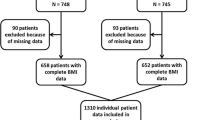
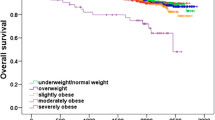
Abstract
Purpose of Review
Obesity is a recognized risk factor for the development of breast cancer and recurrence even when patients are treated appropriately. We reviewed the literature that addresses the impact of obesity on diagnosis and the individual therapeutic interventions, and present a summary of the findings.
Recent Findings
Compared to non-obese women with breast cancer, obese women with breast cancer have a worse disease-free and overall survival despite appropriate local and systemic therapies. In brief, obese breast cancer patients experience more complications related to surgery, radiation, and chemotherapy. Further, obese patients are at increased risk for local recurrence compared to normal-weight women. Similarly, systemic chemotherapy is less effective, even when dosed appropriately on the basis of actual weight. Overall, endocrine therapy is less effective in obese women, and there is a suggestion that aromatase inhibitors may be selectively less effective than tamoxifen. Obese women are less likely to undergo breast reconstruction than normal-weight women, and those who do have surgery experience more surgical complications.
Summary
The efficacy of cancer treatments is significantly lower in obese breast cancer survivors, posing greater challenges in patient care and disease management in this patient population. Further investigations are warranted to assess the effects on treatment outcomes and optimize therapeutic mechanisms in order to successfully target breast cancer associated with obesity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
We are in the midst of an obesity epidemic occurring in a society with an increasing incidence of breast cancer. A better understanding of the relationship between obesity and breast cancer is critical because there are unique diagnostic and treatment challenges in this patient population. Obesity was first reported to have an impact on breast cancer diagnosis and outcome in 1976 when Abe et al. reported that obese breast cancer patients, defined as > 20% body weight over their standard weight (height in cm − 100) × 0.9, had larger primary tumors, higher rates of lymphatic invasion, and worse overall survival compared to normal-weight patients [1]. Obesity is associated with worse breast cancer-specific survival [2]. A recent meta-analysis noted that obese women with breast cancer experience up to an 11% decrease in overall survival, regardless of menopausal status [2]. Further complicating survival outcomes, obesity is an established risk factor for metabolic syndrome, type 2 diabetes, and cardiovascular events [3], with the latter continuing to be the main cause of mortality in women with early-stage breast cancer [4]. The reasons for the adverse effects of obesity on breast cancer are numerous and complex. The purpose of this review is to provide an overview of the physical, psychosocial, and biological factors that contribute to a higher incidence of breast cancer and worse clinical outcomes in the overweight and obese patient population.
Breast Cancer Diagnosis
Obesity impacts the breast cancer trajectory as early as diagnosis. In general, overweight and obese women have poorer compliance with healthy habits and are less likely to comply with screening recommendations such as mammography. A meta-analysis of 16 studies addressed the relationship between body mass index (BMI) and mammography in women aged > 40 and found that overweight women were less likely than normal-weight women to have had a mammogram in the prior 2 years [5]. This was especially true in women with the highest BMI and in Caucasian women, but not in African American women. In looking at barriers to screening to mammography in women aged 50–69, a Kaiser Permanente study found that non-compliance was higher in obese women and that they were twice as likely to cite pain with the procedure as a reason for non-compliance [6].
One might hypothesize that obese women have more fatty tissue in their breasts and that this should decrease breast density, making the mammographic identification of a cancer more obvious. In contrast, it has been shown that the sensitivity of mammography is similar in obese and non-obese women. Older series reported that false-positive findings were 20% higher in obese women compared to normal- or low-weight women [7]. However, a prospective study using mammography registries from seven states conducted on 287,115 postmenopausal women who were not on hormone replacement therapy addressed the impact of screening mammography on breast cancer incidence. Between 1996 and 2005, 614,562 exams were performed and 4446 women were diagnosed with breast cancer. The incidence of hormone receptor (HR)-positive, but not HR-negative, breast cancer increased with increasing BMI. While the false-negative rate was comparable among women with the entire range of BMIs, screen-detected cancers were identified more often and at a more advanced stage in obese women [8]. From this well-designed trial, it was concluded that neither patterns of use nor mammographic accuracy contributed to the increased incidence in obese women. Their findings were further reinforced by the publication of a more recent series. Taken together, the data suggest that obesity is an independent risk for breast cancer [9•].
Patient and Physician-Related Barriers
Psychosocial factors contribute to the challenge of diagnosing and treating obese women with breast cancer. In general, women of lower socioeconomic status are more likely to be obese than women of higher socioeconomic status and this may complicate access to medical care [10]. The stigma of obesity results in fear, fatalism, alienation, low self-esteem, and embarrassment, all of which contribute to lower adherence to screening and treatment guidelines [11,12,13]. Friedman et al. qualitatively examined the reasons why some obese breast cancer patients undergo cancer screening. In-depth interviews of 33 women with a mean weight of 263 pounds (sd 45) were recorded for 60–90 min [14]. Friedman et al. verified that obese breast cancer patients share many of the barriers previously identified in the general population including embarrassment, fear of pain, fear of cancer, and competing demands on their time, suggesting that these barriers are not unique to obese breast cancer survivors. A major finding in this study was that an individual’s personality is an important mediator of health behavior. Those participants who followed through on cancer screenings shared certain personality traits compared with unscreened women, such as conscientiousness or self-regulatory ability, which allowed them to complete difficult or feared tasks. The attitudes of healthcare providers may also be a contributing factor in the creation of barriers to healthcare among obese patients. All too frequently, healthcare providers may reflect negative societal attitudes toward obese individuals and this may impact perceptions, judgments, and decision making [15].
Surgery
Lumpectomy and Radiation
Patients who are obese are at increased risk for complications with anesthesia as they may be more difficult to intubate and maintain ventilatory support than normal-weight women. An increase in the number of comorbid conditions associated with obesity further increases the risk of general anesthesia [16, 17]. Breast-conserving surgery (BCS) or lumpectomy has the advantage of requiring less time in the operating room and therefore less time requiring anesthesia compared to mastectomy. Following BCS, patients receive radiation therapy to the breast and, depending on their stage, may receive regional nodal radiation as well. While some series identify comparable local control rates in obese and normal-weight women undergoing lumpectomy and radiation therapy, others suggest that local recurrence is significantly higher in obese patients [18, 19•, 20]. What is clearer is that the cosmetic outcome with breast conservation (lumpectomy and radiation) may not be as good in obese women compared to normal-weight women. Serial assessments of breast cosmesis following lumpectomy and radiation therapy have identified persistent changes in the appearance of the breast. Two years after radiation, 31.1% of women with large breasts have documented adverse changes in the breast compared to women with medium-sized (16.3%) and small-sized breasts (4.8%) [21]. Even though more favorable cosmetic outcomes have generally been reported for BCS than mastectomy, as many as 40% of all patients who underwent BCS were dissatisfied with the asymmetry and deformity and these outcomes deteriorate over time [22, 23]. While BCS is associated with a positive body image [24], patients with a BMI > 30 kg/m2 were noted to have more postoperative breast asymmetry and less favorable esthetic results [25, 26].
Mastectomy
While small series from individual cancer centers have reported comparable rates of surgical complications in obese and normal-weight women undergoing mastectomy, larger series which include multiple institutions suggest otherwise. Recent data from the ACS-NSQIP database of 7202 women found that obesity was associated with an increase in both minor and major surgical complications, specifically increased risk of bleeding complications and surgical site infections [27].
Sentinel Node Mapping
Surgical staging of the axilla is determined by sentinel node mapping in which the primary lymph nodes involved in lymphatic drainage of the breast are removed in order to determine if breast cancer has spread beyond the breast. This procedure has significantly reduced the number of women requiring axillary lymph node dissections, thereby decreasing the incidence of lymphedema [28,29,30]. However, data from large multi-institutional studies indicate that sentinel lymph node (SLN) identification rates are lower in obese women, and are associated with a higher failure to map rate [31,32,33,34].
Reconstruction
With the rise in both rates of obesity and breast cancer, more obese women are seeking reconstruction following mastectomy. Obese women may not be candidates for reconstruction due to limited reconstructive options or due to comorbidities. A recent systematic review of 29 studies demonstrated that obese women were significantly more likely to have surgical and medical complications following reconstruction compared to normal-weight women. The rate of surgical complications was 2.3 times higher than in non-obese patients, with wound dehiscence being 2.5 times more likely in obese patients. Additionally, obese patients were 2.8 times more likely to have medical complications and have 1.9 times higher risk of reoperation [35•]. Complication rates are also higher in autologous-based (flap) reconstructions in obese patients and include flap failure (both partial and complete), hematomas, necrosis, donor site infections, delayed healing of donor site, seromas, and hernias [36,37,38,39]. A separate systematic review evaluating the risk of complications in obese women undergoing breast reconstruction identified that a BMI of 40 was a threshold at which the rate of complications became prohibitively high [39]. Breast reconstruction is ultimately an elective procedure and the high risk of complications in this population may make surgery inadvisable. Additionally, obese patients undergoing breast reconstruction are more likely to be disappointed with the esthetic results [40].
Radiotherapy
Patients with large breasts may receive increased doses of radiotherapy to critical organs such as the heart or lungs owing to the positioning of the breasts on the chest wall when the patient lies supine [41]. One way to minimize the toxicities associated with radiotherapy in patients with higher BMI and/or large breasts is to use prone whole-breast radiation, which has been found to result in favorable toxicity profiles [42]. Additionally, hypo-fractionated radiotherapy is possible in patients with large breast volumes; however, moist desquamation was found to be four times higher in patients with large breasts compared to those with smaller breasts [43].
Lymphedema
Lymphedema (LE) is a dreaded complication of treatment for breast cancer. There are known risk factors associated with of the development of LE including the number of lymph nodes removed during surgery, development of surgical complications such as infection or seroma, use of chemotherapy, radiation therapy, and comorbid medical conditions, including obesity. In a recent study, lymphedema rates were also found to be higher in patients undergoing axillary lymph node dissection, and among those with a more advanced stage of disease, in addition to having a higher BMI [12, 14, 32]. Additionally, not only are obese women at greater risk for developing post-operative lymphedema, but obese women are also at a higher risk of pre-operative lymphedema [44•, 45].
Systemic Therapy
In the adjuvant setting, full doses of chemotherapy are associated with a greater improvement in overall survival [46]. The appropriate dosing of cytotoxic agents for obese women is challenging and underdosing may impact disease outcomes in obese women. Recently, the American Society of Clinical Oncology published guidelines for the “Appropriate Chemotherapy Dosing for Obese Adult Patients With Cancer” [47]. These guidelines were based upon a systematic review in adult survivors with breast, ovarian, colon, or lung cancer, and suggested that reduced doses of chemotherapy may result in poorer disease-free and overall survival. Importantly, there is no clear evidence that short- or long-term toxicities from chemotherapy are increased in obese breast cancer patients receiving full-weight–based doses. The use of fixed-dose chemotherapy is only applicable for certain chemotherapeutic agents (e.g., carboplatin and bleomycin) [47]. Obese patients require higher doses of chemotherapy to achieve therapeutic levels of tumor suppression [48•, 49]. Ewertz et al. reported the 30-year follow-up of 53,816 women enrolled in clinical trials within the Danish Breast Cancer Cooperative Group (DBCCG), of which 18,979 patients had information about BMI [18]. In this study, obesity was associated with an increased risk for developing distant metastatic disease and of dying of breast cancer. Furthermore, the benefits of adjuvant chemotherapy and/or endocrine therapy were significantly less in the obese population, even among women who received appropriate doses of chemotherapy. These findings were independent of tumor size, nodal status, and known prognostic factors, including HR status of the primary tumor [50].
The therapeutic dose of individual endocrine agents is fixed, regardless of weight or body surface area. The Austrian Breast and Colorectal Cancer Study Group (ABCSG) 12 trial randomized 1804 premenopausal women with early-stage breast cancer treated with a luteinizing hormone-releasing hormone (LHRH) agonist for ovarian suppression, to receive adjuvant tamoxifen or anastrozole with a second randomization to zoledronic acid or not [51•]. They reported that overweight women assigned anastrozole had a 60% increase risk of disease recurrence (HR, 1.60; 95% CI, 1.06 to 2.41; P = 0.02) and death (HR, 2.14; 95% CI, 1.17 to 3.92; P = 0.01), compared to normal-weight survivors. Importantly, the benefits of adjuvant chemotherapy and/or endocrine therapy were significantly less in the obese population and were independent of tumor size, nodal status, and known prognostic factors, including hormone receptor status.
In addition to the DBCCG study, other smaller series have reported less benefit from endocrine therapy in the obese population, independent of tumor size, nodal status, and known prognostic factors, including HR status [18]. In the Arimidex, Tamoxifen Alone or in Combination (ATAC) trial, Sestak et al. reported that postmenopausal breast cancer survivors with a high BMI (BMI > 35 kg/m2) at baseline had a significantly higher rate of breast cancer recurrence after 100 months follow-up compared to normal BMI survivors (HR, 1.39; 95% CI, 1.07 to 1.61; P = 0.01) [52]. Breast cancer survivors who received anastrozole had a 27% lower recurrence rate than women who received tamoxifen, and the benefit of anastrozole was greater in lower BMI survivors. In contrast, the efficacy of anastrozole was significantly (P < 0.01) less effective (higher recurrence rate) in postmenopausal breast cancer survivors with a BMI higher than 30 kg/m2 (HR, 1.30; 95% CI, 0.91 to 1.85) compared to those women with a BMI lower than 28 kg/m2 (HR, 1.29; 95% CI, 0.92 to 1.81). These data collectively suggest a worse prognosis for overweight and obese women treated with endocrine agents and suggest that aromatase inhibitors may be less effective than tamoxifen in this population [52].
Biological Factors
Fat is a metabolically active tissue with high levels of the aromatase enzyme which converts androgen to estrogen. Excess estrogen production from expanded adipose tissue has been proposed as a possible mechanism for the adverse outcomes in obese women with breast cancer. However, obesity is associated with adverse disease outcomes in obese women with hormone-sensitive and hormone-resistant cancers [18]. Furthermore, obesity is a risk factor for developing triple-negative breast cancer, which suggests that higher endogenous estrogens may not be the only mechanism contributing to a higher risk of recurrence [53]. Obesity produces inflammation in adipose tissue, and activated macrophages in adipose tissues of obese individuals produce pro-inflammatory mediators such as TNFα and IL-6. Dysregulated inflammation in adipose tissues results in an accumulation of pro-inflammatory T cells and reduction in T regs, which contributes to obesity-related insulin resistance. STAT3 activity is increased in visceral adipose tissues and ablation of STAT3 in T cells has been shown to improve insulin sensitivity and glucose tolerance and reduce inflammation in visceral adipose tissues [54]. JAK2/STAT3 is a regulator of lipid metabolism and promotes breast cancer cell “stemness” and chemoresistance [55]. The biologic basis for the differences in the natural history of breast cancer in obese women is not completely clear. However, biologically active adipose tissues appear to be a contributing factor to the unique pathophysiology in obese women with breast cancer.
Conclusion
Obese women with breast cancer represent a unique patient population. They are at increased risk for the development of breast cancer and may experience more complications related to surgery and radiation. Despite appropriate local disease treatment, obese women are at increased risk for local recurrence compared to normal-weight women. Similarly, systemic chemotherapy appears to be less effective, even when dosed appropriately on the basis of actual weight. In addition, endocrine therapy may be less effective in obese women, and there is a suggestion that tamoxifen may be more effective than the aromatase inhibitors in this population. Taken together, these data suggest a unique and aggressive biology that is likely due to a tumor environment metabolically activated by adipose tissues. Overall, it is clear that the efficacy of cancer treatments is significantly lower in obese breast cancer survivors, posing greater challenges in patient care and disease management in this patient population. Based upon these challenges, further investigations are warranted to assess the effective diagnostic and treatment mechanisms needed to successfully target breast cancer within the obese patient population.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Abe R, et al. Biological characteristics of breast-cancer in obesity. Tohoku J Exp Med. 1976;120(4):351–9.
Protani M, Coory M, Martin JH. Effect of obesity on survival of women with breast cancer: systematic review and meta-analysis. Breast Cancer Res Treat. 2010;123(3):627–35.
Smith CJ, Ryckman KK. Epigenetic and developmental influences on the risk of obesity, diabetes, and metabolic syndrome. Diabetes Metab Syndr Obes. 2015;8:295–302.
Bradshaw PT, Stevens J, Khankari N, Teitelbaum SL, Neugut AI, Gammon MD. Cardiovascular disease mortality among breast cancer survivors. Epidemiology. 2016;27(1):6–13.
Maruthur NM, Bolen S, Brancati FL, Clark JM. Obesity and mammography: a systematic review and meta-analysis. J Gen Intern Med. 2009;24(5):665–77.
Feldstein AC, Perrin N, Rosales AG, Schneider J, Rix MM, Glasgow RE. Patient barriers to mammography identified during a reminder program. J Women's Health. 2011;20(3):421–8.
Elmore JG, Carney PA, Abraham LA, Barlow WE, Egger JR, Fosse JS, et al. The association between obesity and screening mammography accuracy. Arch Intern Med. 2004;164(10):1140–7.
Kerlikowske K, Walker R, Miglioretti DL, Desai A, Ballard-Barbash R, Buist DSM. Obesity, mammography use and accuracy, and advanced breast cancer risk. JNCI J Natl Cancer Inst. 2008;100(23):1724–33.
• Engmann NJ, et al. Population-attributable risk proportion of clinical risk factors for breast cancer. JAMA Oncol. 2017;3(9):1228–36. This study identified that breast cancer could potentially be averted if obese women attained a body mass index of less than 25.
Newton S, Braithwaite D, Akinyemiju TF. Socio-economic status over the life course and obesity: systematic review and meta-analysis. PLoS One. 2017;12(5):e0177151.
Hum S, Wu M, Pruthi S, Heisey R. Physician and patient barriers to breast cancer preventive therapy. Curr Breast Cancer Rep. 2016;8(3):158–64.
Phelan SM, Griffin JM, Hellerstedt WL, Sayer NA, Jensen AC, Burgess DJ, et al. Perceived stigma, strain, and mental health among caregivers of veterans with traumatic brain injury. Disabil Health J. 2011;4(3):177–84.
Link BG, Struening EL, Neese-Todd S, Asmussen S, Phelan JC. Stigma as a barrier to recovery: the consequences of stigma for the self-esteem of people with mental illnesses. Psychiatr Serv. 2001;52(12):1621–6.
Friedman AM, Hemler JR, Rossetti E, Clemow LP, Ferrante JM. Obese women’s barriers to mammography and Pap smear: the possible role of personality. Obesity. 2012;20(8):1611–7.
Phelan SM, Burgess DJ, Yeazel MW, Hellerstedt WL, Griffin JM, van Ryn M. Impact of weight bias and stigma on quality of care and outcomes for patients with obesity. Obes Rev. 2015;16(4):319–26.
Shearer ES III. Obesity anaesthesia: the dangers of being an apple. Br J Anaesth. 2013;110(2):172–4.
Pandit JJ, Andrade J, Bogod DG, Hitchman JM, Jonker WR, Lucas N, et al. 5th National Audit Project (NAP5) on accidental awareness during general anaesthesia: summary of main findings and risk factors†‡. Br J Anaesth. 2014;113(4):549–59.
Ewertz M, Jensen MB, Gunnarsdóttir KÁ, Højris I, Jakobsen EH, Nielsen D, et al. Effect of obesity on prognosis after early-stage breast cancer. J Clin Oncol. 2011;29(1):25–31.
• Bergom C, et al. The association of local-regional control with high body mass index in women undergoing breast conservation therapy for early stage breast cancer. Int J Radiat Oncol Biol Phys. 2016;96(1):65–71. This study identified that BMI is significantly associated with the locoregional recurrence-free interval and distant recurrence-free interval/overall survival, which implies that a higher BMI predicts a worse distant recurrence-free interval and overall survival.
Warren LEG, Ligibel JA, Chen YH, Truong L, Catalano PJ, Bellon JR. Body mass index and locoregional recurrence in women with early-stage breast cancer. Ann Surg Oncol. 2016;23(12):3870–9.
Moody AM, Mayles WPM, Bliss JM, A'Hern RP, Owen JR, Regan J, et al. The influence of breast size on late radiation effects and association with radiotherapy dose inhomogeneity. Radiother Oncol. 1994;33(2):106–12.
Waljee JF, Hu ES, Ubel PA, Smith DM, Newman LA, Alderman AK. Effect of esthetic outcome after breast-conserving surgery on psychosocial functioning and quality of life. J Clin Oncol. 2008;26(20):3331–7.
D'Aniello C, Grimaldi L, Barbato A, Bosi B, Carli A. Cosmetic results in 242 patients treated by conservative surgery for breast cancer. Scand J Plast Reconstr Surg Hand Surg. 1999;33(4):419–22.
Vrieling C, Collette L, Fourquet A, Hoogenraad WJ, Horiot JC, Jager JJ, et al. The influence of the boost in breast-conserving therapy on cosmetic outcome in the EORTC “boost versus no boost” trial. EORTC Radiotherapy and Breast Cancer Cooperative Groups. European Organization for Research and Treatment of Cancer. Int J Radiat Oncol Biol Phys. 1999;45(3):677–85.
Johansen J, Overgaard J, Rose C, Engelholm SA, Gadeberg CC, Kjaer M, et al. Cosmetic outcome and breast morbidity in breast-conserving treatment--results from the Danish DBCG-82TM national randomized trial in breast cancer. Acta Oncol. 2002;41(4):369–80.
Kern P, Zarth F, Kimmig R, Rezai M. Impact of age, obesity and smoking on patient satisfaction with breast implant surgery - a unicentric analysis of 318 implant reconstructions after mastectomy. Geburtshilfe Frauenheilkd. 2015;75(6):597–604.
Garland M, Hsu FC, Clark C, Chiba A, Howard-McNatt M. The impact of obesity on outcomes for patients undergoing mastectomy using the ACS-NSQIP data set. Breast Cancer Res Treat. 2018;168(3):723–6.
Krag D, Weaver D, Ashikaga T, Moffat F, Klimberg VS, Shriver C, et al. The sentinel node in breast cancer — a multicenter validation study. N Engl J Med. 1998;339(14):941–6.
Mansel RE, Fallowfield L, Kissin M, Goyal A, Newcombe RG, Dixon JM, et al. Randomized multicenter trial of sentinel node biopsy versus standard axillary treatment in operable breast cancer: the ALMANAC trial. J Natl Cancer Inst. 2006;98(9):599–609.
Krag DN, Anderson SJ, Julian TB, Brown AM, Harlow SP, Costantino JP, et al. Sentinel-lymph-node resection compared with conventional axillary-lymph-node dissection in clinically node-negative patients with breast cancer: overall survival findings from the NSABP B-32 randomised phase 3 trial. Lancet Oncol. 2010;11(10):927–33.
Derossis AM, Fey JV, Cody HS III, Borgen PI. Obesity influences outcome of sentinel lymph node biopsy in early-stage breast cancer. J Am Coll Surg. 2003;197(6):896–901.
Cox CE, Dupont E, Whitehead GF, Ebert MD, Nguyen K, Peltz ES, et al. Age and body mass index may increase the chance of failure in sentinel lymph node biopsy for women with breast cancer. Breast J. 2002;8(2):88–91.
Lyman GH, Giuliano AE, Somerfield MR, Benson AB 3rd, Bodurka DC, Burstein HJ, et al. American Society of Clinical Oncology guideline recommendations for sentinel lymph node biopsy in early-stage breast cancer. J Clin Oncol. 2005;23(30):7703–20.
Lyman GH, Somerfield MR, Bosserman LD, Perkins CL, Weaver DL, Giuliano AE. Sentinel lymph node biopsy for patients with early-stage breast cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2017;35(5):561–4.
• Panayi AC, et al. Impact of obesity on outcomes in breast reconstruction: a systematic review and meta-analysis. J Reconstr Microsurg. 2018;34(05):363–75. This study identified that BMI is significantly associated with the locoregional recurrence-free interval and distant recurrence-free interval/overall survival, which implies that a higher BMI predicts a worse distant recurrence-free interval and overall survival.
Fischer JP, Nelson JA, Kovach SJ, Serletti JM, Wu LC, Kanchwala S. Impact of obesity on outcomes in breast reconstruction: analysis of 15,937 patients from the ACS-NSQIP datasets. J Am Coll Surg. 2013;217(4):656–64.
Fischer JP, Nelson JA, Serletti JM, Kanchwala S. Breast reconstruction in the morbidly obese patient: assessment of 30-day complications using the 2005 to 2010 National Surgical Quality Improvement Program Data Sets. Plast Reconstr Surg. 2014;133(6):882e–3e.
Quinn TT, Miller GS, Rostek M, Cabalag MS, Rozen WM, Hunter-Smith DJ. Prosthetic breast reconstruction: indications and update. Gland Surg. 2016;5(2):174–86.
Schaverien MV, Mcculley SJ. Effect of obesity on outcomes of free autologous breast reconstruction: a meta-analysis. Microsurgery. 2014;34(6):484–97.
Kiebert GM, de Haes JC, van de Velde CJ. The impact of breast-conserving treatment and mastectomy on the quality of life of early-stage breast cancer patients: a review. J Clin Oncol. 1991;9(6):1059–70.
Allen AM, et al. Body mass index predicts the incidence of radiation pneumonitis in breast cancer patients. Cancer J. 2005;11(5):390–8.
Krengli M, Masini L, Caltavuturo T, Pisani C, Apicella G, Negri E, et al. Prone versus supine position for adjuvant breast radiotherapy: a prospective study in patients with pendulous breasts. Radiat Oncol. 2013;8(1):232.
Dorn PL, Corbin KS, al-Hallaq H, Hasan Y, Chmura SJ. Feasibility and acute toxicity of hypofractionated radiation in large-breasted patients. Int J Radiat Oncol Biol Phys. 2012;83(1):79–83.
• Erdogan IZ, et al. Preoperative lymphedema-related risk factors in early-stage breast cancer. Lymphat Res Biol. 2018;16(1):28–35. This study identified that BMI is significantly associated with the locoregional recurrence-free interval and distant recurrence-free interval/overall survival, which implies that a higher BMI predicts a worse distant recurrence-free interval and overall survival.
Helyer LK, et al. Obesity is a risk factor for developing postoperative lymphedema in breast cancer patients. Breast J. 2010;16(1):48–54.
Bonadonna G, Valagussa P. Dose-response effect of adjuvant chemotherapy in breast cancer. N Engl J Med. 1981;304(1):10–5.
Griggs JJ, Mangu PB, Anderson H, Balaban EP, Dignam JJ, Hryniuk WM, et al. Appropriate chemotherapy dosing for obese adult patients with cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2012;30(13):1553–61.
• Carroll JP, et al. Toxicity and tolerability of adjuvant breast cancer chemotherapy in obese womenMed Oncol. 2014;31(4). This study showed that obese women received chemotherapy with a proportionally lower mean relative dose intensity than non-obese women (94 vs. 97% of reference dose, P = 0.03), which mainly suggests that obese women receive different chemotherapy regimens and relatively lower chemotherapy doses, compared to non-obese women.
Griggs JJ, Culakova E, Sorbero MES, van Ryn M, Poniewierski MS, Wolff DA, et al. Effect of patient socioeconomic status and body mass index on the quality of breast cancer adjuvant chemotherapy. J Clin Oncol. 2007;25(3):277–84.
Early Breast Cancer Trialists’ Collaborative, G, et al. Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet. 2011;378(9804):1707–16.
• Pfeiler G, Königsberg R, Fesl C, Mlineritsch B, Stoeger H, Singer CF, et al. Impact of body mass index on the efficacy of endocrine therapy in premenopausal patients with breast cancer: an analysis of the prospective ABCSG-12 trial. J Clin Oncol. 2011;29(19):2653–9. This is one of the largest randomized controlled trial showing that overweight women assigned anastrozole had a 60% increased risk of disease recurrence and death, compared to normal-weight survivors.
Sestak I, Distler W, Forbes JF, Dowsett M, Howell A, Cuzick J. Effect of body mass index on recurrences in tamoxifen and anastrozole treated women: an exploratory analysis from the ATAC trial. J Clin Oncol. 2010;28(21):3411–5.
Yang XR, Chang-Claude J, Goode EL, Couch FJ, Nevanlinna H, Milne RL, et al. Associations of breast cancer risk factors with tumor subtypes: a pooled analysis from the Breast Cancer Association Consortium studies. J Natl Cancer Inst. 2011;103(3):250–63.
Priceman SJ, Kujawski M, Shen S, Cherryholmes GA, Lee H, Zhang C, et al. Regulation of adipose tissue T cell subsets by Stat3 is crucial for diet-induced obesity and insulin resistance. Proc Natl Acad Sci. 2013;110(32):13079–84.
Wang TY, et al. JAK/STAT3-regulated fatty acid beta-oxidation is critical for breast cancer stem cell self-renewal and chemoresistance. Cell Metab. 2018;27(1):136–150.e5.
Acknowledgements
The authors thank Nicola M. Solomon, PhD, for editorial assistance and critical review of the manuscript.
Author information
Authors and Affiliations
Contributions
Conception and design of the study: Kyuwan Lee and Joanne E. Mortimer
Drafting the article or revising it critically for important intellectual content: Kyuwan Lee, Joanne E. Laura Kruper, Christina Dieli-Conwright
Final approval of the version to be submitted: Joanne E. Mortimer
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Breast Cancer
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Lee, K., Kruper, L., Dieli-Conwright, C.M. et al. The Impact of Obesity on Breast Cancer Diagnosis and Treatment. Curr Oncol Rep 21, 41 (2019). https://doi.org/10.1007/s11912-019-0787-1
Published:
DOI: https://doi.org/10.1007/s11912-019-0787-1