Abstract
Osteoarthritis (OA) pain is poorly understood and managed, as current analgesics have only limited efficacy and unwanted side effect profiles. A broader understanding of the pathological mechanisms driving OA joint pain is vital for the development of improved analgesics. Both clinical and preclinical data suggest an association between joint levels of the sensory neuropeptide calcitonin gene-related peptide (CGRP) and pain during OA. Whether a direct causative link exists remains an important unanswered question. Given the recent development of small molecule CGRP receptor antagonists with clinical efficacy against migraine pain, the interrogation of the role of CGRP in OA pain mechanisms is extremely timely. In this article, we provide the background to the importance of CGRP in pain mechanisms and review the emerging clinical and preclinical evidence implicating a role for CGRP in OA pain. We suggest that the CGRP receptor antagonists developed for migraine pain warrant further investigation in OA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoarthritis (OA) is a chronic degenerative joint disease characterized by cartilage degeneration, synovial inflammation and subchondral bone changes including sclerosis and osteophyte formation, leading to a decline in joint function [1]. Chronic pain is the main clinical symptom of OA and is the key factor in patients seeking medical attention. The underlying mechanisms driving OA pain are not fully understood, and as a consequence many of the commonly prescribed analgesic drugs used to treat OA pain, such as nonsteroidal anti-inflammatory drugs (NSAIDs) and weak opioids, provide variable symptomatic pain relief [2]. A lack of disease modifying drugs for OA has led to a growing demand for more effective analgesics. A better understanding of the mechanisms leading to changes within the nociceptive system during joint degeneration will help drive forward the development of new and improved analgesics that may exert beneficial effects beyond symptom modification.
Clinical evidence indicates that changes in the peripheral nociceptive system at the level of the OA joint make an important contribution to the pain experienced. Surgical joint replacement [3] and intra-articular local anesthetic [4] alleviate pain in around 60–80 % of patients. Further, experimental studies in OA patients indicate sensitization of joint innervating nociceptors (peripheral sensitization) during OA [5••]. The peripheral mediators of this sensitization have not yet been identified.
Calcitonin gene-related peptide (CGRP) is a pro-inflammatory peptide expressed by sensory neurons, including those that innervate joints [6]. CGRP has long been known to be important in the generation of migraine pain and recent clinical trials have demonstrated efficacy of small molecule CGRP receptor antagonists against migraine pain [7]. Like migraine, elevated levels of CGRP, activation of peripheral neurons, and peripheral sensitization are all features of OA, suggesting that CGRP receptor antagonists may have clinical utility in the treatment of OA pain. However, the importance of CGRP to OA pain mechanisms and the question of whether these antagonists might modulate OA pain are only just beginning to be addressed. This article will provide the background on the importance of CGRP in pain mechanisms as well as evidence for the emerging role of CGRP and its receptor system in painful OA joints.
CGRP
CGRP is a 37-amino acid neuropeptide formed from the alternative splicing of the calcitonin gene [8]. CGRP belongs to a large family of peptides that includes calcitonin, amylin and adrenomedullin, and is one of the most abundant peptides within the nervous system. The predominant form of CGRP is known as α-CGRP. A second isoform of CGRP, β-CGRP, is produced from a separate gene, but shares high sequence homology (three amino acids different in man and one in the rat) [9]. Whilst both isoforms display similar biological and pharmacological activities, they exhibit significant differences in their expression patterns; α-CGRP is expressed in both the peripheral and central nervous system, whilst β-CGRP is primarily expressed in the enteric nervous system [10]. Little is known about the physiological significance of the two CGRP isoforms. CGRP plays a role in a diverse range of biological functions, but is best known for its role as a potent vasodilator [11]. Injection of CGRP causes a persistent and long lasting increase in microvascular blood flow in the skin [11] and other tissues such as the knee joint [12]. However, CGRP also has well-established roles in neurogenic inflammation and nociception [13•].
CGRP-like immunoreactivity (CGRP-LI) is found in 40–50 % of all types of sensory neuronal somata (Aα, Aβ, Aδ, and C-type fibers) of the dorsal root ganglia (DRG), being particularly enriched in small nociceptive neurons [14], including those that innervate joints [6]. Many of these neurons co-express substance P (SP) [15]. A significant number of CGRP-LI neurons are non-nociceptive mechanoreceptors [16]. CGRP-LI nerve fiber terminals synapse onto lamina I-II and lamina V of the spinal cord (laminae associated with nociceptive processing) across a number of species [17], consistent with the expression of CGRP-LI in mainly small nociceptive sensory neurons.
The release of CGRP from the peripheral and central terminals of sensory neurons is Ca2+ dependent, and can be initiated by noxious mechanical stimulation of the skin [18], TRPV1 activation [19, 20], electrical stimulation [19, 21, 22•], noxious heat [23] or protein kinase C (PKC) activation [23]. The release of CGRP into the spinal cord can facilitate the release of excitatory neurotransmitters, such as glutamate and aspartate, strengthening connections in the dorsal horn and promoting synaptic transmission [24]. Thus, the release of CGRP following noxious stimulation and the promotion of excitatory neurotransmission in the dorsal horn of the spinal cord suggests that CGRP has an important role in the transmission of painful stimuli.
The CGRP Receptor System
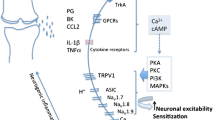
Until relatively recently, there was believed to be two CGRP receptor subtypes in existence, differing in their sensitivity to the classical CGRP receptor antagonist CGRP8-37 [25]. However, following the discovery of the molecular identity of the composition of the CGRP receptor, it is now widely held that only one CGRP receptor exists (Fig. 1). The CGRP receptor was identified following the discovery that when expressed in HEK293 cells, the orphan Family B GPCR, calcitonin receptor-like receptor (CLR), showed high affinity binding sites for CGRP and was pharmacologically responsive to CGRP administration [26]. Shortly after, it was discovered that to fully function, CLR required a single transmembrane protein called receptor activity modifying protein 1 (RAMP1), to aid the transport of CLR to the cell surface [27] (Fig. 1). To date, three isoforms of RAMPs have been identified, with CLR and RAMP2/3 complexes forming adrenomedullin receptors [28]. Thus, the formation of multiple calcitonin receptors is possible, raising the intriguing possibility that under certain conditions (i.e. disease), there may be a dynamic regulation of RAMP isoform expression governing tissue pharmacology.
CGRP receptor composition. The CGRP receptor is a hetero-trimeric protein comprised of the GPCR calcitonin receptor-like receptor (CLR), coupled to a receptor activity modifying protein (RAMP). There are three RAMP isoforms that govern receptor pharmacology; RAMP1 containing receptors are CGRP-binding, whilst RAMP2/3 containing receptors are adrenomedullin-binding .The intracellular receptor component protein (RCP) facilitates secondary messenger signaling. Different tissues/cells can express multiple RAMP isoforms, resulting in a complex pharmacological profile
A third, intracellular receptor component protein (RCP) is also required to facilitate CGRP receptor signaling and couple the receptor to secondary messenger pathways [29] (Fig. 1). CGRP receptor activation results in an increase in intracellular cAMP and subsequent protein kinase A (PKA) activation, mediated by the G protein Gαs [30]. However, signaling through other G proteins is possible, and CGRP receptor signaling has been shown to result in the activation of nitric oxide (NO) [31], mitogen activated protein kinases (MAPK) [32], phospholipase C [33], PKA and PKC [34].
CGRP receptor components are expressed in tissues at all levels of the pain pathway [35–39]. Receptor component expression is primarily on small-medium sized Aδ and C-type DRG neurons, some of which co-express CGRP [36, 38]. These neurons are typically thought to be nociceptive, highlighting a potential role for the CGRP receptor in the modulation of peripheral nociception. Interestingly, RAMP1 has been shown to be functionally rate limiting for the effects of CGRP on trigeminal ganglia, and elevated levels in man may sensitize to the effects of CGRP during migraine [40]. The expression of CGRP receptors on CGRP expressing sensory neurons indicates the existence of CGRP auto-receptors, which may regulate the neurogenic inflammatory and nociceptive properties of CGRP. Whether CGRP receptor components are expressed by sensory neurons innervating joints is currently unknown. Within the spinal cord dorsal horn, CLR and RAMP1 expression is primarily in laminae I and II where Aδ and C-type nociceptors terminate [38]. It is not known whether this CLR and RAMP1 immunoreactivity originates from spinal neurons or primary afferent terminals.
The Role of CGRP in Acute Nociceptive Transmission
Ever since the demonstration of expression of CGRP on small nociceptive afferents and within the dorsal horn of the spinal cord 30 years ago [41], there has been growing interest in the role of CGRP in the transmission of nociceptive information and the generation of pain. The role of CGRP in modulating nociceptive transmission at central sites is well accepted. Although, within the brain, the effects of CGRP receptor activation on pain responses appears to be site specific; CGRP receptor activation within the amygdala evokes pain behavior and facilitates excitatory synaptic transmission [42, 43], whilst CGRP applied to the periaqueductal grey, nucleus accumbens and nucleus raphe magnus is antinociceptive [44–49].
Spinally applied CGRP facilitates excitatory synaptic transmission at the level of the spinal cord, potentiating spinal N-methyl-D-aspartate (NMDA) responses [50] and sensitizing spinal neurons [34, 51, 52], increasing their discharge frequency to mechanical and electrical stimulation and potentiating their responses to SP [52–54]. Behaviorally, intrathecal CGRP in the rat produces mechanical and thermal hyperalgesia as well as mechanical allodynia [34, 54, 55]. Some groups have however published conflicting data on the spinal role of CGRP, demonstrating a lack of effect of CGRP on the excitability of spinal cord neurons [56] and lack of hyperalgesic effect of intrathecal CGRP [57, 58]. These discrepancies could relate to methodological differences (e.g. type of CGRP used with respect to α vs. β analogs).
Despite this compelling evidence, the role of CGRP in the modulation of peripheral nociceptive responses is less well accepted. In vitro studies have provided evidence that CGRP can directly modulate nociceptor excitability. CGRP depolarizes cultured sensory neurons [59] and enhances tetrodotoxin resistant (TTX-R) voltage gated sodium currents (TTX-R NaVs) via a PKA-dependent and PKC-dependent phosphorylation of NaV1.8 [60]. Administration of CGRP to cultured DRG neurons lowered their activation thresholds and increased their excitability by enhancing TTX-R NaV current density, lowering the amount of current required to evoke an action potential and shifting voltage and conductance curves in a hyperpolarized direction [60]. These effects are neurophysiologic hallmarks of peripheral sensitization, suggesting that CGRP has the potential to drive maladaptive processes in peripheral nerves, leading to pain. Despite this in vitro evidence for a direct modulation of nociceptor function, evidence for a direct effect of CGRP on primary afferent nociceptors in vivo is lacking. CGRP may indirectly contribute to sensitization of nociceptors by evoking the release of pro-inflammatory mediators from immune cells and nerves, such as bradykinin and prostaglandins, which can directly excite and sensitize nociceptors [61–63]. Collectively, these studies provide putative mechanisms by which CGRP may modulate peripheral nociception.
In vivo studies have provided support for an important role of CGRP in peripheral nociceptive mechanisms. The differential sensitivity of multiple strains of mice to noxious heat has been attributed to variations in CGRP expression levels in sensory neurons [64]. These compelling findings suggest that peripheral CGRP expression is a critical determinant of noxious heat sensitivity. Intra-plantar injection of CGRP evokes mechanical allodynia, highlighting a sensitizing effect of CGRP on cutaneous afferents [65]. However, the allodynic effects of CGRP were tenfold less potent than SP. In a separate study, a single injection of CGRP into the rat hind paw produced only a weak hyperalgesic response (compared to neurokinin A and SP). However, repeated sub-thresholds injections of CGRP appeared to prime primary afferent fibers to a subsequent challenging dose of CGRP, and induced an intense and long-lasting hyperalgesic response [66]. The potentially important implications of this are that during certain maladaptive disease processes associated with prolonged release of CGRP (e.g. OA), the hyperalgesic priming potential of CGRP may be important for the transition from acute to chronic pain [67].
Antinociceptive Effects of CGRP Receptor Antagonists
Blocking the effects of endogenous CGRP within the spinal cord supports a role for CGRP in spinal nociceptive transmission. Intrathecal administration of the truncated peptide antagonist of CGRP, CGRP8-37, inhibits capsaicin induced central sensitization of spinal dorsal horn wide dynamic range (WDR) neurons [68] and reduces WDR neuron activity to electrical stimulation of the hind paw [53, 69]. Behaviorally, intrathecal CGRP8-37 inhibits SP-induced [57] and thermal injury-induced [70] mechanical and thermal hyperalgesia, and anti-serum raised against CGRP is antinociceptive when administered by the intrathecal [56, 71] or intravenous [72] route.
Blocking the actions of CGRP at the level of the spinal cord is hyperalgesic in models of inflammatory pain. In the kaolin and carrageenan model of arthritis, spinal administration of CGRP8-37 inhibits electrically evoked spinal neuronal hyperexcitability, but has no effect on normal transmission [51], and spinal administration of the small molecule CGRP antagonist BIBN4096BS inhibits the responses of amygdala neurons to mechanical stimulation of the inflamed joint, but not to stimulation of the normal control joint [45]. Following the induction of inflammatory arthritis, levels of RCP in the dorsal horn of the spinal cord are elevated [36]. This increased expression of RCP may facilitate intracellular signaling through the CGRP receptor, contributing to the increased sensitivity to CGRP during inflammatory arthritis. BIBN4096BS inhibits mechanical evoked activity of WDR neurons in the Complete Freund’s Adjuvant (CFA) model of cutaneous inflammatory pain following both systemic and topical (hind paw) administration, but has no effect when applied directly to the spinal cord [13•]. Although the authors did not directly investigate the site of action, the pharmacokinetics of the compound and the equal effectiveness of both topical and systemic applications suggest a peripheral site of action. These preclinical animal model studies suggest that CGRP receptor antagonists may have therapeutic potential for the treatment of pain during inflammation. In addition, the periphery may be an important site of action of these compounds.
CGRP and Migraine Pain
Migraine is considered a chronic neurovascular disorder characterized by recurrent severe headaches, associated with nausea, vomiting, photophobia and phonophobia, which may be preceded by an aura [73]. Studies have shown that the pain arising during migraine is generated by small unmyelinated fibers, originating from the trigeminal nerve, which surround cerebral blood vessels [74]. These fibers contain CGRP and SP [75]. CGRP is released from trigeminal ganglia neurons in vitro following their stimulation [76]. A number of key data support an important role for CGRP in the generation of migraine pain; 1) electrical stimulation of the trigeminovascular system results in the release of CGRP from nociceptive trigeminal afferents [77]; 2) CGRP levels are elevated in blood samples taken from patients during migraine attacks [78]; 3) intravenous CGRP infusion causes headaches and migraines in migraineurs [79]; and 4) small molecule CGRP receptor antagonist are successful at alleviating pain during acute migraine attacks [80, 81]. The effects of these small molecule CGRP receptor antagonists have been investigated in animal models of migraine pain, revealing a complex mechanism of action involving both peripheral and central sites. CGRP receptor antagonists reduce spontaneous and evoked neuronal activity from the spinal trigeminal nucleus (STN; thought to have an important role in centrally mediated migraine pain) [82] and inhibit nitric oxide induced sensitization of the STN [83]. In addition, CGRP receptor antagonists inhibit electrically evoked activity of nociceptors of the trigeminocervical complex [84] and reduce CGRP mediated trigeminovascular neurogenic inflammation [85]. It appears that within the setting of migraine pain, CGRP has a role in modulating nociceptive responses both at peripheral and central levels. The balance of the relative importance of CGRP at these sites is unknown [7].
Evidence for a Role of CGRP in Osteoarthritis Pain
Over recent years, both clinical and preclinical evidence indicating an important association between joint levels of CGRP and pain during OA has been accumulating [6, 13•, 20, 22•, 86–92]. Sensory neurons expressing CGRP innervate most joint structures [93], particularly the synovial membrane, ligaments and subchondral bone [6, 86, 94]. Clinical data from knee and hip OA patients support an association between joint CGRP and pain. Perivascular nerve fibers showing CGRP-LI in the outer region of the menisci of knee OA patients are increased in density in patients with high chondropathy and joint pathology [87]. The vascularization and consequent innervation of CGRP expressing nociceptors in joint structures close to aneural tissue (e.g. cartilage) may provide a mechanism for pain in these patients. Patients with painful OA of the hip have an increased density of CGRP (and SP)-containing nerve fibers in the joint capsule compared to asymptomatic controls [88]. Furthermore, patients with a painless failed hip arthroplasty completely lacked CGRP-LI fibers surrounding the joint. The number of synovial CGRP-LI free nerve fibers and endings is greatest in the medial joint compartment [89]. This compartment is where pain is localized and synovitis and joint pathology are the greatest. Although these human data provide evidence for an association between altered joint CGRP levels and pain during OA, whether there is a direct causative link remains an important unanswered question. Studies in preclinical animal models of OA are beginning to address this question.
The monosodium iodoacetate (MIA) model of OA pain has gathered favor recently, as a robust and reliable model of OA that mirrors key components of human knee OA, including joint pathology [95], pain responses [96, 97], inflammation [96, 98] and analgesic responsiveness [96, 97]. These changes are associated with increases in excitability of joint-associated nociceptors; specifically, an increase in C-fiber spontaneous activity and an increase in the mechanosensitivity of Aδ-fibers [99]. This sensitization is likely to be an important contributor to the ongoing pain in this model of OA, and there is evidence to suggest that CGRP may play an important role in the establishment and maintenance of these sensitized responses. Using back-labeling techniques to identify knee joint afferent cell bodies in DRGs, it has been shown that CGRP expression is increased in these neurons following MIA treatment [6, 90, 91]. Following MIA, CGRP expression increases specifically in medium to large diameter neurons that give rise to Aδ-fiber and Aβ-fiber joint afferents, respectively [91]. Concomitant with this increase in the expression of CGRP, there is also an increase in the basal and evoked release of CGRP from primary afferent fibers into the spinal cord [20, 22•]. The increased expression and release of CGRP during MIA-induced OA may sensitize TTX-R NaV1.8 channels (see above). Indeed, blocking NaV1.8 on knee joint-associated afferents reduces mechanical hyperalgesia in the MIA model [100]. Ablation of CGRP expressing nerve terminals by capsaicin treatment prior to MIA injection reduces pain behavior and bone pathology [92]. The ablated afferents also express other nociceptive neuropeptides and neurotransmitters, so further work is needed to investigate whether there is a direct link between reduced joint CGRP levels and protection against OA pain.
A recent behavioral study carried out in the MIA model demonstrated analgesic effects of the small molecule receptor antagonist BIBN4096BS. Sub-cutaneous treatment with BIBN4096BS reversed MIA-induced weight bearing asymmetry as effectively as morphine [13•]. Given the relatively poor capacity for BIBN4096BS to penetrate the blood brain barrier, the authors hypothesized that the reversal of pain behavior was peripherally mediated, although the investigation of a peripheral site was not further examined in this model and BIBN4096BS effects were only assessed during the acute inflammatory phase (day 3 post-MIA) before OA-like changes are established. Thus, further investigations of the role of CGRP in driving peripheral nociceptive inputs during OA are required. These preclinical studies in the MIA model highlight the potential importance of CGRP in driving aberrant nociceptive inputs from the joint and pain in this model.
Conclusions
There is now compelling evidence to suggest that elevated peripheral CGRP expression and release during joint degeneration may play an important role in the establishment of peripheral sensitization and the pain associated with OA. Augmented release of CGRP from the peripheral terminals of nociceptors within the degenerating joint may activate CGRP receptors expressed by these neurons, directly activating them and driving their sensitization via downstream phosphorylation of ion channels, and indirectly activating them by promoting the release of proinflammatory and sensitizing molecules. The concomitant augmented release of CGRP from the central terminals of nociceptors within the dorsal horn would also be expected to facilitate nociceptive transmission. Similar to migraine pain, it appears that CGRP may have important roles at both peripheral and central levels during OA pain.
Further studies are required to fully understand the role that CGRP and associated regulatory proteins play in the generation of OA pain. Dissecting the complex role of CGRP, including the relative importance of CGRP at peripheral and central sites, may lead to more effective and targeted therapeutics. Blocking the interaction of CGRP and its receptor complex in the periphery may be an attractive therapeutic strategy for the alleviation of OA pain without the side effects of NSAIDs. Vital to this will be a broader understanding of the role that the CGRP receptor system plays in the development of peripheral sensitization and pain in OA.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Dray A, Read SJ. Arthritis and pain. Future targets to control osteoarthritis pain. Arthritis Res Ther. 2007;9:212.
Breivik H, Collett B, Ventafridda V, et al. Survey of chronic pain in Europe: prevalence, impact on daily life, and treatment. Eur J Pain. 2006;10:287–333.
Kosek E, Ordeberg G. Lack of pressure pain modulation by heterotopic noxious conditioning stimulation in patients with painful osteoarthritis before, but not following, surgical pain relief. Pain. 2000;88:69–78.
Creamer P, Hunt M, Dieppe P. Pain mechanisms in osteoarthritis of the knee: effect of intraarticular anesthetic. J Rheumatol. 1996;23:1031–6.
•• Arendt-Nielsen L, Nie H, Laursen MB, et al. Sensitization in patients with painful knee osteoarthritis. Pain. 2010;149:573–81. Demonstration of peripheral sensitization in patients with osteoarthritis.
Fernihough J, Gentry C, Bevan S, et al. Regulation of calcitonin gene-related peptide and TRPV1 in a rat model of osteoarthritis. Neurosci Lett. 2005;388:75–80.
Doods H, Arndt K, Rudolf K, et al. CGRP antagonists: unravelling the role of CGRP in migraine. Trends Pharmacol Sci. 2007;28:580–7.
Amara SG, Jonas V, Rosenfeld MG, et al. Alternative RNA processing in calcitonin gene expression generates mRNAs encoding different polypeptide products. Nature. 1982;298:240–4.
Steenbergh PH, Hoppener JW, Zandberg J, et al. A second human calcitonin/CGRP gene. FEBS Lett. 1985;183:403–7.
Mulderry PK, Ghatei MA, Spokes RA, et al. Differential expression of alpha-CGRP and beta-CGRP by primary sensory neurons and enteric autonomic neurons of the rat. Neuroscience. 1988;25:195–205.
Brain SD, Williams TJ, Tippins JR, et al. Calcitonin gene-related peptide is a potent vasodilator. Nature. 1985;313:54–6.
Cambridge H, Brain SD. Calcitonin gene-related peptide increases blood flow and potentiates plasma protein extravasation in the rat knee joint. Br J Pharmacol. 1992;106:746–50.
• Hirsch S, Corradini L, Just S, et al. The CGRP receptor antagonist BIBN4096BS peripherally alleviates inflammatory pain in rats. Pain. 2013;154:700–7. First study to demonstrate alleviation of pain in an animal model of OA by a CGRP receptor antagonist.
McCarthy PW, Lawson SN. Cell type and conduction velocity of rat primary sensory neurons with calcitonin gene-related peptide-like immunoreactivity. Neuroscience. 1990;34:623–32.
Lee Y, Takami K, Kawai Y, et al. Distribution of calcitonin gene-related peptide in the rat peripheral nervous system with reference to its coexistence with substance P. Neuroscience. 1985;15:1227–37.
Lawson SN, Crepps B, Perl ER. Calcitonin gene-related peptide immunoreactivity and afferent receptive properties of dorsal root ganglion neurones in guinea-pigs. J Physiol. 2002;540:989–1002.
Gibson S, Polak J, Bloom S, et al. Calcitonin gene-related peptide immunoreactivity in the spinal cord of man and of eight other species. J Neurosci. 1984;4:3101–11.
Morton CR, Hutchison WD. Release of sensory neuropeptides in the spinal cord: studies with calcitonin gene-related peptide and galanin. Neuroscience. 1989;31:807–15.
Sauer SK, Bove GM, Averbeck B, et al. Rat peripheral nerve components release calcitonin gene-related peptide and prostaglandin E2 in response to noxious stimuli: evidence that nervi nervorum are nociceptors. Neuroscience. 1999;92:319–25.
Puttfarcken PS, Han P, Joshi SK, et al. A-995662 [(R)-8-(4-methyl-5-(4-(trifluoromethyl)phenyl)oxazol-2-ylamino)-1,2,3,4-te trahydronaphthalen-2-ol], a novel, selective TRPV1 receptor antagonist, reduces spinal release of glutamate and CGRP in a rat knee joint pain model. Pain. 2010;150:319–26.
Schaible HG, Freudenberger U, Neugebauer V, et al. Intraspinal release of immunoreactive calcitonin gene-related peptide during development of inflammation in the joint in vivo—a study with antibody microprobes in cat and rat. Neuroscience. 1994;62:1293–305.
• Ogbonna AC, Clark AK, Gentry C, et al. Pain-like behaviour and spinal changes in the monosodium iodoacetate model of osteoarthritis in C57Bl/6 mice. Eur J Pain. 2013;17:514–26. Demonstrated reduced mechanical allodynia in an animal model of OA following administration of an intrathecal CGRP receptor antagonist.
Kessler F, Habelt C, Averbeck B, et al. Heat-induced release of CGRP from isolated rat skin and effects of bradykinin and the protein kinase C activator PMA. Pain. 1999;83:289–95.
Kangrga I, Larew JSA, Randic M. The effects of substance P and calcitonin gene-related peptide on the efflux of endogenous glutamate and aspartate from the rat spinal dorsal horn in vitro. Neurosci Lett. 1990;108:155–60.
Quirion R, Van Rossum D, Dumont Y, et al. Characterization of CGRP1 and CGRP2 Receptor Subtypesa. Ann N Y Acad Sci. 1992;657:88–105.
Aiyar N, Rand K, Elshourbagy NA, et al. A cDNA encoding the calcitonin gene-related peptide type 1 receptor. J Biol Chem. 1996;271:11325–9.
McLatchie LM, Fraser NJ, Main MJ, et al. RAMPs regulate the transport and ligand specificity of the calcitonin-receptor-like receptor. Nature. 1998;393:333–9.
Hay DL, Howitt SG, Conner AC, et al. CL/RAMP2 and CL/RAMP3 produce pharmacologically distinct adrenomedullin receptors: a comparison of effects of adrenomedullin22–52, CGRP8–37 and BIBN4096BS. Br J Pharmacol. 2003;140:477–86.
Evans BN, Rosenblatt MI, Mnayer LO, et al. CGRP-RCP, a novel protein required for signal transduction at calcitonin gene-related peptide and adrenomedullin receptors. J Biol Chem. 2000;275:31438–43.
Walker CS, Conner AC, Poyner DR, et al. Regulation of signal transduction by calcitonin gene-related peptide receptors. Trends Pharmacol Sci. 2010;31:476–83.
Brain SD, Grant AD. Vascular actions of calcitonin gene-related peptide and adrenomedullin. Physiol Rev. 2004;84:903–34.
Parameswaran N, Disa J, Spielman WS, et al. Activation of multiple mitogen-activated protein kinases by recombinant calcitonin gene-related peptide receptor. Eur J Pharmacol. 2000;389:125–30.
Drissi H, Lasmoles F, Le Mellay V, et al. Activation of phospholipase C-beta1 via Galphaq/11 during calcium mobilization by calcitonin gene-related peptide. J Biol Chem. 1998;273:20168–74.
Sun RQ, Tu YJ, Lawand NB, et al. Calcitonin gene-related peptide receptor activation produces PKA- and PKC-dependent mechanical hyperalgesia and central sensitization. J Neurophysiol. 2004;92:2859–66.
Chakravarty P, Suthar TP, Coppock HA, et al. CGRP and adrenomedullin binding correlates with transcript levels for calcitonin receptor-like receptor (CRLR) and receptor activity modifying proteins (RAMPs) in rat tissues. Br J Pharmacol. 2000;130:189–95.
Ma W, Chabot JG, Powell KJ, et al. Localization and modulation of calcitonin gene-related peptide-receptor component protein-immunoreactive cells in the rat central and peripheral nervous systems. Neuroscience. 2003;120:677–94.
Cottrell GS, Alemi F, Kirkland JG, et al. Localization of calcitonin receptor-like receptor (CLR) and receptor activity-modifying protein 1 (RAMP1) in human gastrointestinal tract. Peptides. 2012;35:202–211.
Cottrell GS, Roosterman D, Marvizon J-C, et al. Localization of calcitonin receptor-like receptor and receptor activity modifying protein 1 in enteric neurons, dorsal root ganglia, and the spinal cord of the rat. J Comp Neurol. 2005;490:239–55.
Pokabla MJ, Dickerson IM, Papka RE. Calcitonin gene-related peptide-receptor component protein expression in the uterine cervix, lumbosacral spinal cord, and dorsal root ganglia. Peptides. 2002;23:507–14.
Zhang Z, Winborn CS, Marquez de Prado B, et al. Sensitization of calcitonin gene-related peptide receptors by receptor activity-modifying protein-1 in the trigeminal ganglion. J Neurosci. 2007;27:2693–703.
Rosenfeld MG, Mermod JJ, Amara SG, et al. Production of a novel neuropeptide encoded by the calcitonin gene via tissue-specific RNA processing. Nature. 1983;304:129–35.
Han JS, Adwanikar H, Li Z, et al. Facilitation of synaptic transmission and pain responses by CGRP in the amygdala of normal rats. Mol Pain. 2010;6:10.
Han JS, Li W, Neugebauer V. Critical role of calcitonin gene-related peptide 1 receptors in the amygdala in synaptic plasticity and pain behavior. J Neurosci. 2005;25:10717–28.
Xu W, Lundeberg T, Wang YT, et al. Antinociceptive effect of calcitonin gene-related peptide in the central nucleus of amygdala: activating opioid receptors through amygdala-periaqueductal gray pathway. Neuroscience. 2003;118:1015–22.
Adwanikar H, Ji G, Li W, et al. Spinal CGRP1 receptors contribute to supraspinally organized pain behavior and pain-related sensitization of amygdala neurons. Pain. 2007;132:53–66.
Li N, Lundeberg T, Yu LC. Involvement of CGRP and CGRP1 receptor in nociception in the nucleus accumbens of rats. Brain Res. 2001;901:161–6.
Huang Y, Brodda-Jansen G, Lundeberg T, et al. Anti-nociceptive effects of calcitonin gene-related peptide in nucleus raphe magnus of rats: an effect attenuated by naloxone. Brain Res. 2000;873:54–9.
Yu LC, Weng XH, Wang JW, et al. Involvement of calcitonin gene-related peptide and its receptor in anti-nociception in the periaqueductal grey of rats. Neurosci Lett. 2003;349:1–4.
Pecile A, Guidobono F, Netti C, et al. Calcitonin gene-related peptide: antinociceptive activity in rats, comparison with calcitonin. Regul Pept. 1987;18:189–99.
Leem JW, Gwak YS, Lee EH, et al. Effects of iontophoretically applied substance P, calcitonin gene-related peptide on excitability of dorsal horn neurones in rats. Yonsei Med J. 2001;42:74–83.
Bird G, Han J, Fu Y, et al. Pain-related synaptic plasticity in spinal dorsal horn neurons: role of CGRP. Mol Pain. 2006;2:31.
Miletic V, Tan H. Iontophoretic application of calcitonin gene-related peptide produces a slow and prolonged excitation of neurons in the cat lumbar dorsal horn. Brain Res. 1988;446:169–72.
Yu Y, Lundeberg T, Yu LC. Role of calcitonin gene-related peptide and its antagonist on the evoked discharge frequency of wide dynamic range neurons in the dorsal horn of the spinal cord in rats. Regul Pept. 2002;103:23–7.
Oku R, Satoh M, Fujii N, et al. Calcitonin gene-related peptide promotes mechanical nociception by potentiating release of substance P from the spinal dorsal horn in rats. Brain Res. 1987;403:350–4.
Cridland RA, Henry JL. Effects of intrathecal administration of neuropeptides on a spinal nociceptive reflex in the rat: VIP, galanin, CGRP, TRH, somatostatin and angiotensin II. Neuropeptides. 1988;11:23–32.
Kawamura M, Kuraishi Y, Minami M, et al. Antinociceptive effect of intrathecally administered antiserum against calcitonin gene-related peptide on thermal and mechanical noxious stimuli in experimental hyperalgesic rats. Brain Res. 1989;497:199–203.
Yu L-C, Hansson P, Lundeberg T. The calcitonin gene-related peptide antagonist CGRP8-37 increases the latency to withdrawal responses in rats. Brain Res. 1994;653:223–30.
Gamse R, Saria A. Nociceptive behavior after intrathecal injections of substance P, neurokinin A and calcitonin gene-related peptide in mice. Neurosci Lett. 1986;70:143–7.
Segond von Banchet G, Pastor A, Biskup C, et al. Localization of functional calcitonin gene-related peptide binding sites in a subpopulation of cultured dorsal root ganglion neurons. Neuroscience. 2002;110:131–45.
Natura G, von Banchet GS, Schaible HG. Calcitonin gene-related peptide enhances TTX-resistant sodium currents in cultured dorsal root ganglion neurons from adult rats. Pain. 2005;116:194–204.
Schaible HG, Schmidt RF. Excitation and sensitization of fine articular afferents from cat's knee joint by prostaglandin E2. J Physiol. 1988;403:91–104.
Birrell GJ, McQueen DS, Iggo A, et al. PGI2-induced activation and sensitization of articular mechanonociceptors. Neurosci Lett. 1991;124:5–8.
Wang H, Ehnert C, Brenner GJ, et al. Bradykinin and peripheral sensitization. Biol Chem. 2006;387:11–4.
Mogil JS, Miermeister F, Seifert F, et al. Variable sensitivity to noxious heat is mediated by differential expression of the CGRP gene. Proc Natl Acad Sci. 2005;102:12938–43.
Shi X, Wang L, Li X, et al. Neuropeptides contribute to peripheral nociceptive sensitization by regulating interleukin-1beta production in keratinocytes. Anesth Analg. 2011;113:175–83.
Nakamura-Craig M, Gill BK. Effect of neurokinin A, substance P and calcitonin gene related peptide in peripheral hyperalgesia in the rat paw. Neurosci Lett. 1991;124:49–51.
Ferrari LF, Bogen O, Levine JD. Nociceptor subpopulations involved in hyperalgesic priming. Neuroscience. 2010;165:896–901.
Sun RQ, Lawand NB, Lin Q, et al. Role of calcitonin gene-related peptide in the sensitization of dorsal horn neurons to mechanical stimulation after intradermal injection of capsaicin. J Neurophysiol. 2004;92:320–6.
Yan Y, Yu LC. Involvement of opioid receptors in the CGRP8-37-induced inhibition of the activity of wide-dynamic-range neurons in the spinal dorsal horn of rats. J Neurosci Res. 2004;77:148–52.
Lofgren O, Yu LC, Theodorsson E, et al. Intrathecal CGRP(8-37) results in a bilateral increase in hindpaw withdrawal latency in rats with a unilateral thermal injury. Neuropeptides. 1997;31:601–7.
Kuraishi Y, Nanayama T, Ohno H, et al. Antinociception induced in rats by intrathecal administration of antiserum against calcitonin gene-related peptide. Neurosci Lett. 1988;92:325–9.
Bowler KE, Worsley MA, Broad L, et al. Evidence for anti-inflammatory and putative analgesic effects of a monoclonal antibody to calcitonin gene-related peptide. Neuroscience. 2013;228:271–82.
Eftekhari S, Edvinsson L. Possible sites of action of the new calcitonin gene-related peptide receptor antagonists. Ther Adv Neurol Disord. 2010;3:369–78.
Goadsby PJ, Charbit AR, Andreou AP, et al. Neurobiology of migraine. Neuroscience. 2009;161:327–41.
Uddman R, Edvinsson L, Ekman R, et al. Innervation of the feline cerebral vasculature by nerve fibers containing calcitonin gene-related peptide: trigeminal origin and co-existence with substance P. Neurosci Lett. 1985;62:131–6.
Fischer MJM. Calcitonin gene-related peptide receptor antagonists for migraine. Expet Opin Investig Drugs. 2010;19:815–23.
Goadsby PJ, Edvinsson L, Ekman R. Release of vasoactive peptides in the extracerebral circulation of humans and the cat during activation of the trigeminovascular system. Ann Neurol. 1988;23:193–6.
Goadsby PJ, Edvinsson L, Ekman R. Vasoactive peptide release in the extracerebral circulation of humans during migraine headache. Ann Neurol. 1990;28:183–7.
Lassen L, Haderslev P, Jacobsen V, et al. Cgrp may play a causative role in migraine. Cephalalgia. 2002;22:54–61.
Olesen J, Diener HC, Husstedt IW, et al. Calcitonin gene-related peptide receptor antagonist BIBN 4096 BS for the acute treatment of migraine. N Engl J Med. 2004;350:1104–10.
Ho TW, Mannix LK, Fan X, et al. Randomized controlled trial of an oral CGRP receptor antagonist, MK-0974, in acute treatment of migraine. Neurology. 2008;70:1304–12.
Fischer MJ, Koulchitsky S, Messlinger K. The nonpeptide calcitonin gene-related peptide receptor antagonist BIBN4096BS lowers the activity of neurons with meningeal input in the rat spinal trigeminal nucleus. J Neurosci. 2005;25:5877–83.
Kulchitsky S, Fischer M, Messlinger K. Calcitonin gene-related peptide receptor inhibition reduces neuronal activity induced by prolonged increase in nitric oxide in the rat spinal trigeminal nucleus. Cephalalgia. 2009;29:408–17.
Storer RJ, Akerman S, Goadsby PJ. Calcitonin gene-related peptide (CGRP) modulates nociceptive trigeminovascular transmission in the cat. Br J Pharmacol. 2004;142:1171–81.
Doods H, Hallermayer G, Wu D, et al. Pharmacological profile of BIBN4096BS, the first selective small molecule CGRP antagonist. Br J Pharmacol. 2000;129:420–3.
Ahmed AS, Li J, Erlandsson-Harris H, et al. Suppression of pain and joint destruction by inhibition of the proteasome system in experimental osteoarthritis. Pain. 2012;153:18–26.
Ashraf S, Wibberley H, Mapp PI, et al. Increased vascular penetration and nerve growth in the meniscus: a potential source of pain in osteoarthritis. Ann Rheum Dis. 2011;70:523–9.
Saxler G, Loer F, Skumavc M, et al. Localization of SP- and CGRP-immunopositive nerve fibers in the hip joint of patients with painful osteoarthritis and of patients with painless failed total hip arthroplasties. Eur J Pain. 2007;11:67–74.
Saito T, Koshino T. Distribution of neuropeptides in synovium of the knee with osteoarthritis. Clin Orthop Relat Res. 2000;376:172-82.
Orita S, Ishikawa T, Miyagi M, et al. Pain-related sensory innervation in monoiodoacetate-induced osteoarthritis in rat knees that gradually develops neuronal injury in addition to inflammatory pain. BMC Musculoskelet Disord. 2011;12:134.
Ferreira-Gomes J, Adaes S, Sarkander J, et al. Phenotypic alterations of neurons that innervate osteoarthritic joints. Arthritis Rheum. 2010;62:3677–3685.
Kalff KM, El Mouedden M, van Egmond J, et al. Pre-treatment with capsaicin in a rat osteoarthritis model reduces the symptoms of pain and bone damage induced by monosodium iodoacetate. Eur J Pharmacol. 2010;641:108–13.
Marshall KW, Theriault E, Homonko DA. Distribution of substance P and calcitonin gene related peptide immunoreactivity in the normal feline knee. J Rheumatol. 1994;21:883–9.
Schwab W, Bilgicyildirim A, Funk RH. Microtopography of the autonomic nerves in the rat knee: a fluorescence microscopic study. Anat Rec. 1997;247:109–18.
Guzman R, Evans M, Bove S, et al. Mono-iodoacetate-induced histologic changes in subchondral bone and articular cartilage of rat femorotibial joints: an animal model of osteoarthritis. Toxicol Pathol. 2003;31:619–24.
Fernihough J, Gentry C, Malcangio M, et al. Pain related behaviour in two models of osteoarthritis in the rat knee. Pain. 2004;112:83–93.
Ivanavicius S, Ball A, Heapy C, et al. Structural pathology in a rodent model of osteoarthritis is associated with neuropathic pain: increased expression of ATF-3 and pharmacological characterisation. Pain. 2007;128:272–82.
Bove S, Calcaterra S, Brooker R, et al. Weight bearing as a measure of disease progression and efficacy of anti-inflammatory compounds in a model of monosodium iodoacetate-induced osteoarthritis. Osteoarthr Cartil. 2003;11:821–30.
Kelly S, Dunham JP, Murray F, et al. Spontaneous firing in C-fibers and increased mechanical sensitivity in A-fibers of knee joint-associated mechanoreceptive primary afferent neurones during MIA-induced osteoarthritis in the rat. Osteoarthr Cartil. 2012;20:305–13.
Schuelert N, McDougall JJ. Involvement of Nav 1.8 sodium ion channels in the transduction of mechanical pain in a rodent model of osteoarthritis. Arthritis Res Ther. 2012;14:R5.
Acknowledgments
The authors’ research is supported by grants from University of Nottingham and Arthritis Research UK (grant number 18769).
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
Mr. Craig M. Bullock has been awarded a PhD studentship by the University of Nottingham.
Dr. Sara Kelly has received a grant funded by Arthritis Research UK (#18769) and BBSRC and Pain Relief Foundation. Dr. Kelly is employed as a lecturer at the University of Nottingham.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Pain Aspects of Arthritis
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Bullock, C.M., Kelly, S. Calcitonin Gene-Related Peptide Receptor Antagonists: Beyond Migraine Pain—A Possible Analgesic Strategy for Osteoarthritis?. Curr Pain Headache Rep 17, 375 (2013). https://doi.org/10.1007/s11916-013-0375-2
Published:
DOI: https://doi.org/10.1007/s11916-013-0375-2