patients with an ICD can safely participate in exercise [56]; however, data from an adult study suggests that patients with HCM who receive ICD interventions have increased anxiety with exercise compared to HCM patients without ICD events [57]; therefore, anxiety symptoms should be monitored closely. Furthermore, according to the 2020 AHA/ACC guidelines in managing HCM, it is considered a class III recommendation (more harm than benefit) to place an ICD purely for sports participation in a patient who does not otherwise meet the traditional guidelines for ICD placement.
Shared decision making in a pediatric setting
The AHA/ACC guidelines for managing HCM recommend engaging in shared decision-making regarding exercise and sports participation. This includes an agreement between three main parties, (1) the patient, (2) parents, and (3) the coach, trainers, and/or schoolteachers. An understanding of the patient’s developmental status and ability to appreciate risk should be considered. Parents may share a different level of concern around risk than their children. Young athletes with HCM may perceive the benefits of participating in their sport to far outweigh the risks of their disease; therefore, it is also important to understand the patient’s values and to identify any competing factors or conflicts of interest (such as scholarships). Coaches, trainers, and teachers should also be engaged in the conversation, with a clear understanding of the athlete’s limits. The latter group should also ensure that an automated external defibrillator (AED) is accessible at their facility and should be trained in performing cardiopulmonary resuscitation (CPR) [58, 59].
Cardiac rehabilitation
Recent data has repeatedly demonstrated the safety and efficacy of cardiac rehabilitation programs in patients with HCM [36, 60, 61], with structured programs resulting in improved peak VO2. In experienced pediatric centers, cardiac rehabilitation should remain a strong consideration for improving the health of children with HCM and for promoting long term activity habits. While there is currently wide variability in the use of cardiac rehab in children with HCM, there are initiatives to standardize these practices. Further research is necessary to clearly delineate the safety, feasibility, and implementation strategies of cardiac rehabilitation in pediatric patients with HCM.
Summary
Patients with HCM are at an elevated risk of SCD; however, the degree to which activity increases this risk is unclear and likely patient specific. Therefore, appropriate risk stratification and shared decision making should be engaged when counseling on exercise or sports participation. CPET with stress echocardiography as appropriate, ambulatory rhythm monitoring and cardiac MRI are helpful tools for risk stratification, along with validated tools such as the primacy calculator. Overall, the physical and mental health benefits of physical activity outweigh the risk of SCD in many pediatric patients with HCM, and safe and effective forms of activity should be found for children with HCM whenever possible.
Dilated cardiomyopathy
Dilated cardiomyopathy (DCM) is defined as a reduction in ventricular function with ventricular dilation, without a volume or pressure loading component. Inherited forms of DCM include sarcomeric mutations, muscular dystrophy, or mitochondrial disease, and acquired forms include myocarditis, toxin exposure, and inflammatory diseases. DCM is associated with malignant arrhythmias, sudden death, and heart failure [30, 62, 63]. There is currently no curative therapy for DCM; therefore, patients are managed pharmacologically with guideline-directed medical therapy, mechanical support, or by cardiac transplantation [30].
Current knowledge of exercise and sports participation
Patients with DCM have a low peak VO2 and an elevated Ve/VCO2 slope [64,65,66]. The mechanism for low peak VO2 is likely multifactorial, including LV dysfunction, low chronotropic reserve, low stroke volume reserve, and physical deconditioning. The Ve/VCO2 slope has been shown to correlate with capillary wedge pressure [67] and is likely elevated in the setting of LV dilation with elevated LV diastolic pressures. There is prognostic utility of CPET in children with DCM as well. In a single center, retrospective study by Chen et al., the peak VO2, percent predicted peak VO2, peak workload, VO2 at anaerobic threshold, and Ve/VCO2 slope all correlated with the composite outcome (death before or after listing for heart transplant, hospitalization for worsening heart failure needing intravenous inotropic support, mechanical ventilation, or mechanical circulatory support) [66]. Additionally, in a single center, retrospective study by Giardini et al., the percent predicted peak VO2, Ve/VCO2 slope, and peak systolic BP associated with death without heart transplantation, or clinical deterioration requiring urgent listing for heart transplantation [65]. While further research is needed to delineate the mechanisms for these associations, there appears to be good prognostic value with CPET for children with DCM.
With regards to sports participation, in 2019, the European Society for Cardiology (ESC) published guidance for counseling individuals with DCM [68]. In brief, those with DCM who have reduced LVEF (EF < 40%), and/or frequent/complex ventricular tachyarrhythmias on ambulatory rhythm monitoring and exercise testing, or a history of unexplained syncope should be limited from competitive sports but can safely engage in leisurely activities. Participating in competitive sports for these patients is considered a class III recommendation (more harm than benefit). Furthermore, patients with Lamin A/C or Filamin C gene mutations are at high risk for life-threatening arrhythmias and, therefore, should not engage in competitive sports. There is limited published data on exercise and sports participation for children with DCM. In a notable pediatric study, Wittekind et al. retrospectively analyzed children with DCM who underwent cardiac rehabilitation at a single institution, revealing overall safety with exercise, and modest improvements in 6-min walk distance (6MWD) accompanied by a slight reduction in waist circumference [69]. This is an important finding, as the 6MWD is correlated with transplant free survival in children with DCM [70]. Furthermore, McBride et al. demonstrated that patients with DCM who are admitted to the hospital while awaiting heart transplantation were able to participate in cardiac rehabilitation, including those on continuous inotropic infusions, without any adverse events [71]. Overall, the limited evidence among children with DCM suggests that exercise and competitive sports are safe for certain patients and provide cardiovascular benefit (Fig. 1); however, further studies are needed, including prospective analyses of cardiac rehabilitation programs.
Anthracycline-induced cardiomyopathy
Patients who undergo chemotherapy are at risk for many cardiovascular diseases, including CM from anthracycline chemotherapy agents [72]. Anthracyclines induce cellular apoptosis, which can result in myocardial growth arrest, with resultant ventricular restrictive physiology or DCM with ventricular dysfunction [73, 74]. Additionally, patients who undergo chemotherapy are at risk for hypertension and insulin resistance [75]; therefore, engaging in physical activity and exercise is very important. Furthermore, higher levels of physical activity are linked to better rates of cancer remission [76, 77], making exercise a disease-modifying intervention that should be strongly encouraged in all cancer survivors.
Current knowledge of exercise capacity and barriers to exercise
Survivors of childhood cancer exposed to anthracyclines are known to have reduced exercise capacity [78,79,80,81]. The mechanism of this is unclear, although some data suggests that a small LV with reduced stroke volume may play a role [81, 82] along with chronic illness, physical deconditioning, and sarcopenia [83]. Despite the traditional concern for safety with exercise, various studies in cancer survivors reveal that exercise is safe and can improve cardiovascular health [84, 85]. In 2019, the American College of Sports medicine published a consensus statement endorsing routine exercise in cancer-survivors given its many health benefits and provided an abundance of safety data [86]. Additionally in 2019, the AHA published a statement regarding the utility of a multi-disciplinary cardio-oncology rehabilitation (CORE) service, citing many studies confirming cardiovascular and mental health benefits of exercise in cancer survivors, including those with chemotherapy-related CM [87]. Furthermore, there are many mental health and psychosocial challenges faced by cancer survivors, including difficulty with interpersonal relationships, poor self-esteem, depression, post-traumatic stress disorder, poor body image, mood swings, substance abuse, and suicidal ideation [88]. Exercise can provide patients with a sense of control, autonomy, and can boost self-esteem, and therefore is a highly advantageous in this vulnerable population. Further studies regarding the feasibility of exercise programs for pediatric cancer survivors are needed.
Patients with a ventricular assist device
Patients with advanced heart failure due to CM may require ventricular assist device (VAD) support while awaiting cardiac transplantation, and this population is rapidly growing [89]. While VAD use can improve symptoms and QOL in pediatric patients with heart failure [90], there is less pediatric literature about the safety of exercise among patients with VADs. However, recent data has demonstrated safe exercise practices and increased 6MWD with VAD support [91]. In fact, a study from the Children’s Hospital of Philadelphia revealed that exercise and rehabilitation are safe in children on VAD support [92]. Pre-exercise titrations to VAD output can be done safely to improve cardiac output with exercise [93]. Given the increasing number of pediatric patients being supported with VADs prior to cardiac transplant [89, 94] and the importance of minimizing frailty prior to transplant [95], developing standardized and effective cardiac rehabilitation programs for pediatric VAD patients should be a major goal of CM care.
General guidance for exercise and sports participation
For children with CM, there is a broad range of functional capacities and a spectrum of what defines exercise and sports participation. For some, the ability to perform activities of daily living may be the maximum of their capacity, while for others, competitive sports may be the goal. The ESC has developed a useful gradation system for various types of exercise, grading them on a scale of dynamic and strength domains [96].
We propose the following framework for exercise in children with CM, summarized in Fig. 2. First, a safe and effective form of exercise can and should be identified in all children with CM. This may require creativity in determining safe types of exercise, and consultation with a sports cardiologist or exercise physiologist can be helpful. To safely prescribe exercise, a general risk assessment should be performed, which may include CPET, advanced imaging such as cardiac MRI, and an ambulatory rhythm monitor. Exercise goals should be identified, and the intensity of exercise should match the patient’s risk profile and disease severity. Parents and patients should be counseled on safe exercise practices. In general, patients should try to exercise in a group setting or with a partner, always maintain adequate hydration, slowly increase their intensity toward a goal level, and perform stretches prior to, and after activity. For schools and coaches, CPR training is preferred, along with access to, and training in the use of an AED. Athletes with CM should be given free range to take breaks if symptoms occur, or for water breaks as needed. At each clinic visit, concerning symptoms such as chest pain, shortness of breath, palpitations, or syncope should be reviewed, and counseling toward self-awareness of one’s symptoms is essential.
In conclusion, we believe that a safe form of exercise can and should be pursued in all children with CM. There is no “one size fits all” approach when counseling children with CM toward sports and exercise, as there is a large range in disease risk and sports competitiveness. Participation in cardiac rehabilitation, leisurely exercise, and competitive sports can provide numerous physical and psychological benefits and can improve the lives of children with CM in general. Image created with BioRender.com.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance
Li VL, He Y, Contrepois K, Liu H, Kim JT, Wiggenhorn AL, et al. An exercise-inducible metabolite that suppresses feeding and obesity. Nature. 2022;606(7915):785–90.
Eriksson KF, Lindgarde F. Prevention of type 2 (non-insulin-dependent) diabetes mellitus by diet and physical exercise. The 6-year Malmö feasibility study. Diabetologia. 1991;34(12):891–8.
Baumann FT, Bloch W, Beulertz J. Clinical exercise interventions in pediatric oncology: A systematic review. Vol. 74, Pediatr Res. 2013;366–74.
Thijssen DHJ, Redington A, George KP, Hopman MTE, Jones H. Association of exercise preconditioning with immediate cardioprotection: A review. Vol. 3, JAMA Cardiology. Am Med Assoc. 2018;169–76.
Booth FW, Roberts CK, Laye MJ. Lack of exercise is a major cause of chronic diseases. Compr Physiol. 2012;2(2):1143–211.
Kvam S, Kleppe CL, Nordhus IH, Hovland A. Exercise as a treatment for depression: A meta-analysis. Vol. 202, Journal of Affective Disorders. Elsevier B.V.; 2016. p. 67–86.
Penedo FJ, Dahn JR, Williams L. Exercise and well-being: A review of mental and physical health benefits associated with physical activity. Vol. 18, Curr Opin Psychiatry. 2005.
Williams CF, Bustamante EE, Waller JL, Davis CL. Exercise effects on quality of life, mood, and self-worth in overweight children: The SMART randomized controlled trial. Transl Behav Med. 2019;9(3):451–9.
Kemppainen J, Stolen K, Kalliokoski KK, Salo T, Karanko H, Viljanen T, et al. Exercise training improves insulin stimulated skeletal muscle glucose uptake independent of changes in perfusion in patients with dilated cardiomyopathy. J Card Fail. 2003;9(4):286–95.
Liang X, Tang J, Chao F lei, Zhang Y, Chen L mu, Wang F fei, et al. Exercise improves depressive symptoms by increasing the number of excitatory synapses in the hippocampus of CUS-induced depression model rats. Behav Brain Res. 2019;18:374.
Schuch FB, Vancampfort D, Rosenbaum S, Richards J, Ward PB, Stubbs B. Exercise improves physical and psychological quality of life in people with depression: A meta-analysis including the evaluation of control group response. Vol. 241, Psychiatry Research. Elsevier Irel Ltd. 2016;47–54.
McCaskie C, Siafarikas A, Cochrane Wilkie J, Sutton V, Chivers P, Hart NH, et al. The benefits to bone health in children and pre-school children with additional exercise interventions: A systematic review and meta-analysis. Vol. 15, Nutrients MDPI. 2023.
Santos-Parker JR, LaRocca TJ, Seals DR. Aerobic exercise and other healthy lifestyle factors that influence vascular aging. Adv Physiol Educ. 2014;38(4):296–307.
Christian S, Somerville M, Taylor S, Spence JC, Giuffre M, Atallah J. The impact of physical activity modification on the well-being of a cohort of children with an inherited arrhythmia or cardiomyopathy. Cardiol Young. 2020;30(5):692–7.
Khoury M, Wittekind S, Lal AK, Conway J, Bansal N, Lorts A, et al. Significant variation in exercise recommendations for youth with cardiomyopathies or Fontan circulation: An advanced cardiac therapies improving outcomes network learning survey. Circ Heart Fail. 2021.
Whitehill RD, Balaji S, Kelleman M, Chandler SF, Abrams DJ, Mao C, et al. Exercise recommendations in pediatric HCM: Variation and influence of provider characteristics. Pediatr Cardiol. 2022;43(1):132–41.
Burstein DS, McBride M, Lorts A, Rosenthal D, Peng DM, Lantz J, et al. Variation in cardiac rehabilitation for pediatric ventricular assist device recipients across North America. ASAIO J. 2021;67(9):1045–50.
Kumar S, Kelly AS. Review of childhood obesity: From epidemiology, etiology, and comorbidities to clinical assessment and treatment. Vol. 92, Mayo Clinic Proceedings. Elsevier Ltd. 2017;251–65.
Ogden CL, Carroll MD, Fryar CD, Flegal KM. Key findings data from the national health and nutrition examination survey. 2011.
Conway J, Pidborochynski T, Khoury M, Dhillon SS, Vaujois L, Dallaire F, et al. Cardiovascular disease profile in children with HCM (aspire-HCM): A Canadian cross-sectional study. Circulation. 2022.
Edelson JB, Stanley HM, Min J, Burstein DS, Lane-Fall M, O’Malley S, et al. Cardiopulmonary exercise testing in pediatric patients with hypertrophic cardiomyopathy. JACC: Advances. 2022;1(4):100107.
Hebert JJ, Klakk H, Møller NC, Grøntved A, Andersen LB, Wedderkopp N. The prospective association of organized sports participation with cardiovascular disease risk in children (the CHAMPS study-DK). Mayo Clin Proc. 2017;92(1):57–65.
Spanaki A, O’Curry S, Winter-Beatty J, Mead-Regan S, Hawkins K, English J, et al. Psychosocial adjustment and quality of life in children undergoing screening in a specialist paediatric hypertrophic cardiomyopathy clinic. Cardiol Young. 2016;26(5):961–7.
Heron M. National vital statistics reports, volume 62, Number 6, December 20, 2013 [Internet]. 2010. Available from: http://www.cdc.gov/nchs/nvss/mortality_tables.htm.
Ullal AJ, Abdelfattah RS, Ashley EA, Froelicher VF. Hypertrophic cardiomyopathy as a cause of sudden cardiac death in the young: A meta-analysis. Am J Med. 2016;129(5):486-496.e2.
Lipshultz SE, Orav EJ, Wilkinson JD, Towbin JA, Messere JE, Lowe AM, et al. Risk stratification at diagnosis for children with hypertrophic cardiomyopathy: An analysis of data from the Pediatric Cardiomyopathy Registry. The Lancet. 2013;382(9908):1889–97.
Maron BJ, Gardin JM, Flack JM, Gidding SS, Kurosaki TT, Bild DE. Prevalence of hypertrophic cardiomyopathy in a general population of young adults . Circulation. 1995.
Husser D, Ueberham L, Jacob J, Heuer D, Riedel-Heller S, Walker J, et al. Prevalence of clinically apparent hypertrophic cardiomyopathy in Germany—An analysis of over 5 million patients. PLoS One. 2018;13(5).
Ommen SR, Mital S, Burke MA, Day SM, Deswal A, Elliott P, et al. Diagnosis and management of hypertrophic cardiomyopathy: Updated guidelines from the ACC/AHA. Am Fam Physician. 2022;105(2):207–9.
Lee TM, Hsu DT, Kantor P, Towbin JA, Ware SM, Colan SD, et al. Pediatric cardiomyopathies. Circ Res. 2017;121(7):855–73.
Shah M. Hypertrophic cardiomyopathy. Vol. 27, Cardiology in the young. Cambridge University Press. 2017;S25–30.
Weissler-Snir A, Allan K, Cunningham K, Connelly KA, Lee DS, Spears DA, et al. Hypertrophic cardiomyopathy-related sudden cardiac death in young people in Ontario. Circulation. 2019;140(21):1706–16.
Hwang J won, Lee SC, Kim D, Kim J, Kim EK, Chang SA, et al. Determinants of exercise capacity in patients with hypertrophic cardiomyopathy. J Korean Med Sci. 2022;37(8).
Maron BJ, Doerer JJ, Haas TS, Tierney DM, Mueller FO. Sudden deaths in young competitive athletes analysis of 1866 deaths in the united states, 1980–2006. Circulation. 2009;119(8):1085–92.
Finocchiaro G, Papadakis M, Robertus JL, Dhutia H, Steriotis AK, Tome M, et al. Etiology of sudden death in sports insights from a United Kingdom Regional Registry. 2016.
Saberi S, Wheeler M, Bragg-Gresham J, Hornsby W, Agarwal PP, Attili A, et al. Effect of moderate-intensity exercise training on peak oxygen consumption in patients with hypertrophic cardiomyopathy: A randomized clinical trial. JAMA - Journal of the American Medical Association. 2017;317(13):1349–57.
• Lampert R, Ackerman MJ, Marino BS, Burg M, Ainsworth B, Salberg L, et al. Vigorous exercise in patients with hypertrophic cardiomyopathy. JAMA Cardiol [Internet]. 2023. Available from: https://jamanetwork.com/journals/jamacardiology/fullarticle/2805064. This paper highlights that vigorous exercise is safe in patients with HCM, contrary to previous literature.
Reineck E, Rolston B, Bragg-Gresham JL, Salberg L, Baty L, Kumar S, et al. Physical activity and other health behaviors in adults with hypertrophic cardiomyopathy. Am J Cardiol. 2013;111(7):1034–9.
Przybylski R, Fischer IR, Gauvreau K, Alexander ME, Shafer KM, Colan SD, et al. Assessment of exercise function in children and young adults with hypertrophic cardiomyopathy and correlation with transthoracic echocardiographic parameters. Pediatr Cardiol. 2022;43(5):1037–45.
Gallo G, Mastromarino V, Limongelli G, Calcagni G, Ragno L, Valente F, et al. Insights from cardiopulmonary exercise testing in pediatric patients with hypertrophic cardiomyopathy [Internet]. Available from: https://academic.oup.com/eurheartj/article/42/Supplement_1/ehab724.1768/6391822.
• Conway J, Min S, Villa C, Weintraub RG, Nakano S, Godown J, et al. The prevalence and association of exercise test abnormalities with sudden cardiac death and transplant-free survival in childhood hypertrophic cardiomyopathy. Circulation. 2022. This is a large multi-center study, that provided new insights, into the prognostic utility of CPET, specifically for children with HCM.
Yasui K, Shibata T, Nishizawa T, Yokoyama U, Takigiku K, Sakon T, et al. Response of the stroke volume and blood pressure of young patients with nonobstructive hypertrophic cardiomyopathy to exercise. Jpn Circ J. 2001;65:300–4.
Rowland T. Cardiopulmonary exercise testing in children and adolescents. Champaign, IL: Human Kinetics; 2017. p. 44–44.
MacNamara JP, Dias KA, Hearon CM, Hieda M, Turer AT, Link MS, et al. Limits to submaximal and maximal exercise in patients with hypertrophic cardiomyopathy. J Appl Physiol. 2022;133(4):787–97.
Jhaveri S, Komarlu R, Worley S, Shahbah D, Gurumoorthi M, Zahka K. Left atrial strain and function in pediatric hypertrophic cardiomyopathy. J Am Soc Echocardiogr. 2021;34(9):996–1006.
Kjaergaard J, Johnson BD, Pellikka PA, Cha SS, Oh JK, Ommen SR. Left atrial index is a predictor of exercise capacity in patients with hypertrophic cardiomyopathy. J Am Soc Echocardiogr. 2005;18(12):1373–80.
Magrì D, Santolamazza C. Cardiopulmonary exercise test in hypertrophic cardiomyopathy. In: Annals of the American Thoracic Society. Am Thoracic Soc. 2017. p. S102–9.
• Sobierajski F, Storey K, Bird M, Anthony S, Pol S, Pidborochynski T, et al. Use of photovoice to explore pediatric patients with hypertrophic cardiomyopathy and their parents’ perceptions of a heart-healthy lifestyle. J Am Heart Assoc. 2022;11(7). This study provides new information of the specific psychological factors that teenagers with HCM face regarding exercise and sports participation.
Balaji S, DiLorenzo MP, Fish FA, Etheridge SP, Aziz PF, Russell MW, et al. Risk factors for lethal arrhythmic events in children and adolescents with hypertrophic cardiomyopathy and an implantable defibrillator: An international multicenter study. Heart Rhythm. 2019;16(10):1462–7.
Kaski JP, Norrish G, Ding T, Field E, Ziółkowska L, Olivotto I, et al. Development of a novel risk prediction model for sudden cardiac death in childhood hypertrophic cardiomyopathy (HCM risk-kids). JAMA Cardiol. 2019;4(9):918–27.
Miron A, Lafreniere-Roula M, Steve Fan CP, Armstrong KR, Dragulescu A, Papaz T, et al. A validated model for sudden cardiac death risk prediction in pediatric hypertrophic cardiomyopathy. Circulation. 2020;142(3):217–29.
Wittlieb-Weber CA, Cohen MS, McBride MG, Paridon SM, Morrow R, Wasserman M, et al. Elevated left ventricular outflow tract velocities on exercise stress echocardiography may be a normal physiologic response in healthy youth. J Am Soc Echocardiogr. 2013;26(12):1372–8.
Dejgaard LA, Haland TF, Lie OH, Ribe M, Bjune T, Leren IS, et al. Vigorous exercise in patients with hypertrophic cardiomyopathy. Int J Cardiol. 2018;1(250):157–63.
Burstein DS, Gaynor JW, Griffis H, Ritter A, Connor MJO, Rossano JW, et al. Genetic variant burden and adverse outcomes in pediatric cardiomyopathy. Pediatr Res. 2021;89(6):1470–6.
Pollmann K, Kaltenecker E, Schleihauf J, Ewert P, Görlach A, Wolf CM. Compound mutation in cardiac sarcomere proteins is associated with increased risk for major arrhythmic events in pediatric onset hypertrophic cardiomyopathy. J Clin Med. 2021;10(22).
Lampert R, Olshansky B, Heidbuchel H, Lawless C, Saarel E, Ackerman M, et al. Safety of sports for athletes with implantable cardioverter-defibrillators: Results of a prospective, multinational registry. Circulation. 2013;127(20):2021–30.
Maron BJ, Casey SA, Olivotto I, Sherrid M V., Semsarian C, Autore C, et al. Clinical course and quality of life in high-risk patients with hypertrophic cardiomyopathy and implantable cardioverter-defibrillators. Circ Arrhythm Electrophysiol. 2018;11(4).
de Feria AE, Lin KY, Day SM. Applying shared decision making to sports participation for a patient with hypertrophic cardiomyopathy. JACC Case Rep. 2021;3(1):6–9.
Etheridge SP, Saarel EV, Martinez MW. Exercise participation and shared decision-making in patients with inherited channelopathies and cardiomyopathies. Heart Rhythm. 2018;15(6):915–20.
Wasserstrum Y, Barbarova I, Lotan D, Kuperstein R, Shechter M, Freimark D, et al. Efficacy and safety of exercise rehabilitation in patients with hypertrophic cardiomyopathy. J Cardiol. 2019;74(5):466–72.
McBride MG, Burstein DS, Edelson JB, Paridon SM. Cardiopulmonary rehabilitation in pediatric patients with congenital and acquired heart disease. Vol. 40, Journal of Cardiopulmonary Rehabilitation and Prevention. Lippincott Williams and Wilkins; 2020;370–7.
Schultheiss HP, Fairweather DL, Caforio ALP, Escher F, Hershberger RE, Lipshultz SE, et al. Dilated cardiomyopathy. Nat Rev Dis Primers. 2019;5(1).
Lynn J, Eries J, Rey J, Towbin A. Dilated cardiomyopathy [Internet]. Vol. 375, www/thelancet/com. 2010. Available from: https://www.thelancet.com.
Sinagra G, Iorio A, Merlo M, Cannatà A, Stolfo D, Zambon E, et al. Prognostic value of cardiopulmonary exercise testing in idiopathic dilated cardiomyopathy. Int J Cardiol. 2016;15(223):596–603.
Giardini A, Fenton M, Andrews RE, Derrick G, Burch M. Peak oxygen uptake correlates with survival without clinical deterioration in ambulatory children with dilated cardiomyopathy. Circulation. 2011;124(16):1713–8.
Chen CK, Manlhiot C, Russell JL, Kantor PF, McCrindle BW, Conway J. The utility of cardiopulmonary exercise testing for the prediction of outcomes in ambulatory children with dilated cardiomyopathy. Transplantation. 2017;101(10):2455–60.
Klaassen SHC, Liu LCY, Hummel YM, Damman K, van der Meer P, Voors AA, et al. Clinical and hemodynamic correlates and prognostic value of VE/VCO2 slope in patients with heart failure with preserved ejection fraction and pulmonary hypertension. J Card Fail. 2017;23(11):777–82.
Pelliccia A, Solberg EE, Papadakis M, Adami PE, Biffi A, Caselli S, et al. Recommendations for participation in competitive and leisure time sport in athletes with cardiomyopathies, myocarditis, and pericarditis: Position statement of the sport cardiology section of the European Association of Preventive Cardiology (EAPC). Eur Heart J. 2019;40(1):19–33.
Wittekind SG, Gerdes Y, Mays W, Chin C, Jefferies JL. Cardiac rehabilitation improves cardiometabolic health: In young patients with nonischemic dilated cardiomyopathy. Tex Heart Inst J. 2018;45(1):27–30.
den Boer SL, Flipse DHK, van der Meulen MH, Backx APCM, du Marchie Sarvaas GJ, Ten Harkel ADJ, et al. Six-minute walk test as a predictor for outcome in children with dilated cardiomyopathy and chronic stable heart failure. Pediatr Cardiol. 2017;38(3):465–71.
Mcbride MG, Binder TJ, Paridon SM. Safety and feasibility of inpatient exercise training in pediatric heart failure: A preliminary report [Internet]. Available from: https://journals.lww.com/jcrjournal/pages/default.aspx.
Mulrooney DA, Yeazel MW, Kawashima T, Mertens AC, Mitby P, Stovall M, et al. Cardiac outcomes in a cohort of adult survivors of childhood and adolescent cancer: Retrospective analysis of the childhood cancer survivor study cohort. BMJ (Online). 2009;339(7736):34.
Jefferies JL, Mazur WM, Howell CR, Plana JC, Ness KK, Li Z, et al. Cardiac remodeling after anthracycline and radiotherapy exposure in adult survivors of childhood cancer: A report from the St Jude Lifetime Cohort Study. Cancer. 2021;127(24):4646–55.
Jordan JH, Castellino SM, Meléndez GC, Klepin HD, Ellis LR, Lamar Z, et al. Left ventricular mass change after anthracycline chemotherapy. Circ Heart Fail. 2018;11(7).
Oeffinger KC, Mertens AC, Sklar CA, Kawashima T, Hudson MM, Meadows AT, et al. Chronic health conditions in adult survivors of childhood cancer. N Engl J Med [Internet]. 2006;355(15):1572–82. Available from: https://doi.org/10.1056/NEJMsa060185.
Thomas R, Kenfield SA, Yanagisawa Y, Newton RU. Why exercise has a crucial role in cancer prevention, risk reduction and improved outcomes. Vol. 139, British Medical Bulletin. Oxford University Press. 2021;100–19.
Ornish D, Weidner G, Fair WR, Marlin R, Pettengill EB, Raisin CJ, et al. Intensive lifestyle changes may affect the progression of prostate cancer. J Urol. 2005;174(3):1065–70.
Wolf CM, Reiner B, Kühn A, Hager A, Müller J, Meierhofer C, et al. Subclinical cardiac dysfunction in childhood cancer survivors on 10-years follow-up correlates with cumulative anthracycline dose and is best detected by cardiopulmonary exercise testing, circulating serum biomarker, speckle tracking echocardiography, and tissue Doppler imaging. Front Pediatr. 2020;31:8.
Ness KK, Plana JC, Joshi VM, Luepker R V, Durand JB, Daniel, et al. Exercise intolerance, mortality, and organ system impairment in adult survivors of childhood cancer. J Clin Oncol [Internet]. 2019;38:29–42.
Christiansen JR, Kanellopoulos A, Lund MB, Massey R, Dalen H, Kiserud CE, et al. Impaired exercise capacity and left ventricular function in long-term adult survivors of childhood acute lymphoblastic leukemia. Pediatr Blood Cancer. 2015;62(8):1437–43.
Masood IR, Rezvan PH, Lee K, Vervaet H, Kuo C, Loss K, et al. Left ventricle size correlates with peak exercise capacity in pediatric cancer survivors exposed to anthracycline chemotherapy. Pediatr Cardiol [Internet]. 2023. Available from: https://link.springer.com/10.1007/s00246-023-03192-z.
Tsuda T, Kernizan D, Glass A, D’Aloisio G, Hossain J, Quillen J. Cardiopulmonary exercise testing characterizes silent cardiovascular abnormalities in asymptomatic pediatric cancer survivors. Pediatr Cardiol. 2022.
Bas O, Erdemir AG, Onur MR, Ozer N, Sener YZ, Aksu S, et al. Sarcopenia and anthracycline cardiotoxicity in patients with cancer. BMJ Support Palliat Care [Internet]. 2021;0:1–9. Available from: http://spcare.bmj.com/.
Smith WA, Ness KK, Joshi V, Hudson MM, Robison LL, Green DM. Exercise training in childhood cancer survivors with subclinical cardiomyopathy who were treated with anthracyclines. Pediatr Blood Cancer. 2013. https://doi.org/10.1002/pbc.24850.
Dieli-Conwright CM, Courneya KS, Demark-Wahnefried W, Sami N, Lee K, Sweeney FC, et al. Aerobic and resistance exercise improves physical fitness, bone health, and quality of life in overweight and obese breast cancer survivors: A randomized controlled trial. Breast Cancer Res. 2018;20(1).
Campbell KL, Winters-Stone KM, Wiskemann J, May AM, Schwartz AL, Courneya KS, et al. Exercise guidelines for cancer survivors: Consensus statement from international multidisciplinary roundtable. Med Sci Sports Exerc. 2019;51(11):2375–90.
Gilchrist SC, Barac A, Ades PA, Alfano CM, Franklin BA, Jones LW, et al. Cardio-oncology rehabilitation to manage cardiovascular outcomes in cancer patients and survivors: A scientific statement from the American Heart Association. Circulation. 2019;139(21):E997-1012.
Friend AJ, Feltbower RG, Hughes EJ, Dye KP, Glaser AW. Mental health of long-term survivors of childhood and young adult cancer: A systematic review. Vol. 143, International Journal of Cancer. Wiley-Liss Inc. 2018. p. 1279–86.
Rossano JW, Vanderpluym CJ, Peng DM, Hollander SA, Maeda K, Adachi I, et al. The Society of Thoracic Surgeons Pedimacs Annual report fifth annual pediatric interagency registry for mechanical circulatory support (Pedimacs) report. Ann Thorac Surg [Internet]. 2021;112:1763–74. Available from: https://doi.org/10.1016/j.athoracsur.2021.10.001.
Peng DM, Yu S, Lowery R, Ventresco C, Cousino MK, St. Louis JD, et al. Self-reported quality of life in children with ventricular assist devices. Pediatr Transplant. 2022;26(4).
Goldberg JF, Schlosser RS, Walrath JG, Morris SA, Elias BA, Price JF, et al. Improved 6 minute walk distance and brain-type natriuretic peptide after continuous-flow ventricular assist device placement in children. ASAIO J. 2019;65(7):725–30.
Burstein DS, McBride MG, Edelson JB, Rossano JW, O’Connor MJ, Lin KY, et al. Safety and feasibility of exercise rehabilitation in children with ventricular assist devices. Pediatr Cardiol. 2022;43(5):1029–36.
Burstein DS, McBride MG, Rossano JW, O’Connor MJ, Lin KY, Mascio CE, et al. Increasing pump speed during exercise training improves exercise capacity in children with ventricular assist devices. ASAIO J. 2021;67(4):449–56.
Yarlagadda V, Maeda K, Zhang Y, Chen S, Dykes JC, Gowen MA, et al. Temporary circulatory support in U.S. children awaiting heart transplantation. 2017.
Hermsen JA, Opotowsky AR, Powell AW, Mays WA, Chin C, Shertzer JD, et al. Physical frailty is modifiable in young cardiac rehabilitation patients. Pediatr Cardiol. 2022;43(8):1799–810.
Budts W, Pieles GE, Roos-Hesselink JW, de La Garza MS, D’Ascenzi F, Giannakoulas G, et al. Recommendations for participation in competitive sport in adolescent and adult athletes with congenital heart disease (CHD): Position statement of the sports cardiology exercise section of the European Association of Preventive Cardiology (EAPC), the European Society of Cardiology (ESC) working group on adult congenital heart disease and the sports cardiology, physical activity and prevention working group of the Association for European Paediatric and Congenital Cardiology (AEPC). Eur Heart J. 2020;41(43):4191–9.
Mehra MR, Cleveland JC, Uriel N, Cowger JA, Hall S, Horstmanshof D, et al. Primary results of long-term outcomes in the MOMENTUM 3 pivotal trial and continued access protocol study phase: A study of 2200 HeartMate 3 left ventricular assist device implants. Eur J Heart Fail. 2021;23(8):1392–400.
Funding
Open access funding provided by SCELC, Statewide California Electronic Library Consortium.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Jonathan Edelson report payment or honoraria from Abbott Technologies. Dr. Masood has no conflicts of interest to report.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Masood, I.R., Edelson, J.B. Exercise and Sports Participation in Children with Cardiomyopathy: A Review. Curr Treat Options Cardio Med 25, 543–559 (2023). https://doi.org/10.1007/s11936-023-01006-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11936-023-01006-2