Abstract
Purpose of Review
In this review, we summarize the current understanding of consciousness including its neuroanatomic basis. We discuss major theories of consciousness, physical exam-based and electroencephalographic metrics used to stratify levels of consciousness, and tools used to shed light on the neural correlates of the conscious experience. Lastly, we review an expanded category of ‘disorders of consciousness,’ which includes disorders that impact either the level or experience of consciousness.
Recent Findings
Recent studies have revealed many of the requisite EEG, ERP, and fMRI signals to predict aspects of the conscious experience. Neurological disorders that disrupt the reticular activating system can affect the level of consciousness, whereas cortical disorders from seizures and migraines to strokes and dementia may disrupt phenomenal consciousness. The recently introduced memory theory of consciousness provides a new explanation of phenomenal consciousness that may explain better than prior theories both experimental studies and the neurologist’s clinical experience.
Summary
Although the complete neurobiological basis of consciousness remains a mystery, recent advances have improved our understanding of the physiology underlying level of consciousness and phenomenal consciousness.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Definition
The term “consciousness” often carries different definitions. In this article, we define “consciousness” as having two core components:
-
1.
Phenomenal consciousness: our personal, subjective experience of the present moment [1]. Understanding the neural basis of phenomenal consciousness is referred to by Chalmers as “The hard problem” [2].
-
2.
Level of consciousness: the state of wakefulness assessed by various scales described in terms such as comatose, stuporous, obtunded, delirious, confused, and alert. Level of consciousness can be thought of as a volume dial, upon which phenomenal consciousness is dependent.
Theories of Consciousness
There are at least twenty-two supported neurobiological explanations [see REF 3•] for the basis of consciousness. In this review, we will reference a few of the major theories.
Higher Order Theory (HOT) claims that consciousness depends on meta-representations, representations that occur higher in a processing hierarchy. For example, whereas the lower order representation of visual stimulus of a painting is represented in visual cortex as “red and green areas,” it only achieves consciousness when relayed to higher areas—specifically the pre-frontal cortex—allowing for the self-conscious statement, “I am having the experience of seeing a painting” [4, 5].
Global Workspace Theory (GWT) postulates a centralized resource shared by multiple independent threads of processing. Mental states are conscious if and only if they are available to a wide range of cognitive processing, which depends on a frontoparietal network accessible to multiple local processing networks throughout the cortex [6,7,8].
Integrated Information Theory (IIT) suggests that consciousness is defined by a system’s structural capability to produce irreducible, integrated information, a measurable and unidimensional quantity referred to as “Φ.” The brain regions with the highest Φ value are thought to be a temporo-parieto-occipital area, the “posterior hot zone” [9, 10].
Local Recurrency Theory (LRT) argues that recurrent processing in sensory cortices is sufficient to generate conscious experience while parietal and frontal regions are required only for reports and judgements pertaining to a stimulus [11, 12].
The Memory Theory of Consciousness (MToC) proposes that the phenomenonal consciousness evolved from, and functions as a part of, episodic memory and other explicit memory systems (sensory, working, and semantic memory) [13••]. This theory can explain long-standing puzzles in consciousness such as postdictive effects—the finding that conscious perception of a stimulus can be affected not only by conditions preceding a stimulus, but also by the conditions following a stimulus. The entire cerebral cortex and hippocampus function together to serially integrate and time stamp parallel unconscious brain processes to form a linear, coherent stream of conscious experiences. This theory proposes that the experience of consciousness is actually a memory, preceded temporally by unconscious processes of sensation, decisions, and/or actions. The theory also proposes that different cortical areas produce different aspects of consciousness, each with its own neural correlate [13••].
Each of these theories works towards understanding the neural correlates of consciousness—the neurobiological mechanisms necessary and sufficient to generate a conscious experience. A better understanding of consciousness and its neurobiology may improve the clinical care of patients with disorders of consciousness, including not only anoxia and severe traumatic brain injury but also delirium, epilepsy, migraine aura, cortical strokes, and cortical dementias.
Clinical Metrics of Levels of Consciousness
Level of consciousness, that is, the state of wakefulness, is commonly defined clinically through descriptors of psychomotor activity in response to stimulus. The Glasgow coma scale (GCS) is perhaps the most widely used clinical scale of consciousness, favored for its three metric simplicity (eye opening, verbal response, motor response). However, in recent years, it has drawn criticism for lack of content validity, standardization, and inter-examiner reliability [14, 15]. A simpler measure favored in the intensive care unit (ICU) setting is the Richmond Agitation-Sedation Scale, which plots wakefulness on a single dimension from combative behavior to complete unresponsiveness [16]. Another scale with high inter-examiner reliability, content validity, standardization, and prognostic capability is the Coma Recovery Scale Revised (CRS-S) [17,18,19]. See [REF 6] for other scales.
Patients may, however, be clinically unresponsive and maintain electroencephalographic (EEG) or functional MRI (fMRI) activity that suggests underlying consciousness [20,21,22,23]. In a large, ICU-based study of brain activity, Claasen et al. used EEG patterns to detect occult brain activity in response to spoken motor commands in patients with acute brain injury [24••], observing that about 15% of clinically unresponsive patients have a “conscious” EEG pattern in response to spoken commands. Follow-up investigations found that 41% of patients with covert consciousness make a complete recovery versus only 10% of patients without covert consciousness [25•].
EEG Activity as a Correlate of Consciousness
EEG is the gold standard in determining the sleep-wake phase and is a useful tool in assessing the level of consciousness. The Bispectral Index (BIS) is used by anesthesiologists to assist in the titration of anesthetics. Raw EEG data is collected from four electrodes on the forehead and analyzed by a proprietary, algorithm which outputs a unidimensional score, the BIS, which quantifies the level of wakefulness from 0 (comatose—flat line EEG) to 100 (awake—low amplitude, fast frequency) [26].
EEG frequency ranges and measures of connectivity have been considered as potential correlates of phenomenal consciousness (or at least aspects of it). The alpha frequency (8–12 hz), canonically considered to be an idling rhythm, has recently been thought to have a functional role in the inhibition of task-irrelevant information [27,28,29,30]. Pre-stimulus alpha activity predicts poor conscious perception; occipital alpha power might filter out which stimuli will be perceived consciously, and which stimuli will be deemed “task irrelevant” and ignored [31,32,33,34].
For example, Krasich et al. showed that about halfway between the maximum positive and negative peaks of occipital alpha activity, a stimulus is mostly likely to be consciously perceived [35••]. In the post-stimulus period, a drop in parietooccipital alpha power has been suggested as a marker of conscious perception [36]. Hutchinson et al. studied a paradigm of inattentional blindness and confirmed that alpha power 250–500 ms post stimulus is negatively associated with conscious perception [37].
ERP Activity as a Correlate of Consciousness
Event-related potentials (ERPs) are EEG changes time locked to sensory, motor, or cognitive events that provide a means to study electrophysiologic correlates of mental processes. ERPs provide excellent temporal resolution and are a practical (non-invasive, relatively inexpensive) method of studying cognitive functions [38].
The conscious perception of a stimulus depends on a variety of factors including internal features of the stimulus itself (such as its salience). External factors that affect stimulus perception include observer state (such as attentional) and context (such as masking) which may affect perception preceding, simultaneously, or following the stimulus [39]. Each of these factors can be experimentally controlled and modified in ERP study paradigms to determine the brain signals that are together necessary and sufficient for conscious perception—the neural correlates of consciousness [40].
Early ERP components, peaking within 100 ms after stimulus, are known as “sensory” or “exogenous,” as they depend mostly on the physical parameters of the stimulus itself. The middle and late components, generated 100–300 ms and 300–600 ms after stimulus, respectively, reflect stimulus evaluation and are termed “cognitive” or “endogenous” potentials [41].
Early ERP Components (P50, N100, and Others)
In their historic work, Libet et al. reported in 1967 that early ERP sensory evoked potentials over the primary somatosensory cortex are not always sufficient to cause perceptual experience [42]. This finding, replicated and expanded upon by others [43,44,45,46], started the search for other early potentials to determine the first signals sufficient to predict sensory experience, also known as perception.
Middle ERP components (P100, N140, ERN/Pe)
P100
The P100, a positive occipital ERP component occurring 100 ms after stimulus presentation, has been implicated as the earliest ERP involved in conscious perception and associated tasks [39, 47, 48]. Other work, however, found the P100 in both perceived and unperceived conditions [49].
N140
The N140 is a negative ERP peaking around 140 ms. Multiple studies highlight the N140 as important for perception but there is debate as to whether the signal should be interpreted as awareness, perception, detection, or attention [50,51,52]. Given the prolongation of the N140 in patients with clinical deficits in contralateral spatial attention, we favor this specifically as a marker of somatosensory hemi-attention rather than conscious perception [53].
Late ERP Components (late Pe, P300, VAN)
Our review of early and middle ERP components suggests that they are mainly correlated with attention or unconscious sensory processes. Later, ERP components may be more likely to represent true neural correlates of consciousness.
Positivity Performance Error (Pe)
The late positivity performance error (Pe) component (400–600 ms) has been associated with conscious awareness of error detection [54].
P300
The P300 is a posterior ERP observed 290–450 ms after a stimulus [55]. Historically, the P300, which includes components P3a (central maximum) and P3b (parietal maximum), has been thought to represent a gold-standard marker of conscious perception [56,57,58]. Recently, however, the P300 has been favored to specifically represent post perceptual processing, rather than phenomenal conscious awareness [See REF 59, 60• for in-depth review].
VAN
The visual awareness negativity (VAN) is a negative difference wave, observed 200 ms after stimulus presentation, most prominently over occipitotemporal sites, which differentiates aware from unaware states [61]. The VAN may be the earliest reliable electrophysiologic marker that correlates with aware perception after the presentation of a visual stimulus. There is an analogous auditory awareness negativity leading to recent proposals to consider these potentials as parts of a broader termed “perceptual awareness negativity” [62].
Connectivity as a Correlate of Consciousness
In the conscious patient, a TMS pulse sets off a complex chain of events as information reverberates through cortical brain regions, but in the unconscious patient, a similar pulse may induce only a short-lived local effect [63,64,65,66,67]. This finding supports and provides a testable metric for IIT that applies a connectivity approach to evaluate levels of consciousness.
Studying EEG recordings in patients with diminished levels of consciousness as determined by GCS, Toker et al. proposed that state of consciousness correlates with the proximity of slow cortical oscillatory potentials to a critical pattern described as the edge-of-chaos [68]. In a serial awakening paradigm, exploring EEG phase connectivity as a correlate of consciousness, patient states were differentiated across sleep stages by measures of EEG phase connectivity in posterior brain regions [69•].
Similarly, using fMRI, Demetrezi et al. found that compared to healthy controls, those with decreased level of consciousness showed impaired baseline connectivity in bilateral executive control, default mode, and auditory networks [70]. Luppi et al. employed resting-state fMRI with measures of integration and entropy to identify consciousness-specific patterns of brain function including temporal states of high integration and functional diversity [71••].
Aspects of Consciousness
If we assume that consciousness is unitary, prompting us to look for a single neural correlate of consciousness, then it is difficult to make sense of these divergent results from various studies using different measures of brain physiology. If, however, we take the approach suggested by the MToC—that there are various aspects of consciousness such as visual, auditory, emotional, phonological, motor, and more—then each study may reveal the neural correlate of consciousness for one aspect of consciousness but not others [13••]. This modular view of consciousness is entirely consistent with the disorders of consciousness encountered daily by the neurologist.
Disorders of Consciousness
Classically, this group included only disorders that directly impact the level of consciousness, such as TBI and cardiac arrest. Following the MToC, however, we broaden this definition to include disorders that affect phenomenal consciousness [13••].
Seizure
Epileptic seizures offer a window into the requisite anatomy and function for maintaining consciousness as different seizure subtypes affect consciousness differently. A generalized onset seizure includes bilateral motor involvement, loss of consciousness, and involvement of bilaterally distributed networks [72]. The fact that complete loss of consciousness in seizure requires generalized involvement of both hemispheres argues strongly that level of consciousness is a diffuse cortical phenomenon, consistent with MToC but not HOT, GWT, IIT, or LRT. Focal seizures are subclassified as impaired awareness or unimpaired awareness [73]. Focal seizures with altered awareness also impair phenomenal consciousness, as manifested by, for example, amnesia, automatisms, and behavioral arrest [74].
Migraines
Although migraine does not generally affect the level of consciousness, Budson et al. argued that migraine aura is, in fact, a focal disruption of phenomenal consciousness [13••]. Rarely, in the case of the basilar sub-type, migraine can affect the level of consciousness, causing confusion. More commonly, migraine aura results in the impairment of various aspects of phenomenal consciousness, such as visual associated with posterior onset of cortical spreading depression [75]. Positive followed by negative visual phenomena (scintillating scotoma) are common changes associated with focally impaired conscious awareness.
Strokes and Other Discrete Brain Lesions
Stroke, discrete in border and stable over time, is the prototype brain lesion and offers a means to evaluate the neuroanatomic basis of consciousness. We will first discuss how strokes affect the level of consciousness, and then how strokes can affect various aspects of phenomenal consciousness.
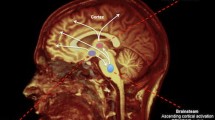
At onset, level of consciousness is impaired in about 36% of patients with large hemispheric infarcts and 14% of stroke patients as a whole [76, 77]. Clinical attention to level of consciousness is critical as early decreased consciousness may portend a 2.2-fold risk of in-hospital mortality [37]. Outside of infarcts that affect wide distributions of cortex (directly or indirectly through edema and hydrocephalus), the best described lesions affecting the level of consciousness are those within the reticular activating system (bi-thalamic and brainstem) [78,79,80]. In a highly powered brain mapping study of penetrating head lesions, no individual cortical regions were associated with loss of consciousness while lesioned region connectivity to the dorsal midbrain (i.e., reticular activating system) did correlate with loss of consciousness [81•].
Cortical strokes, especially when occurring bilaterally, may cause impairments to various aspects of phenomenal consciousness. For example, unilateral occipital infarcts impair hemifield visual consciousness and bilateral occipital infarcts may cause cortical blindness and a complete loss of conscious visual perception (despite some intact unconscious function) [82]. However, no single or bilateral cortical infarct, unless involving most of the cortex, leads to permanent unconsciousness. If there were a “hot zone” of consciousness (as suggested by HOT, GWT, IIT, and LRT), there should be a cortical stroke syndrome that devastates consciousness; no such cortical lesion has been described. By contrast, MToC suggests that cortical strokes will impair aspects of phenomenal consciousness without affecting the level of consciousness.
Delirium
Delirium is a syndrome characterized by an alteration in attention, awareness, and cognition [83•]. Budson et al. suggested that delirium is a state of individuals being awake but unconscious, noting that they do not appear to have conscious control of their own actions [13••]. The prevalence of delirium in hospitalized patients is high: up to 23% of hospitalized patients and 88% of palliative care inpatients [84, 85]. Despite the high prevalence of delirium, the underlying pathophysiology of delirium is unclear. Recent studies have approached the neural substrate of delirium from a connectivity and functional state perspective. Fleishmann et al. suggested that theta band hyperconnectivity and alpha band dysconnectivity may underly delirium [86]. Montfort et al. support the theory of disintegrate resting state in delirium and revealed a specific loss of function in the posterior cingulate cortex among delirious patients [87].
Dementia
Although not generally considered a disorder of level of consciousness, dementia almost always affects aspects of the phenomenal conscious experience [13••]. Recent studies have attempted to answer the question of how conscious experience might be affected by the dementias.
Patients with Alzheimer’s disease demonstrated poor ratings in visual imagery, auditory imagery, and spatiotemporal specificity—all core components of the phenomenal conscious experience [88]. In a study of ERPs, latency of the P300 (a candidate correlate of consciousness discussed above and supported by MToC as a marker of one aspect of phenomenal consciousness) showed the highest correlation with Alzheimer’s disease pathology, supporting previous studies using the P300 as a marker of cognitive decline [89,90,91].
Corticobasal syndrome and alien-limb phenomenon present a disconnection between action and volitional, conscious control. In 2019, the first MRI-based group analysis of alien limb phenomenon in corticobasal degeneration revealed that CBS with alien-limb phenomenon can be differentiated from CBS patients without alien-limb syndrome based on structural differences in the cingulate gyrus as well as the frontomedian cortex, post central gyrus, and temporoparietooccipital regions [92•]. This finding supports previous literature that the anterior cingulate gyrus is critical for conscious control of motor functions [93].
Not all effects of dementia dampen consciousness. In some patients with FTD, a domain of consciousness can be augmented. The fascinating emergence of artistic creativity in some FTD patients has been described since 1998 [94]. Recent systematic review suggests that the relative preservation of the occipital cortex, which may be less inhibited by degenerating frontotemporal systems, is strongly correlated with visual artistic creativity [95].
Lastly, the absence of level of conscious deficits in neurodegenerative diseases raises questions about the validity of some theories of consciousness. If meta-cognition in the frontal lobe is essential for consciousness, as stated in HOT, one would expect that patients with moderate FTD to become unconscious and lack any conscious awareness of their surroundings. Similarly, if the posterior brain and its connectivity is the center of consciousness as maintained by proponents of IIT and RPT, then patients with posterior cortical atrophy would be expected to have impairments beyond the visuospatial domain and become unconscious. Impairments in patients with dementia, therefore, are best explained by MToC, which argues for impaired aspects of phenomenal consciousness with dementia.
Conclusions
Although the complete neurobiological basis of consciousness remains a mystery, recent advances have improved our understanding of the disorders of levels of consciousness and of aspects of phenomenal consciousness. The idea espoused by Budson et al. in the MToC—that the substrate of consciousness is the cerebral cortex, which enables various aspects of phenomenal consciousness—is consistent with both the neurological literature and the experience of clinical neurologists [13••]. Different ERPs likely correspond to different aspects of phenomenal consciousness—not all of consciousness—which may explain some of the disagreements in the literature. Neurological disorders that disrupt the reticular activating system can affect the level of consciousness, whereas cortical disorders from seizures and migraines to strokes and dementia may disrupt phenomenal consciousness. We believe that consideration of these neurological disorders argues against local theories of consciousness and best support theories of consciousness as distributed through a large cortical network, such as MToC. The future of consciousness research will be furthered by the application of experiential paradigms assessed by EEG, ERP, and fMRI to patients with disorders of consciousness. Such research may improve clinical care and reveal the neurological basis of consciousness.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Block N. On a confusion about a function of consciousness. Behav Brain Sci. 1995;18:227–47.
Chalmers D. Facing up to the problem of consciousness. J Consciousness Studies. 1995;2:200–19.
•• Seth AK, Bayne T. Theories of consciousness. Nat Rev Neurosci. 2022;23:439–52. Comprehensive review of the current neurobiological theories of consciousness
Rosenthal D. Consciousness and mind. Clarendon Press; 2005.
Brown R, Lau H, LeDoux J. Understanding the higher-order approach to consciousness. Trends Cogn Sci. 2019;23:754–68.
Baars BA. Cognitive theory of consciousness. Cambridge Univ, Press; 1988.
Dehaene S, Changeux J. Experimental and theoretical approaches to conscious processing. Neuron. 2011;70:200–27.
Mashour G, Roelfsema P, Changeux J, Dehaene S. Conscious processing and the global neuronal workspace hypothesis. Neuron. 2020;105:776–98.
Tononi G. Consciousness as integrated information: a provisional manifesto. Biol Bull. 2008;215:216–42.
Tononi G, Boly M, Koch C. Integrated information theory: from consciousness to its physical substrate. Nat Rev Neurosci. 2016;17:450–61.
Lamme VA. Towards a true neural stance on consciousness. Trends Cogn Sci. 2006;10:494–501.
Lamme VA. How neuroscience will change our view on consciousness. Cogn Neurosci. 2010;1:204–20.
•• Budson AE, Richman KA, Kensinger EA. Consciousness as a memory system. Cogn Behav Neurol. 2022;35:263–97. A novel theory of consciousness as a memory system
American Congress of Rehabilitation Medicine, Brain-Injury Interdisciplinary Special Interest Group, Disorders of Consciousness Task Force, Seel R, Sherer M, Whyte J, Katz D, Giacino J, Rosenbaum A, Hammond F, Kalmar K, Pape TL, Zafonte R, Biester R, Kaelin D, Kean J, Zasler N. Assessment scales for disorders of consciousness: evidence-based recommendations for clinical practice and research. Arch Phys Med Rehab. 2010;91
Yan X, Xiao L, Liao M, Huang J, He Z, Yan T. The validity, reliability and feasibility of four instruments for assessing the consciousness of stroke patients in a neurological intensive care unit compared. BMC Med Res Methodol. 2022;22
Sessler C, Gosnell M, Grap M, Brophy G, O’Neal P, Keane K, et al. The Richmond agitation–sedation scale validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med. 2002;166:1338–44.
American Congress of Rehabilitation Medicine, Brain Injury-Interdisciplinary Special Interest, Group, Disorders of Consciousness Task Force, Seel RT, et al. Assessment scales for disorders of consciousness: evidence-based recommendations for clinical practice and research. Arch Phys Med Rehab. 2010;91:1795–831.
Bodien YG, Carlowicz CA, Chatelle C, Giacino JT. Sensitivity and specificity of the coma recovery scale–revised total score in detection of conscious awareness. Arch Phys Med Rehab. 2015;97:490–2.
Frigerio S, Molteni E, Colombo K, Pastore V, Fedeli C, Galbiati S, et al. Neuropsychological assessment through Coma Recovery Scale-Revised and Coma/Near Coma Scale in a sample of pediatric patients with disorder of consciousness. J Neurol. 2022;270:1019–29.
Owen AM, Coleman MR, Boly M, Davis MH, Laureys S, Pickard JD. Detecting awareness in the vegetative state. Science. 2006;313:1402.
Goldfine AM, Victor JD, Conte MM, Bardin JC, Schiff ND. Determination of awareness in patients with severe brain injury using EEG power spectral analysis. Clin Neurophysiol. 2011;122:2157–68.
Monti MM, Vanhaudenhuyse A, Coleman MR, et al. Willful modulation of brain activity in disorders of consciousness. N Engl J Med. 2010;362:579–89.
Edlow BL, Chatelle C, Spencer CA, et al. Early detection of consciousness in patients with acute severe traumatic brain injury. Brain. 2017;140:2399–414.
•• Classen J, et al. Detection of brain activation in patients with ABI. N Engl J Med. 2019;380:2497–505. EEG verbal command paradigm to detect cognitive motor dissociation
• Egbebike J, Shen Q, Doyle K, Der-Nigoghossian C, Panicker L, Gonzales I, et al. Cognitive-motor dissociation and time to functional recovery in patients with acute brain injury in the USA: a prospective observational cohort study. Lancet Neurol. 2022;21:704–13. Cognitive-motor dissociation in patients with decreased level of consciousness is an important prognostic factor
Lee H-C, Ryu H-G, Park Y, Yoon SB, Yang SM, Oh H-W, Jung C-W. Data Driven Investigation of Bispectral Index Algorithm. Sci Rep. 2019:9.
Klimesch W, Sauseng P, Hanslmayr S. EEG alpha oscillations: the inhibition-timing hypothesis. Brain Res Rev. 2007;53:63–88.
Klimesh W. Evoked alpha and early access to the knowledge system: the P1 inhibition timing hypothesis. Brain Res. 2011;1408:52–71.
Jensen O, Mazaheri A. Shaping functional architecture by oscillatory alpha activity: gating by inhibition. Front Hum Neurosci. 2010;4:186.
Solis-Vivanco R, Mathilde B, Jensen O. Top-down control of alpha phase adjustment inanticipation of predictable visual stimuli. J Cog Neurosci. 2018;30:1–13.
Ergenoglu T, Demiralp T, Bayraktaroglu Z, Ergen M, Beydagi H, Uresin Y. Alpha rhythm of the EEG modulates visual detection performance in humans. Cog Brain Res. 2004;20:376–83.
Hanslmayr S, Aslan A, Staudigl T, Klimesch W, Herrmann CS, Bäuml K-H. Prestimulus oscillations predict visual perception performance between and within subjects. Neuroimage. 2007;37:1465–73.
Hv D, Schoffelen J-M, Oostenveld R, Jensen O. Prestimulus oscillatory activity in the alpha band predicts visual discrimination ability. J Neurosci. 2008;28:1816–23.
Benwell CS, Tagliabue CF, Veniero D, Cecere R, Savazzi S, Thut G. Prestimulus EEG power predicts conscious awareness but not objective visual performance. eNeuro. 2017;4:e0182-17.2017 1–17.
•• Krasich K, Simmons C, O’Neill K, Giattino CM, Brigard FD, Sinnott-Armstrong W, Mudrik L, Woldorff MG. Prestimulus oscillatory brain activity interacts with evoked recurrent processing to facilitate conscious visual perception. Sci Rep. 2022:12. Stimulus timing within alpha phase predicts conscious perception
Babiloni C, Vecchio F, Bultrini A, Romani GL, Rossini PM. Pre- and poststimulus alpha rhythms are related to conscious visual perception: a high-resolution EEG study. Cerebral Cortex. 2006;16:1690–700.
Hutchinson B, Pammer K, Jack B. Pre-stimulus alpha predicts inattentional blindness. Conscious Cogn. 2021;87
Voss JL, Paller KA. Learning and memory: a comprehensive reference (Second Edition). Academic Press; 2017.
Sergent C, Wyart V, Babo-Rebelo M, Cohen L, Naccache L, Tallon-Baudry C. Cueing attention after the stimulus is gone can retrospectively trigger conscious perception. Curr Biol. 2013;23:150–5.
Crick F, Koch C. Towards a neurobiological theory of consciousness. Semin Neurosci. 1990;2:263–75.
Sur S, Sinha VK. Event-related potential: an overview. Ind Psychiatry J. 2009;18:70–3.
Libet B, Alberts WW, Wright EW Jr, Feinstein B. Responses of human somatosensory cortex to stimuli below threshold for conscious sensation. Science. 1967;158:1597–600.
Schubert R, Blankenburg F, Lemm S, Villringer A, Curio G. Now you feel it—now you don’t: ERP correlates of somatosensory awareness. Psychophysiology. 2006;43:31–40.
Allison T, Wood C, McCarthy G. The relationship between human long-latency somatosensory evoked potentials recorded from the cortical surface and from the scalp. EEG and Clin Neurophys. 1992;84:301–14.
Maguière F, Merlet I, Forss N, Vanni S, Jousmaki V, Adeleine P, et al. Activation of a distributed somatosensory cortical network in the human brain. A dipole modeling study of magnetic fields evoked by median nerve stimulation. Part I: Location and activation timing of SEF sources. EEG and Clin Neurophys. 1992;104:281–9.
Forschack N, Nierhaus T, Müller MM, Villringer A. Dissociable neural correlates of stimulation intensity and detection in somatosensation. Neuroimage. 2020;217:116908.
Forster B, Tziraki M, Jones A. The attentive homunculus: ERP evidence for somatotopic allocation of attention in tactile search. Neuropsychologia. 2016;84:158–66.
Kida T, Wasaka T, Nakata H, Akatsuka K, Kakigi. Active attention modulates passive attention-related neural responses to sudden somatosensory input against a silent background. Exp Brain Res. 2006;175:609–17.
Gusso MM, Christison-Lagay KL, Zuckerman D, Chandrasekaran G, Kronemer SI, Ding JZ, et al. More than a feeling: scalp EEG and eye signals in conscious tactile perception. Conscious Cogn. 2022;105
Al E, Iliopoulos F, Forschack N, Nierhaus T, Grund M, Motyka P, et al. Heart-brain interactions shape somatosensory perception and evoked potentials. Proc Natl Acad Sci USA. 2020;117:10575–84.
Forschack N, Nierhaus T, Müller M, Villringer A. Dissociable neural correlates of stimulation intensity and detection in somatosensation. Neuroimage. 2020;217:116908.
Schröder P, Nierhaus T, Blankenburg F. Dissociating perceptual awareness and postperceptual processing: the P300 is not a reliable marker of somatosensory target detection. J Neurosci. 2021;41:4686–96.
Ueno T, Hada Y, Shimizu Y, Yamada T. Relationship between somatosensory event-related potential N140 aberrations and hemispatial agnosia in patients with stroke: a preliminary study. Int J Neurosci. 2017;128:487–94.
Endrass T, Reuter B, Kathmann N. ERP correlates of conscious error recognition: aware and unaware errors in an antisaccade task. Eur J Neurosci. 2007;26:1714–20.
Van Dinteren R, Arns M, Jongsma ML, Kessels RP. P300 Development across the lifespan: a systematic review and meta-analysis. PLoS One. 2014;9:e87347.
Sutton S, Braren M, Zubin J, John ER. Evoked-potential correlates of stimulus uncertainty. Science. 1965;150:1187–8.
Lamy D, Salti M, Bar-Haim Y. Neural correlates of subjective awareness and unconscious processing: an ERP study. J Cogn Neurosci. 2009;21:1435–46.
Sergent C, Baillet S, Dehaene S. Timing of the brain events underlying access to consciousness during the attentional blink. Nat Neurosci. 2005;8:391–1400.
Koch C, Massimini M, Boly M, Tononi G. Neural correlates of consciousness: progress and problems. Nat Rev Neurosci. 2016;17:307–21.
• Förster J, Koivisto M, Revonsuo A. ERP and MEG correlates of visual consciousness: the second decade. Conscious Cogn. 2020;80 Review that concludes that the P300 is a signal of post sensory cognitive processing and the VAN may be the earliest marker of awareness
Railo H, Koivisto M, Revonsuo A. Tracking the processes behind conscious perception: a review of event-related potential correlates of visual consciousness. Conscious Cogn. 2011;20:972–83.
Dembski C, Koch C, Pitts M. Perceptual awareness negativity: a physiological correlate of sensory consciousness. Trends Cogn Sci. 2021;25(8):660–70.
Balduzzi D. Estimating integrated information with TMS pulses during wakefulness, sleep, and under anesthesia. Annu Int Conf IEEE Eng Med Biol Soc. 2011;2011:4717–20.
Ferrarelli F, Massimini M, Sarasso S, Casali A, Riedner BA, Angelini G, Tononi G, Pearce RA. Breakdown in cortical effective connectivity during midazolam-induced loss of consciousness. Proc Natl Acad Sci USA. 2010;107:2681–6.
Massimini M, Ferrarelli F, Huber R, Esser SK, Singh H, Tononi G. Breakdown of cortical effective connectivity during sleep. Science. 2005;309:2228–32.
Casali AG, Gosseries O, Rosanova M, Boly M, Sarasso S, Casali KR, et al. A theoretically based index of consciousness independent of sensory processing and behavior. Sci Transl Med. 2013;5:198ra05.
Rosanova M, Gosseries O, Casarotto S, Boly M, Casali AG, Bruno MA, Mariotti M, Boveroux P, Tononi G, Laureys S, Massimini M. Recovery of cortical effective connectivity and recovery of consciousness in vegetative patients. Brain. 2012;135:1308–20.
Toker D, Pappas I, Lendner JD, Frohlich J, Mateos DM, Muthukumaraswamy S, et al. Consciousness is supported by near-critical slow cortical electrodynamics. Proc Natl Acad Sci USA. 2022;119:e2024455119.
• Lee M, Baird B, Gosseries O, Nieminen JO, Boly M, Postle BR, et al. Connectivity differences between consciousness and unconsciousness in non-rapid eye movement sleep: a TMS–EEG study. Sci Rep. 2019;9:5175. Connectivity differences most notable in the posterior cortex define consciousness and unconsciousness in sleep
Demertzi A, Gómez F, Crone JS, Vanhaudenhuyse A, Tshibanda L, Noirhomme Q, et al. Multiple fMRI system-level baseline connectivity is disrupted in patients with consciousness alterations. Cortex. 2014;52:35–46.
•• Luppi AI, Craig MM, Pappas I, Finoia P, Williams GB, Allanson J, et al. Consciousness-specific dynamic interactions of brain integration and functional diversity. Nat Commun. 2019;10:4616. High integration across cortical regions in fMRI define level of conscoiusness
Scheffer IE, Berkovic S, Capovilla G, Connolly MB, French J, Guilhoto L, et al. LAE classification of the epilepsies: position paper of the ILAE Commission for Classification and Terminology. Epilepsia. 2017;58:512–21.
Scheffer IE, Berkovic S, Capovilla G, Connolly MB, French J, Guilhoto L, et al. LAE classification of the epilepsies: position paper of the ILAE Commission for Classification and Terminology. Epilepsia. 2017;58:512–21.
Englot DJ, Yang L, Hamid H, Danielson N, Bai X, Marfeo A, et al. Impaired consciousness in temporal lobe seizures: role of cortical slow activity. Brain. 2010;133:3764–77.
Harriott AM, Takizawa T, Chung DY, Chen S-P. Spreading depression as a preclinical model of migraine. J Headache Pain. 2019;20:45.
Li J, Zhang P, Wu S, Yuan R, Liu J, Tao W, et al. Impaired consciousness at stroke onset in large hemisphere infarction: incidence, risk factors and outcome. Sci Rep. 2020;10:13170.
Alkhachroum A, Bustillo AJ, Asdaghi N, Ying H, Marulanda-Londono E, Gutierrez CM, et al. Association of acute alteration of consciousness in patients with acute ischemic stroke with outcomes and early withdrawal of care. Neurology. 2022;98:1470–8.
Bogousslavsky J, Regli F, Uske A. Thalamic infarcts: clinical syndromes, etiology, and prognosis. Neurology. 1988;38:837–47.
Amici S. Thalamic infarcts and hemorrhages. Front Neurol Neurosci. 2012;30:132–6.
Jang SH, Chang CH, Jung YJ, Kim JH, Kwon YH. Relationship between impaired consciousness and injury of ascending reticular activating system in patients with intracerebral hemorrhage. Stroke. 2019;50:2234–7.
• Snider SB, Hsu J, Darby RR, Cooke D, Fischer D, Cohen AL, et al. Cortical lesions causing loss of consciousness are anticorrelated with the dorsal brainstem. Hum Brain Mapp. 2020;41:1520–31. Highly powered brain mapping study of the distributed cortical system required for consciousness
Das JM, Naqvi IA. Anton Syndrome. In: StatPearls. StatPearls Publishing; 2021.
• Wilson JE, Mart MF, Cunningham C, Shehabi Y, Girard TD, AM ML, et al. Delirium. Nat Rev Dis Primers. 2020;6:90. Comprehensive delirium neuropathology review
Gibb K, Seeley A, Quinn T, Siddiqi N, Shenkin S, Rockwood K, et al. The consistent burden in published estimates of delirium occurrence in medical inpatients over four decades: a systematic review and meta-analysis study. Age Aging. 2020;49:352–60.
Hosie A, Davidson PM, Agar M, Sanderson CR, Phillips J. Delirium prevalence, incidence, and implications for screening in specialist palliative care inpatient settings: a systematic review. Palliat Med. 2013;27:486–98.
Fleischmann R, Traenkner S, Kraft A, Schmidt S, Schreiber SJ, Brandt SA. Delirium is associated with frequency band specific dysconnectivity in intrinsic connectivity networks: preliminary evidence from a large retrospective pilot case-control study. Pilot Feasibility Stud. 2019;5:2.
Van Montfort SJ, Van Dellen E, Van Den Bosch AMR, Otte WM, Schutte MJL, Choi S-H, et al. Resting-state fMRI reveals network disintegration during delirium. NeuroImage Clin. 2018;20:35–41.
Moustafa AA, Haj ME. Phenomenological characteristics of future thinking in Alzheimer’s disease. J Alzheimers Dis. 2018;2018:1279–87.
Fruehwirt W, Dorffner G, Roberts S, Gerstgrasser M, Grossegger D, Schmidt R, et al. Associations of event-related brain potentials and Alzheimer’s disease severity: a longitudinal study. Prog Neuropsychopharmacol Biol Psychiatry. 2019;92:31–8.
Porcaro C, Balsters J, Mantini D, Robertson I, Wenderoth N. P3b amplitude as a signature of cognitive decline in the older population: an EEG study enhanced by Functional Source Separation. Neuroimage. 2019;184:535–46.
Pedroso RV, Fraga FJ, Corazza DI, Andrezza C, Andreatto A, Coelho FGM, et al. P300 latency and amplitude in Alzheimer’s disease: a systematic review. Brazil. J Otorhinolaryngology. 2012;78:126–32.
• Albrecht F, Mueller K, Ballarini T, Lampe L, Diehl-Schmid J, Fassbender K, et al. Unraveling corticobasal syndrome and alien limb syndrome with structural brain imaging. Cortex. 2019;117:33–40. Reveals the structural nature of corticobasal syndrome and the anatomic nature of volitional movement
Paus T. Primate anterior cingulate cortex: where motor control, drive and cognition interface. Nat Rev Neurosci. 2001;2:417–24.
Miller BL, Cummings J, Mishkin F, Boone K, Prince F, Ponton M, et al. Emergence of artistic talent in frontotemporal dementia. Neurology. 1998;51:978–82.
Geser F, Jellinger K, Fellner L, Wenning G, Yilmazer-Hanke D, Haybaeck J. Emergent creativity in frontotemporal dementia. J Neural Transm. 2021;128:279–93.
Funding
NIH/NIA P30 (grant number: AG072978) titled “Boston University Alzheimer’s Disease Core Center.” The principal investigator is Ann McKee, MD.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Friedman, G., Turk, K.W. & Budson, A.E. The Current of Consciousness: Neural Correlates and Clinical Aspects. Curr Neurol Neurosci Rep 23, 345–352 (2023). https://doi.org/10.1007/s11910-023-01276-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11910-023-01276-0