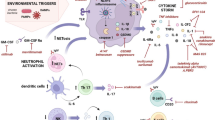
Abstract
Experimental models of rheumatoid arthritis have contributed immensely to our understanding of the pathogenesis as well as the treatment of this debilitating autoimmune disease. Significant progress has been made in the past few years in defining the role of newer cytokines and regulatory T cells, of inflammation-mediated bone and cartilage damage, and of the cholinergic anti-inflammatory pathway in modulating the disease process in arthritis. Furthermore, new therapeutic targets, including specific tyrosine kinases and proteasome subunits, have been explored. These advances offer renewed optimism for continued improvements in the management of rheumatoid arthritis.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Zenz R, Eferl R, Kenner L, et al. Psoriasis-like skin disease and arthritis caused by inducible epidermal deletion of Jun proteins. Nature. 2005;437:369–75.
Binstadt BA, Patel PR, Alencar H, et al. Particularities of the vasculature can promote the organ specificity of autoimmune attack. Nat Immunol. 2006;7:284–92.
• Murakami M, Okuyama Y, Ogura H, et al. Local microbleeding facilitates IL-6- and IL-17-dependent arthritis in the absence of tissue antigen recognition by activated T cells. J Exp Med. 2011;208:103–14. Mice with an activating IL-6R mutation and transgenic T-cell receptors recognizing nonjoint antigens develop arthritis that is dependent on IL-17 and IL-6. Arthritis also can be induced by the combination of joint “microbleeding” and adoptive transfer of Th17 cells.
Chu CQ, Swart D, Alcorn D, Tocker J, Elkon KB. Interferon-gamma regulates susceptibility to collagen-induced arthritis through suppression of interleukin-17. Arthritis Rheum. 2007;56:1145–51.
Kim EY, Chi HH, Bouziane M, Gaur A, Moudgil KD. Regulation of autoimmune arthritis by the pro-inflammatory cytokine interferon-gamma. Clin Immunol (Orlando, Fla). 2008;127:98–106.
•• Rajaiah R, Puttabyatappa M, Polumuri SK, Moudgil KD. Interleukin-27 and Interferon-{gamma} are involved in regulation of autoimmune arthritis. J Biol Chem. 2011;286:2817–25. This study unravels differences in the temporal kinetics of IL-17, IFN-γ, and IL-27 following an arthritogenic challenge in rat strains of the same major histocompatibility complex haplotype, but with differential susceptibility to AA. Supporting these profiles, treatment of susceptible rats with IL-27 or IFN-γ suppressed AA. Also, IFN-γ upregulates IL-27, revealing a novel interplay between these two cytokines that regulate IL-17.
•• Doodes PD, Cao Y, Hamel KM, et al. IFN-gamma regulates the requirement for IL-17 in proteoglycan-induced arthritis. J Immunol. 2010;184:1552–9. This study describes the relative contribution of IFN-γ and IL-17 to the pathogenesis of PGIA. The induction of PGIA is predominantly IFN-γ dependent in wild-type mice, but IL-17 dependent in mice deficient in IFN-γ. Also, T-box transcription factor regulates IL-17, pointing to the IFN-γ–mediated regulation of IL-17.
Lubberts E, Koenders MI, Oppers-Walgreen B, et al. Treatment with a neutralizing anti-murine interleukin-17 antibody after the onset of collagen-induced arthritis reduces joint inflammation, cartilage destruction, and bone erosion. Arthritis Rheum. 2004;50:650–9.
Bush KA, Farmer KM, Walker JS, Kirkham BW. Reduction of joint inflammation and bone erosion in rat adjuvant arthritis by treatment with interleukin-17 receptor IgG1 Fc fusion protein. Arthritis Rheum. 2002;46:802–5.
Ishiguro A, Akiyama T, Adachi H, Inoue J, Nakamura Y. Therapeutic potential of anti-interleukin-17A aptamer: suppression of interleukin-17A signaling and attenuation of autoimmunity in two mouse models. Arthritis Rheum. 2011;63:455–66.
Cao Y, Doodes PD, Glant TT, Finnegan A. IL-27 induces a Th1 immune response and susceptibility to experimental arthritis. J Immunol. 2008;180:922–30.
Niedbala W, Cai B, Wei X, et al. Interleukin 27 attenuates collagen-induced arthritis. Ann Rheum Dis. 2008;67:1474–9.
Goldberg R, Wildbaum G, Zohar Y, Maor G, Karin N. Suppression of ongoing adjuvant-induced arthritis by neutralizing the function of the p28 subunit of IL-27. J Immunol. 2004;173:1171–8.
Veenbergen S, Smeets RL, Bennink MB, et al. The natural soluble form of IL-18 receptor beta exacerbates collagen-induced arthritis via modulation of T-cell immune responses. Ann Rheum Dis. 2010;69:276–83.
Palmer G, Talabot-Ayer D, Lamacchia C, et al. Inhibition of interleukin-33 signaling attenuates the severity of experimental arthritis. Arthritis Rheum. 2009;60:738–49.
Hartgring SA, Willis CR, Alcorn D, et al. Blockade of the interleukin-7 receptor inhibits collagen-induced arthritis and is associated with reduction of T cell activity and proinflammatory mediators. Arthritis Rheum. 2010;62:2716–25.
Kochetkova I, Golden S, Holderness K, Callis G, Pascual DW. IL-35 stimulation of CD39+ regulatory T cells confers protection against collagen II-induced arthritis via the production of IL-10. J Immunol. 2010;184:7144–53.
• Notley CA, McCann FE, Inglis JJ, Williams RO. Anti-CD3 therapy expands the numbers of CD4+ and CD8+ Treg cells and induces sustained amelioration of collagen-induced arthritis. Arthritis Rheum. 2010;62:171–8. Treatment of mice with anti-CD3 caused attenuation of CIA. Anti-CD3 induced the generation of CD8 + CD25 + FoxP3 + T cells while expanding the number of natural Tregs of the CD4 + CD25 + FoxP3 + subset. CD8 + CD25 + FoxP3 + T cells suppressed the proliferation of CD4 + T cells and the production of IFN-γ and IL-17.
Nakatsukasa H, Tsukimoto M, Tokunaga A, Kojima S. Repeated gamma irradiation attenuates collagen-induced arthritis via up-regulation of regulatory T cells but not by damaging lymphocytes directly. Radiat Res. 2010;174:313–24.
Weng L, Williams RO, Vieira PL, Screaton G, Feldmann M, Dazzi F. The therapeutic activity of low-dose irradiation on experimental arthritis depends on the induction of endogenous regulatory T cell activity. Ann Rheum Dis. 2010;69:1519–26.
Nguyen LT, Jacobs J, Mathis D, Benoist C. Where FoxP3-dependent regulatory T cells impinge on the development of inflammatory arthritis. Arthritis Rheum. 2007;56:509–20.
Salazar L, Aravena O, Abello P, et al. Modulation of established murine collagen-induced arthritis by a single inoculation of short-term lipopolysaccharide-stimulated dendritic cells. Ann Rheum Dis. 2008;67:1235–41.
Jaen O, Rulle S, Bessis N, Zago A, Boissier MC, Falgarone G. Dendritic cells modulated by innate immunity improve collagen-induced arthritis and induce regulatory T cells in vivo. Immunology. 2009;126:35–44.
• Adamopoulos IE, Chao CC, Geissler R, et al. Interleukin-17A upregulates receptor activator of NF-kappaB on osteoclast precursors. Arthritis Res Ther. 2010;12:R29. This study draws attention to the direct action of IL-17 on osteoclast precursors affecting the upregulation of RANK on these cells in vitro. Increased RANK expression in turn increases sensitivity to RANKL signaling, osteoclast differentiation, and bone loss. Previous reports described an indirect role of IL-17 via upregulation of RANKL on the stromal cells.
Rifas L, Weitzmann MN. A novel T cell cytokine, secreted osteoclastogenic factor of activated T cells, induces osteoclast formation in a RANKL-independent manner. Arthritis Rheum. 2009;60:3324–35.
van Hamburg JP, Mus AM, de Bruijn MJ, et al. GATA-3 protects against severe joint inflammation and bone erosion and reduces differentiation of Th17 cells during experimental arthritis. Arthritis Rheum. 2009;60:750–9.
• Soderstrom K, Stein E, Colmenero P, et al. Natural killer cells trigger osteoclastogenesis and bone destruction in arthritis. Proc Natl Acad Sci U S A. 2010;107:13028–33. This study shows that synovial NK cells express both RANKL and macrophage colony-stimulating factor, and that these cells can induce the differentiation of monocytes into osteoclasts. Thus, NK cells may trigger bone damage in arthritis.
Zaiss MM, Frey B, Hess A, et al. Regulatory T cells protect from local and systemic bone destruction in arthritis. J Immunol. 2010;184:7238–46.
Schett G, Stolina M, Dwyer D, et al. Tumor necrosis factor alpha and RANKL blockade cannot halt bony spur formation in experimental inflammatory arthritis. Arthritis Rheum. 2009;60:2644–54.
Cejka D, Hayer S, Niederreiter B, et al. Mammalian target of rapamycin signaling is crucial for joint destruction in experimental arthritis and is activated in osteoclasts from patients with rheumatoid arthritis. Arthritis Rheum. 2010;62:2294–302.
Buttgereit F, Zhou H, Kalak R, et al. Transgenic disruption of glucocorticoid signaling in mature osteoblasts and osteocytes attenuates K/BxN mouse serum-induced arthritis in vivo. Arthritis Rheum. 2009;60:1998–2007.
•• Ishii M, Egen JG, Klauschen F, et al. Sphingosine-1-phosphate mobilizes osteoclast precursors and regulates bone homeostasis. Nature. 2009;458:524–8. This interesting study addressed a novel aspect of bone resorption, namely the trafficking of osteoclast precursors to and from the bone surface. It is reported here that S1P induces chemotaxis of osteoclast precursors, thus controlling their migratory behavior. Therefore, S1P-mediated events could serve as therapeutic targets.
Lee DM, Kiener HP, Agarwal SK, et al. Cadherin-11 in synovial lining formation and pathology in arthritis. Science. 2007;315:1006–10.
Shahrara S, Pickens SR, Mandelin 2nd AM, et al. IL-17-mediated monocyte migration occurs partially through CC chemokine ligand 2/monocyte chemoattractant protein-1 induction. J Immunol. 2010;184:4479–87.
• Ruth JH, Park CC, Amin MA, et al. Interleukin-18 as an in vivo mediator of monocyte recruitment in rodent models of rheumatoid arthritis. Arthritis Res Ther. 2010;12:R118. This study shows that IL-18 induces monocyte recruitment to the grafted synovial tissue in mice. Also shown was the role of IL-18 in initiating the production of other proinflammatory cytokines relevant for arthritis.
Connolly M, Marrelli A, Blades M, et al. Acute serum amyloid A induces migration, angiogenesis, and inflammation in synovial cells in vitro and in a human rheumatoid arthritis/SCID mouse chimera model. J Immunol. 2010;184:6427–37.
Ablin JN, Entin-Meer M, Aloush V, et al. Protective effect of eotaxin-2 inhibition in adjuvant-induced arthritis. Clin Exp Immunol. 2010;161:276–83.
Doodes PD, Cao Y, Hamel KM, et al. CCR5 is involved in resolution of inflammation in proteoglycan-induced arthritis. Arthritis Rheum. 2009;60:2945–53.
Zhou HF, Chan HW, Wickline SA, Lanza GM, Pham CT. Alphavbeta3-targeted nanotherapy suppresses inflammatory arthritis in mice. FASEB J. 2009;23:2978–85.
Choi EY, Chavakis E, Czabanka MA, et al. Del-1, an endogenous leukocyte-endothelial adhesion inhibitor, limits inflammatory cell recruitment. Science. 2008;322:1101–4.
Tu-Rapp H, Pu L, Marques A, et al. Genetic control of leucocyte–endothelial cell interaction in collagen-induced arthritis. Ann Rheum Dis. 2010;69:606–10.
• van Maanen MA, Lebre MC, van der Poll T, et al. Stimulation of nicotinic acetylcholine receptors attenuates collagen-induced arthritis in mice. Arthritis Rheum. 2009;60:114–22. This study highlights the role of the cholinergic anti-inflammatory pathways in CIA. It was reported that cervical vagotomy aggravated arthritis, whereas oral administration of nicotine afforded protection against arthritis. The effects of nicotine treatment on clinical arthritis correlated with reduction in both bone damage and TNF-α expression in the synovial tissue.
Westman M, Saha S, Morshed M, Lampa J. Lack of acetylcholine nicotine alpha 7 receptor suppresses development of collagen-induced arthritis and adaptive immunity. Clin Exp Immunol. 2010;162:62–7.
• Yu H, Yang YH, Rajaiah R, Moudgil KD. Nicotine-induced differential modulation of autoimmune arthritis in the Lewis rat involves changes in IL-17 and anti-cyclic citrullinated peptide antibodies. Arthritis and rheumatism 2011;63:981–91. This study revealed that the timing of nicotine treatment (in relation to the onset of arthritis) differentially modulated AA in the Lewis rat. Nicotine pretreatment exacerbated arthritis, while post-treatment attenuated AA. Also, this is the first report on the detection of aCCP antibodies during the natural course of AA. The level of aCCP correlated well with disease severity.
Uysal H, Bockermann R, Nandakumar KS, et al. Structure and pathogenicity of antibodies specific for citrullinated collagen type II in experimental arthritis. J Exp Med. 2009;206:449–62.
Pine PR, Chang B, Schoettler N, et al. Inflammation and bone erosion are suppressed in models of rheumatoid arthritis following treatment with a novel Syk inhibitor. Clin Immunol (Orlando, Fla). 2007;124:244–57.
Jakus Z, Simon E, Balazs B, Mocsai A. Genetic deficiency of Syk protects mice from autoantibody-induced arthritis. Arthritis Rheum. 2010;62:1899–910.
• Lin TH, Hegen M, Quadros E, et al. Selective functional inhibition of JAK-3 is sufficient for efficacy in collagen-induced arthritis in mice. Arthritis Rheum. 2010;62:2283–93. This study shows that selective inhibition of JAK3 was sufficient to disrupt signaling of the common γ-chain cytokines leading to the suppression of CIA. The use of a selective inhibitor of JAK3 would offer advantages over the use of inhibitors that also disrupt the signaling events mediated via other related members, such as JAK1, JAK2, and Tyk2.
Ahmed AS, Li J, Ahmed M, et al. Attenuation of pain and inflammation in adjuvant-induced arthritis by the proteasome inhibitor MG132. Arthritis Rheum. 2010;62:2160–9.
Muchamuel T, Basler M, Aujay MA, et al. A selective inhibitor of the immunoproteasome subunit LMP7 blocks cytokine production and attenuates progression of experimental arthritis. Nat Med. 2009;15:781–7.
Acknowledgment
This work was supported by grants (R01 AT004321 and PO1 AT002605) from the National Institutes of Health.
Disclosure
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moudgil, K.D., Kim, P. & Brahn, E. Advances in Rheumatoid Arthritis Animal Models. Curr Rheumatol Rep 13, 456–463 (2011). https://doi.org/10.1007/s11926-011-0200-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11926-011-0200-z
Keywords
- Rheumatoid arthritis (RA)
- Animal models
- Collagen-induced arthritis (CIA)
- Adjuvant arthritis (AA)
- Glucose-6-phosphate isomerase (GPI)
- Proteoglycan-induced arthritis (PGIA)
- Cytokines
- Interleukin
- Tumor necrosis factor-alpha (TNF-alpha)
- Interferon-γ (IFN-γ)
- CD4 + CD25+ regulatory T cells (Treg)
- Dendritic cells (DC)
- Inflammation
- Receptor activator of nuclear factor-κ B ligand (RANKL)
- Osteoprotegerin (OPG)
- Chemokines
- Developmental endothelial locus-1 (Del-1)
- Anticyclic citrullinated peptide (aCCP) antibody
- α7-nicotinic acetylcholine receptors (α7nAChRs)
- Sphingosine-1-phosphate (S1P)
- Spleen tyrosine kinase (Syk)
- Janus kinase (JAK)-Interleukin-1 receptor antagonist (IL-1Ra)