Abstract
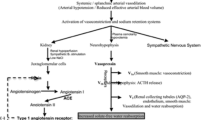
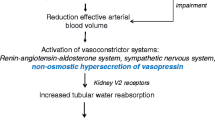
Cirrhosis is associated with high morbidity rates due to complications of portal hypertension. Ascites is the most frequent complication in patients with cirrhosis, and refractory ascites will develop in approximately 10 % of patients during follow-up. Currently, the first-line treatment for patients with refractory ascites is large-volume paracentesis with albumin supplementation. In more advanced stages of the disease, dilutional hyponatremia may develop as the result of nonosmotic hypersecretion of vasopressin. Although vaptans have shown promising results as a potential pharmacologic approach to treating hypervolemic hyponatremia, their results on long-term efficacy and safety in patients with cirrhosis are not conclusive. Moreover, because of concerns regarding side effects, the US Food and Drug Administration recently removed the indication of tolvaptan for use in patients with cirrhosis. Finally, in late stages of the disease, intense renal vasoconstriction occurs and leads to the development of hepatorenal syndrome. The treatment of choice for patients with hepatorenal syndrome is vasoconstrictor drugs followed by liver transplantation.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Ginès P, Cárdenas A, Solà E, Schrier RW. Liver disease and the kidney. In: Coffman TM, Falk RJ, Molitoris BA, Neilson EG, Schrier RW, editors. Schrier’s diseases of the kidney. 9th ed. Philadelphia: Lippincott Williams & Wilkins; 2012. This is an updated and extensive chapter on the pathophysiology and treatment of functional kidney abnormalities and complications of cirrhosis.
Arroyo V, Ginès P, Gerbes A, et al. Definition and diagnostic criteria of refractory ascites and hepatorenal syndrome in cirrhosis. International Ascites Club. Hepatology. 1996;23:164–76.
Arroyo V, Fernandez-Esparrach G, Gines P. Diagnostic approach to the cirrhotic patient with ascites. J Hepatol. 1996;25 Suppl 1:35–40.
EASL clinical practice guidelines on the management of ascites, spontaneous bacterial peritonitis, and hepatorenal syndrome in cirrhosis. J Hepatol. 2010;53(3):397–417.
Gines P et al. Management of cirrhosis and ascites. N Engl J Med. 2004;350(16):1646–54.
Salerno F et al. Survival and prognostic factors of cirrhotic patients with ascites: a study of 134 outpatients. Am J Gastroenterol. 1993;88(4):514–9.
Runyon BA. Management of adult patients with ascites due to cirrhosis. Hepatology. 2004;39(3):841–56.
Moore KP, Aithal GP. Guidelines on the management of ascites in cirrhosis. Gut. 2006;55 Suppl 6:vi1–vi12.
Boyer TD, Haskal ZJ. The role of transjugular intrahepatic portosystemic shunt in the management of portal hypertension. Hepatology. 2005;41(2):386–400.
Ochs A et al. The transjugular intrahepatic portosystemic stent-shunt procedure for refractory ascites. N Engl J Med. 1995;332(18):1192–7.
Wong F et al. Transjugular intrahepatic portosystemic stent shunt: effects on hemodynamics and sodium homeostasis in cirrhosis and refractory ascites. Ann Intern Med. 1995;122(11):816–22.
Huonker M et al. Cardiac function and haemodynamics in alcoholic cirrhosis and effects of the transjugular intrahepatic portosystemic stent shunt. Gut. 1999;44(5):743–8.
Guevara M et al. Transjugular intrahepatic portosystemic shunt in hepatorenal syndrome: effects on renal function and vasoactive systems. Hepatology. 1998;28(2):416–22.
Lebrec D et al. Transjugular intrahepatic portosystemic shunts: comparison with paracentesis in patients with cirrhosis and refractory ascites: a randomized trial. French Group of Clinicians and a Group of Biologists. J Hepatol. 1996;25(2):135–44.
Rossle M et al. A comparison of paracentesis and transjugular intrahepatic portosystemic shunting in patients with ascites. N Engl J Med. 2000;342(23):1701–7.
Gines P et al. Transjugular intrahepatic portosystemic shunting versus paracentesis plus albumin for refractory ascites in cirrhosis. Gastroenterology. 2002;123(6):1839–47.
Sanyal AJ et al. The North American Study for the Treatment of Refractory Ascites. Gastroenterology. 2003;124(3):634–41.
Salerno F et al. Randomized controlled study of TIPS versus paracentesis plus albumin in cirrhosis with severe ascites. Hepatology. 2004;40(3):629–35.
Albillos A et al. A meta-analysis of transjugular intrahepatic portosystemic shunt versus paracentesis for refractory ascites. J Hepatol. 2005;43(6):990–6.
Deltenre P et al. Transjugular intrahepatic portosystemic shunt in refractory ascites: a meta-analysis. Liver Int. 2005;25(2):349–56.
D'Amico G et al. Uncovered transjugular intrahepatic portosystemic shunt for refractory ascites: a meta-analysis. Gastroenterology. 2005;129(4):1282–93.
Saab S et al. TIPS versus paracentesis for cirrhotic patients with refractory ascites. Cochrane Database Syst Rev. 2006;4, CD004889.
Salerno F et al. Transjugular intrahepatic portosystemic shunt for refractory ascites: a meta-analysis of individual patient data. Gastroenterology. 2007;133(3):825–34.
Bellot P et al. Automated low flow pump system for the treatment of refractory ascites: a multi-center safety and efficacy study. J Hepatol. 2013;58(5):922–7. This was the first study to assess the efficacy and safety of a new device for the management of refractory ascites. The results show that the system is efficacious in treating ascites, although the adverse event rate is relatively high.
Decaux G, Soupart A, Vassart G. Non-peptide arginine-vasopressin antagonists: the vaptans. Lancet. 2008;371(9624):1624–32.
Gines P et al. Effects of satavaptan, a selective vasopressin V(2) receptor antagonist, on ascites and serum sodium in cirrhosis with hyponatremia: a randomized trial. Hepatology. 2008;48(1):204–13.
Wong F, Ginès P, Watson H, et al. Effects of a selective vasopressin V2 receptor antagonist, satavaptan, on ascites recurrence after paracentesis in patients with cirrhosis. J Hepatol. 2010;53:283–90.
Wong F et al. Satavaptan for the management of ascites in cirrhosis: efficacy and safety across the spectrum of ascites severity. Gut. 2012;61(1):108–16. This analysis evaluated the efficacy and safety of satavaptan in three randomized double-blind studies comparing satavaptan with placebo in three different populations of patients with cirrhosis and ascites. Satavaptan, alone or in combination with diuretics, was not superior to placebo in controlling ascites in any of the populations studied.
Hernández-Gea V, Aracil C, Colomo A, Garupera I, Poca M, Torras X, et al. Development of ascites in compensated cirrhosis with severe portal hypertension treated with β-blockers. Am J Gastroenterol. 2012;107:418–27. This study shows that β-blocker treatment, by inducing a decrease in hepatic venous pressure gradient, may reduce the risk of ascites development in patients with compensated cirrhosis.
Sersté T, Melot C, Francoz C, Durand F, Rautou PE, Valla D, et al. Deleterious effects of beta-blockers on survival in patients with cirrhosis and refractory ascites. Hepatology. 2010;52:1017–22.
Ginès P, Berl T, Bernardi M, et al. Hyponatremia in cirrhosis: from pathogenesis to treatment. Hepatology. 1998;28:851–64.
Ginès P, Guevara M. Hyponatremia in cirrhosis: pathogenesis, clinical significance and management. Hepatology. 2008;48:1002–10.
Solà E, Watson H, Graupera I, Turon F, Barreto R, Rodriguez E, et al. Factors related to quality of life in patients with cirrhosis and ascites: relevance of serum sodium concentration and leg edema. J Hepatol. 2012;57:1199–206. This study investigated factors related to quality of life in patients with cirrhosis and ascites. Results of this study indicate that serum sodium concentration is one of the most important independent factors predicting impaired health-related quality of life in patients with cirrhosis and ascites.
Ahluwalia V, Wade JB, Thacker L, Kraft KA, Sterling RK, Stravitz RT, et al. Differential impact of hyponatremia and hepatic encephalopathy on health-related quality of life and brain metabolite abnormalities in cirrhosis. J Hepatol. 2013;59:467–73. This recent study analyzed the effects of hyponatremia and hepatic encephalopathy on health-related quality of life and cognition in patients with cirrhosis. The study shows that hyponatremia has a negative impact on health-related quality of life, independent of the presence of hepatic encephalopathy.
Angeli P, Wong F, Watson H, Gines P. Hyponatremia in cirrhosis: results of a patient population survey. Hepatology. 2006;44:1535–42.
Jalan R, Mookerjee R, Cheshire L, Williams R, et al. Albumin infusion for severe hyponatremia in patients with refractory ascites: a randomized clinical trial. J Hepatol. 2007;46:232A.
McCormick PA, Mistry P, Kaye G, Burroughs AK, McIntyre N. Intravenous albumin infusion is an effective therapy for hyponatraemia in cirrhotic patients with ascites. Gut. 1990;31:204–7.
Wong F, Blei AT, Blendis LM, Thuluvath PJ. A vasopressin receptor antagonist (VPA-985) improves serum sodium concentration in patients with hyponatremia: a multicenter, randomized, placebo-controlled trial. Hepatology. 2003;37:182–91.
Gerbes AL, Gulberg V, Ginès P, et al. VPA Study Group. Therapy of hyponatremia in cirrhosis with a vasopressin receptor antagonist: a randomized double-blind multicenter trial. Gastroenterology. 2003;124:933–9.
Schrier RW, Gross P, Gheorghiade M, et al. Tolvaptan, a selective oral vasopressin V2-receptor antagonist, for hyponatremia. N Engl J Med. 2006;355:2099–112.
Cárdenas A, Ginès P, Marotta P, et al. The safety and efficacy of tolvaptan, an oral vasopressin antagonist in the treatment of hyponatremia in cirrhosis. J Hepatol. 2012;56:571–8. This is the only study investigating the efficacy and safety of tolvaptan specifically in patients with cirrhosis.
Fagundes C, Ginès P. Hepatorenal syndrome: a severe, but treatable, cause of kidney failure in cirrhosis. Am J Kidney Dis. 2012;59:874–85. This article is a recent and updated review of the pathophysiology, diagnosis, and management of HRS.
Salerno F, Gerbes A, Ginès P, Wong F, Arroyo V. Diagnosis, prevention and treatment of hepatorenal syndrome in cirrhosis. Gut. 2007;56:1310–8.
Fagundes C, Pepin MN, Guevara, Barreto R, Casals G, Solà E, et al. Urinary neutrophil gelatinase-lipocalin as biomarker in the differential diagnosis of impairment of kidney function in cirrhosis. J Hepatol. 2012;57:267–73. This study evaluated the usefulness of uNGAL in the differential diagnosis of kidney injury in cirrhosis. It shows that in patients with cirrhosis, uNGAL is significantly higher in those with ATN than in those with HRS.
Verna EC, Brown RS, Farrand E, Pichardo EM, Forster CS, Sola-Del Valle DA, et al. Urinary neutrophil gelatinase-associated lipocalin predicts mortality and identifies acute kidney injury in cirrhosis. Dig Dis Sci. 2012;57:2362–70. This is another study evaluating the usefulness of uNGAL in the differential diagnosis of kidney injury in cirrhosis. Like the previous study, it shows that in patients with cirrhosis, uNGAL is significantly higher in those with ATN than in those with HRS.
Sanyal A, Boyer T, Garcia-Tsao G, et al. A prospective, randomized, double blind, placebo-controlled trial of terlipressin for type 1 hepatorenal syndrome (HRS). Gastroenterology. 2008;134:1360–8.
Martin-Llahi M, Pepin MN, Guevara G, et al. Terlipressin and albumin vs albumin in patients with cirrhosis and hepatorenal syndrome: a randomized study. Gastroenterology. 2008;134:1352–9.
Gluud LL, Christensen K, Christensen E, Krag A. Systematic review of randomized trials on vasoconstrictor drugs for hepatorenal syndrome. Hepatology. 2010;51:576–84.
Piano S, Morando F, Fasolato S, et al. Continuous recurrence of type 1 hepatorenal syndrome and long-term treatment with terlipressin and albumin: a new exception to MELD score in the allocation system to liver transplantation? J Hepatol. 2011;55:491–6. This study shows that terlipressin administered as a continuous IV infusion is safe and effective in patients with type 1 HRS.
Rodriguez E, Elia C, Solà E, Barreto R, Graupera I, Andrealli A, et al. Terlipressin and albumin for type 1 hepatorenal syndrome associated with sepsis. J Hepatol. 2014;60(5):955–61. This proof-of-concept study was the first to investigate the efficacy and safety of terlipressin and albumin treatment for patients with type 1 HRS associated with ongoing sepsis. It found that treatment is safe and effective.
Singh V, Ghosh S, Singh B, Kumar P, Sharma N, Bhalla A, et al. Noradrenaline vs terlipressin in the treatment of hepatorenal syndrome: a randomized study. J Hepatol. 2012;56:1293–8. This randomized study compared the efficacy and safety of terlipressin and noradrenaline for type 1 HRS. It suggests that noradrenaline is as safe and effective as terlipressin in treating type 1 HRS.
Esrailian E, Pantangco ER, Kyulo NL, Hu KQ, Runyon BA. Octreotide/midodrine therapy significantly improves renal function and 30-day survival in patients with type 1 hepatorenal syndrome. Dig Dis Sci. 2007;52:742–8.
Skagen C, Einstein M, Lucey MR, Said A. Combination treatment with octreotide, midodrine, and albumin improves survival in patients with type 1 and type 2 hepatorenal syndrome. J Clin Gastroenterol. 2009;43:680–5.
Kribben A, Gerken G, Haag S, Herget-Rosenthal S, et al. Effects of fractionated plasma separation and adsorption on survival in patients with acute-on-chronic liver failure. Gastroenterology. 2012;142(4):782–9. This randomized trial investigated the effects of fractionated plasma separation and adsorption (FPSA) on survival in patients with acute-on-chronic liver failure. The study did not show a survival benefit in patients treated with FPSA compared with standard medical therapy. Moreover, there were no differences in kidney function during follow-up between the FPSA and standard medical therapy groups.
Mitzner SR, Stange J, Klammt S, et al. Improvement of hepatorenal syndrome with extracorporeal albumin dialysis MARS: results of a prospective, randomized, controlled clinical trial. Liver Transpl. 2000;6:277–86.
Charlton MR, Wall WJ, Ojo AO, Ginès P, et al. Report of the First International Liver Transplantation Society Expert Panel Consensus Conference on Renal Insufficiency in Liver Transplantation. Liver Transpl. 2009;15(11):S1–S34.
Angeli P, Ginès P. Hepatorenal syndrome, MELD score and liver transplantation: an evolving issue with relevant implications for clinical practice. J Hepatol. 2012;57:1135–40. This article reviews the impact of HRS and its pharmacologic therapy in patients on the waiting list for liver transplantation. It analyses the predictive value of the MELD score and its impact on prioritization for organ allocation.
Acknowledgments
Some of the studies reported in this review were performed with the support of grants from the Fondo de Investigación Sanitaria (FIS PI12/00330 and EC/90077) and Ciber de Enfermedades Hepáticas y Digestivas (CIBERehd). CIBERehd is funded by the Instituto de Salud Carlos III, Ministerio de Sanidad, España.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
Elsa Solà, and Isabel Graupera declare that they have no conflicts of interest. Pere Ginès reports grants from Oprhan Therapeutics, and advisory board consultancy from Ferring Pharmaceuticals, outside the submitted work.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Solà, E., Graupera, I. & Ginès, P. From Refractory Ascites to Dilutional Hyponatremia and Hepatorenal Syndrome: Current Options for Treatment. Curr Hepatology Rep 13, 189–197 (2014). https://doi.org/10.1007/s11901-014-0240-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11901-014-0240-8