Abstract
Purpose of Review
We review how understanding the fitness and comorbidity burden of patients, and molecular landscape of underlying acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS) at the time of diagnosis is now integral to treatment.
Recent Findings
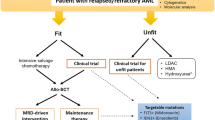
The upfront identification of patients’ fitness and molecular profile facilitates selection of targeted and novel agents, enables risk stratification, allows consideration of allogeneic hematopoietic cell transplantation in high-risk patients, and provides treatment selection for older (age ≥ 75) or otherwise unfit patients who may not tolerate conventional treatment. The use of measurable residual disease (MRD) assessment improves outcome prediction and can also guide therapeutic strategies such as chemotherapy maintenance and transplant. In recent years, several novel drugs have received FDA approval for treating patients with AML with or without specific mutations. A doublet and triplet combination of molecular targeted and other novel treatments have resulted in high response rates in early trials. Following the initial success in AML, novel drugs are undergoing clinical trials in MDS.
Summary
Unprecedented advances have been made in precision medicine approaches in AML and MDS. However, lack of durable responses and long-term disease control in many patients still present significant challenges, which can only be met, to some extent, with innovative combination strategies throughout the course of treatment from induction to consolidation and maintenance.



Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Genomic and epigenomic landscapes of adult de novo acute myeloid leukemia. N Engl J Med. 2013;368(22):2059–2074.
Papaemmanuil E, et al. Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med. 2016;374(23):2209–21.
UpadhyayBanskota S, Khanal N, Bhatt VR. A precision medicine approach to management of acute myeloid leukemia in older adults. Curr Opin Oncol. 2020;32(6):650–5.
Koenig K, et al. The changing landscape of treatment in acute myeloid leukemia. Am Soc Clin Oncol Educ Book. 2020;40:1–12.
Khanal N, UpadhyayBanskota S, Bhatt VR. Novel treatment paradigms in acute myeloid leukemia. Clin Pharmacol Ther. 2020;108(3):506–14.
Bhatt VR. Personalizing therapy for older adults with acute myeloid leukemia role of geriatric assessment and genetic profiling. Cancer Treat Rev. 2019;75:52–61.
Dhakal P, et al. Preferences of adults with cancer for systemic cancer treatment: do preferences differ based on age? Future Oncol. 2022;18(3):311–21.
Bhatt VR, et al. Integrating geriatric assessment and genetic profiling to personalize therapy selection in older adults with acute myeloid leukemia. J Geriatr Oncol 2022.
Perl AE. The role of targeted therapy in the management of patients with AML. Hematology. 2017;2017(1):54–65.
Schlenk RF, et al. Differential impact of allelic ratio and insertion site in FLT3-ITD-positive AML with respect to allogeneic transplantation. Blood. 2014;124(23):3441–9.
Daver N, Venugopal S, Ravandi F. FLT3 mutated acute myeloid leukemia: 2021 treatment algorithm. Blood Cancer J. 2021;11(5):104.
•• Stone RM, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454–64. A phase 3 trial that established addition of midostaurin to intensive chemotherapy as a standard in FLT3 mutated AML.
Rollig C, et al. The addition of sorafenib to standard AML treatment results in a substantial reduction in relapse risk and improved survival. Updated results from long-term follow-up of the randomized-controlled Soraml trial. Blood. 2017;130(Supplement 1):721–721.
•• Perl AE, et al. Gilteritinib or chemotherapy for relapsed or refractory FLT3-mutated AML. N Engl J Med. 2019;381(18):1728–40. A phase 3 trial that demonstrated gilteritinib to be superior than salvage intensive chemotherapy relapsed or refractory FLT3 mutated AML.
Xuan L, et al. Sorafenib maintenance in patients with FLT3-ITD acute myeloid leukaemia undergoing allogeneic haematopoietic stem-cell transplantation: an open-label, multicentre, randomised phase 3 trial. Lancet Oncol. 2020;21(9):1201–12.
Short NJ, et al. A triplet combination of azacitidine, venetoclax and gilteritinib for patients with FLT3-mutated acute myeloid leukemia: results from a phase I/II study. Blood. 2021;138(Supplement 1):696–696.
Maiti A, et al. Triplet therapy with venetoclax, FLT3 inhibitor and decitabine for FLT3-mutated acute myeloid leukemia. Blood Cancer J. 2021;11(2):25.
Malaise M, Steinbach D, Corbacioglu S. Clinical implications of c-Kit mutations in acute myelogenous leukemia. Curr Hematol Malig Rep. 2009;4(2):77–82.
Ayatollahi H, et al. Prognostic importance of C-KIT mutations in core binding factor acute myeloid leukemia: a systematic review. Hematol Oncol Stem Cell Ther. 2017;10(1):1–7.
Marcucci G, et al. Combination of dasatinib with chemotherapy in previously untreated core binding factor acute myeloid leukemia: CALGB 10801. Blood Adv. 2020;4(4):696–705.
Paschka P, et al. Adding dasatinib to intensive treatment in core-binding factor acute myeloid leukemia—results of the AMLSG 11–08 trial. Leukemia. 2018;32(7):1621–30.
DeAngelo DJ, et al. Safety and efficacy of avapritinib in advanced systemic mastocytosis: the phase 1 EXPLORER trial. Nat Med. 2021;27(12):2183–91.
Gotlib J, et al. Efficacy and safety of avapritinib in advanced systemic mastocytosis: interim analysis of the phase 2 PATHFINDER trial. Nat Med. 2021;27(12):2192–9.
Xue, S., et al., Rapid response to avapritinib of acute myeloid leukemia with t(8;21) and KIT mutation relapse post allo-HSCT. Leukemia & Lymphoma, 2022 1–4.
• DiNardo CD, et al. Durable remissions with ivosidenib in IDH1-mutated relapsed or refractory AML. N Engl J Med. 2018;378(25):2386–98. This early phase trial demonstrated the role of ivosidenib in relapsed or refractory IDH1 mutated AML.
• Stein EM, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130(6):722–31. This early phase trial demonstrated the role of enasidenib in relapsed or refractory IDH1 mutated AML.
• Roboz GJ, et al. Ivosidenib induces deep durable remissions in patients with newly diagnosed IDH1-mutant acute myeloid leukemia. Blood. 2020;135(7):463–71. This early phase trial demonstrated the role of ivosidenib as an initial treated in newly diagnosed IDH1 mutated AML.
Amatangelo MD, et al. Enasidenib induces acute myeloid leukemia cell differentiation to promote clinical response. Blood. 2017;130(6):732–41.
Choe S, et al. Molecular mechanisms mediating relapse following ivosidenib monotherapy in IDH1-mutant relapsed or refractory AML. Blood Adv. 2020;4(9):1894–905.
•• Montesinos P, et al. Ivosidenib and azacitidine in IDH1-mutated acute myeloid leukemia. New England Journal of Medicine. 2022;386(16):1519–31. This phase 3 trial demonstrated that a combination of ivosidenib and azacitidine is superior to azacitidine alone in newly diagnosed IDH1 mutated AML.
DiNardo CD, et al. Enasidenib plus azacitidine versus azacitidine alone in patients with newly diagnosed, mutant-IDH2 acute myeloid leukaemia (AG221-AML-005): a single-arm, phase 1b and randomised, phase 2 trial. Lancet Oncol. 2021;22(11):1597–608.
Venugopal S, et al. Efficacy and safety of enasidenib and azacitidine combination in patients with IDH2 mutated acute myeloid leukemia and not eligible for intensive chemotherapy. Blood Cancer J. 2022;12(1):10.
Falini B, et al. NPM1-mutated acute myeloid leukemia: from bench to bedside. Blood. 2020;136(15):1707–21.
•• Schlenk RF, et al. Gemtuzumab ozogamicin in NPM1-mutated acute myeloid leukemia: early results from the prospective randomized AMLSG 09–09 phase III study. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2020;38(6):623–32. This phase 3 trial demonstrated that the addition of gemtuzumab ozogamicin can reduce the risk of relapse in NPM1 mutated AML.
Kapp-Schwoerer S, et al. Impact of gemtuzumab ozogamicin on MRD and relapse risk in patients with NPM1-mutated AML: results from the AMLSG 09–09 trial. Blood. 2020;136(26):3041–50.
DiNardo CD, et al. Molecular patterns of response and treatment failure after frontline venetoclax combinations in older patients with AML. Blood. 2020;135(11):791–803.
Otoukesh S, et al. The efficacy of venetoclax and hypomethylating agents in acute myeloid leukemia with extramedullary involvement. Leuk Lymphoma. 2020;61(8):2020–3.
Issa GC, et al. Therapeutic implications of menin inhibition in acute leukemias. Leukemia. 2021;35(9):2482–95.
Kuykendall A, et al. Acute myeloid leukemia: the good, the bad, and the ugly. Am Soc Clin Oncol Educ Book. 2018;38:555–73.
Hunter AM, Sallman DA. Current status and new treatment approaches in TP53 mutated AML. Best Pract Res Clin Haematol. 2019;32(2):134–44.
Welch JS, et al. TP53 and decitabine in acute myeloid leukemia and myelodysplastic syndromes. N Engl J Med. 2016;375(21):2023–36.
•• DiNardo CD, et al. Azacitidine and venetoclax in previously untreated acute myeloid leukemia. N Engl J Med. 2020;383(7):617–29. This phase 3 trial demonstrated that a combination of venetoclax and azacitidine is superior to azacitidine alone in newly diagnosed AML in older adults.
Sallman DA, et al. Phase 1b/2 combination study of APR-246 and azacitidine (AZA) in patients with TP53 mutant myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML). Blood. 2018;132:3091.
Sallman DA, et al. The first-in-class anti-CD47 antibody magrolimab (5F9) in combination with azacitidine is effective in MDS and AML patients: ongoing phase 1b results. Blood. 2019;134:569.
• Heuser M, et al. Clinical benefit of glasdegib plus low-dose cytarabine in patients with de novo and secondary acute myeloid leukemia: long-term analysis of a phase II randomized trial. Ann Hematol. 2021;100(5):1181–94. This phase 2 trial demonstrated that a combination of glasdegib and low-dose cytarabine is superior to low-dose cytarabine alone in newly diagnosed AML in older adults.
Cortes JE, et al. Survival outcomes and clinical benefit in patients with acute myeloid leukemia treated with glasdegib and low-dose cytarabine according to response to therapy. J Hematol Oncol. 2020;13(1):92.
•• Lancet JE, et al. CPX-351 versus 7+3 cytarabine and daunorubicin chemotherapy in older adults with newly diagnosed high-risk or secondary acute myeloid leukaemia: 5-year results of a randomised, open-label, multicentre, phase 3 trial. Lancet Haematol. 2021;8(7):e481–91. This phase 3 trial demonstrated that CPX351 is superior to 7+3 in newly diagnosed high risk or secondary AML in older adults.
Linenberger ML. CD33-directed therapy with gemtuzumab ozogamicin in acute myeloid leukemia: progress in understanding cytotoxicity and potential mechanisms of drug resistance. Leukemia. 2005;19(2):176–82.
Burnett AK, et al. Identification of patients with acute myeloblastic leukemia who benefit from the addition of gemtuzumab ozogamicin: results of the MRC AML15 trial. J Clin Oncol. 2011;29(4):369–77.
Schuurhuis GJ, et al. Minimal/measurable residual disease in AML: a consensus document from the European LeukemiaNet MRD Working Party. Blood. 2018;131(12):1275–91.
Heuser M, et al. 2021 Update on MRD in acute myeloid leukemia: a consensus document from the European LeukemiaNet MRD Working Party. Blood. 2021;138(26):2753–67.
Short NJ, et al. Association of measurable residual disease with survival outcomes in patients with acute myeloid leukemia: a systematic review and meta-analysis. JAMA Oncol. 2020;6(12):1890–9.
Hourigan CS, et al. Measurable residual disease testing in acute myeloid leukaemia. Leukemia. 2017;31(7):1482–90.
•• Roboz GJ, et al. Oral azacitidine prolongs survival of patients with AML in remission independently of measurable residual disease status. Blood. 2022;139(14):2145–55. This phase 3 trial demonstrated that oral azacitidine maintenance results in survival benefit.
Venditti A, et al. GIMEMA AML1310 trial of risk-adapted, MRD-directed therapy for young adults with newly diagnosed acute myeloid leukemia. Blood. 2019;134(12):935–45.
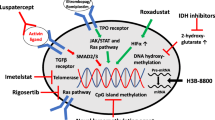
Lee P, et al. Molecular targeted therapy and immunotherapy for myelodysplastic syndrome. Int J Mol Sci. 2021;22(19):10232.
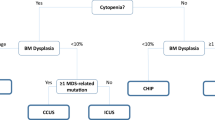
Radakovich N, et al. A geno-clinical decision model for the diagnosis of myelodysplastic syndromes. Blood Adv. 2021;5(21):4361–9.
Cazzola M. Myelodysplastic syndromes. N Engl J Med. 2020;383(14):1358–74.
Nagata Y, et al. Machine learning demonstrates that somatic mutations imprint invariant morphologic features in myelodysplastic syndromes. Blood. 2020;136(20):2249–62.
Sebert M, et al. Ivosidenib monotherapy is effective in patients with IDH1 mutated myelodysplastic syndrome (MDS): the Idiome phase 2 study by the GFM group. Blood. 2021;138:62.
Cortes JE, et al. Olutasidenib (FT-2102) induces rapid remissions in patients with IDH1-mutant myelodysplastic syndrome: results of phase 1/2 single agent treatment and combination with azacitidine. Blood. 2019;134:674.
Venugopal S, et al. MDS-090: phase II study of the IDH2 inhibitor enasidenib in patients with high-risk IDH2-mutated myelodysplastic syndromes (MDS). Clinical Lymphoma Myeloma and Leukemia. 2021;21:S339.
Cumbo C, et al. TP53 in myelodysplastic syndromes: recent biological and clinical findings. Int J Mol Sci. 2020;21(10):3432.
Sallman DA, et al. Eprenetapopt (APR-246) and azacitidine in TP53-mutant myelodysplastic syndromes. J Clin Oncol. 2021;39(14):1584–94.
Daver N, et al. FLT3 mutations in myelodysplastic syndrome and chronic myelomonocytic leukemia. Am J Hematol. 2013;88(1):56–9.
Strati P, et al. Phase I/II trial of the combination of midostaurin (PKC412) and 5-azacytidine for patients with acute myeloid leukemia and myelodysplastic syndrome. Am J Hematol. 2015;90(4):276–81.
Macdonald DA, et al. A phase I/II study of sorafenib in combination with low dose cytarabine in elderly patients with acute myeloid leukemia or high-risk myelodysplastic syndrome from the National Cancer Institute of Canada Clinical Trials Group trial IND186. Leukemia & Lymphoma. 2013;54(4):760–6.
• Garcia-Manero G, et al. Oral cedazuridine/decitabine for MDS and CMML: a phase 2 pharmacokinetic/pharmacodynamic randomized crossover study. Blood. 2020;136(6):674–83. This trial supported the approval of oral decitabine.
Pagliuca S, Gurnari C, Visconte V. Molecular targeted therapy in myelodysplastic syndromes: new options for tailored treatments. Cancers. 2021;13(4):784.
Zeidan AM, et al. A phase 1b study evaluating the safety and efficacy of venetoclax as monotherapy or in combination with azacitidine for the treatment of relapsed/refractory myelodysplastic syndrome. Blood. 2019;134:565.
Garcia JS, et al. Molecular responses are observed across mutational spectrum in treatment-naïve higher-risk myelodysplastic syndrome patients treated with venetoclax plus azacitidine. Blood. 2021;138:241.
Sasaki K, et al. De novo acute myeloid leukemia: a population-based study of outcome in the United States based on the Surveillance, Epidemiology, and End Results (SEER) database, 1980 to 2017. Cancer. 2021;127(12):2049–61.
Döhner H, Wei AH, Löwenberg B. Towards precision medicine for AML. Nat Rev Clin Oncol. 2021;18(9):577–90.
Terwijn M, et al. High prognostic impact of flow cytometric minimal residual disease detection in acute myeloid leukemia: data from the HOVON/SAKK AML 42A study. J Clin Oncol. 2013;31(31):3889–97.
Ngai LL, et al, MRD tailored therapy in AML: what we have learned so far. Front Oncol. 2021:10.
Roloff GW, Griffiths EA. When to obtain genomic data in acute myeloid leukemia (AML) and which mutations matter. Blood Adv. 2018;2(21):3070–80.
Burd A, et al. Precision medicine treatment in acute myeloid leukemia using prospective genomic profiling: feasibility and preliminary efficacy of the Beat AML Master Trial. Nat Med. 2020;26(12):1852–8.
Duncavage EJ, et al. Genome sequencing as an alternative to cytogenetic analysis in myeloid cancers. New Eng J Med. 2021;384(10):924–35.
Bhatt VR. Advances and unanswered questions in management of acute myeloid leukemia in older adults: a glimpse into the future. J Geriatr Oncol. 2021;12(6):980–4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
VRB reports participating in Safety Monitoring Committee for Protagonist, and receiving consulting fees from Genentech, Incyte, Servier Pharmaceuticals LLC, and AbbVie; research funding (institutional) from AbbVie, Pfizer, Incyte, Jazz, and National Marrow Donor Program; and drug support (institutional) from Oncoceutics for a trial. All other authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This is not an original research article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Health Economics.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Upadhyay Banskota, S., Khanal, N., Marar, R.I. et al. Precision Medicine in Myeloid Malignancies: Hype or Hope?. Curr Hematol Malig Rep 17, 217–227 (2022). https://doi.org/10.1007/s11899-022-00674-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11899-022-00674-4