Abstract
Background
The discovery of vaccines significantly reduced morbidity and mortality of infectious diseases and led to the elimination and eradication of some. Development of safe and effective vaccines is a critical step to the control of infectious diseases; however, there is the need to address vaccine hesitancy because of its potential impact on vaccine uptake.
Methods
We conducted a narrative review of studies on interventions to address measles and human papillomavirus vaccine hesitancy. We discussed how lessons learned from these studies could be applied towards COVID-19 and future human immunodeficiency virus vaccines.
Results
We found that there are several successful approaches to improving vaccine acceptance. Interventions should be context specific and build on the challenges highlighted in various settings.
Conclusion
Strategies could be used alone or in combination with others. The most successful interventions directly targeted the population for vaccination. Use of financial incentives could be a potential tool to improve vaccine uptake.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There have been major global efforts to address two highly significant epidemics—human immunodeficiency virus (HIV) and SARS-CoV-2. One of those efforts is the development of vaccines to prevent severe diseases that could result from these infections. Historically, vaccines are the most effective means of preventing illness and death from infectious diseases. The discovery of vaccines has led to reduced morbidity and mortality from several infectious diseases and the elimination and eradication of some [1]. Development of safe and effective vaccines is a huge step to the control of epidemics; however, there is the need to address vaccine hesitancy because of its potential impact on vaccine uptake. Vaccine acceptance is critical in the successful control of pandemics. Successful immunization programs have resulted in the prevention of 4–5 million deaths among all age groups annually, making it one of the most successful and cost-effective public health interventions [2].
WHO defines vaccine hesitancy as a “delay in acceptance or refusal of vaccines despite availability of vaccination services.” The resurgence of infectious diseases in recent years can be attributed to vaccine refusals [3,4,5,6]. Vaccine hesitancy are among the 10 threats to global health in 2019 [7,8,9,10,11]. Many individuals are left unvaccinated, leaving them and others around them vulnerable to diseases. With the introduction of the COVID-19 vaccines, there is a greater potential to save millions of lives with vaccines [12]. However, the spread of vaccine misinformation and anti-information has resulted in an increase in vaccine mistrust in the US. False vaccination information is spreading rapidly through social media, causing general distrust and safety concerns, thereby worsening vaccination hesitancy [13] and disease outcomes [14]. There is scientific evidence that anti-vaccination activities organized via social media is predictive of the belief that vaccines are not safe and the prevalence of vaccine disinformation predicts a decrease in mean vaccination coverage over time [15]. Vaccination misinformation may have significant and lasting impacts on vaccine acceptance overall. Poor vaccination rates mean high cases of preventable illnesses, suffering, death [16,17,18,19,20,21,22], and huge economic burden [9, 23]. Poor vaccine coverage and the persistent high burden of vaccine-preventable diseases despite proven vaccine safety and effectiveness imply the need to identify drivers of vaccine hesitance and effective intervention strategies to reduce both among this population. Reducing the spread of false vaccine information or the proportion of people who believe them could reduce the effects of these harmful messages [14]. As the world undertakes the largest vaccination campaign in human history to combat COVID-19 and HIV, past research on strategies for vaccine acceptance and hesitancy can inform our understanding of these issues. Lessons learned from interventions to improve measles and human papillomavirus (HPV) vaccine uptake could be applied to improve uptake of COVID-19 vaccine and potential future HIV vaccine(s).
Human Immunodeficiency Virus (HIV) Vaccine
Since HIV was identified as the cause of acquired immune deficiency syndrome (AIDS) in 1984, millions of people have been affected worldwide. HIV/AIDS incurs huge morbidity, mortality, and economic burden globally. The introduction of the anti-retroviral therapy (ART) resulted in a decrease in the public health and economic burden [24]; however, with a slower decrease in number of new cases, prevalence of the infection continues to rise. Currently, more than 36 million people are infected with HIV and more than 39 million AIDS-related deaths have occurred globally [25]. This is a major public health problem for years to come.
There is currently no vaccine for HIV, but scientists have been working on developing one for over 30 years [26]. The challenges with producing a HIV vaccine lies in the structure and characteristics of the HIV to mutate rapidly and evade the human immune system. This means that a vaccine for one HIV subtype may prevent that specific subtype but be completely ineffective against another HIV subtype. However, extensive progress has been made. The National Institutes for Health (NIH) is investing in a safe and effective preventive HIV vaccine [27]. There is evidence that an upcoming HIV vaccine trial looks promising.
COVID-19 Vaccine
In December 2019, a cluster of cases of pneumonia was reported in Wuhan, Hubei Province [28]. A novel coronavirus (SARS-CoV-2) was identified as the cause and the disease was called coronavirus disease 2019 (COVID-19). In January 2020, the World Health Organization (WHO) declared COVID-19 a public health emergency of international concern, the highest level of alarm under international law. As of May 2021, more than 160 million confirmed cases of SARS-CoV-2 infections and 3.5 million confirmed deaths had been recorded [29]. In December 2020, the US Food and Drug Administration (FDA) issued the first Emergency Use Authorization (EUA) for the use of a vaccine to protect against COVID-19 and by February 2021, there were three authorized COVID-19 vaccines [30]—Pfizer-BioNTech, Moderna, and Johnson and Johnson vaccines.
Vaccination is a crucial part of ending an epidemic; therefore, widespread vaccine uptake is critical for the control of the global pandemic through vaccine. COVID-19 vaccine has the potential to save lives if its uptake is optimal. Studies show a mixed response to COVID-19 vaccine acceptance globally [31,32,33,34] and even among healthcare workers [35, 36]. A study conducted in early 2020 among adults in the United States (US) found a COVID-19 vaccine acceptance rate of 67% [37]. Current data show that as of August 2021, 70% of US adults have received at least one dose of the COVID-19 vaccine [38]; however, there continues to be a geographic and racial/ethnic disparity in the acceptance of the vaccine, with Blacks and Hispanics having persistently low vaccination coverage [39]. In low-income countries, only 1.1% have received at least one dose [40]. Poor COVID-19 vaccine coverage could have negative impacts on the efforts to control the pandemic. There is a need for public health officials and policymakers to prioritize effective strategies to promote acceptance and uptake of COVID-19 vaccines and potential HIV vaccines, especially for those who are most vulnerable. We conducted a narrative review of studies on interventions to address measles and HPV vaccine hesitancy. We discussed how these strategies could be applied towards COVID-19 and potential HIV vaccines.
Search Strategy
We used a subset of the studies that were retrieved for a different systematic review for this review. We searched the database Medline on the platform Ovid, using the Medline All segment, which includes non-Medline PubMed records. Relevant material from journals not indexed in Medline was identified through citation chaining.
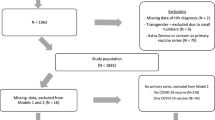
Documents from before 1990 and documents indexed as addressing animals without also addressing humans were not retrieved. When run on March 12, 2020, the search retrieved 14,753 records, of which 47 were later identified as duplicates. The records were uploaded to Covidence for screening. In addition to the database searching, 412 additional potentially relevant studies were identified through citation chaining and uploaded to Covidence for screening. The final search terms used for searching translated to PubMed search query is as follows:
PubMed translation of final search terms |
(vaccines[mh] OR immunization[mh] OR vaccin*[tw] or immunis*[tw] or immuniz*[tw] or inoculat*[tw]) AND (intervention[tw] or interventions[tw] or treatment[tw] or treatments[tw] or group[tw] or groups[tw] or trial[tw] or trials[tw] or program[tw] or programs[tw] or programme[tw] or programmes[tw] or evaluat*[tiab] or experiment*[tiab]) AND (behavior[mh] OR behav*[tw] OR incentiv*[tw] OR psychology[subheading] OR motivate*[tw] OR motivation[mh]) NOT (animals[mh:noexp] NOT humans[mh]) AND (1990:3000[pdat]) |
Study Selection
For this review, we included those articles that discussed a behavioral insights intervention on vaccination and excluded those that were explanatory, were not intervention-based, or did not look at vaccine uptake, intent, knowledge, or attitudes as an outcome measure, or were not in English. We also excluded systematic reviews and meta-analyses. We restricted the studies to those that mentioned either measles or HPV in the title or abstract and those that were published after 2005 when the HPV vaccine became available. Two reviewers independently reviewed the records selected for manual screening to identify final studies to include. We included 152 studies on vaccine promotion strategies with diverse contents that were implemented in different settings and targeting various populations (Fig. 1).
Study Outcomes of Interest
The primary outcome of interest for this review was vaccine uptake with vaccine knowledge, attitudes, and intent as secondary outcomes of interest. Outcome was defined as per each study’s criteria. Some studies classified outcomes based on self-reports while others used clinical or insurance records.
The studies were classified under the 9 types of vaccine promotion intervention domains. These include Education campaigns (providing education on vaccination, disease, and how vaccine work); On-site vaccination (providing vaccine at workplace or places of worship); Incentives (offering financial incentives for vaccination); Free vaccination (providing vaccination free of cost); Institutional recommendation (a recommendation made by the institution that person works at especially for healthcare providers); Provider recommendation (recommendation by doctor/nurse); Reminder and recall (reminders for vaccination); Message framing (gain vs loss framing of the vaccine), and Vaccine champion (institutionally appointed champion that encouraged vaccination). If more than one type of domain was employed, we classified the study as multi-component.
HPV Vaccine Promotion
We identified 131 articles conducted from 2006 to 2020 that discussed behavioral insights interventions to promote HPV vaccinations (Table 1). Most of the studies were conducted in the US (Table 2). A majority focused on educational interventions, either alone or in combination with one or more strategies. Educational interventions included use of pamphlets, movies, workshops, consulting sessions, and live/virtual sessions to educate on HPV topics and the role of vaccination. The most common target population for the interventions was females, either as HPV vaccine–eligible individuals or their parents/guardians. Some interventions were targeted towards adolescents only [41,42,43,44,45,46,47,48,49,50,51,52,53], young adults only [54,55,56,57,58,59,60], or a combination of both [61,62,63,64,65], while the rest targeted key decision-makers parents/guardians only [47, 52, 66,67,68,69,70,71,72,73,74], parent/child dyads [75,76,77,78,79], or parents/healthcare staff/school staff [80]. None of the interventions focused on adolescent boys only. There were also educational interventions that focused on educating healthcare workers [46, 49,50,51, 62, 81,82,83,84,85]. Some studies used multi-component strategies, i.e., a combination of two or more interventions. Many programs were implemented in schools, universities, or healthcare settings to target adolescents and/or adults.
Some authors focused on improving HPV vaccine uptake for specific populations such as girls only [43, 44, 47, 72, 75, 76, 78, 80, 86,87,88,89], young men only [55, 56, 90], or young women only [54, 91,92,93,94,95,96,97], college students [55, 58, 90,91,92, 97, 98], gay and bisexual men [80, 99, 100], female entertainment sex workers [101], and ethnicity/races [59, 60, 62, 76, 77, 89] (Table 3). Fewer studies targeted specific groups such as a race/ethnicity or a gender. The common outcome variables studied include intention to get HPV vaccine, actual HPV vaccine uptake, HPV or HPV vaccine awareness or knowledge, risk perception for HPV, attitudes towards HPV vaccination, perceived norm, and self-efficacy to get vaccinated against HPV.
Overall, educational interventions improved HPV or HPV vaccine awareness and knowledge [41,42,43,44, 66, 67, 75, 86,87,88], beliefs [41], risk perception for HPV [87], perceived effectiveness of HPV vaccine [69], attitudes towards HPV vaccination [41,42,43, 45, 67, 75, 86, 88], self-efficacy to get vaccinated against HPV, perceived norms [88], acceptance or intention to get vaccinated against HPV [43,44,45, 54, 55, 66, 67, 75, 88], and actual HPV vaccine uptake [46, 68, 75, 81, 82]. Improved awareness and knowledge of HPV and HPV vaccine were usually associated with intention to get vaccinated or actual vaccine uptake; however, some studies found non-significant difference in HPV vaccine acceptability after an educational intervention [71]. Educational interventions designed to target specific populations were effective in improving vaccine knowledge and this was usually associated with improved vaccine acceptance. Parents and caregivers were more confident in HPV vaccine when they were exposed to messages that addressed lack of knowledge about the vaccine [52, 67, 72, 74, 75, 77,78,79, 83]. Some of the educational interventions targeted providers, while others targeted young adults as catch-up populations.
The second most common intervention was message framing [64, 102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119]. This involves describing the benefits of receiving (gain-framed message) or the costs of not receiving (loss-framed message) HPV vaccine to the participants. This type of intervention was employed mainly for adults. The effect of message framing on HPV vaccine acceptance was moderated by perceived susceptibility [106, 110, 112] in some cases. Overall, message framing improved HPV knowledge and HPV vaccine acceptance; however, this depended on the other variables in the study and use of appropriately framed messages for target populations. Some authors found that HPV vaccination intention and uptake were not influenced by message framing [102, 111]. Furthermore, some authors found some types of message framing to have negative effects on vaccine acceptance [103]. Gainforth and Latimer found that participants (women) who were exposed to high-risk information perceived HPV vaccine as having higher response cost and were less motivated to receive HPV vaccine compared to women who received low-risk information. The authors observed that this negative effect of high-risk information may be mitigated by appropriately framed messages. Increase in vaccination intentions and uptake significantly improved when messages were tailored to the target population [90].
The use of financial incentives improved HPV vaccine initiation and completion in England [120] and the US [121]. Impacts were not moderated by deprivation level and decision quality was unaffected by the intervention. Offering financial incentives to the participants was associated with significant increase in both the initiation and completion of HPV vaccines series.
Free-of-charge HPV vaccine was associated with increased vaccine uptake in some settings. In a study conducted in Australia, when HPV vaccine was offered free of charge, there was a significant increase in vaccine coverage from 42.6 to 73.2% among gay and bisexual men [122]. In Denmark, free vaccination increased coverage for adolescent girls to 80% for dose 1, 75% for dose 2, and 62% for dose 3 [122]. High-risk participants or those who perceived themselves to be at risk were more likely to accept the free vaccine [100]. On the other hand, the study in the US found that the proportion of adolescent and adult females completing three on-time HPV4 doses was 21% vs. 18% respectively. No adolescent receiving free HPV4 vaccine completed three doses. Grant sponsorship of at least one HPV4 dose among adults did not predict three dose on-time completion (OR = 1.56, 95%CI: 0.80, 3.06). Neither was adult grant sponsorship of HPV4 significant when analyzing exclusive payor sources vs. a combination of payor sources (OR = 0.72, 95%CI: 0.10, 5.17).
Most of the studies that employed multi-component strategies included education of target populations in addition to one or more other domains, e.g., on-site vaccination [123,124,125,126,127], vaccine champion [124, 128, 129], free vaccination [124, 125], reminder and recall [129,130,131,132,133,134], incentives [135, 136], provider recommendation [126, 130, 137, 138], and provider/staff training sessions [138]. Some studies combined message framing with one or more strategies such as free vaccine [139], and reminder and recall [140]. Another popular intervention was provider recommendation, either alone [141] or in combination with one or more strategies such as provider education [142, 143], reminder and recall [144,145,146,147], on site vaccination [148,149,150], incentive [146], vaccine champion [147], and free vaccination [147]. Tull et al. combined reminder and recall strategy with vaccination champion and on-site vaccination [151]. Multi-component strategies were used in variety of settings including school-based [124, 125, 127, 128, 133, 139, 142, 148, 151], healthcare [129, 130, 132, 134, 136, 137, 145,146,147, 150], and public settings [126, 131, 135, 144]. Target populations for multi-component strategies include adults as either vaccine decision-makers for adolescents or catch-up populations [123, 142], adolescents [123, 126, 128, 130,131,132, 134, 137, 138, 145, 146, 148, 150, 151], adolescent girls only [125, 127, 135, 140, 144], both adults and adolescents [129, 136, 147, 152], and healthcare providers [137]. Multi-component strategies were successful in improving HPV knowledge among adults and adolescents [131, 139], healthcare providers’ attitudes, subjective norms about HPV vaccination, and their perceived behavioral control to recommend HPV vaccination [137], as well as vaccine acceptance among parents/guardians [126, 127, 130, 131, 140, 144, 150] and adolescents [126, 131, 139, 148]. It significantly improved vaccine initiation and completion among girls [124, 125, 127, 135, 144], young adults [123, 142], adolescents [123, 126, 128, 131,132,133,134, 138, 145, 146, 150, 151], and adults and adolescents [126, 129, 136, 147, 152]. Our findings indicate that multi-component strategies were efficient for all study settings and target populations, and significantly had positive effects on outcome variables.
The use of patient reminder system alone was acceptable to parents [153, 154] and increased rates of vaccine initiation and completion among adolescent girls [155, 156], adolescents [153, 156,157,158,159], young adult females [156, 160,161,162], and young adults [163]. It increased vaccine intention in adults [154, 163]. However, some authors found no significant increase in completion rates [160] and no significant difference in the completion rates between intervention and control groups [153, 158, 160, 161]. The effect of this strategy appeared to be stronger in girls aged 9–17 years compared with young women aged 18–26 years at the first dose and in blacks compared with whites [156].
Use of vaccine champions were found to be successful in improving HPV vaccine coverage up to the 80 s or 90 s percentage among adolescent girls [125, 127, 164,165,166].
Measles Vaccine Promotion
We identified 22 studies focused on measles vaccine promotion interventions of which 6 focused on education only, 6 on message framing, 2 on health systems only, 2 on financial incentives only, 1 on reminder and recall only, 1 on on-site/closer vaccination only, and 1 on provider recommendation only. Three of the studies employed multi-component strategies (Table 4). Studies focused on educational interventions found that use of a decision aid [167], using spiritual leaders [168], and combining education with on-site vaccination [169] (campaigns) can increase measles vaccination coverage. Studies on messaging found that both gain-framed and loss-framed messaging and messages focused on disease risk can improve intent to vaccinate their children in parents [170,171,172,173]. Financial incentives both at the level of the provider and at the level of the consumer can improve vaccine uptake [174,175,176,177]. There is also some evidence suggesting that there might be a dose response relationship between vaccine uptake and monetary amount for incentives being offered to the consumer [177]. Reminder and recall [178] strategy alone did not improve vaccine uptake but when paired with financial incentives [175, 177], significantly improved vaccination rates. Studies that focused on improving health system performance and quality also led to improved vaccination coverage [179, 180]. Distance to healthcare facilities is a predictor of service usage including vaccination coverage and can also exacerbate inequities. One study [181] focused on optimal location of vaccination service delivery found that after placing outreach points for vaccination, there was no relationship between distance to health facility and vaccination coverage for measles highlighting the importance of on-site/closer vaccination points (Tables 5 and 6).
Conclusion
Lessons for COVID-19 and Potential HIV Vaccines
Review of the studies on improving measles and HPV vaccinations shows that the most promising strategies include use of multi-component interventions especially when education is included. Educational interventions focused on disease risk can be used to improve coverage of COVID-19 and a future HIV vaccine in diverse populations. Message framing is a powerful tool for vaccine promotion; however, messages should be carefully framed and should be targeted to the population of interest. Financial incentives, free-of-charge vaccines, and use of vaccine champions should be considered in future vaccine promotions as they were successful in increasing both measles and HPV vaccine coverage rates. Interventions should be context specific, be designed to target populations, and build on the challenges highlighted in various settings.
Abbreviations
- HPV:
-
Human papillomavirus
- HIV:
-
Human immunodeficiency virus
- AIDS:
-
Acquired immune deficiency syndrome
- COVID-19:
-
Coronavirus disease-2019
- SARS-CoV-2:
-
Severe acute respiratory syndrome coronavirus-2
- MMR:
-
Measles, mumps, and rubella
References
Greenwood B. The contribution of vaccination to global health: past, present and future. Philos Trans R Soc Lond B Biol Sci. 2014 [cited 2021 May 27];369. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4024226/.
World Health Organization. Immunization. World Health Organization. 2019 [cited 2021 Jul 2]. Available from: https://www.who.int/news-room/facts-in-pictures/detail/immunization
Kubin L. Is there a resurgence of vaccine preventable diseases in the U.S.? J Pediatr Nurs. 2019 [cited 2021 Apr 5];44:115–8. Available from: https://www.sciencedirect.com/science/article/pii/S0882596318303646.
Phadke VK, Bednarczyk RA, Omer SB. Vaccine refusal and measles outbreaks in the US. JAMA. 2020 [cited 2021 Apr 5];Oct 6;324(13):1344-45. https://doi.org/10.1001/jama.2020.14828.
Phadke VK, Bednarczyk RA, Salmon DA, Omer SB. Association between vaccine refusal and vaccine-preventable diseases in the United States. JAMA. 2016 [cited 2021 Apr 22];315(11):1149–1158. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5007135/
Omer SB, Salmon DA, Orenstein WA, deHart MP, Halsey N. Vaccine refusal, mandatory immunization, and the risks of vaccine-preventable diseases. N Engl J Med. 2009 [cited 2021 Mar 28];360(19):1981–1988. Available from: https://doi.org/10.1056/NEJMsa0806477.
Wang X, Ren ZJ. Why does misinformation spread so quickly? Calif Manage Rev. 2020 [cited 2021 Mar 28];63(1). Available from: https://cmr.berkeley.edu/2020/04/misinformation/.
Vaccinate your family. Outbreaks of vaccine-preventable diseases. Vaccinate Your Family. 2021 [cited 2021 Apr 6]. Available from: https://vaccinateyourfamily.org/questions-about-vaccines/outbreaks-of-vaccine-preventable-diseases/
Vaccinate Your Family. State of the Immunion: a report on vaccine-preventable diseases in the U.S. 2018 [cited 2021 Apr 6]. Available from: https://vaccinateyourfamily.org/wp-content/uploads/2018/07/FINALSOTIReport_2018-1.pdf.
Omer SB, Betsch C, Leask J. Mandate vaccination with care. Nature. 2019 [cited 2021 Apr 6];571(7766):469–72. https://doi.org/10.1038/d41586-019-02232-0.
World Health Organization. Ten health issues WHO will tackle this year. World Health Organization. 2019 [cited 2021 Jul 31]. Available from: https://www.who.int/news-room/spotlight/ten-threats-to-global-health-in-2019.
World Health Organization. COVID-19 vaccines. 2020 [cited 2021 Mar 287]. Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/covid-19-vaccines.
Burki T. Vaccine misinformation and social media. Lancet Digit Health. 2019 [cited 2021 Apr 6];1(6):e258–9. Available from: https://www.thelancet.com/journals/landig/article/PIIS2589-7500(19)30136-0/abstract.
Brainard J, Hunter PR. Misinformation making a disease outbreak worse: outcomes compared for influenza, monkeypox, and norovirus. Simulation. 2020;96(4):365–74. https://doi.org/10.1177/0037549719885021. [cited 2021 Apr 6]. Available from: https://journals.sagepub.com/doi/pdf/10.1177/0037549719885021.
Wilson SL, Wiysonge C. Social media and vaccine hesitancy. BMJ Global Health. 2020. [cited 2021 Jul 31];5:e004206. Available from: https://gh.bmj.com/content/5/10/e004206.
Centers for Disease Control and Prevention. What you should know for the 2017-2018 influenza season. Centers for Disease Control and Prevention. 2019; [cited 2021 Apr 5]. Available from: https://www.cdc.gov/flu/about/season/flu-season-2017-2018.htm.
Centers for Disease Control and Prevention. Estimated influenza illnesses, medical visits, hospitalizations, and deaths in the United States — 2018–2019 influenza season. 2020 [cited 2021 Apr 5]. Available from: https://www.cdc.gov/flu/about/burden/2018-2019.html.
Elam-Evans LD. National, regional, state, and selected local area vaccination coverage among adolescents aged 13–17 Years — United States, 2019. MMWR Morb Mortal Wkly Rep. 2020 [cited 2021 May 17]:69. Available from: https://www.cdc.gov/mmwr/volumes/69/wr/mm6933a1.htm.
Sriram S, Ranganathan R. Why human papillomavirus vaccination coverage is low among adolescents in the US? A study of barriers for vaccination uptake. J Family Med Prim Care. 2019 [cited 2021 May 15];8:866–70. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6482788/.
Isenor JE, Edwards NT, Alia TA, Slayter KL, MacDougall DM, McNeil SA, Bowles SK, et al. Impact of pharmacists as immunizers on vaccination rates: a systematic review and meta-analysis. Vaccine. 2016 [cited 2021 May 15];34(47):5708–23. https://doi.org/10.1016/j.vaccine.2016.08.085.
Centers for Disease Control and Prevention. Measles cases and outbreaks. Centers for Disease Control and Prevention. 2021; [cited 2021 Apr 5]. Available from: https://www.cdc.gov/measles/cases-outbreaks.html.
Centers for Disease Control and Prevention. Measles (Rubeola): cases and outbreaks. 2020 [cited 2021 Apr 5]. Available from: https://www.cdc.gov/measles/cases-outbreaks.html.
Vaccinate Your Family. Vaccines are cost saving. Vaccinate Your Family. 2020; [cited 2021 Apr 5]. Available from: https://vaccinateyourfamily.org/why-vaccinate/vaccine-benefits/costs-of-disease-outbreaks/.
National Institute of Allergy and Infectious Diseases. Antiretroviral drug discovery and development. National Institute of Allergy and Infectious Diseases. 2018; [cited 2021 May 27]. Available from: https://www.niaid.nih.gov/diseases-conditions/antiretroviral-drug-development.
Pandey A, Galvani AP. The global burden of HIV and prospects for control. Lancet HIV. 2019 Dec [cited 2021 May 27];6(12):e809–11. https://doi.org/10.1016/S2352-3018(19)30230-9. Available from: https://www.thelancet.com/journals/lanhiv/article/PIIS2352-3018(19)30230-9/abstract.
White B. Exciting progress in the development of an HIV vaccine. 2021 [cited 2021 Jul 5]. Available from: https://www.legacycommunityhealth.org/exciting-progress-in-the-development-of-an-hiv-vaccine/.
National Institute of Allergy and Infectious Diseases (NIH). HIV vaccine development. 2019 [cited 2021 May 27]. Available from: https://www.niaid.nih.gov/diseases-conditions/hiv-vaccine-development.
World Health Organization. Archived: WHO timeline - COVID-19. [cited 2021 May 26]. Available from: https://www.who.int/news/item/27-04-2020-who-timeline%2D%2D-covid-19
World Health Organization. Coronavirus disease (COVID-19) – World Health Organization. 2021 [cited 2021 May 26]. Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019.
The US Food and Drug Administration. COVID-19 vaccines. FDA; 2021 [cited 2021 May 26];. Available from: https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-covid-19/covid-19-vaccines.
Sallam M. COVID-19 vaccine hesitancy worldwide: a concise systematic review of vaccine acceptance rates. Vaccines (Basel). 2021 Feb 16 [cited 2021 Mar 24];9(2):160. https://doi.org/10.3390/vaccines9020160. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7920465/.
Desmon S. COVID-19 vaccine acceptance falling globally and in U.S. The Hub. 2021 [cited 2021 Jul 3]. Available from: https://hub.jhu.edu/2021/02/24/vaccine-acceptance-rate-falling-globally/
Lazarus JV, Ratzan SC, Palayew A, Gostin LO, Larson HJ, Rabin K, et al. A global survey of potential acceptance of a COVID-19 vaccine. Nat Med. 2021 [cited 2021 Jul 3];27:225–8. Available from: https://www.nature.com/articles/s41591-020-1124-9.
Solís Arce JS, Warren SS, Meriggi NF, et al. COVID-19 vaccine acceptance and hesitancy in low- and middle-income countries. Nat Med. 2021 [cited 2021 Mar 24];27:1385–94. https://doi.org/10.1038/s41591-021-01454-y.
Dzieciolowska S, Hamel D, Gadio S, Dionne M, Gagnon D, Robitaille L, et al. COVID-19 vaccine acceptance, hesitancy, and refusal among Canadian healthcare workers: A multicenter survey. Am J Infect Control. 2021 [cited 2021 Jul 3];. Available from: https://www.sciencedirect.com/science/article/pii/S0196655321002741.
Lee JT, Althomsons SP, Wu H, et al. Disparities in COVID-19 vaccination coverage among health care personnel working in long-term care facilities, by job category. National Healthcare Safety Network — United States, March 2021. MMWR Morb Mortal Wkly Rep. 2021 [cited 2021 Aug 1];70:1036–9. https://doi.org/10.15585/mmwr.mm7030a2.
Malik AA, McFadden SM, Elharake J, Omer SB. Determinants of COVID-19 vaccine acceptance in the US. EClinicalMedicine. 2020 Sep [cited 2021 Aug 1];26:100495. https://doi.org/10.1016/j.eclinm.2020.100495.
Centers for Disease Control and Prevention. COVID data tracker. Centers for Disease Control and Prevention. 2021; [cited 2021 Aug 1]. Available from: https://covid.cdc.gov/covid-data-tracker.
Ndugga N, Hill L, Artiga S, Parker N. Latest data on COVID-19 vaccinations by race/ethnicity. KFF. 2021; [cited 2021 Aug 2]. Available from: https://www.kff.org/coronavirus-covid-19/issue-brief/latest-data-on-covid-19-vaccinations-race-ethnicity/.
Our World in Data. Share of people who received at least one dose of COVID-19 vaccine. Our World in Data. 2021; [cited 2021 Aug 2]. Available from: https://ourworldindata.org/grapher/share-people-vaccinated-covid.
Marek E, Dergez T, Rebek-Nagy G, Szilard I, Kiss I, Ember I, et al. Effect of an educational intervention on Hungarian adolescents’ awareness, beliefs and attitudes on the prevention of cervical cancer. Vaccine. 2012 [cited 2021 Jun 30];30:6824–32. Available from: https://www.sciencedirect.com/science/article/pii/S0264410X12013229.
Gottvall M, Tydén T, Höglund AT, Larsson M. Knowledge of human papillomavirus among high school students can be increased by an educational intervention. Int J STD AIDS. 2010 [cited 2021 Jun 30];21:558–62.
Lai CY, Wu WW, Tsai SY, Cheng SF, Lin KC, Liang SY. The effectiveness of a Facebook-assisted teaching method on knowledge and attitudes about cervical cancer prevention and HPV vaccination intention among female adolescent students in Taiwan. Health Educ Behav. 2015 Jun [cited 2021 Jul 1];42(3):352-360. https://doi.org/10.1177/1090198114558591. Available from: https://doi.org/10.1177/1090198114558591
Brabin L, Stretch R, Roberts SA, Elton P, Baxter D, McCann R. Survey of girls' recall of a film providing information on human papillomavirus and cervical cancer 6 months after an offer of vaccination. Vaccine. 2010 [cited 2021 Jun 30];28(25):4210–4. https://doi.org/10.1016/j.vaccine.2010.03.077. Available from: https://www.sciencedirect.com/science/article/pii/S0264410X10004779.
Lloyd GP, Marlow LA, Waller J, Miles A, Wardle J. An experimental investigation of the emotional and motivational impact of HPV information in adolescents. J Adolesc Health. 2009 [cited 2021 Jun 30;45(5):532–4. https://doi.org/10.1016/j.jadohealth.2009.06.003. Available from: https://www.jahonline.org/article/S1054-139X(09)00236-5/abstract.
Cates JR, Crandell JL, Diehl SJ, Coyne-Beasley T. Immunization effects of a communication intervention to promote preteen HPV vaccination in primary care practices. Vaccine. 2018 [cited 2021 Jul 7];36(1):122–7. https://doi.org/10.1016/j.vaccine.2017.11.025. Available from: https://www.sciencedirect.com/science/article/pii/S0264410X17315864.
Kennedy A, Sapsis KF, Stokley S, Curtis CR, Gust D. Parental attitudes toward human papillomavirus vaccination: evaluation of an educational intervention, 2008. J Health Commun. 2011 [cited 2021 Jun 30];16(3):300–13. https://doi.org/10.1080/10810730.2010.532296.
Wetzel C, Tissot A, Kollar LM, Hillard PA, Stone R, Kahn JA. Development of an HPV educational protocol for adolescents. J Pediatr Adolesc Gynecol. 2007;20(5):281–7. https://doi.org/10.1016/j.jpag.2006.12.002.
Zimmerman RK, Moehling KK, Lin CJ, Zhang S, Raviotta JM, Reis EC, Humiston SG, Nowalk MP. Improving adolescent HPV vaccination in a randomized controlled cluster trial using the 4 Pillars™ practice Transformation Program. Vaccine. 2017 Jan 3 [cited 2021 Jul 7];35(1):109–17. https://doi.org/10.1016/j.vaccine.2016.11.018. Available from: https://www.sciencedirect.com/science/article/pii/S0264410X16310659.
Bonville CA, Domachowske JB, Suryadevara M. A quality improvement education initiative to increase adolescent human papillomavirus (HPV) vaccine completion rates. Hum Vaccin Immunother. 2019 [cited 2021 Jul 8];15(7-8):1570–6. https://doi.org/10.1080/21645515.2019.1627822.
Dempsey AF, Pyrznawoski J, Lockhart S, Barnard J, Campagna EJ, Garrett K, Fisher A, Dickinson LM, O'Leary ST. Effect of a health care professional communication training intervention on adolescent human papillomavirus vaccination: A Cluster Randomized Clinical Trial. JAMA Pediatr. 2018 [cited 2021 Jul 8];172(5):e180016. https://doi.org/10.1001/jamapediatrics.2018.0016.
Dixon BE, Zimet GD, Xiao S, Tu W, Lindsay B, Church A, Downs SM. An educational intervention to improve HPV vaccination: a cluster randomized trial. Pediatrics. 2019 Jan [cited 2021 Jul 8];143(1):e20181457. https://doi.org/10.1542/peds.2018-1457. Available from: https://pediatrics.aappublications.org/content/143/1/e20181457.
Liu C-R, Liang H, Zhang X, Pu C, Li Q, Li Q-L, et al. Effect of an educational intervention on HPV knowledge and attitudes towards HPV and its vaccines among junior middle school students in Chengdu, China. BMC Public Health. 2019 [cited 2021 Jul 8];19:488. Available from: https://doi.org/10.1186/s12889-019-6823-0.
Baxter CE, Barata PC. The paradox of HPV vaccines: how to reach sexually inexperienced women for protection against a sexually transmitted infection. Womens Health Issues. 2011 May-Jun [cited 2021 Jun 30];21(3):239–45. https://doi.org/10.1016/j.whi.2010.11.007. Available from: https://www.whijournal.com/article/S1049-3867(10)00156-8/abstract.
Bonafide KE, Vanable PA. Male human papillomavirus vaccine acceptance is enhanced by a brief intervention that emphasizes both male-specific vaccine benefits and altruistic motives. Sex Transm Dis. 2015 [cited 2021 Jul 1];42(2):76–80. https://doi.org/10.1097/OLQ.0000000000000226. Available from: https://journals.lww.com/stdjournal/Fulltext/2015/02000/Male_Human_Papillomavirus_Vaccine_Acceptance_Is.5.aspx.
DiClemente RJ, Crosby RA, Salazar LF, Nash R, Younge S. Is male intent to be vaccinated against HPV a function of the promotion message? Int J STD AIDS. 2011 [cited 2021 Jun 30];22(6):332–4. https://doi.org/10.1258/ijsa.2011.010429.
Gualano MR, Thomas R, Stillo M, Mussa MV, Quattrocolo F, Borraccino A, et al. What is the most useful tool in HPV vaccine promotion? Results from an experimental study. Hum Vaccines Immunother. 2019;15(7–8):1607–14.
Hayes KN, Pan I, Kunkel A, McGivney MS, Thorpe CT. Evaluation of targeted human papillomavirus vaccination education among undergraduate college students. J Am Coll Health. Taylor & Francis. 2019 [cited 2021 Jul 8];67(8):781–9. https://doi.org/10.1080/07448481.2018.1515742.
Reiter PL, Katz ML, Bauermeister JA, Shoben AB, Paskett ED, McRee A-L. Increasing human papillomavirus vaccination among young gay and bisexual men: a randomized pilot trial of the outsmart HPV intervention. LGBT Health. 2018 [cited 2021 Jul 8];5(5):325–9. https://doi.org/10.1089/lgbt.2018.0059.
Chan A, Brown B, Sepulveda E, Teran-Clayton L. Evaluation of fotonovela to increase human papillomavirus vaccine knowledge, attitudes, and intentions in a low-income Hispanic community. BMC Res Notes. 2015 [cited 2021 Jul 8];8:615–625. https://doi.org/10.1186/s13104-015-1609-7. Available from: https://www.ncbi.nlm.nih.gov/pubmed/26514184.
Keim-Malpass J, Mitchell EM, Camacho F. HPV vaccination series completion and co-vaccination: pairing vaccines may matter for adolescents. Vaccine. 2015 [cited 2021 Jul 8];33(43):5729–32. Available from: https://www.sciencedirect.com/science/article/pii/S0264410X15013535.
Dempsey AF, Maertens J, Sevick C, Jimenez-Zambrano A, Juarez-Colunga E. A randomized, controlled, pragmatic trial of an iPad-based, tailored messaging intervention to increase human papillomavirus vaccination among Latinos. Hum Vaccin Immunother. 2019 [cited 2021 Jul 8];15(7-8):1577–84. https://doi.org/10.1080/21645515.2018.1559685.
Roussos-Ross K, Foster L, Peterson HV, Decesare J. Do educational seminars for the human papillomavirus vaccine improve attitudes toward the value of vaccination? J Pediatr Adolesc Gynecol. 2017;30(4):456–9. https://doi.org/10.1016/j.jpag.2016.12.003. Available from: https://www.sciencedirect.com/science/article/pii/S1083318816302145.
Leader AE, Weiner JL, Kelly BJ, Hornik RC, Cappella JN. Effects of information framing on human papillomavirus vaccination. J Women’s (Larchmt). 2009 Feb [cited 2021 Jun 30];18(2):225–33. https://doi.org/10.1089/jwh.2007.0711.
Suarez Mora A, Madrigal JM, Jordan L, Patel A. Effectiveness of an educational intervention to increase human papillomavirus knowledge in high-risk minority women. J Low Genit Tract Dis 22(4):288-l. https://doi.org/10.1097/lgt.0000000000000386. [cited 2021 Jul 8]. Available from: https://journals.lww.com/jlgtd/Fulltext/2018/10000/Effectiveness_of_an_Educational_Intervention_to.5.aspx.
Chapman E, Venkat P, Ko E, Orezzoli JP, Carmen MD, EIO G. Use of multimedia as an educational tool to improve human papillomavirus vaccine acceptability—A pilot study. Gynecol Oncol. 2010 [cited 2021 Jun 30];118(2):103–7. https://doi.org/10.1016/j.ygyno.2010.04.010. Available from: https://www.gynecologiconcology-online.net/article/S0090-8258(10)00320-3/abstract.
Shah PD, Calo WA, Gilkey MB, Boynton MH, Dailey SA, Todd KG, et al. Questions and concerns about HPV vaccine: a communication experiment. Pediatrics. American Academy of Pediatrics. 2019 [cited 2021 Jul 7];143(2):e20181872. https://doi.org/10.1542/peds.2018-1872. Available from: https://pediatrics.aappublications.org/content/143/2/e20181872.
Baldwin AS, Denman DC, Sala M, Marks EG, Shay LA, Fuller S, et al. Translating self-persuasion into an adolescent HPV vaccine promotion intervention for parents attending safety-net clinics. Patient Educ Couns. 2017 [cited 2021 Jul 8];100(4):736–41. https://doi.org/10.1016/j.pec.2016.11.014. Available from: https://www.sciencedirect.com/science/article/pii/S0738399116305419.
Casillas A, Singhal R, Tsui J, Glenn BA, Bastani R, Mangione CM. The impact of social communication on perceived HPV vaccine effectiveness in a low-income, minority population. Ethn Dis. 2011;21(4):495–501. Available from https://pubmed.ncbi.nlm.nih.gov/22428357/.
Cox DS, Cox AD, Sturm LA, Zimet G. Behavioral interventions to increase HPV vaccination acceptability among mothers of young girls. Health Psychology. 2010 [cited 2021 Jun 30];29(1):29–39. https://doi.org/10.1037/a0016942. Available from: https://doi.apa.org/doiLanding?doi=10.1037%2Fa0016942.
Dempsey AF, Zimet GD, Davis RL, Koutsky L. Factors that are associated with parental acceptance of human papillomavirus vaccines: a randomized intervention study of written information about HPV. Pediatrics. 2006 [cited 2021 Jun 30];117(5):1486–93. https://doi.org/10.1542/peds.2005-1381.
Spleen AM, Kluhsman BC, Clark AD, Dignan MB, Lengerich EJ, ACTION Health Cancer Task Force. An increase in HPV-related knowledge and vaccination intent among parental and non-parental caregivers of adolescent girls, age 9-17 years, in Appalachian Pennsylvania. J Cancer Educ. 2012 [cited 2021 Jun 30];27(2):312-9. https://doi.org/10.1007/s13187-011-0294-z. Available from: https://doi.org/10.1007/s13187-011-0294-z.
Cipriano JJ, Scoloveno R, Kelly A. Increasing parental knowledge related to the human papillomavirus (HPV) vaccine. J Pediatr Health Care Off Publ Natl Assoc Pediatr Nurse Assoc Pract. 2018;32(1):29–35. https://doi.org/10.1016/j.pedhc.2017.06.006.
Rickert VI, Auslander BA, Cox DS, Rosenthal SL, Rupp RE, Zimet GD. School-based HPV immunization of young adolescents: Effects of two brief health interventions. Hum Vaccin Immunother,. Available from. 2015 [cited 2021 Jul 1];11(2):315–21. https://doi.org/10.1080/21645515.2014.1004022, https://doi.org/10.1080/21645515.2014.1004022.
Wegwarth O, Kurzenhäuser-Carstens S, Gigerenzer G. Overcoming the knowledge–behavior gap: the effect of evidence-based HPV vaccination leaflets on understanding, intention, and actual vaccination decision. Vaccine. 2014 [cited 2021 Jun 30];32(12):1388–93. https://doi.org/10.1016/j.vaccine.2013.12.038. Available from: https://www.sciencedirect.com/science/article/pii/S0264410X13017696.
Lee H, Kim M, Cooley ME, Kiang PN-C, Kim D, Tang S, et al. Using narrative intervention for HPV vaccine behavior change among Khmer mothers and daughters: a pilot RCT to examine feasibility, acceptability, and preliminary effectiveness. Appl Nurs Res. 2018;40:51–60. https://doi.org/10.1016/j.apnr.2017.12.008.
Obulaney PA, Gilliland I, Cassells H. Increasing cervical cancer and human papillomavirus prevention knowledge and HPV vaccine uptake through mother/daughter education. J Community Health Nurs. 2016 [cited 2021 Jul 1];33(1):54–66. quiz 66-7. https://doi.org/10.1080/07370016.2016.1120595.
Abuelo CE, Levinson KL, Salmeron J, Sologuren CV, Fernandez MJV, Belinson JL. The Peru Cervical Cancer Screening Study (PERCAPS): The design and implementation of a mother/daughter screen, treat, and vaccinate program in the Peruvian jungle. J Community Health. 2014 [cited 2021 Jun 30];39(3):409–15. https://doi.org/10.1007/s10900-013-9786-6.
Katz ML, Oldach BR, Goodwin J, Reiter PL, Ruffin MT, Paskett ED. Development and initial feedback about a human papillomavirus (HPV) vaccine comic book for adolescents. Journal of Cancer Education. 2014 Jun [cited 2021 Jun 30;29(2):318–24. https://doi.org/10.1007/s13187-013-0604-8.
Reiter PL, Stubbs B, Panozzo CA, Whitesell D, Brewer NT. HPV and HPV vaccine education intervention: effects on parents, healthcare staff, and school staff. Cancer Epidemiol Biomarkers Prev. 2011 [cited 2021 Jun 30;20(11):2354–61. https://doi.org/10.1158/1055-9965.EPI-11-0562.
Levinson KL, Abuelo C, Chyung E, Salmeron J, Belinson SE, Sologuren CV, et al. The Peru Cervical Cancer Prevention Study (PERCAPS): community-based participatory research in Manchay, Peru. Int J Gynecol Cancer. BMJ Specialist Journals. 2013 [cited 2021 Jun 30];23:141–7. Available from: https://ijgc.bmj.com/content/23/1/141.
Brewer NT, Hall ME, Malo TL, Gilkey MB, Quinn B, Lathren C. Announcements versus conversations to improve HPV vaccination coverage: a randomized trial. Pediatrics. 2017 [cited 2021 Jun 30];139(1):e20161764. https://doi.org/10.1542/peds.2016-1764.
Groom HC, Irving SA, Caldwell J, Larsen R, Beaudrault S, Luther LM, et al. Implementing a multipartner HPV vaccination assessment and feedback intervention in an integrated health system. J Public Health Manag Pract. 2017 [cited 2021 Jul 7];23(6):589–92. https://doi.org/10.1097/PHH.0000000000000562.
Kumar MM, Boies EG, Sawyer MH, Kennedy M, Williams C, Rhee KE. A brief provider training video improves comfort with recommending the human papillomavirus vaccine. Clin Pediatr (Phila). 2019[cited 2021 Jul 7];58(1):17–23. https://doi.org/10.1177/0009922818805217.
Choi N, Curtis CR, Loharikar A, Fricchione M, Jones E, Balzer E, et al. Successful use of interventions in combination to improve human papillomavirus vaccination coverage rates among adolescents—Chicago, 2013 to 2015. Acad Pediatr. 2018 [cited 2021 Jul 7];18(2S):S93–S100. https://doi.org/10.1016/j.acap.2017.09.016.
Chan SSC, Ng BHY, Lo WK, Cheung TH, Chung TKH. Adolescent girls’ attitudes on human papillomavirus vaccination. J Pediatr Adolesc Gynecol. 2009 [cited 2021 Jun 30];22(2):85–90. Available from: https://doi.org/10.1016/j.jpag.2007.12.007.
Steckelberg A, Albrecht M, Kezle A, Kasper J, Mühlhauser I. Impact of numerical information on risk knowledge regarding human papillomavirus (HPV) vaccination among schoolgirls: a randomised controlled trial. German Med Sci; 2013 [cited 2021 Jun 30];11:Doc15. https://doi.org/10.3205/000183.
Kwan TTC, Tam K, Lee PWH, Chan KKL, HYS N. The effect of school-based cervical cancer education on perceptions towards human papillomavirus vaccination among Hong Kong Chinese adolescent girls. Patient Educ Couns. 2011 [cited 2021 Jun 30];84(1):118–22. Available from: https://www.sciencedirect.com/science/article/pii/S0738399110003800.
DiClemente RJ, Murray CC, Graham T, Still J. Overcoming barriers to HPV vaccination: A randomized clinical trial of a culturally tailored, media intervention among African American girls. Hum Vaccin Immunother. 2015 [cited 2021 Jun 30];11(12):2883–94. https://doi.org/10.1080/21645515.2015.1070996.
Gerend MA, Barley J. Human papillomavirus vaccine acceptability among young adult men. Sexually Transmitted Diseases. 2009 [cited 2021 Jun 30];36(1):58–62. https://doi.org/10.1097/OLQ.0b013e31818606fc.
Hopfer S. Effects of a narrative HPV vaccination intervention aimed at reaching college women: a randomized controlled trial. Prev Sci. 2012 [cited 2021 Jun 30];13(2):173–82. https://doi.org/10.1007/s11121-011-0254-1.
Kim S, Hmielowski JD. The influence of self-efficacy in medical drama television programming on behaviors and emotions that promote cervical cancer prevention. Am J Health Behav. 2017;41(6):719–727. https://doi.org/10.5993/AJHB.41.6.6.
Krakow M, Yale RN, Perez Torres D, Christy K, Jensen JD. Death narratives and cervical cancer: impact of character death on narrative processing and HPV vaccination. Health Psychol. 2017;36(12):1173–80. https://doi.org/10.1037/hea0000498.
Le AT, Tran VT, Dinh HT-P, Dau CX, Pham BQ, Nguyen HT, et al. Effectiveness of community intervention program on knowledge and practice of HPV prevention among married females in Vietnam rural areas. Cancer Control. 2019[cited 2021 Jul 8]. 26(1):1073274819862792. https://doi.org/10.1177/1073274819862792.
Gerend MA, Shepherd MA, Lustria ML. Increasing human papillomavirus vaccine acceptability by tailoring messages to young adult women’s perceived barriers. Sex Transm Dis. 2013 May;40(5):401–5. https://doi.org/10.1097/OLQ.0b013e318283c8a8.
Chang IJ, Huang R, He W, Zhang S-K, Wang S-M, Zhao F-H, et al. Effect of an educational intervention on HPV knowledge and vaccine attitudes among urban employed women and female undergraduate students in China: a cross-sectional study. BMC Public Health. 2013 [cited 2021 Jun 30];13:916. Available from: https://doi.org/10.1186/1471-2458-13-916
Perez GK, Cruess DG, Strauss NM. A brief information-motivation-behavioral skills intervention to promote human papillomavirus vaccination among college-aged women. Psychol Res Behav Manag. 2016 [cited 2021 Jun 30];9:285–96. https://doi.org/10.2147/PRBM.S112504.
Bennett AT, Patel DA, Carlos RC, Zochowski MK, Pennewell SM, Chi AM, et al. Human papillomavirus vaccine uptake after a tailored, online educational intervention for female university students: a randomized controlled trial. J Women’s Health. 2015 [cited 2021 Jul 1];24(11):950–7. https://doi.org/10.1089/jwh.2015.5251.
McRee A-L, Shoben A, Bauermeister JA, Katz ML, Paskett ED, Reiter PL. Outsmart HPV: acceptability and short-term effects of a web-based HPV vaccination intervention for young adult gay and bisexual men. Vaccine. 2018 [cited 2021 Jul 7];36(52):8158–64. https://doi.org/10.1016/j.vaccine.2018.01.009.
McGrath L, Fairley CK, Cleere EF, Bradshaw CS, Chen MY, Chow EPF. Human papillomavirus vaccine uptake among young gay and bisexual men who have sex with men with a time-limited targeted vaccination programme through sexual health clinics in Melbourne in 2017. Sex Transm Infect. 2019 [cited 2021 Jul 7];95(3):181–6. https://doi.org/10.1136/sextrans-2018-053619.
Wadhera P, Evans JL, Stein E, Gandhi M, Couture M-C, Sansothy N, et al. Human papillomavirus knowledge, vaccine acceptance, and vaccine series completion among female entertainment and sex workers in Phnom Penh, Cambodia: the young women’s health study. Int J STD AIDS. 2015 [cited 2021 Jul 1];26(12):893–902. https://doi.org/10.1177/0956462414563626.
Juraskova I, Bari RA, O’Brien MT, McCaffery KJ. HPV vaccine promotion: does referring to both cervical cancer and genital warts affect intended and actual vaccination behavior? Womens Health Issues. 2011 [cited 2021 Jun 30];21(1):71–9. https://doi.org/10.1016/j.whi.2010.08.004.
Gainforth HL, Latimer AE. Risky business: Risk information and the moderating effect of message frame and past behaviour on women’s perceptions of the Human Papillomavirus vaccine - Journal of Health Psychology. 2012 Sep [cited 2021 Jun 30] 17(6):896-905; quiz 905-6. https://doi.org/10.1177/1359105311431173.
Gainforth HL, Cao W, Latimer-Cheung AE. Message framing and parents’ intentions to have their children vaccinated against hpv. Public Health Nursing. 2012 [cited 2021 Jun 30];29(6):542–52. https://doi.org/10.1111/j.1525-1446.2012.01038.x.
Gesser-Edelsburg A, Walter N, Shir-Raz Y, Green MS. Voluntary or mandatory? The valence framing effect of attitudes regarding HPV vaccination. J Health Commun. 2015 [cited 2021 Jul 1];20(11):1287–93. https://doi.org/10.1080/10810730.2015.1018642.
Nan X, Madden K, Richards A, Holt C, Wang MQ, Tracy K. Message framing, perceived susceptibility, and intentions to vaccinate children against HPV among African American parents. Health Communication. 2016[cited 2021 Jul 1];31(7):798–805. https://doi.org/10.1080/10410236.2015.1005280.
Bell RA, McGlone MS, Dragojevic M. Vicious viruses and vigilant vaccines: effects of linguistic agency assignment in health policy advocacy. J Health Commun. 2014 [cited 2021 Jun 30];19(10):1178–1195. Available from: https://doi.org/10.1080/10810730.2013.811330.
Bigman CA, Cappella JN, Hornik RC. Effective or ineffective: attribute framing and the human papillomavirus (HPV) vaccine. Patient Educ Couns. 2010 [cited 2021 Jun 30;81(Suppl):S70–6. https://doi.org/10.1016/j.pec.2010.08.014.
Donahue K, Hendrix K, Sturm L, Zimet G. Provider communication and mothers’ willingness to vaccinate against human papillomavirus and influenza: a randomized health messaging trial. Acad Pediatr. 2018 [cited 2021 Jul 7];18(2):145–53. https://doi.org/10.1016/j.acap.2017.07.007.
Gerend MA, Shepherd JE. Using message framing to promote acceptance of the human papillomavirus vaccine. Health Psychol. 2007 Nov;26(6):745–52. https://doi.org/10.1037/0278-6133.26.6.745.
Gerend MA, Shepherd JE. Predicting human papillomavirus vaccine uptake in young adult women: comparing the health belief model and theory of planned behavior. Ann Behav Med. 2012 [cited 2021 Jun 30];44(2):171–80. https://doi.org/10.1007/s12160-012-9366-5 Erratum in: Ann Behav Med. 2012 Oct;44(2):293.
Gerend MA, Shepherd JE, Monday KA. Behavioral frequency moderates the effects of message framing on HPV vaccine acceptability. Ann Behav Med. 2008 Apr;35(2):221–9. https://doi.org/10.1007/s12160-008-9024-0.
Juraskova I, O’Brien M, Mullan B, Bari R, Laidsaar-Powell R, McCaffery K. HPV vaccination and the effect of information framing on intentions and behaviour: an application of the theory of planned behaviour and moral norm. IntJ Behav Med. 2012 [cited 2021 Jun 30];19(4):518–25. Available from: https://doi.org/10.1007/s12529-011-9182-5
Krieger JL, Sarge MA. A serial mediation model of message framing on intentions to receive the human papillomavirus (HPV) vaccine: Revisiting the role of threat and efficacy perceptions. Health Communication. Routledge. 2013 [cited 2021 Jun 30];28(1):5–19. Available from. https://doi.org/10.1080/10410236.2012.734914.
Lechuga J, Swain GR, Weinhardt LS. Impact of framing on intentions to vaccinate daughters against HPV: a cross-cultural perspective. Annals of Behavioral Medicine. 2011 [cited 2021 Jun 30];42(2):221–6. Available from: https://doi.org/10.1007/s12160-011-9273-1
McRee AL, Reiter PL, Chantala K, Brewer NT. Does framing human papillomavirus vaccine as preventing cancer in men increase vaccine acceptability? Cancer Epidemiol Biomarkers Prev. 2010 Aug;19(8):1937–44. https://doi.org/10.1158/1055-9965.EPI-09-1287.
Park S-Y. The effects of message framing and risk perceptions for HPV vaccine campaigns: focus on the role of regulatory fit. Health Marketing Quarterly. Routledge; 2012 [cited 2021 Jun 30];29:283–302. Available from: https://doi.org/10.1080/07359683.2012.732847
Liu S, Yang JZ, Chu H. Now or future? Analyzing the effects of message frame and format in motivating Chinese females to get HPV vaccines for their children. Patient Educ Couns. 2019 Jan [cited 2021 Jul 8];102(1):61–67. Available from: https://doi.org/10.1016/j.pec.2018.09.005.
Lee MJ, Cho J. Promoting HPV vaccination online: message design and media choice. Health Promot Pract. 2017 [cited 2021 Jul 8];18(5):645–53. https://doi.org/10.1177/1524839916688229.
Mantzari E, Vogt F, Marteau TM. Financial incentives for increasing uptake of HPV vaccinations: a randomized controlled trial. Health Psychol. 2015[cited 2021 Jul 8];34(2):160–71. 2015;34:160–71. https://doi.org/10.1037/hea0000088.
Caskey R, Sherman EG, Beskin K, Rapport R, Xia Y, Schwartz A. A behavioral economic approach to improving human papillomavirus vaccination. J Adolesc Health. 2017 [cited 2021 Jul 8]; 61(6):755–760. Available from: https://doi.org/10.1016/j.jadohealth.2017.07.020.
Widgren K, Simonsen J, Valentiner-Branth P, Mølbak K. Uptake of the human papillomavirus-vaccination within the free-of-charge childhood vaccination programme in Denmark. Vaccine. 2011 [cited 2021 Jun 30];29(52):9663–9667. Available from: https://doi.org/10.1016/j.vaccine.2011.10.021.
Piedimonte S, Leung A, Zakhari A, Giordano C, Tellier P-P, Lau S. Impact of an HPV education and vaccination campaign among Canadian university students. J Obstet Gynaecol Can. 2018[cited 2021 Jul 7];40(4):440–6. Available from https://doi.org/10.1016/j.jogc.2017.07.028.
Botha MH, van der Merwe FH, Snyman LC, Dreyer G. The vaccine and cervical cancer screen (VACCS) project: acceptance of human papillomavirus vaccination in a school-based programme in two provinces of South Africa. South Afr Med J. 2015 [cited 2021 Jun 30];105(1):40–3. https://doi.org/10.7196/samj.8419.
Fregnani JHTG, Carvalho AL, Eluf-Neto J, de CB RK, de Kuil LM, da Silva TA, et al. A school-based human papillomavirus vaccination program in Barreto, Brazil: final results of a demonstrative study. PloS One. 2013 [cited 2021 Jun 30]. 8(4):e62647. https://doi.org/10.1371/journal.pone.0062647.
Gilkey MB, Calo WA, Moss JL, Shah PD, Marciniak MW, Brewer NT. Provider communication and HPV vaccination: the impact of recommendation quality. Vaccine. 2016 Feb 24 [cited 2021 Jul 7];34(9):1187–1192. Available from: https://doi.org/10.1016/j.vaccine.2016.01.023.
LaMontagne DS, Barge S, Le NT, Mugisha E, Penny ME, Gandhi S, et al. Human papillomavirus vaccine delivery strategies that achieved high coverage in low- and middle-income countries. Bull World Health Organ. 2011 Nov 1 [cited 2021 Jul 7]. 89(11):821–830B. https://doi.org/10.2471/BLT.11.089862.
Poscia A, Pastorino R, Boccia S, Ricciardi W, Spadea A. The impact of a school-based multicomponent intervention for promoting vaccine uptake in Italian adolescents: a retrospective cohort study. Ann Ist Super Sanita. 2019 Apr-Jun [cited 2021 Jul 7];55(2):124–30. https://doi.org/10.4415/ANN_19_02_04.
Meyer AF, Borkovskiy NL, Brickley JL, Chaudhry R, Franqueira A, Furst JW, et al. Impact of electronic point-of-care prompts on human papillomavirus vaccine uptake in retail clinics. Am J Prev Med. 2018[cited 2021 Jul 7];55(6):822–9. https://doi.org/10.1016/j.amepre.2018.06.027.
Reno JE, Thomas J, Pyrzanowski J, Lockhart S, O’Leary ST, Campagna EJ, et al. Examining strategies for improving healthcare providers’ communication about adolescent HPV vaccination: evaluation of secondary outcomes in a randomized controlled trial. Hum Vaccines Immunother. 2019 [cited 2021 Jul 8];15(7–8):1592–1598. Available from: https://doi.org/10.1080/21645515.2018.1547607
Lee HY, Koopmeiners JS, McHugh J, Raveis VH, Ahluwalia JS. mHealth pilot study: text messaging intervention to promote HPV vaccination. Am J Health Behav. 2016;40(1):67–76.
Berenson AB, Rupp R, Dinehart EE, Cofie LE, Kuo Y-F, Hirth JM. Achieving high HPV vaccine completion rates in a pediatric clinic population. Hum Vaccin Immunother. 2019 Aug 3 [cited 2021 Jul 8];15(7–8):1562–1569. Available from: https://doi.org/10.1080/21645515.2018.1533778
Whelan NW, Steenbeek A, Martin-Misener R, Scott J, Smith B, AD’Angelo-Scott H. Engaging parents and schools improves uptake of the human papillomavirus (HPV) vaccine: examining the role of the public health nurse. Vaccine. 2014 [cited 2021 Jun 30];32(36):4665–71. Available from: https://doi.org/10.1016/j.vaccine.2014.06.026.
Rand CM, Schaffer SJ, Dhepyasuwan N, Blumkin A, Albertin C, Serwint JR, et al. Provider communication, prompts, and feedback to improve HPV vaccination rates in resident clinics. Pediatrics. 2018 [cited 2021 Jul 8];141(4): e20170498. Available from: https://doi.org/10.1542/peds.2017-0498.
Lefevere E, Hens N, De Smet F, Beutels P. The impact of non-financial and financial encouragements on participation in non school-based human papillomavirus vaccination: a retrospective cohort study. Eur J Health Econ. 2016 [cited 2021 Jul 7];17(3):305–15. Available from: https://doi.org/10.1007/s10198-015-0680-2
Perkins RB, Zisblatt L, Legler A, Trucks E, Hanchate A, Gorin SS. Effectiveness of a provider-focused intervention to improve HPV vaccination rates in boys and girls. Vaccine. 2015;33(9):1223–9.
Malo TL, Hall ME, Brewer NT, Lathren CR, Gilkey MB. Why is announcement training more effective than conversation training for introducing HPV vaccination? A theory-based investigation. Implement Sci. 2018 [cited 2021 Jul 7];13(1):57. Available from: https://doi.org/10.1186/s13012-018-0743-8
Sanderson M, Canedo JR, Khabele D, Fadden MK, Harris C, Beard K, et al. Pragmatic trial of an intervention to increase human papillomavirus vaccination in safety-net clinics. BMC Public Health. 2017 [cited 2021 Jul 8];17(1):158. Available from: https://doi.org/10.1186/s12889-017-4094-1.
Tu Y-C, Lin Y-J, Fan L-W, Tsai T-I, Wang H-H. Effects of multimedia framed messages on human papillomavirus prevention among adolescents. West J Nurs Res. 2019 [cited 2021 Jul 8];41(1):58–77. Available from: https://doi.org/10.1177/0193945918763873
McGlone MS, Stephens KK, Rodriguez SA, Fernandez ME. Persuasive texts for prompting action: agency assignment in HPV vaccination reminders. Vaccine. 2017 [cited 2021 Jul 8];35(34):4295–7. Available from: https://www.sciencedirect.com/science/article/pii/S0264410X17308770.
Finney Rutten LJ, St. Sauver JL, Beebe TJ, Wilson PM, Jacobson DJ, Fan C, et al. Association of both consistency and strength of self-reported clinician recommendation for HPV vaccination and HPV vaccine uptake among 11- to 12-year-old children. Vaccine. 2017 [cited 2021 Jul 8];35(45):6122–8. Available from: https://www.sciencedirect.com/science/article/pii/S0264410X1731304X.
Wermers R, Ostroski T, Hagler D. Health care provider use of motivational interviewing to address vaccine hesitancy in college students. J Am Assoc Nurse Pract. 2021 [cited 2021 Jul 8];33(1):86–93. Available from: https://journals.lww.com/jaanp/Fulltext/2021/01000/Health_care_provider_use_of_motivational.13.aspx.
Joseph NP, Bernstein J, Pelton S, Belizaire M, Goff G, Horanieh N, et al. Brief client-centered motivational and behavioral intervention to promote HPV vaccination in a hard-to-reach population: a pilot randomized controlled trial. Clin Pediatr. 2016 [cited 2021 Jul 1];55(9):851–9. Available from: https://doi.org/10.1177/0009922815616244
Moss JL, Reiter PL, Rimer BK, Brewer NT. Collaborative patient-provider communication and uptake of adolescent vaccines. Soc Sci Med. 2016 [cited 2021 Jul 1];159:100–107. Available from: https://doi.org/10.1016/j.socscimed.2016.04.030.
Zimet G, Dixon BE, Xiao S, Tu W, Kulkarni A, Dugan T, et al. Simple and elaborated clinician reminder prompts for human papillomavirus vaccination: a randomized clinical trial. Acad Pediatr. 2018 [cited 2021 Jul 8];18(2, Supplement):S66–71. Available from: https://www.sciencedirect.com/science/article/pii/S1876285917305636.
Goleman MJ, Dolce M, Morack J. Quality improvement initiative to improve human papillomavirus vaccine initiation at 9 years of Age. Acad Pediatr. 2018 [cited 2021 Jul 8];18(7):769–75. Available from: https://www.sciencedirect.com/science/article/pii/S1876285918303097
Deshmukh U, Oliveira CR, Griggs S, Coleman E, Avni-Singer L, Pathy S, et al. Impact of a clinical interventions bundle on uptake of HPV vaccine at an OB/GYN clinic. Vaccine. 2018 [cited 2021 Jul 8];36(25):3599–605. Available from: https://doi.org/10.1016/j.vaccine.2018.05.039.
Grandahl M, Rosenblad A, Stenhammar C, Tydén T, Westerling R, Larsson M, et al. School-based intervention for the prevention of HPV among adolescents: a cluster randomised controlled study. BMJ Open. 2016 [cited 2021 Jul 7];6(1):e009875. Available from: https://bmjopen.bmj.com/content/6/1/e009875.
Tan LJ, VanOss R, Ofstead CL, Wetzler HP. Maximizing the impact of, and sustaining standing orders protocols for adult immunization in outpatient clinics. Am J Infect Control. 2020 1 [cited 2021 Jul 7];48(3):290–6. Available from: https://www.ajicjournal.org/article/S0196-6553(19)30740-0/abstract
Fenton AT, Eun TJ, Clark JA, Perkins RB. Indicated or elective? The association of providers’ words with HPV vaccine receipt. Hum Vaccines Immunother. 2018 Oct 3 [cited 2021 Jul 7];14(10):2503–9. Available from: https://doi.org/10.1080/21645515.2018.1480237
Tull F, Borg K, Knott C, Beasley M, Halliday J, Faulkner N, et al. Short message service reminders to parents for increasing adolescent human papillomavirus vaccination rates in a secondary school vaccine program: a randomized control trial. J Adolesc Health. 2019 [cited 2021 Jul 8];65(1):116–23. Available from: https://www.sciencedirect.com/science/article/pii/S1054139X19300199
Berenson AB, Rahman M, Hirth JM, Rupp RE, Sarpong KO. A human papillomavirus vaccination program for low-income postpartum women. Am J Obstet Gynecol. 2016 Sep;215(3):318.e1–9. https://doi.org/10.1016/j.ajog.2016.02.032.
Henrikson NB, Zhu W, Baba L, Nguyen M, Berthoud H, Gundersen G, et al. Outreach and reminders to improve human papillomavirus vaccination in an integrated primary care system. Clin Pediatr (Phila). 2018 Nov;57(13):1523–31. https://doi.org/10.1177/0009922818787868.
Mayne S, Karavite D, Grundmeier RW, Localio R, Feemster K, DeBartolo E, et al. The implementation and acceptability of an HPV vaccination decision support system directed at both clinicians and families. AMIA Annu Symp Proc AMIA Symp. 2012;2012:616–24.
Suh C, Saville A, Daley MF, Glazner JE, Barrow J, Stokley S. et al. Effectiveness and net cost of reminder/recall for adolescent immunizations | American Academy of Pediatrics. Pediatrics. 2012 [cited 2021 Jun 30];129(6):e1437–45. Available from: https://pediatrics.aappublications.org/content/129/6/e1437.
Chao C, Preciado M, Slezak J, Xu L. A randomized intervention of reminder letter for human papillomavirus vaccine series completion. J Adolesc Health. 2015 Jan 1 [cited 2021 Jun 30];56(1):85–90. Available from: https://www.jahonline.org/article/S1054-139X(14)00346-2/abstract.
Szilagyi PG, Serwint JR, Humiston SG, Rand CM, Schaffer S, Vincelli P, et al. Effect of provider prompts on adolescent immunization rates: a randomized trial. Acad Pediatr. 2015 [cited 2021 Jun 30];15(2):149–57. https://doi.org/10.1016/j.acap.2014.10.006.
Kempe A, O’Leary ST, Shoup JA, Stokley S, Lockhart S, Furniss A, et al. Parental choice of recall method for hpv vaccination: a pragmatic trial. Pediatrics. 2016 [cited 2021 Jul 8];137(3). Available from: https://pediatrics.aappublications.org/content/137/3/e20152857.
Kempe A, Barrow J, Stokley S, Saville A, Glazner JE, Suh C, et al. Effectiveness and cost of immunization recall at school-based health centers. Pediatrics. 2012 [cited 2021 Jun 30];129(6):e1446–52. Available from: https://pediatrics.aappublications.org/content/129/6/e1446.
Patel A, Stern L, Unger Z, Debevec E, Roston A, Hanover R, et al. Staying on track: A cluster randomized controlled trial of automated reminders aimed at increasing human papillomavirus vaccine completion. Vaccine. 2014 [cited 2021 Jun 30;32(21):2428–33. Available from: https://www.sciencedirect.com/science/article/pii/S0264410X14003351.
Richman AR, Maddy L, Torres E, Goldberg EJ. A randomized intervention study to evaluate whether electronic messaging can increase human papillomavirus vaccine completion and knowledge among college students. J Am Coll Health. 2016 [cited 2021 Jul 1];64(4):269–78. Available from: https://doi.org/10.1080/07448481.2015.1117466
Yee LM, Martinez NG, Nguyen AT, Hajjar N, Chen MJ, Simon MA. Using a patient navigator to improve postpartum care in an urban women’s health clinic. Obstet Gynecol. 2017 [cited 2021 Jun 30];129(5):925–33. https://doi.org/10.1097/AOG.0000000000001977.
Kreuter MW, McQueen A, Boyum S, Fu Q. Unmet basic needs and health intervention effectiveness in low-income populations. Prev Med. 2016 [cited 2021 Jul 8];91:70–75. Available from: https://doi.org/10.1016/j.ypmed.2016.08.006.
Lee C-C, Chen T-S, Wu T-Z, Huang L-M. A human papillomavirus public vaccination program in Taiwan: The Kinmen County experience. J Formos Med Assoc. 2012 [cited 2021 Jun 30];111(12):682–5. Available from: https://www.sciencedirect.com/science/article/pii/S0929664612005013.
Binagwaho A, Wagner CM, Gatera M, Karema C, Nutt CT, Ngabo F. Achieving high coverage in Rwanda’s national human papillomavirus vaccination programme. Bull World Health Organ. 2012;90(8):623–628. https://doi.org/10.2471/BLT.11.097253.
Hayashi Y, Shimizu Y, Netsu S, Hanley S, Konno R. High HPV vaccination uptake rates for adolescent girls after regional governmental funding in Shiki City, Japan. Vaccine. 2012 [cited 2021 Jun 30];30(37):5547–50. Available from: https://www.sciencedirect.com/science/article/pii/S0264410X12008936.
Shourie S, Jackson C, Cheater F, Bekker H, Edlin R, Tubeuf S, et al. A cluster randomised controlled trial of a web based decision aid to support parents’ decisions about their child’s Measles Mumps and Rubella (MMR) vaccination. Vaccine. 2013;31(50):6003–10. https://doi.org/10.1016/j.vaccine.2013.10.025.
Rahman M, Elam LB, Balat MI, Berenson AB. Well-woman visit of mothers and human papillomavirus vaccine intent and uptake among their 9-17 year old children. Vaccine. 2013;31(47):5544–8. https://doi.org/10.1016/j.vaccine.2013.09.017.
Uddin MJ, Adhikary G, Ali MW, Ahmed S, Shamsuzzaman M, Odell C, et al. Evaluation of impact of measles rubella campaign on vaccination coverage and routine immunization services in Bangladesh. BMC Infect Dis. 2016 [cited 2021 Jul 10];16(1):411. Available from: https://doi.org/10.1186/s12879-016-1758-x
Abhyankar P, O’Connor DB, Lawton R. The role of message framing in promoting MMR vaccination: evidence of a loss-frame advantage. Psychol Health Med. 2008;13(1):1–16. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18066916.
Hendrix KS, Finnell SME, Zimet GD, Sturm LA, Lane KA, Downs SM. Vaccine message framing and parents’ intent to immunize their infants for MMR. Pediatrics. 2014 [cited 2021 Jul 10];134(3):e675–83. Available from: https://pediatrics.aappublications.org/content/134/3/e675.
Horne Z, Powell D, Hummel JE, Holyoak KJ. Countering antivaccination attitudes. Proc Natl Acad Sci. 2015 [cited 2021 Jul 10];112(33):10321–4. https://doi.org/10.1073/pnas.1504019112.
Powell-Jackson T, Fabbri C, Dutt V, Tougher S, Singh K. Effect and cost-effectiveness of educating mothers about childhood DPT vaccination on immunisation uptake, knowledge, and perceptions in Uttar Pradesh, India: A randomised controlled trial. PLOS Med. 2018 [cited 2021 Jul 10];15(3):e1002519. Available from: https://journals.plos.org/plosmedicine/article?id=10.1371/journal.pmed.1002519.
Barham T, Maluccio JA. Eradicating diseases: the effect of conditional cash transfers on vaccination coverage in rural Nicaragua. J Health Econ. 2009 [cited 2021 Jul 10];28(3):611–21. Available from: https://www.sciencedirect.com/science/article/pii/S0167629609000022.
Cockman P, Dawson L, Mathur R, Hull S. Improving MMR vaccination rates: herd immunity is a realistic goal. BMJ. 2011 [cited 2021 Jul 10];343:d5703. Available from: https://doi.org/10.1136/bmj.d5703.
Merilind E, Salupere R, Västra K, Kalda R. The influence of performance-based payment on childhood immunisation coverage. Health Policy. 2015 Jun 1 [cited 2021 Jul 10];119(6):770–7. Available from: https://www.sciencedirect.com/science/article/pii/S0168851015000317.
Gibson DG, Ochieng B, Kagucia EW, Were J, Hayford K, Moulton LH, et al. Mobile phone-delivered reminders and incentives to improve childhood immunisation coverage and timeliness in Kenya (M-SIMU): a cluster randomised controlled trial. Lancet Glob Health. 2017 [cited 2021 Jul 10];5(4):e428–38. Available from: https://www.thelancet.com/journals/langlo/article/PIIS2214-109X(17)30072-4/abstract.
Hofstetter AM, DuRivage N, Vargas CY, Camargo S, Vawdrey D, Fisher A, et al. Text message reminders for timely routine MMR vaccination: a randomized controlled trial. Vaccine. 2015;33(43):5741–6. https://doi.org/10.1016/j.vaccine.2015.09.042.
Fu LY, Weissman M, McLaren R, Thomas C, Campbell J, Mbafor J, Doshi U, Cora-Bramble D. Improving the quality of immunization delivery to an at-risk population: a comprehensive approach. Pediatrics. 2012 [cited 2021 Jul 10];129(2):e496–e503. Available from: https://doi.org/10.1542/peds.2010-3610.
Goel S, Dogra V, Gupta SK, Lakshmi PVM, Varkey S, Pradhan N, et al. Effectiveness of Muskaan Ek Abhiyan (The Smile Campaign) for strengthening routine immunization in Bihar, India. Indian Pediatr. 2012 [cited 2021 Jul 10];49(2):103–8. Available from: https://doi.org/10.1007/s13312-012-0023-7
Sasaki S, Igarashi K, Fujino Y, Comber AJ, Brunsdon C, Muleya CM, et al. The impact of community-based outreach immunisation services on immunisation coverage with GIS network accessibility analysis in peri-urban areas, Zambia. J Epidemiol Community Health. 2011 [cited 2021 Jul 10];65(12):1171–8. Available from: https://jech.bmj.com/content/65/12/1171.
Acknowledgements
We would like to acknowledge Erin James and Kate Nyhan from Yale University for their support with the search and review of the literature.
Funding
This review was supported by a grant from Bill and Melinda Gates Foundation (GRANT AGREEMENT INVESTMENT ID: INV-001288) and Yale Institute for Global Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Global Health
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aguolu, O.G., Malik, A.A., Ahmed, N. et al. Overcoming Vaccine Hesitancy for Future COVID-19 and HIV Vaccines: Lessons from Measles and HPV Vaccines. Curr HIV/AIDS Rep 19, 328–343 (2022). https://doi.org/10.1007/s11904-022-00622-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11904-022-00622-0