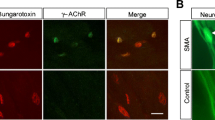
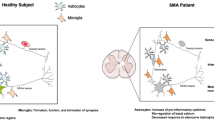
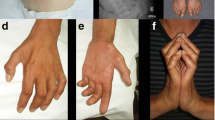
Abstract
Spinal muscular atrophy (SMA) is a common and often fatal neurodegenerative disease that primarily afflicts infants and young children. SMA is caused by abnormally low levels of the survival motor neuron (SMN) protein resulting from a combination of recessively inherited mutations in the SMN1 gene and the presence of an almost identical but partially functional copy gene, SMN2. Absence of the uniquely human SMN2 gene in SMA patients has never been reported because the SMN protein is indispensable for cell survival. Modeling SMA in animals therefore poses a challenge. This review describes the different strategies used to overcome this hurdle and model SMA in mice. We highlight new and emerging insights regarding SMA gained by studying the mice and illustrate how the animals serve as important tools to understand and eventually treat the human disease.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Monani UR: Spinal muscular atrophy: a deficiency in a ubiquitous protein; a motor neuron specific disease. Neuron 2005, 48:885–895.
Lefebvre S, Burglen L, Reboullet S, et al.: Identification and characterization of a spinal muscular atrophy-determining gene. Cell 1995, 80:155–165.
Monani UR, Lorson CL, Parsons DW, et al.: A single nucleotide difference that alters splicing patterns distinguishes the SMA gene SMN1 from the copy gene SMN2. Hum Mol Genet 1999, 8:1177–1183.
Cartegni L, Krainer AR: Disruption of an SF2/ASF-dependent exonic splicing enhancer in SMN2 causes spinal muscular atrophy in the absence of SMN1. Nat Genet 2002, 30:377–384.
Kashima T, Manley JL: A negative element in SMN2 exon 7 inhibits splicing in spinal muscular atrophy. Nat Genet 2003, 34:460–463.
Coovert DD, Le TT, McAndrew PE, et al.: The survival motor neuron protein in spinal muscular atrophy. Hum Mol Genet 1997, 6:1205–1214.
Feldkotter M, Schwarzer V, Wirth R, et al.: Quantitative analyses of SMN1 and SMN2 based on real-time lightCycler PCR: fast and highly reliable carrier testing and prediction of severity of spinal muscular atrophy. Am J Hum Genet 2003, 70:358–368.
Liu Q, Fischer U, Wang F, Dreyfuss G: The spinal muscular atrophy disease gene product, SMN, and its associated protein SIP1 are in a complex with spliceosomal snRNP proteins. Cell 1997, 90:1013–1021.
Fischer U, Liu Q, Dreyfuss G: The SMN-SIP1 complex has an essential role in spliceosomal snRNP biogenesis. Cell 1997, 90:1023–1029.
Pellizzoni L, Kataoka N, Charroux B, Dreyfuss G: A novel function for SMN, the spinal muscular atrophy disease gene product, in pre-mRNA splicing. Cell 1998, 95:615–624.
Chan YB, Miguel-Aliaga I, Franks C, et al.: Neuromuscular defects in a Drosophila survival motor neuron gene mutant. Hum Mol Genet 2003, 12:1367–1376.
McWhorter ML, Monani UR, Burghes AH, Beattie CE: Knockdown of the survival motor neuron (Smn) protein in zebrafish causes defects in motor axon outgrowth and pathfinding. J Cell Biol 2003, 162:919–931.
Miguel-Aliaga I, Culetto E, Walker DS, et al.: The Caenorhabditis elegans orthologue of the human gene responsible for spinal muscular atrophy is a maternal product critical for germline maturation and embryonic viability. Hum Mol Genet 1999, 8:2133–2143.
Briese M, Esmaeili B, Fraboulet S, et al.: Deletion of smn-1, the Caenorhabditis elegans ortholog of the spinal muscular atrophy gene, results in locomotor dysfunction and reduced lifespan. Hum Mol Genet 2009, 18:97–104.
Schmitt-John T, Drepper C, Mussmann A, et al.: Mutation of Vps54 causes motor neuron disease and defective spermiogenesis in the wobbler mouse. Nat Genet 2005, 37:1213–1215.
Schmidt WM, Kraus C, Höger H, et al.: Mutation in the Scyl1 gene encoding amino-terminal kinase-like protein causes a recessive form of spinocerebellar neurodegeneration. EMBO Rep 2007, 8:691–697.
Martin N, Jaubert J, Gounon P, et al.: A missense mutation in Tbce causes progressive motor neuronopathy in mice. Nat Genet 2002, 32:443–447.
DiDonato CJ, Chen XN, Noya D, et al.: Cloning, characterization, and copy number of the murine survival motor neuron gene: homolog of the spinal muscular atrophy-determining gene. Gen Res 1997, 7:339–352.
Schrank B, Gotz R, Gunnersen JM, et al.: Inactivation of the survival motor neuron gene, a candidate gene for human spinal muscular atrophy, leads to massive cell death in early mouse embryos. Proc Natl Acad Sci U S A 1997, 94:9920–9925.
Frugier T, Tiziano FD, Cifuentes-Diaz C, et al.: Nuclear targeting defect of SMN lacking the C-terminus in a mouse model of spinal muscular atrophy. Hum Mol Genet 2000, 9:849–858.
Cifuentes-Diaz C, Frugier T, Tiziano FD, et al.: Deletion of murine SMN exon 7 directed to skeletal muscle leads to severe muscular dystrophy. J Cell Biol 2001, 152:1107–1114.
Monani UR, Sendtner M, Coovert, DD, et al.: The human centromeric survival motor neuron gene (SMN2) rescues embryonic lethality in Smn (-/-) mice and results in a mouse with spinal muscular atrophy. Hum Mol Genet 2000, 9:333–339.
Hsieh-Li HM, Chang JG, Jong YJ, et al.: A mouse model for spinal muscular atrophy. Nat Genet 2000, 24:66–70.
Le TT, Pham LT, Butthbach ME, et al.: SMN Delta7, the major product of the centromeric survival motor neuron (SMN2) gene, extends survival in mice with spinal muscular atrophy and associates with full-length SMN. Hum Mol Genet 2005, 14:845–857.
Monani UR, Pastore MT, Gavrilina TO, et al.: A transgene carrying an A2G missense mutation in the SMN gene modulates phenotypic severity in mice with severe (type I) spinal muscular atrophy. J Cell Biol 2003, 160:41–52.
Kerr DA, Nery JP, Traystman RJ, et al.: Survival motor neuron protein modulates neuron-specific apoptosis. Proc Natl Acad Sci U S A 2000, 97:13312–13317.
• Kariya S, Park G-H, Maeno-Hikichi Y, et al.: Reduced SMN protein impairs maturation of the neuromuscular junctions in mouse models of spinal muscular atrophy. Hum Mol Genet 2008, 17:2552–2569. This study describes a previously unappreciated hallmark of human SMA—profound defects of the distal motor unit—using severe as well as mildly affected model mice. Both pre- and postsynaptic specializations were shown to display abnormalities.
• Kong L, Wang X, Choe DW, et al.: Impaired synaptic vesicle release and immaturity of neuromuscular junctions in spinal muscular atrophy mice. J Neurosci 2009, 29:842–851. This study independently reported distal defects of the motor unit in severe SMA model mice and showed for the first time that synaptic vesicle release may be perturbed in the human disease.
Bromberg MB, Swoboda KJ: Motor unit number estimation in infants and children with spinal muscular atrophy. Muscle Nerve 2002, 25:445–447.
McGovern VL, Gavrilina TO, Beattie CE, Burghes AH: Embryonic motor axon development in the severe SMA mouse. Hum Mol Genet 2008, 18:2900–2909.
• Gavrilina TO, McGovern VL, Workman E, et al.: Neuronal SMN expression corrects spinal muscular atrophy in severe SMA mice while muscle-specific SMN expression has no phenotypic effect. Hum Mol Genet 2008, 17:2900–2909. This report is the first to attempt to define which tissues require wild-type levels of SMN to rescue the SMA phenotype in model mice.
Carrel TL, McWhorter ML, Workman E, et al.: Survival motor neuron function in motor axons is independent of functions required for small nuclear ribonucleoprotein biogenesis. J Neurosci 2006, 26:11014–11020.
Sun Y, Grimmler M, Schwarzer V, et al.: Molecular and functional analysis of intragenic SMN1 mutations in patients with spinal muscular atrophy. Human Mutat 2005, 25:64–71.
Shpargel KB, Matera AG: Gemin proteins are required for efficient assembly of Sm-class ribonucleoproteins. Proc Natl Acad Sci U S A 2005, 102:17372–17377.
•• Workman E, Saieva L, Carrel TL, et al.: A SMN missense mutation complements SMN2 restoring snRNPs and rescuing SMA mice. Hum Mol Genet 2009, 18:2215–2229. This study provides important data suggesting that SMN’s role in snRNP biogenesis is inextricably intertwined with the SMA phenotype. Rescue of snRNP assembly and restoration of minor spliceosome complexes, even if effected by mutant molecules, correlate with disease severity.
•• Gabanella F, Butchbach ME, Saieva L, et al.: Ribonucleoprotein assembly defects correlate with spinal muscular atrophy severity and preferentially affect a subset of spliceosomal snRNPs. PLoS One 2007, 2:e921. These experiments showed for the first time that SMN deficiency selectively affects a subset of spliceosomal snRNPs and that there is a remarkable correlation between assembly of snRNP complexes and disease phenotype.
Chang JG, Hsieh-Li HM, Jong YJ, et al.: Treatment of spinal muscular atrophy by sodium butyrate. Proc Natl Acad Sci U S A 2001, 98:9808–9813.
Tsai LK, Tsai MS, Ting CH, Li H: Multiple therapeutic effects of valproic acid in spinal muscular atrophy model mice. J Mol Med 2008, 86:1243–1254.
Swoboda KJ, Scott CB, Reyna SP, et al.: Phase II open label study of valproic acid in spinal muscular atrophy. PLoS One 2009, 4:e5268.
Narver HL, Kong L, Burnett BG, et al.: Sustained improvement of spinal muscular atrophy mice treated with trichostatin A plus nutrition. Ann Neurol 2008, 64:465–470.
Mattis VB, Ebert AD, Fosso MY, et al.: Delivery of a read-through inducing compound, TC007, lessens the severity of a spinal muscular atrophy animal model. Hum Mol Genet 2009, 18:3906–3913.
Heier CR, DiDonato CJ: Translational readthrough by the aminoglycoside geneticin (G418) modulates SMN stability in vitro and improves motor function in SMA mice in vivo. Hum Mol Genet 2009, 18:1310–1322.
Lunn MR, Root DE, Martino AM, et al.: Indoprofen upregulates the survival motor neuron protein through a cyclooxygenase-independent mechanism. Chem Biol 2004, 11:1489–1493.
Butchbach ME, Singh J, Thornorsteinsdóttir M, et al.: Effects of 2,4-diaminoquinazoline derivatives on SMN expression and phenotype in a mouse model for spinal muscular atrophy. Hum Mol Genet 2009 Nov 25 (Epub ahead of print).
Hua Y, Vickers TA, Okunola HL, et al.: Antisense masking of an hnRNP A1/A2 intronic splicing silencer corrects SMN2 splicing in transgenic mice. Am J Hum Genet 2008, 82:834–848.
Williams JH, Schray RC, Patterson CA, et al.: Oligonucleotide-mediated survival of motor neuron protein expression in CNS improves phenotype in a mouse model of spinal muscular atrophy. J Neurosci 2009, 29:7633–7638.
Baughan TD, Dickson A, Osman EY, Lorson CL: Delivery of bifunctional RNAs that target an intronic repressor and increase SMN levels in an animal model of spinal muscular atrophy. Hum Mol Genet 2009, 18:1600–1611.
Tsai LK, Tsai MS, Ting CH, et al.: Restoring Bcl-x(L) levels benefits a mouse model of spinal muscular atrophy. Neurobiol Dis 2008, 3:361–367.
Tsai MS, Chiu YT, Wang SH, et al.: Abolishing Bax-dependent apoptosis shows beneficial effects on spinal muscular atrophy model mice. Molec Ther 2006, 13:1149–1155.
Sumner CJ, Wee CD, Warsing LC, et al.: Inhibition of myostatin does not ameliorate disease features of severe spinal muscular atrophy mice. Hum Mol Genet 2009, 18:3145–3152.
Rose FF Jr, Mattis VB, Rindt H, Lorson CL: Delivery of recombinant follistatin lessens disease severity in a mouse model of spinal muscular atrophy. Hum Mol Genet 2009, 18:997–1005.
Mack TG, Reiner M, Beirowski B, et al.: Wallerian degeneration of injured axons and synapses is delayed by a Ube4b/Nmnat chimeric gene. Nat Neurosci 2001, 4:1199–1206.
Rose FF Jr, Meehan PW, Coady TH, et al.: The Wallerian degeneration slow (Wld(s)) gene does not attenuate disease in a mouse model of spinal muscular atrophy. Biochem Biophys Res Commun 2008, 375:119–123.
Kariya S, Mauricio R, Dai Y, Monani UR: The neuroprotective factor Wld(s) fails to mitigate distal axonal and neuromuscular junction (NMJ) defects in mouse models of spinal muscular atrophy. Neurosci Lett 2009, 449:246–251.
Chang HC, Dimlich DN, Yokokura T, et al.: Modeling spinal muscular atrophy in Drosophila. PLoS One 2008, 3:e3209.
Corti S, Nizzardo M, Nardini M, et al.: Neural stem cell transplantation can ameliorate the phenotype of a mouse model of spinal muscular atrophy. J Clin Invest 2008, 118:3316–3330.
Setola V, Towne C, Kieran D, et al.: AAV-mediated gene therapy for SMA: in vitro and in vivo effects of FL-SMN and a-SMN [abstract]. Presented at the 13th Annual Spinal Muscular Atrophy Research Group Meeting. Cincinnati, OH; June 18–20, 2009.
Passini M, Bu J, Roskelley EM, et al.: CNS delivery of human SMN1 by adeno-associated virus is highly efficacious in a mouse model of spinal muscular atrophy type I [abstract]. Presented at the 13th Annual Spinal Muscular Atrophy Research Group Meeting. Cincinnati, OH; June 18–20, 2009.
Foust K, Kaspar BK: The kinetics of scAAV9 expression in the spinal cord after neonatal intravenous injection [abstract]. Presented at the 13th Annual Spinal Muscular Atrophy Research Group Meeting. Cincinnati, OH; June 18–20, 2009.
Bowerman M, Anderson CL, Beauvais A, et al.: SMN, profilin IIa and plastin 3: a link between the deregulation of actin dynamics and SMA pathogenesis. Mol Cell Neurosci 2009, 42:66–74.
Acknowledgments
We are grateful to Drs. D. C. De Vivo, S. Przedborski, and C. E. Henderson for advice and to S. Patruni for critically reading this manuscript. Work in the laboratory is funded by National Institute of Neurological Disorders and Stroke (NINDS) R01 NS057482, NINDS P01 NS055923, US Department of Defense (DoD) W81XWH-09-1-0245, DoD W81XWH-08-1-0009, the Motor Neuron Center, Columbia University, and the SMA Foundation, which also was instrumental in coordinating the generation of the newer model mice.
Disclosure
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, GH., Kariya, S. & Monani, U.R. Spinal Muscular Atrophy: New and Emerging Insights from Model Mice. Curr Neurol Neurosci Rep 10, 108–117 (2010). https://doi.org/10.1007/s11910-010-0095-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11910-010-0095-5