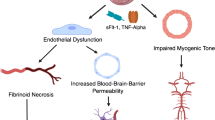
Abstract
Purpose of Review
To provide insight into the mechanisms underlying cerebral pathophysiology and to highlight possible methods for evaluation, screening, and surveillance of cerebral complications in preeclampsia.
Recent Findings
The pathophysiology of eclampsia remains enigmatic. Animal studies show that the cerebral circulation in pregnancy and preeclampsia might be affected with increased permeability over the blood-brain barrier and altered cerebral blood flow due to impaired cerebral autoregulation. The increased blood pressure cannot be the only underlying cause of eclampsia and cerebral edema, since some cases of eclampsia arise without simultaneous hypertension. Findings from animal studies need to be confirmed in human tissues. Evaluation of brain alterations in preeclampsia and eclampsia is challenging and demands a multidisciplinary collaboration, since no single method can accurately and fully describe how preeclampsia affects the brain.
Summary
Cerebral complications of preeclampsia are significant factors in maternal morbidity and mortality worldwide. No single method can accurately describe the full picture of how preeclampsia affects the brain vasculature and parenchyma. We recommend an international and multidisciplinary effort not only to overcome the issue of limited sample availability but also to optimize the quality of research.




Similar content being viewed by others
Abbreviations
- BBB:
-
Blood-brain barrier
- BP:
-
Blood pressure
- CBF:
-
Cerebral blood flow
- CVR:
-
Cerebral vascular resistance
- CSF:
-
Cerebrospinal fluid
- GABA:
-
Gamma amino butyric acid
- hPSCs:
-
Human pluripotent stem cells
- JAMs:
-
Junctional adhesion molecules
- LPS:
-
Lipopolysaccharide
- MgSO4 :
-
Magnesium sulfate
- MRI:
-
Magnetic resonance imaging
- MRS:
-
Magnetic resonance spectroscopy
- H-MRS:
-
Magnetic resonance spectroscopy focused on hydrogen metabolites
- P-MRS:
-
Magnetic resonance spectroscopy focused on phosphorus metabolites
- NfL:
-
Neurofilament light chain
- NSE:
-
Neuron-specific enolase
- PTZ:
-
Pentylenetetrazole
- PRES:
-
Posterior reversible encephalopathy syndrome
- RUPP:
-
Reduced uteroplacental perfusion pressure
- RUPP+HC:
-
Reduced uteroplacental perfusion pressure plus high cholesterol diet
- S100B:
-
S100 calcium-binding protein B
- TEER:
-
Transendothelial electrical resistance
- WML:
-
White matter lesions
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Brown MA, Lindheimer MD, de Swiet M, Assche AV, Moutquin J-M. The classification and diagnosis of the hypertensive disorders of pregnancy: statement from the International Society for the Study of Hypertension in Pregnancy (ISSHP). Hypertens Pregnancy. 2001;20:ix–xiv. https://doi.org/10.3109/10641950109152635.
ACOG TFoHiP. Hypertension in pregnancy. Washington: 2013.
Brown MA, Magee LA, Kenny LC, Karumanchi SA, McCarthy FP, Saito S, et al. The hypertensive disorders of pregnancy: ISSHP classification, diagnosis & management recommendations for international practice. Pregnancy Hypertens. 2018;13:291–310. https://doi.org/10.1016/j.preghy.2018.05.004.
Campbell OM, Graham WJ. Strategies for reducing maternal mortality: getting on with what works. Lancet. 2006;368:1284–99. https://doi.org/10.1016/S0140-6736(06)69381-1.
Say L, Chou D, Gemmill A, Tuncalp O, Moller AB, Daniels J, et al. Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health. 2014;2(6):e323–33. https://doi.org/10.1016/S2214-109X(14)70227-X.
Mol BWJ, Roberts CT, Thangaratinam S, Magee LA, de Groot CJM, Hofmeyr GJ. Pre-eclampsia. Lancet. 2016;387:999–1011. https://doi.org/10.1016/S0140-6736(15)00070-7.
Sibai BM. Magnesium sulfate prophylaxis in preeclampsia: lessons learned from recent trials. Am J Obstet Gynecol. 2004;190:1520–6. https://doi.org/10.1016/j.ajog.2003.12.057.
Sibai B, Dekker G, Kupferminc M. Pre-eclampsia. Lancet. 2005;365(9461):785–99. https://doi.org/10.1016/S0140-6736(05)17987-2.
Duley L. The global impact of pre-eclampsia and eclampsia. Semin Perinatol. 2009;33(3):130–7. https://doi.org/10.1053/j.semperi.2009.02.010.
Sibai BM. Diagnosis, prevention, and management of eclampsia. Obstet Gynecol. 2005;105(2):402–10.
Basit S, Wohlfahrt J, Boyd HA. Pre-eclampsia and risk of dementia later in life: nationwide cohort study. Bmj. 2018:k4109. https://doi.org/10.1136/bmj.k4109.
Brussé I, Duvekot J, Jongerling J, Steegers E, De Koning I. Impaired maternal cognitive functioning after pregnancies complicated by severe pre-eclampsia: a pilot case-control study. Acta Obstet Gynecol Scand. 2008;87:408–12. https://doi.org/10.1080/00016340801915127.
Duley L. Do women with pre-eclampsia, and their babies, benefit from magnesium sulphate? The magpie trial: a randomised placebo-controlled trial. Lancet. 2002;359:1877–90. https://doi.org/10.1016/S0140-6736(02)08778-0.
•• Johnson AC, Tremble SM, Chan SL, Moseley J, LaMarca B, Nagle KJ, et al. Magnesium sulfate treatment reverses seizure susceptibility and decreases neuroinflammation in a rat model of severe preeclampsia. PLoS One. 2014;9(11):e113670. https://doi.org/10.1371/journal.pone.0113670 One of the few animal models for cerebral injury and blood brain barrier alteration in preeclampsia.
Andolf EG, Sydsjo GC, Bladh MK, Berg G, Sharma S. Hypertensive disorders in pregnancy and later dementia: a Swedish National Register Study. Acta Obstet Gynecol Scand. 2017;96(4):464–71. https://doi.org/10.1111/aogs.13096.
Nerenberg KA, Park AL, Vigod SN, Saposnik G, Berger H, Hladunewich MA, et al. Long-term risk of a seizure disorder after eclampsia. Obstet Gynecol. 2017;130(6):1327–33. https://doi.org/10.1097/AOG.0000000000002364.
Aukes AM, De Groot JC, Wiegman MJ, Aarnoudse JG, Sanwikarja GS, Zeeman GG. Long-term cerebral imaging after pre-eclampsia. BJOG. 2012;119(9):1117–22. https://doi.org/10.1111/j.1471-0528.2012.03406.x.
Aukes AM, de Groot JC, Aarnoudse JG, Zeeman GG. Brain lesions several years after eclampsia. Am J Obstet Gynecol. 2009;200(5):504 e1–5. https://doi.org/10.1016/j.ajog.2008.12.033S0002-9378(08)02439-3.
Enzinger C, Fazekas F, Ropele S, Schmidt R. Progression of cerebral white matter lesions - clinical and radiological considerations. J Neurol Sci. 2007;257:5–10. https://doi.org/10.1016/j.jns.2007.01.018.
Prins ND vDE, den Heijer T, et al. Cerebral white matter lesions and the risk of dementia. Arch Neurol. 2004;61:1531–4.
Wiegman MJ, Zeeman GG, Aukes AM, Bolte AC, Faas MM, Aarnoudse JG, et al. Regional distribution of cerebral white matter lesions years after preeclampsia and eclampsia. Obstet Gynecol. 2014;123(4):790–5. https://doi.org/10.1097/AOG.0000000000000162.
Raman MR, Tosakulwong N, Zuk SM, Senjem ML, White WM, Fields JA, et al. Influence of preeclampsia and late-life hypertension on MRI measures of cortical atrophy. J Hypertens. 2017;35:2479–85. https://doi.org/10.1097/HJH.0000000000001492.
• Brewer J, Owens MY, Wallace K, Reeves AA, Morris R, Khan M, et al. Posterior reversible encephalopathy syndrome in 46 of 47 patients with eclampsia. Am J Obstet Gynecol. 2013;208(6):468 e1-6. https://doi.org/10.1016/j.ajog.2013.02.015 One of the largest studies characterizing cerebral edema in eclampsia.
Mielke MM, Milic NM, Weissgerber TL, White WM, Kantarci K, Mosley TH, et al. Impaired cognition and brain atrophy decades after hypertensive pregnancy disorders. Circ Cardiovasc Qual Outcomes. 2016;9:S70–S6. https://doi.org/10.1161/CIRCOUTCOMES.115.002461.
Siepmann T, Boardman H, Bilderbeck A, Griffanti L, Kenworthy Y, Zwager C, et al. Long-term cerebral white and gray matter changes after preeclampsia. Neurology. 2017;88:1256–64. https://doi.org/10.1212/WNL.0000000000003765.
Elharram M, Dayan N, Kaur A, Landry T, Pilote L. Long-term cognitive impairment after preeclampsia: a systematic review and meta-analysis. Obstet Gynecol. 2018;132(2):355–64. https://doi.org/10.1097/AOG.0000000000002686.
Cipolla MJ. Cerebrovascular function in pregnancy and eclampsia. Hypertension. 2007;50:14–24. https://doi.org/10.1161/HYPERTENSIONAHA.106.079442.
Johnson AC, Nagle KJ, Tremble SM, Cipolla MJ. The contribution of normal pregnancy to eclampsia. PLoS One. 2015;10(7):e0133953. https://doi.org/10.1371/journal.pone.0133953.
Cipolla MJ, Kraig RP. Seizures in women with preeclampsia: mechanisms and management. Fetal Matern Med Rev. 2011;22(2):91–108. https://doi.org/10.1017/S0965539511000040.
Coughlin WF, McMurdo SK, Reeves T. MR imaging of postpartum cortical blindness. J Comput Assist Tomogr. 1989;13(4):572–6.
Trommer BL, Homer D, Mikhael MA. Cerebral vasospasm and eclampsia. Stroke. 1988;19(3):326–9.
Busija DW, Heistad DD. Factors involved in the physiological regulation of the cerebral circulation. Rev Physiol Biochem Pharmacol. 1984;101:161–211.
Donaldson JO. Eclamptic hypertensive encephalopathy. Semin Neurol. 1988;8:230–3. https://doi.org/10.1055/s-2008-1041383.
Zeeman GG, Cipolla MJ, Cunningham FG. Cerebrovascular (patho)physiology in preeclampsia/eclampsia. Chesley’s Hypertensive Disorders in Pregnancy. 2009:227–47. https://doi.org/10.1016/B978-0-12-374213-1.00013-6.
Aagaard-Tillery KM, Belfort MA. Eclampsia: morbidity, mortality, and management. Clin Obstet Gynecol. 2005;48:12–23. https://doi.org/10.1097/01.grf.0000153882.58132.ba.
•• van Veen TR, Panerai RB, Haeri S, Griffioen AC, Zeeman GG, Belfort MA. Cerebral autoregulation in normal pregnancy and preeclampsia. Obstet Gynecol. 2013;122:1064–9. https://doi.org/10.1097/AOG.0b013e3182a93fb5 The first study to describe the dynamic cerebral autoregulation in preeclampsia.
Brunner H, Cockcroft JR, Deanfield J, Donald A, Ferrannini E, Halcox J, et al. Endothelial function and dysfunction. Part II: association with cardiovascular risk factors and diseases. A statement by the Working Group on Endothelins and Endothelial Factors of the European Society of Hypertension. J Hypertens. 2005;23(2):233–46.
Roberts JM. Endothelial dysfunction in preeclampsia. Semin Reprod Endocrinol. 1998;16(1):5–15. https://doi.org/10.1055/s-2007-1016248.
Abbott NJ. Evidence for bulk flow of brain interstitial fluid: significance for physiology and pathology. Neurochem Int. 2004;45:545–52. https://doi.org/10.1016/j.neuint.2003.11.006.
Haseloff RF, Dithmer S, Winkler L, Wolburg H, Blasig IE. Transmembrane proteins of the tight junctions at the blood-brain barrier: structural and functional aspects. Semin Cell Dev Biol. 2015;38:16–25. https://doi.org/10.1016/j.semcdb.2014.11.004.
Keaney J, Campbell M. The dynamic blood-brain barrier. FEBS J. 2015;282:4067–79. https://doi.org/10.1111/febs.13412.
Daneman R, Prat A. The blood - brain barrier. Dev Med Child Neurol. 2015;3:311–4. https://doi.org/10.1111/j.1469-8749.1961.tb15323.x.
Decleves X, Jacob A, Yousif S, Shawahna R, Potin S, Scherrmann J-M. Interplay of drug metabolizing CYP450 enzymes and ABC transporters in the blood-brain barrier. Curr Drug Metab. 2011;12:732–41. https://doi.org/10.2174/138920011798357024.
Liao MZ, Gao C, Shireman LM, Phillips B, Risler LJ, Neradugomma NK, et al. P-gp/ABCB1 exerts differential impacts on brain and fetal exposure to norbuprenorphine. Pharmacol Res. 2017;119:61–71. https://doi.org/10.1016/j.phrs.2017.01.018.
•• Warrington JP, Fan F, Murphy SR, Roman RJ, Drummond HA, Granger JP, et al. Placental ischemia in pregnant rats impairs cerebral blood flow autoregulation and increases blood-brain barrier permeability. Physiol Rep. 2014;2(8). https://doi.org/10.14814/phy2.12134 This manuscript shown evidences of placental-derived moleculas that impairs brain circulation.
Clayton AM, Shao Q, Paauw ND, Giambrone AB, Granger JP, Warrington JP. Postpartum increases in cerebral edema and inflammation in response to placental ischemia during pregnancy. Brain Behav Immun. 2018;70:376–89. https://doi.org/10.1016/j.bbi.2018.03.028.
Black KD, Horowitz JA. Inflammatory markers and preeclampsia: a systematic review. Nurs Res. 2018;67:242–51. https://doi.org/10.1097/NNR.0000000000000285.
LaMarca BD, Ryan MJ, Gilbert JS, Murphy SR, Granger JP. Inflammatory cytokines in the pathophysiology of hypertension during preeclampsia. Curr Hypertens Rep. 2007;9:480–5. https://doi.org/10.1007/s11906-007-0088-1.
Warrington JP, Drummond HA, Granger JP, Ryan MJ. Placental ischemia-induced increases in brain water content and cerebrovascular permeability: role of TNFα. Am J Physiol Regul Integr Comp Physiol. 2015:ajpregu.00372.2015. https://doi.org/10.1152/ajpregu.00372.2015.
•• Amburgey OA, Chapman AC, May V, Bernstein IM, Cipolla MJ. Plasma from preeclamptic women increases blood-brain barrier permeability: role of vascular endothelial growth factor signaling. Hypertension. 2010;56(5):1003–8. https://doi.org/10.1161/HYPERTENSIONAHA.110.158931 This manuscript shown evidences of involvment of VEGFR2 in the blood brain barrier alterations induced by plasma from preeclampsia.
Li X, Han X, Bao J, Liu Y, Ye A, Thakur M, et al. Nicotine increases eclampsia-like seizure threshold and attenuates microglial activity in rat hippocampus through the alpha7 nicotinic acetylcholine receptor. Brain Res. 1642;2016:487–96. https://doi.org/10.1016/j.brainres.2016.04.043.
Huang Q, Liu L, Hu B, Di X, Brennecke SP, Liu H. Decreased seizure threshold in an eclampsia-like model induced in pregnant rats with lipopolysaccharide and pentylenetetrazol treatments. PLoS One. 2014;9(2):e89333. https://doi.org/10.1371/journal.pone.0089333.
Eigenmann DE, Xue G, Kim KS, Moses AV, Hamburger M, Oufir M. Comparative study of four immortalized human brain capillary endothelial cell lines, hCMEC/D3, hBMEC, TY10, and BB19, and optimization of culture conditions, for an in vitro blood–brain barrier model for drug permeability studies. Fluids Barriers CNS. 2013:1–17.
Rahman NA, ANaHM R, Meyding-Lamade U, Craemer EM, Diah S, Tuah AA, et al. Immortalized endothelial cell lines for in vitro blood–brain barrier models: a systematic review. Brain Res. 1642;2016:532–45. https://doi.org/10.1016/j.brainres.2016.04.024.
Dauchy S, Dutheil F, Weaver RJ, Chassoux F, Daumas-Duport C, Couraud PO, et al. ABC transporters, cytochromes P450 and their main transcription factors: expression at the human blood-brain barrier. J Neurochem. 2008;107(6):1518–28. https://doi.org/10.1111/j.1471-4159.2008.05720.x.
Dutheil F, Jacob A, Dauchy S, Beaune P, Scherrmann JM, Decleves X, et al. ABC transporters and cytochromes P450 in the human central nervous system: influence on brain pharmacokinetics and contribution to neurodegenerative disorders. Expert Opin Drug Metab Toxicol. 2010;6(10):1161–74. https://doi.org/10.1517/17425255.2010.510832.
Ohtsuki S, Ikeda C, Uchida Y, Sakamoto Y, Miller F, Glacial F, et al. Quantitative targeted absolute proteomic analysis of transporters, receptors and junction proteins for validation of human cerebral microvascular endothelial cell line hCMEC/D3 as a human blood-brain barrier model. Mol Pharm. 2013;10(1):289–96. https://doi.org/10.1021/mp3004308.
Bosworth AM, Faley SL, Bellan LM, Lippmann ES. Modeling neurovascular disorders and therapeutic outcomes with human-induced pluripotent stem cells. Front Bioeng Biotechnol. 2017;5:87. https://doi.org/10.3389/fbioe.2017.00087.
• Nielsen SSE, Siupka P, Georgian A, Preston JE, Tóth AE, Yusof SR, et al. Improved method for the establishment of an in vitro blood-brain barrier model based on porcine brain endothelial cells. J Vis Exp. 2017. https://doi.org/10.3791/56277 Key paper for understanding characteristics of in vitro model of brain blood barrier based in porcine endothelial cells.
Smith M, Omidi Y, Gumbleton M. Primary porcine brain microvascular endothelial cells: biochemical and functional characterisation as a model for drug transport and targeting. J Drug Target. 2007;15:253–68. https://doi.org/10.1080/10611860701288539.
Zhang Y, Li CSW, Ye Y, Johnson K, Poe J, Johnson S, et al. Porcine brain microvessel endothelial cells as an in vitro model to predict in vivo blood-brain barrier permeability. Drug Metab Dispos. 2006;34:1–15. https://doi.org/10.1124/dmd.105.006437.which.
Culot M, Lundquist S, Vanuxeem D, Nion S, Landry C, Delplace Y, et al. An in vitro blood-brain barrier model for high throughput (HTS) toxicological screening. Toxicol in Vitro. 2008;22(3):799–811. https://doi.org/10.1016/j.tiv.2007.12.016.
Helms HC, Hersom M, Kuhlmann LB, Badolo L, Nielsen CU, Brodin B. An electrically tight in vitro blood-brain barrier model displays net brain-to-blood efflux of substrates for the ABC transporters, P-gp, Bcrp and Mrp-1. AAPS J. 2014;16(5):1046–55. https://doi.org/10.1208/s12248-014-9628-1.
Thomsen LB, Burkhart A, Moos T. A triple culture model of the blood-brain barrier using porcine brain endothelial cells, Astrocytes and Pericytes. PLOS ONE. 2015;10:e0134765. https://doi.org/10.1371/journal.pone.0134765.
Helms HC, Waagepetersen HS, Nielsen CU, Brodin B. Paracellular tightness and claudin-5 expression is increased in the BCEC/astrocyte blood-brain barrier model by increasing media buffer capacity during growth. AAPS J. 2010;12(4):759–70. https://doi.org/10.1208/s12248-010-9237-6.
Torres-Vergara P, Penny J. Pro-inflammatory and anti-inflammatory compounds exert similar effects on P-glycoprotein in blood-brain barrier endothelial cells. J Pharm Pharmacol. 2018;70(6):713–22. https://doi.org/10.1111/jphp.12893.
Salmeri M, Motta C, Anfuso CD, Amodeo A, Scalia M, Toscano MA, et al. VEGF receptor-1 involvement in pericyte loss induced by Escherichia coli in an in vitro model of blood brain barrier. Cell Microbiol. 2013;15(8):1367–84. https://doi.org/10.1111/cmi.12121.
Perriere N, Yousif S, Cazaubon S, Chaverot N, Bourasset F, Cisternino S, et al. A functional in vitro model of rat blood-brain barrier for molecular analysis of efflux transporters. Brain Res. 2007;1150:1–13. https://doi.org/10.1016/j.brainres.2007.02.091.
Forster C, Silwedel C, Golenhofen N, Burek M, Kietz S, Mankertz J, et al. Occludin as direct target for glucocorticoid-induced improvement of blood-brain barrier properties in a murine in vitro system. J Physiol. 2005;565(Pt 2):475–86. https://doi.org/10.1113/jphysiol.2005.084038.
Alms D, Fedrowitz M, Romermann K, Noack A, Loscher W. Marked differences in the effect of antiepileptic and cytostatic drugs on the functionality of p-glycoprotein in human and rat brain capillary endothelial cell lines. Pharm Res. 2014;31(6):1588–604. https://doi.org/10.1007/s11095-013-1264-4.
Neuhaus W, Stessl M, Strizsik E, Bennani-Baiti B, Wirth M, Toegel S, et al. Blood-brain barrier cell line PBMEC/C1-2 possesses functionally active P-glycoprotein. Neurosci Lett. 2010;469(2):224–8. https://doi.org/10.1016/j.neulet.2009.11.079.
Neuhaus W, Plattner VE, Wirth M, Germann B, Lachmann B, Gabor F, et al. Validation of in vitro cell culture models of the blood-brain barrier: tightness characterization of two promising cell lines. J Pharm Sci. 2008;97(12):5158–75. https://doi.org/10.1002/jps.21371.
Belfort MA, Saade GR, Yared M, Grunewald C, Herd JA, Varner MA, et al. Change in estimated cerebral perfusion pressure after treatment with nimodipine or magnesium sulfate in patients with preeclampsia. Am J Obstet Gynecol. 1999;181:402–7. https://doi.org/10.1016/S0002-9378(99)70569-7.
Belfort MA, Tooke-Miller C, Allen JC, Dizon-Townson D, Varner MA. Labetalol decreases cerebral perfusion pressure without negatively affecting cerebral blood flow in hypertensive gravidas. Hypertens Pregnancy. 2002;21:185–97. https://doi.org/10.1081/PRG-120015845.
Belfort MA, Varner MW, Dizon-Townson DS, Grunewald C, Nisell H. Cerebral perfusion pressure, and not cerebral blood flow, may be the critical determinant of intracranial injury in preeclampsia: a new hypothesis. Am J Obstet Gynecol. 2002;187:626–34. https://doi.org/10.1067/mob.2002.125241.
Van Veen TR, Panerai RB, Haeri S, Singh J, Adusumalli JA, Zeeman GG, et al. Cerebral autoregulation in different hypertensive disorders of pregnancy. Am J Obstet Gynecol. 2015;212:513.e1-.e7. https://doi.org/10.1016/j.ajog.2014.11.003.
Valdueza JM, Balzer JO, Villringer A, Vogl TJ, Kutter R, Einhaupl KM. Changes in blood flow velocity and diameter of the middle cerebral artery during hyperventilation: assessment with MR and transcranial Doppler sonography. AJNR Am J Neuroradiol. 1997;18(10):1929–34.
Williams DS, Detre JA, Leigh JS, Koretsky AP. Magnetic resonance imaging of perfusion using spin inversion of arterial water. Proc Natl Acad Sci U S A. 1992;89(1):212–6.
Le Bihan D. Intravoxel incoherent motion imaging using steady-state free precession. Magn Reson Med. 1988;7(3):346–51.
Nelander M, Hannsberger D, Sundstrom-Poromaa I, Bergman L, Weis J, Akerud H, et al. Assessment of cerebral perfusion and edema in preeclampsia with intravoxel incoherent motion MRI. Acta Obstet Gynecol Scand. 2018;97(10):1212–8. https://doi.org/10.1111/aogs.13383.
Jansen JF, Backes WH, Nicolay K, Kooi ME. 1H MR spectroscopy of the brain: absolute quantification of metabolites. Radiology. 2006;240(2):318–32. https://doi.org/10.1148/radiol.2402050314.
Oberhaensli RD, Galloway GJ, Hilton-Jones D, Bore PJ, Styles P, Rajagopalan B, et al. The study of human organs by phosphorus-31 topical magnetic resonance spectroscopy. Br J Radiol. 1987;60(712):367–73. https://doi.org/10.1259/0007-1285-60-712-367.
Nelander M, Weis J, Bergman L, Larsson A, Wikstrom AK, Wikstrom J. Cerebral magnesium levels in preeclampsia; a phosphorus magnetic resonance spectroscopy study. Am J Hypertens. 2017;30(7):667–72. https://doi.org/10.1093/ajh/hpx022.
Nelander M, Wikstrom AK, Weis J, Bergman L, Larsson A, Sundstrom-Poromaa I, et al. Cerebral osmolytes and plasma osmolality in pregnancy and preeclampsia: a proton magnetic resonance spectroscopy study. Am J Hypertens. 2018;31(7):847–53. https://doi.org/10.1093/ajh/hpy019.
Rutherford JM, Moody A, Crawshaw S, Rubin PC. Magnetic resonance spectroscopy in pre-eclampsia: evidence of cerebral ischaemia. BJOG. 2003;110(4):416–23.
Sengar AR, Gupta RK, Dhanuka AK, Roy R, Das K. MR imaging, MR angiography, and MR spectroscopy of the brain in eclampsia. AJNR Am J Neuroradiol. 1997;18(8):1485–90.
Marchi N, Cavaglia M, Fazio V, Bhudia S, Hallene K, Janigro D. Peripheral markers of blood-brain barrier damage. Clin Chim Acta. 2004;342(1–2):1–12. https://doi.org/10.1016/j.cccn.2003.12.008.
Kanner AA, Marchi N, Fazio V, Mayberg MR, Koltz MT, Siomin V, et al. Serum S100β: a noninvasive marker of blood-brain barrier function and brain lesions. Cancer. 2003;97:2806–13. https://doi.org/10.1002/cncr.11409.
Schmidt A, Tort A, Amaral O, Schmidt A, Walz R, Vettorazzi-Stuckzynski J, et al. Serum S100B in pregnancy-related hypertensive dis- orders: a case–control study. Clin Chem. 2004;50:435–8. https://doi.org/10.1373/clinchem.2003.027391.
Vettorazzi J, Torres FV, de Avila TT, Martins-Costa SH, Souza DO, Portela LV, et al. Serum S100B in pregnancy complicated by preeclampsia: a case-control study. Pregnancy Hypertens. 2012;2(2):101–5. https://doi.org/10.1016/j.preghy.2011.11.004.
Bergman L, Akhter T, Wikstrom AK, Wikstrom J, Naessen T, Akerud H. Plasma levels of S100B in preeclampsia and association with possible central nervous system effects. Am J Hypertens. 2014;27(8):1105–11. https://doi.org/10.1093/ajh/hpu020.
Artunc-Ulkumen B, Guvenc Y, Goker A, Gozukara C. Maternal serum S100-B, PAPP-A and IL-6 levels in severe preeclampsia. Arch Gynecol Obstet. 2015;292:97–102. https://doi.org/10.1007/s00404-014-3610-0.
Chou SHY, Robertson CS. Monitoring biomarkers of cellular injury and death in acute brain injury. Neurocrit Care. 2014;21:187–214. https://doi.org/10.1007/s12028-014-0039-z.
Bergman L, Akerud H. Plasma levels of the cerebral biomarker, neuron-specific enolase, are elevated during pregnancy in women developing preeclampsia. Reprod Sci. 2016;23(3):395–400. https://doi.org/10.1177/1933719115604732.
Bergman L, Akerud H, Wikström AK, Larsson M, Naessen T, Akhter T. Cerebral biomarkers in women with preeclampsia are still elevated 1 year postpartum. Am J Hypertens. 2016;29:1374–9. https://doi.org/10.1093/ajh/hpw097.
Bogoslovsky T, Gill J, Jeromin A, Davis C, Diaz-Arrastia R. Fluid biomarkers of traumatic brain injury and intended context of use. Diagnostics. 2016;6:1–22. https://doi.org/10.3390/diagnostics6040037.
Randall J, Mörtberg E, Provuncher GK, Fournier DR, Duffy DC, Rubertsson S, et al. Tau proteins in serum predict neurological outcome after hypoxic brain injury from cardiac arrest: results of a pilot study. Resuscitation. 2013;84:351–6. https://doi.org/10.1016/j.resuscitation.2012.07.027.
Evers KS, Atkinson A, Barro C, Fisch U, Pfister M, Huhn EA, et al. Neurofilament as neuronal injury blood marker in preeclampsia. Hypertension. 2018;71(6):1178–84. https://doi.org/10.1161/HYPERTENSIONAHA.117.10314.
• Bergman L, Zetterberg H, Kaihola H, Hagberg H, Blennow K, Akerud H. Blood-based cerebral biomarkers in preeclampsia: plasma concentrations of NfL, tau, S100B and NSE during pregnancy in women who later develop preeclampsia - A nested case control study. PLoS One. 2018;13(5):e0196025. https://doi.org/10.1371/journal.pone.0196025 Cerebral biomarkers are increased in preeclampsia, reinforcing cerebral involvement.
Acknowledgments
The authors would like to thank Emily Gatu for her editorial assistance and the researchers belonging to GRIVAS health for their valuable input.
Funding Disclose
This manuscript was supported by Conicyt grant REDI170373. CE is supported by DIUBB 184309 4/R.
Authors’ Roles
CE and LB: designed and wrote the manuscript. All co-authors included their respective sections according to expertise. JMR, AKW, and JP contributed to the writing of the manuscript and provided a critical revision of its contents. All co-authors approved the final version of this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interest relevant to this manuscript.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Preeclampsia
Electronic Supplementary Material
ESM 1
(DOCX 28 kb)
Rights and permissions
About this article
Cite this article
Bergman, L., Torres-Vergara, P., Penny, J. et al. Investigating Maternal Brain Alterations in Preeclampsia: the Need for a Multidisciplinary Effort. Curr Hypertens Rep 21, 72 (2019). https://doi.org/10.1007/s11906-019-0977-0
Published:
DOI: https://doi.org/10.1007/s11906-019-0977-0