Abstract
Purpose of Review
In this article, we review a range of digital technologies for possible application in heart failure patients, with a focus on lessons learned. We also discuss a future model of heart failure management, as digital technologies continue to become part of standard care.
Recent Findings
Digital technologies are increasingly used by healthcare professionals and those living with heart failure to support more personalised and timely shared decision-making, earlier identification of problems, and an improved experience of care. The COVID-19 pandemic has accelerated the acceptability and implementation of a range of digital technologies, including remote monitoring and health tracking, mobile health (wearable technology and smartphone-based applications), and the use of machine learning to augment data interpretation and decision-making. Much has been learned over recent decades on the challenges and opportunities of technology development, including how best to evaluate the impact of digital health interventions on health and healthcare, the human factors involved in implementation and how best to integrate dataflows into the clinical pathway.
Summary
Supporting patients with heart failure as well as healthcare professionals (both with a broad range of health and digital literacy skills) is crucial to success. Access to digital technologies and the internet remains a challenge for some patients. The aim should be to identify the right technology for the right patient at the right time, in a process of co-design and co-implementation with patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Digital health encompasses the creation and practical use of computerised devices, methods and systems for health and healthcare [1]. It includes mobile health (mHealth: the use of mobile and wireless technologies to support achieving health objectives [2]), health information technology (IT), wearable devices and remote patient management (RPM). Digital health is an integral part of the transformation of healthcare systems towards a more patient-centric preventive model, where resources are focused toward health maintenance rather than only on crisis management.
The vision of a person-centred healthcare system is key to policy makers’ plans for a more modern, flexible and sustainable healthcare system [3, 4], where people living with (or at risk of) cardiovascular disease use a range of digital technologies to ensure optimal experience and outcome of health (and healthcare) decision-making, only interacting with the appropriate healthcare professionals when (and if) this is useful.
The implementation of digital systems in healthcare such as electronic medical records and e-prescribing has accelerated in recent years [5] but technologies to support health maintenance and healthcare decision-making (such as remote monitoring) have seen slower uptake. The COVID-19 pandemic has triggered a rapid wave of adoption and greater acceptance of digital technologies by patients, healthcare professionals (HCP) and systems — a process often referred to as a “tech-celleration” — as change that was expected to take years occurred within a few weeks in response to the need for restricted face-to-face interaction [6•].
This article reviews the lessons learned in the creation, assessment and implementation of digital health technologies for heart failure (HF) management, focusing on remote monitoring and devices to support and improve patient and HCP decision-making (remote patient management, RPM) compared with traditional HF care strategies (Fig. 1). We also share our vision of the future, where the right tool is used at the right time and in the right place to enable optimal outcome and experience of care.
The contrast between the traditional model of heart failure care (left), with a modern digitally-supported patient-centric model of heart failure care (right). The traditional model uses fixed periodic review with a healthcare organisation (HCO) focus on reactive management of patient crises. The modern model of heart failure care uses digital technology to support patients at various stages of their heart failure illness, allowing dynamic management of issues as they arise. Healthcare organisation focus is on pro-active health maintenance. HCP, healthcare professional; HCO, healthcare organisation; CIEDs, cardiac implantable electronic devices; Apps, smartphone applications
Lessons Learned from Remote Monitoring Using Stand-Alone Technologies
The early remote monitoring studies for patients with HF tended to be small, often single centred and of short duration. Patients were typically recruited at (or shortly after) a heart failure hospitalisation (HFH), and the technologies used were simple and stand-alone. Some of the key studies are listed in more detail in (Table 1). Such studies are likely to be subject to publication bias (positive studies more likely to be published than neutral or negative studies) and may not have been representative of more routine practice due to the enthusiasm of the single centres (Fig. 2).
Meta-analysis of these early small randomised studies suggested a powerful impact on both mortality and hospitalisation compared to usual care [13], with a 34% relative risk reduction in mortality (CI 19–46%, p < 0.0001) and 21% relative risk reduction in heart failure hospitalisation (HFH) (CI 6–33%, p = 0.008). How this effect was mediated is unclear but is likely to include improved guideline compliance by HCPs, improved medication and lifestyle compliance by patients, and earlier detection of decompensation with intervention to restabilise the HF syndrome and reduce the need for hospitalisation.
Except for the encouraging results of two early larger trials [7, 19], the next generation of multicentre studies (consequently adequately powered, arguably more likely to be representative of usual practice, and less subject to publication bias) often failed to show a significant difference in “hard” endpoints such as mortality or HFH over 6–20 months of follow-up. This was despite a variety of patient phenotypes and risk, and a wide range of interventions including devices with embedded reminders and assessment of patient-reported symptoms, structured telephone support with nurse specialists and monitoring of weight, blood pressure, heart rate and ECG [8, 9, 20].
TIM-HF2 (Telemedical Interventional Management in patients with Heart Failure) provided the first positive large randomised controlled trial (RCT) in RPM using stand-alone equipment in 710 patients with HF, New York Heart Association (NYHA) class II or III, and a HFH within the preceding 12 months.
TIM-HF2 reported a borderline statistically significant 20% reduction (p = 0.046) in the primary endpoint of percentage of days lost to unplanned admission or death during 12-month follow-up from 6.64 to 4.88%, with no statistically significant reduction in the secondary endpoint of cardiovascular (CV) mortality (p = 0.056) [12••]. However, it required an intensive and well-resourced approach, with daily review of the monitoring data, ability to stream ECGs and a centralised 24/7 telemonitoring centre in Berlin with close connection with the emergency services, local general practitioners and the patient and their family. Assessment of the likely cost-effectiveness of this approach, suggested a small cost saving per patient year (€1760) in the German healthcare system [21].
The neutral results from several other large RCTs, and the selected nature of the population within TIM-HF2, led to the most recent European Society of Cardiology (ESC) guidelines to be lukewarm about its recommendation for routine telemonitoring of patient with HF with stand-alone equipment (class 2 level B — “may be considered”). [22]. Similarly, weak recommendations have been issued by the American, Canadian and Australian HF societies [23,24,25].
Rehabilitation and training programmes in HF have used a combination of supervised and unsupervised sessions in hospital and at home. TELEREH-HF (The Telerehabilation in Heart failure patients) RCT randomised 850 patients up to 6 months after a HFH with NYHA I-III symptoms and reduced ejection fraction [17•]. A total of 425 patients received a 9-week hybrid comprehensive telerehabilitation (HCTR) programme encompassing RPM and individualised tele-rehabilitation. This hybrid approach did not extend the percentage of days alive and out of hospital at extended follow-up: 91.9 (± 19.3) HCTR group vs 92.8 days in the usual care group (± 18.3)(p = 0.74). However, several physical rehabilitation studies have replicated the other findings of TELEREH-HF, with significant but short-term improvement in symptoms, physical performance, functional capacity and depressive symptoms [16, 18, 26, 27] without translating into reductions in mortality or HFH risk [15, 17•]. It is likely that cardiac rehabilitation and physical fitness need to be maintained in order to gain long-term benefits [28] and may be dependent on improving patient’s capacity for self-management [17•]. Ongoing support by digital technology and remote patient management may facilitate these objectives.
Lessons Learned from Remote Monitoring and Remote Patient Management Using Implantable Devices
Cardiac Implantable Electronic Devices (CIEDs)
CIEDs record multiple physiological variables — such as heart rate, heart rate variability, episodes of arrhythmia including atrial fibrillation or fast ventricular rhythms, transthoracic impedance, patient activity, and in some systems sleep apnoea and the intensity of heart sounds. Many of these variables are associated with the risk of HF decompensation [29, 30].
Initial studies (detailed in Table 2) likely placed too much weight on one variable leading to too many false positives. In the DOT-HF (Diagnostic Outcome Trial in Heart Failure) randomised study, an audible alarm was triggered within the device if a threshold of a decreasing trend in transthoracic impedance (reflection of increasing lung water and left ventricular filling pressure) was crossed. This triggered a 79% increase in HF hospitalisation, and the study was stopped early due to this [31]. The human factors triggered by an audible alarm sounding within the device were likely part of the problem: it is difficult for the patient, the family, and the emergency room physicians to ignore an alarm. Many of the admissions were short and with low mortality — suggesting that HF had not truly decompensated, and the admission was often unnecessary.
In the Optilink-HF Study (Optimization of Heart Failure Management using OptiVol™ Fluid Status Monitoring and CareLink™) in just over 1000 patients followed up for an average of 23 months, few actions were taken in response to “alerts” on changes in transthoracic impedance sent by SMS to the responsible physicians, and when action was taken, it was often delayed [42]. There is little point in remotely monitoring a patient if the data collected are not rapidly integrated into the decision-making processes.
With the disappointing results from single parameter monitoring, investigators moved to a more broadly based approach, with “multiparametric” monitoring, often with an algorithm-based approach to stratifying patients into three risk categories (high, medium or low) [33, 38]. In patients stratified into the highest-risk group, the absolute risk of HF decompensation within the next month is low (7% in one analysis [42]), implying that many patients will restabilise even if algorithmic interpretation of the parameters is reliable.
The IN-TIME (Implant-based Multiparameter Telemonitoring of Patients with Heart Failure) study [29] evaluated automatic daily data transmission of multiparametric device data to a single call centre vs. those receiving conventional follow-up through 12 months after ICD or CRT-D implantation in 716 patients enrolled at 36 centres in Australia, Europe and Israel [43]. The odds of the primary endpoint of the composite “Packer” clinical score for HF, (comprising all-cause death, overnight HFH, change in NYHA class, and change in patient global self-assessment) worsening in the intervention group was 0.63 (95% CI 0.43–0.90) compared with the control group. A reduction in a secondary endpoint of all-cause mortality was also reported. The authors interpreted their findings as most likely due to the centralized daily review of all monitored parameters, combined with the protocolised and timely actions that were taken in response to the data [29]. A pooled analysis of remote monitoring of CIEDs using one manufacturer’s technology (including IN-TIME) reported that active remote management was associated with a 36% reduction in HFH (p = 0.007) and a borderline significant reduction in all-cause (but not CV) mortality [44].
In the larger and longer duration REM-HF (REmote Management of Heart Failure using implantable electronic devices) study at nine large UK hospitals, all the remotely collected data from a CIED was reviewed weekly by a team experienced in HF and remote monitoring [35]. They had the time to focus on the remote monitoring processes and worked to a standardised protocol. Despite multiple actions being taken by the monitoring teams in response to the data review of the 1650 patients followed up for a mean of 2.8 years, there was no significant change in either HFH or CV mortality. This study illustrates that without prespecified standard operating procedures, it is not straightforward to translate complex multiparametric monitoring information into “actionable” care strategies to improve clinical outcomes.
More formal approaches to multiparametric monitoring include the Triage-HF studies [39, 40]. In the British Triage-HF plus, a high “Heart Failure Risk Score” (HFRS™) triggered a clinical phone triage system operated by the local HF team. This approach provided high sensitivity but low specificity for worsening HF. The investigators concluded that it was safe to continue to monitor remotely patients with medium or low risk scores, but those with a high-risk score required telephone triage, with 71% being positive for symptoms of worsening HF or an alternative medical problem. Most recently, the HeartLogic™ algorithm has been tested in an FDA-approved study. MANAGE-HF (Multiple cArdiac seNsors for mAnaGEment of Heart Failure) — phase I [45•] uses a scoring system developed and validated from large multicentre datasets, but with additional support for centres to continually review and act on the data and to increase their efforts to persuade patients to act on the advice given. It is clear that if a patient is not willing to change their medication or lifestyle advice compliance in response to remotely collected data, the link between more data and better outcome will be lost.
The role of insertable cardiac monitors (ICMs) in HF care has been limited currently to the diagnosis of arrhythmia such as sub-clinical atrial fibrillation [46]. ALLEVIATE-HF (Algorithm Using LINQ Sensors for Evaluation And Treatment of Heart Failure: NCT04452149), due to report in 2024, will randomise up to 700 patients to an ICM-based risk stratification algorithm (plus a medicines management plan) or usual management. The endpoint is a hierarchical composite of cardiovascular death, HF events, change in Kansas City Cardiomyopathy Questionnaire and change in 6-min walk test distance.
Remote haemodynamic monitoring
Remote haemodynamic monitoring has been examined with a range of technologies in the past two decades (described in detail in Table 3). Most data are related to the implantable pulmonary artery pressure (PAP) monitoring system CardioMEMS™ HF. Patients receive a home Patient Electronics Unit for daily upload of resting, supine PAP information from the sensor to a secure website (Merlin.net™). PAP increases represent an early sign of imminent cardiac decompensation. Uploaded PAP information should be reviewed at least weekly by trained HCPs. Additional PAP reviews are triggered by email notifications of PAP excursions outside the user-defined thresholds automatically issued by the Merlin.net system. The sensor is easily implanted at right heart catheterisation, carries a low risk of technical failure in routine care [47•, 48] and facilitates targeting of a specific PAP range, with adjustment of HF therapies including diuretics to maintain patients within that range, where possible. Most adjustments occur in the first few months of monitoring, and thereafter, trends in the pressures are used to detect signs of decompensation or over-treatment.
The pivotal American CHAMPION trial randomised patients with NYHA class III HF across a range of ejection fraction and reported a 30% reduction in HF hospitalisation risk at 6 months [50]. This study, along with other post-marketing randomised and observational studies in the USA and Europe [47•, 48, 53, 54], has suggested robust evidence of benefit when used in specialist centres.
More recently, two European studies have shown the likely added benefit in NYHA class III patients in routine care within healthcare systems outside the USA: MEMS-HF (CardioMEMS European Monitoring Study for Heart Failure) in Germany [47•] and COAST (CardioMEMS HF System Post-Market Study) in the UK [48], with a 62% and 82% reduction in annualised HF hospitalisation rates, respectively. Such data have facilitated positive reimbursement decision making outside the USA, including most recently in England by the National Institute for Health and Care Excellence (NICE) [55]. In Germany, health authorities are awaiting the results of PASSPORT-HF (Pulmonary Artery Sensor System Pressure Monitoring to Improve Heart Failure Outcomes [56]), a randomised study which compares the efficacy of standard HCP-coordinated HF care (“basic care”) plus PAP-guided management with basic care alone, before a final reimbursement decision is made.
Broadening of the indication for remote PAP monitoring using CardioMEMS™ to less symptomatic patients (NYHA class II) was the purpose of GUIDE-HF (Haemodynamic-guided management of heart failure) [52]. Complicated by the COVID-19 pandemic, with huge shifts in patient care patterns during “lockdowns”, the study failed to reach statistical significance overall. A pre-specified pre-COVID analysis (using the majority of data in the study, prior to the first USA COVID-19-related lockdown) suggested a strong evidence of benefit across this broader range of patients (24% reduction in HF events over 12 months, p = 0.014). The FDA has just approved the expansion of the use of CardioMEMS to include patients with NYHA class II HF, provided they have experienced a recent HFH or have an elevated plasma natriuretic peptide concentration [57].
The most recent update to the European Society of Cardiology (ESC) HF guideline makes the recommendation for implantable remote monitoring (for CIEDs or haemodynamic monitoring) only for those with the CardioMEMS™ system, and limited to those with NYHA Class III symptoms despite optimal medical therapy [22], while Australian and Canadian guidelines have a weak or no recommendation, respectively [23, 24].
Ongoing studies may expand the choice of location of invasive monitoring device beyond PAP sensors, allowing a more personalised approach related to the individual’s physiology. Implant sites currently under investigation in first-in-human safety trials, include the inferior vena cava (FUTURE-HF) and the left atrium (VECTOR-HF). Placed in the inter-atrial septum, preliminary results from the left atrial pressure sensor show it is likely to be safe, the readings show a strong correlation with invasive PAP measurements and there is a signal of improvement in NYHA class [58].
A recurring theme in remote monitoring studies is that the ultimate decision maker remains the patient. Patients should be selected with care. They should be at risk of HFH, must “buy in” to the concept of daily collection of data (often requiring their active participation), and must be willing to comply with treatment or adherence recommendations even if asymptomatic. This care cycle will otherwise be broken — and this is particularly likely to happen if patients are contacted by someone they have not met and with whom they do not have a therapeutic relationship [59]. Similarly, HCPs need to be encouraged to act, and to persuade the patient to change therapy (or to be more adherent to therapy) even if they are initially reluctant. Without these actions the potential benefit of early detection of decompensation may be lost — with time taken to collect and review data but without any action being triggered that may reduce risk [59]. HCPs quickly lose interest in reviewing remotely collected data if they cannot observe the benefits for their patients or the system. These and other key elements of RPM are summarised in Fig. 3.
Key elements to consider for successful remote patient management. Modified with permission from Angermann C, 2019 [59]. AI, artificial intelligence; HCP, healthcare professional
Wearables ( Table 4 )
Wearable medical devices (“wearables”) can provide a variety of data from sensors that typically can be worn on the wrist, clipped to clothing or stuck to the skin [65, 66••]. The most studied are “smart” watches, activity monitors and monitoring patches.
Smartwatches and activity monitors typically combine accelerometers, which track movement, with photoplethysmography (PPG), an optical sensor able to monitor heart rates. PPG traces can also be used to assess cardiac rhythm. The Apple™ Heart study used PPG with an “irregular pulse algorithm” to screen patients for atrial fibrillation (AF); 34% of patients with a positive notification subsequently had AF diagnosed on ECG patch testing [62]. The accuracy of PPG is hindered by a high dropout rate, changes in position, changes in exercise and rapid changes in heart rate [67,68,69], and that it does not produce an ECG [70]. Despite this, the early results from the Fitbit Heart Study add to the Apple™ Heart study showing the potential utility of wrist-worn wearables in screening of asymptomatic individuals, with likely higher relevance for those at higher risk of AF such as patients with heart failure [60], and those at risk of development of the heart failure syndrome due to AF [71].
ECG sensors, such as Kardia™, have a larger footprint for electrodes allowing recording of up to six ECG leads, improving both diagnostic accuracy and time to diagnosis with beneficial reductions in cost when used for high-risk patients in the community or attending primary care [60, 61•]. This technology can be used with a range of smartphones and NICE has recently recommended this as a validated option in patients with suspected paroxysmal AF [72], allowing patients with HF and palpitation or syncope to benefit from high-quality prolonged rhythm detection.
Wearable patch technology is typically placed on the chest wall and can monitor several additional variables including movement, temperature and respiratory rate for a period of days to weeks. This technology greatly increases the diagnostic yield for AF [63] but also provides data for multiparametric scores similar to those used with remote monitoring of CIEDs in HF.
LINK-HF [64••] (Multisensor Non-invasive Remote Monitoring for Prediction of Heart Failure Exacerbation) studied the VitalConnect™ disposable patch sensor (7-day battery life) with a re-usable sensor electronics module. One module can provide months’ of continuous monitoring. In patients recently discharged after a HFH, the technology was able to identify the risk of further HFH with greater than 76% sensitivity and an 85% specificity in a retrospective analysis — similar to the performance of Medtronic’s Heart Failure Risk Score (HFRS™) and Boston Scientific’s Heartlogic™ algorithm in their development and validation studies in CIEDs [33, 37]. Such patch technology may not just be beneficial in monitoring patients at home but also may allow real-time monitoring in hospital and other care settings, although further validation is required [73].
Technologies in Development ( Table 5 )
Examples of novel technologies under investigation to improve RPM in HF include: ReDS (remote dielectric sensor from Sensible Medical), SimpleSense™ (Nanowear), µCor™ (Zoll®) and HearO™ (Cordio Medical).
ReDS uses an electromagnetic based technology developed by the military to provide an instantaneous estimation of lung fluid content. Using ReDS to guide management in recently discharged patients with HF demonstrated a halving of the rate of HFH over 6 months in a non-randomised study [74]. The ReDS-SAFE HF trial is currently randomising up to 240 patients admitted for HF at one US centre, to ReDS-guided discharge or usual care, with a primary composite endpoint of unplanned visit for HF or all-cause mortality (NCT04305717).
SimpleSENSE™ is an FDA-cleared wearable undergarment that monitors several vital signs such as heart rate, heart sounds, respiratory rate, lung volume and physical activity and is connected to a machine-learning platform. Results are awaited from a cohort study developing and validating a multi-parametric algorithm to detect HF decompensation.
µCor is an ECG patch and HF management system that uses radiofrequency technology to measure multiple parameters including thoracic fluid index. A multicentre trial aims to correlate this to the risk of HF events (NCT03476187).
Voice detection algorithms allow the HearO™ mobile phone application to assess fluid status and highlight the difference between pre- and post-dialysis states [84], and through fluid optimisation during a HFH [81].
Factors Affecting the Development and Use of Digital Tools in HF Care
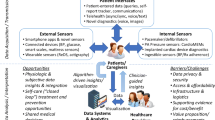
There are an increasing range of digital technologies available to support remote patient management in HF (and other diseases) (Fig. 4) but several factors are pivotal to their optimal development, maintenance, and longer term use. We have expanded on these issues in our commentary in this edition of the journal, entitled The Digital Future of Heart Failure Care. They include issues around the evaluation and regulation of technology, co-design and co-implementation, data security and access, machine learning in support of decision-making and litigation risk. Key to success is a better understanding of the support needs and capabilities of patients living with HF (and their healthcare advisors) including their digital and health literacy and degree of activation.
Right Technology, Right patient, Right time: a Vision of the Future
The traditional model of HF care with periodic clinic review, using only data collected at that time by the healthcare team using “their” technologies, is likely to be replaced (or at least supplemented) by a more patient-centric approach.
Collecting data remotely provides “real” life data — which represent more than 99% of the time when patients are in their own environment — to be used in shared decision-making. Shared decision-making requires a good working relationship between patient and HCP, as well as a patient who is adequately informed, educated and (therefore) motivated to make changes based on remote monitoring data. Access to HCPs and systems will still be needed and must be available flexibly and at potentially short notice, but much of the routine processes of care can be supported by technology and RPM, freeing up time and bandwidth for members of the healthcare team to deal with the more complex, or nuanced, situations where human-to-human interaction adds more value.
In practice, digital remote technologies will support the key activities within the care pathway — ensuring rapid and accurate diagnosis, risk stratification and prognostication, therapeutic decision-making and tailored support including education and care. Active shared decision-making between clinician and patient will decide which technology can best be used to achieve the shared goals, whether it is initial education and support to self-care, identification when another therapy may be beneficial, or helping the patient maintain stability with optimised quality of life at home, without the need to engage with the “official” healthcare system by attending clinic or being hospitalised.
Technology should provide closed-loop interactions with patients, offering advice on fluid intake, diuretic regime, exercise, necessity for blood tests and when to contact an HCP. When patients require input from an HCP they deserve a tailored and responsive interaction based on multiple relevant data points and sources, with decision and sensemaking supported by digital tools (and likely artificial intelligence). Clinical deterioration will be predicted and identified early using remote monitoring and stabilisation facilitated by appropriate escalation of therapy. Admission to hospital will, of course, remain necessary for some patients, but early discharge will be supported with virtual wards (“hospital-at-home”) with monitoring pathways and rehabilitation facilitated remotely.
We highlight the differences between the traditional model of care and this more modern, digitally enabled approach to HF care in Fig. 1.
Conclusion
Digital technologies are already deeply embedded within all aspects of society, including health and healthcare. HF care already relies on many of them, but the clinical pathways and treatment algorithms to successfully integrate digital technologies and decision support into the healthcare system are in their infancy.
Ultimately, digital technologies will become part of the “new normal”, being selected for use when they make sense and help patients and their HCPs achieve shared goals. Due consideration must be paid to ensuring the evidence base is robust, that data flows and human factors are considered, and that the digital technologies are co-designed and implemented to ensure a better outcome and experience of care. Data should not be collected for its own sake — it must support better decision-making and more efficient care.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
U.S. Food and Drug Administration. What is digital health? FDA: Digital Health Center of Excellence. 2020. https://www.fda.gov/medical-devices/digital-health-center-excellence/what-digital-health. Accessed 11 April 2022.
The Global Observatory for eHealth (GOe). mHealth: New horizons for health through mobile technologies. Global Observatory for eHealth series. 2011;Volume 3. https://www.who.int/goe/publications/goe_mhealth_web.pdf. Accessed 11 April 2022.
Digital Health Europe. Recommendations on the European Health Data Space: Supporting responsible health data sharing and use through governance, policy and practice. . July 2021. https://digitalhealtheurope.eu/wp-content/uploads/DHE_recommendations_on_EHDS_July_2021.pdf. Accessed 11 April 2022.
European Commission. Communication from the commission to the European parliament, the council, the European economic and social committee and the committee of the regions on enabling the digital transformation of health and care in the digital single market; empowering citizens and building a healthier society. April 2018. https://www.eumonitor.eu/9353000/1/j9tvgajcor7dxyk_j9vvik7m1c3gyxp/vknuqttcz4zd. Accessed 11 April 2022.
Statista Research department. Projected growth in global healthcare data volume 2020. 2020. https://www.statista.com/statistics/1037970/global-healthcare-data-volume/. Accessed 11 April 2022.
Cowie MR, Lam CSP. Remote monitoring and digital health tools in CVD management. Nat Rev Cardiol. Jul 2021;18(7):457–458. https://doi.org/10.1038/s41569-021-00548-xConcise opinion piece on digital health in CVD management
Cleland JG, Louis AA, Rigby AS, Janssens U, Balk AH. Noninvasive home telemonitoring for patients with heart failure at high risk of recurrent admission and death: the Trans-European Network-Home-Care Management System (TEN-HMS) study. J Am Coll Cardiol. 2005;45(10):1654–64. https://doi.org/10.1016/j.jacc.2005.01.050.
Dar O, Riley J, Chapman C, Dubrey SW, Morris S, Rosen SD, et al. A randomized trial of home telemonitoring in a typical elderly heart failure population in North West London: results of the Home-HF study. Eur J Heart Fail. 2009;11(3):319–25. https://doi.org/10.1093/eurjhf/hfn050.
Chaudhry SI, Mattera JA, Curtis JP, Spertus JA, Herrin J, Lin Z, et al. Telemonitoring in patients with heart failure. N Engl J Med. 2010;363(24):2301–9. https://doi.org/10.1056/NEJMoa1010029.
Koehler F, Winkler S, Schieber M, Sechtem U, Stangl K, Böhm M, et al. Telemedical interventional monitoring in heart failure (TIM-HF), a randomized, controlled intervention trial investigating the impact of telemedicine on mortality in ambulatory patients with heart failure: study design. Eur J Heart Fail. 2010;12(12):1354–62. https://doi.org/10.1093/eurjhf/hfq199.
Ong MK, Romano PS, Edgington S, Aronow HU, Auerbach AD, Black JT, et al. Effectiveness of remote patient monitoring after discharge of hospitalized patients with heart failure: the better effectiveness after transition – heart failure (BEAT-HF) randomized clinical trial. JAMA Intern Med. 2016;176(3):310–8. https://doi.org/10.1001/jamainternmed.2015.7712.
Koehler F, Koehler K, Deckwart O, Prescher S, Wegscheider K, Kirwan BA, et al. Efficacy of telemedical interventional management in patients with heart failure (TIM-HF2): a randomised, controlled, parallel-group, unmasked trial. Lancet. Sep 22 2018;392(10152):1047–1057. https://doi.org/10.1016/s0140-6736(18)31880-4This is the first positive large randomised controlled trial of remote monitoring
Inglis SC, Clark RA, McAlister FA, Ball J, Lewinter C, Cullington D, et al. Structured telephone support or telemonitoring programmes for patients with chronic heart failure. Cochrane Database Syst Rev. 2010;(8) https://doi.org/10.1002/14651858.CD007228.pub2
Zhu Y, Gu X, Xu C. Effectiveness of telemedicine systems for adults with heart failure: a meta-analysis of randomized controlled trials. Heart Fail Rev. 2020;25(2):231–243. https://doi.org/10.1007/s10741-019-09801-5This recent meta-analysis shows a significant reduction in mortality when using remote monitoring
O’Connor CM, Whellan DJ, Lee KL, Keteyian SJ, Cooper LS, Ellis SJ, et al. Efficacy and safety of exercise training in patients with chronic heart failure: HF-ACTION randomized controlled trial. JAMA. 2009;301(14):1439–50. https://doi.org/10.1001/jama.2009.454.
Dalal HM, Taylor RS, Jolly K, Davis RC, Doherty P, Miles J, et al. The effects and costs of home-based rehabilitation for heart failure with reduced ejection fraction: the REACH-HF multicentre randomized controlled trial. Eur J Prev Cardiol. 2019;26(3):262–72. https://doi.org/10.1177/2047487318806358.
Piotrowicz E, Pencina MJ, Opolski G, Zareba W, Banach M, Kowalik I, et al. Effects of a 9-week hybrid comprehensive telerehabilitation program on long-term outcomes in patients with heart failure: the telerehabilitation in heart failure patients (TELEREH-HF) randomized clinical trial. JAMA Cardiol. Mar 1 2020;5(3):300–308. https://doi.org/10.1001/jamacardio.2019.5006Large randomised controlled study of a modern hybrid approach of rehabilitation and remote patient monitoring
Kitzman DW, Whellan DJ, Duncan P, Pastva AM, Mentz RJ, Reeves GR, et al. Physical rehabilitation for older patients hospitalized for heart failure. N Engl J Med. 2021;385(3):203–16. https://doi.org/10.1056/NEJMoa2026141.
Angermann CE, Störk S, Gelbrich G, Faller H, Jahns R, Frantz S, et al. Mode of action and effects of standardized collaborative disease management on mortality and morbidity in patients with systolic heart failure: the Interdisciplinary Network for Heart Failure (INH) study. Circ Heart Fail. 2012;5(1):25–35. https://doi.org/10.1161/circheartfailure.111.962969.
Koehler F, Winkler S, Schieber M, Sechtem U, Stangl K, Böhm M, et al. Telemedical Interventional Monitoring in Heart Failure (TIM-HF), a randomized, controlled intervention trial investigating the impact of telemedicine on mortality in ambulatory patients with heart failure: study design. Eur J Heart Fail. 2010;12(12):1354–62.
Sydow H, Prescher S, Koehler F, Koehler K, Dorenkamp M, Spethmann S, et al. Cost-effectiveness of noninvasive telemedical interventional management in patients with heart failure: health economic analysis of the TIM-HF2 trial. Clin Res Cardiol. Dec 11 2021; https://doi.org/10.1007/s00392-021-01980-2
McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) With the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2021;42(36):3599–726. https://doi.org/10.1093/eurheartj/ehab368.
Atherton JJ, Sindone A, De Pasquale CG, Driscoll A, Macdonald PS, Hopper I, et al. National Heart Foundation of Australia and Cardiac Society of Australia and New Zealand: Guidelines for the prevention, detection, and management of heart failure in australia 2018. Heart Lung Circ. 2018;27(10):1123–208. https://doi.org/10.1016/j.hlc.2018.06.1042.
Ezekowitz JA, O’Meara E, McDonald MA, Abrams H, Chan M, Ducharme A, et al. 2017 Comprehensive update of the Canadian Cardiovascular Society Guidelines for the management of heart failure. Can J Cardiol. 2017;33(11):1342–433. https://doi.org/10.1016/j.cjca.2017.08.022.
Maddox TM, Januzzi JL, Allen LA, Breathett K, Butler J, Davis LL, et al. 2021 Update to the 2017 ACC expert consensus decision pathway for optimization of heart failure treatment: answers to 10 pivotal issues about heart failure with reduced ejection fraction. J Am Coll Cardiol. 2021;77(6):772–810. https://doi.org/10.1016/j.jacc.2020.11.022.
Mueller S, Winzer EB, Duvinage A, Gevaert AB, Edelmann F, Haller B, et al. Effect of high-intensity interval training, moderate continuous training, or guideline-based physical activity advice on peak oxygen consumption in patients with heart failure with preserved ejection fraction: a randomized clinical trial. JAMA. 2021;325(6):542–51. https://doi.org/10.1001/jama.2020.26812.
Blumenthal JA, Babyak MA, O’Connor C, Keteyian S, Landzberg J, Howlett J, et al. Effects of exercise training on depressive symptoms in patients with chronic heart failure: the HF-ACTION randomized trial. JAMA. 2012;308(5):465–74. https://doi.org/10.1001/jama.2012.8720.
Ekelund L-G, Haskell WL, Johnson JL, Whaley FS, Criqui MH, Sheps DS. Physical fitness as a predictor of cardiovascular mortality in asymptomatic North American Men. N Engl J Med. 1988;319(21):1379–84. https://doi.org/10.1056/nejm198811243192104.
Hindricks G, Taborsky M, Glikson M, Heinrich U, Schumacher B, Katz A, et al. Implant-based multiparameter telemonitoring of patients with heart failure (IN-TIME): a randomised controlled trial. Lancet. 2014;384(9943):583–90. https://doi.org/10.1016/s0140-6736(14)61176-4.
Yu C-M, Wang L, Chau E, Chan RH-W, Kong S-L, Tang M-O, et al. Intrathoracic impedance monitoring in patients with heart failure. Circulation. 2005;112(6):841–8. https://doi.org/10.1161/circulationaha.104.492207.
Braunschweig F, Ford I, Conraads V, Cowie MR, Jondeau G, Kautzner J, et al. Can monitoring of intrathoracic impedance reduce morbidity and mortality in patients with chronic heart failure? Rationale and design of the diagnostic outcome trial in heart failure (DOT-HF). Eur J Heart Fail. 2008;10(9):907–16. https://doi.org/10.1016/j.ejheart.2008.06.016.
Conraads VM, Tavazzi L, Santini M, Oliva F, Gerritse B, Yu C-M, et al. Sensitivity and positive predictive value of implantable intrathoracic impedance monitoring as a predictor of heart failure hospitalizations: the SENSE-HF trial. Eur Heart J. 2011;32(18):2266–73. https://doi.org/10.1093/eurheartj/ehr050.
Cowie MR, Sarkar S, Koehler J, Whellan DJ, Crossley GH, Tang WH, et al. Development and validation of an integrated diagnostic algorithm derived from parameters monitored in implantable devices for identifying patients at risk for heart failure hospitalization in an ambulatory setting. Eur Heart J. 2013;34(31):2472–80. https://doi.org/10.1093/eurheartj/eht083.
Böhm M, Drexler H, Oswald H, Rybak K, Bosch R, Butter C, et al. Fluid status telemedicine alerts for heart failure: a randomized controlled trial. Eur Heart J. 2016;37(41):3154–63. https://doi.org/10.1093/eurheartj/ehw099.
Morgan JM, Kitt S, Gill J, McComb JM, Ng GA, Raftery J, et al. Remote management of heart failure using implantable electronic devices. Eur Heart J. 2017;38(30):2352–60. https://doi.org/10.1093/eurheartj/ehx227.
Boriani G, Da Costa A, Quesada A, Ricci RP, Favale S, Boscolo G, et al. Effects of remote monitoring on clinical outcomes and use of healthcare resources in heart failure patients with biventricular defibrillators: results of the MORE-CARE multicentre randomized controlled trial. Eur J Heart Fail. 2017;19(3):416–25. https://doi.org/10.1002/ejhf.626.
Boehmer JP, Hariharan R, Devecchi FG, Smith AL, Molon G, Capucci A, et al. A multisensor algorithm predicts heart failure events in patients with implanted devices: results from the MultiSENSE Study. JACC Heart Fail. 2017;5(3):216–25. https://doi.org/10.1016/j.jchf.2016.12.011.
Gardner RS, Singh JP, Stancak B, Nair DG, Cao M, Schulze C, et al. HeartLogic multisensor algorithm identifies patients during periods of significantly increased risk of heart failure events: results from the MultiSENSE Study. Circ Heart Fail. 2018;11(7):e004669. https://doi.org/10.1161/CIRCHEARTFAILURE.117.004669.
Virani SA, Sharma V, McCann M, Koehler J, Tsang B, Zieroth S. Prospective evaluation of integrated device diagnostics for heart failure management: results of the TRIAGE-HF study. ESC Heart Fail. 2018;5(5):809–17. https://doi.org/10.1002/ehf2.12309.
Ahmed FZ, Taylor JK, Green C, Moore L, Goode A, Black P, et al. Triage-HF Plus: a novel device-based remote monitoring pathway to identify worsening heart failure. ESC Heart Fail. 2020;7(1):107–16. https://doi.org/10.1002/ehf2.12529.
Calo L, Bianchi V, Ferraioli D, Santini L, Dello Russo A, Carriere C, et al. Multiparametric implantable cardioverter-defibrillator algorithm for heart failure risk stratification and management: an analysis in clinical practice. Circ Heart Fail. 2021;14(10): e008134. https://doi.org/10.1161/CIRCHEARTFAILURE.120.008134.
Böhm M, Drexler H, Oswald H, Rybak K, Bosch R, Butter C, et al. Fluid status telemedicine alerts for heart failure: a randomized controlled trial. Eur Heart J. 2016;37(41):3154–63. https://doi.org/10.1093/eurheartj/ehw099.
Packer M. Proposal for a new clinical end point to evaluate the efficacy of drugs and devices in the treatment of chronic heart failure. J Card Fail. 2001;7(2):176–82. https://doi.org/10.1054/jcaf.2001.25652.
Hindricks G, Varma N, Kacet S, Lewalter T, Søgaard P, Guédon-Moreau L, et al. Daily remote monitoring of implantable cardioverter-defibrillators: insights from the pooled patient-level data from three randomized controlled trials (IN-TIME, ECOST, TRUST). Eur Heart J. 2017;38(22):1749–55. https://doi.org/10.1093/eurheartj/ehx015.
A Hernandez NA, L Allen, R Ahmed, V Averina, J Boehmer, C Chien, M Galvao, L Klein, M Kwan, C Lam, S Ruble, C Stolen, K Stein. Multiple cardiac sensors for management of heart failure (MANAGE-HF)- Phase I. Jour Card fail. 2022; page numbers to be added First phase of important trial suggesting safe and successful integration of remote monitoring into clinical care
Kort RSS, Tuininga YS, Bosker HA, Janssen M, Tukkie R. Telemonitoring with an implantable loop recorder in outpatient heart failure care : one year follow-up report from a prospective observational Dutch multicentre study. Neth Heart J. 2019;27(1):46–51. https://doi.org/10.1007/s12471-018-1198-x.
Angermann CE, Assmus B, Anker SD, Asselbergs FW, Brachmann J, Brett ME, et al. Pulmonary artery pressure-guided therapy in ambulatory patients with symptomatic heart failure: the CardioMEMS European Monitoring Study for Heart Failure (MEMS-HF). Eur J Heart Fail. Oct 2020;22(10):1891–1901. https://doi.org/10.1002/ejhf.1943First study to suggest generalisability of US CardioMEMS findings to rest of western healthcare system
Cowie MR, Flett A, Cowburn P, Foley P, Chandrasekaran B, Loke I, et al. Real-world evidence in a national health service: results of the UK CardioMEMS HF System Post-Market Study. ESC Heart Fail. 2022;9(1):48–56. https://doi.org/10.1002/ehf2.13748.
Bourge RC, Abraham WT, Adamson PB, Aaron MF, Aranda JM Jr, Magalski A, et al. Randomized controlled trial of an implantable continuous hemodynamic monitor in patients with advanced heart failure: the COMPASS-HF study. J Am Coll Cardiol. 2008;51(11):1073–9. https://doi.org/10.1016/j.jacc.2007.10.061.
Abraham WT, Adamson PB, Bourge RC, Aaron MF, Costanzo MR, Stevenson LW, et al. Wireless pulmonary artery haemodynamic monitoring in chronic heart failure: a randomised controlled trial. Lancet. 2011;377(9766):658–66. https://doi.org/10.1016/s0140-6736(11)60101-3.
Abraham WT, Stevenson LW, Bourge RC, Lindenfeld JA, Bauman JG, Adamson PB. Sustained efficacy of pulmonary artery pressure to guide adjustment of chronic heart failure therapy: complete follow-up results from the CHAMPION randomised trial. The Lancet. 2016;387(10017):453–61. https://doi.org/10.1016/S0140-6736(15)00723-0.
Lindenfeld J, Zile MR, Desai AS, Bhatt K, Ducharme A, Horstmanshof D, et al. Haemodynamic-guided management of heart failure (GUIDE-HF): a randomised controlled trial. The Lancet. 2021;398(10304):991–1001. https://doi.org/10.1016/S0140-6736(21)01754-2.
Shavelle DM, Desai AS, Abraham WT, Bourge RC, Raval N, Rathman LD, et al. Lower rates of heart failure and all-cause hospitalizations during pulmonary artery pressure-guided therapy for ambulatory heart failure: one-year outcomes from the CardioMEMS Post-Approval Study. Circ Heart Fail. 2020;13(8): e006863. https://doi.org/10.1161/circheartfailure.119.006863.
Abraham WT, Stevenson LW, Bourge RC, Lindenfeld JA, Bauman JG, Adamson PB. Sustained efficacy of pulmonary artery pressure to guide adjustment of chronic heart failure therapy: complete follow-up results from the CHAMPION randomised trial. Lancet. 2016;387(10017):453–61. https://doi.org/10.1016/s0140-6736(15)00723-0.
National Institute for Health and Care Excellence. Percutaneous implantation of pulmonary artery pressure sensors for monitoring treatment of chronic heart failure: interventional procedures guidance [IPG711]. NICE; 2021. https://www.nice.org.uk/guidance/IPG711. Accessed 11 April 2022.
Störk S, Bernhardt A, Böhm M, Brachmann J, Dagres N, Frantz S, et al. Pulmonary artery sensor system pressure monitoring to improve heart failure outcomes (PASSPORT-HF): rationale and design of the PASSPORT-HF multicenter randomized clinical trial. Clin Res Cardiol. Mar 4 2022:1–11. https://doi.org/10.1007/s00392-022-01987-3
Abbott Press release. Abbott’s CardioMEMS HF system receives FDA approval to support patients battling earlier-stage heart failure. Abbott media. 2022. https://abbott.mediaroom.com/2022-02-21-Abbotts-CardioMEMS-TM-HF-System-Receives-FDA-Approval-to-Support-Patients-Battling-Earlier-Stage-Heart-Failure. Accessed 11 April 2022.
Perl L, Meerkin D, D’Amario D, Avraham BB, Hasin T, Ince H, et al. TCT-130 Initial results from the VECTOR-HF Trial—a system for remote left atrial pressure monitoring for patients with heart failure. J Am Coll Cardiol. 2021;78(19_Supplement_S):B55–B55. https://doi.org/10.1016/j.jacc.2021.09.980.
Angermann CE. The trialist’s perspective: what do you need to prove for remote monitoring devices to be approved? Eur Heart J Suppl. 2019;21(Suppl M):M57-m60. https://doi.org/10.1093/eurheartj/suz214.
Lowres N, Neubeck L, Salkeld G, Krass I, McLachlan AJ, Redfern J, et al. Feasibility and cost-effectiveness of stroke prevention through community screening for atrial fibrillation using iPhone ECG in pharmacies. The SEARCH-AF study Thromb Haemost. 2014;111(6):1167–76. https://doi.org/10.1160/th14-03-0231.
Halcox JPJ, Wareham K, Cardew A, Gilmore M, Barry JP, Phillips C, et al. Assessment of remote heart rhythm sampling using the alivecor heart monitor to screen for atrial fibrillation. Circulation. 2017;136(19):1784–1794. https://doi.org/10.1161/CIRCULATIONAHA.117.030583Regular rhythm recording in at risk patients is beneficial in the detection of asymptomatic atrial fibrillation
Perez MV, Mahaffey KW, Hedlin H, Rumsfeld JS, Garcia A, Ferris T, et al. Large-Scale Assessment of a smartwatch to identify atrial fibrillation. N Engl J Med. 2019;381(20):1909–17. https://doi.org/10.1056/NEJMoa1901183.
Gladstone DJ, Wachter R, Schmalstieg-Bahr K, Quinn FR, Hummers E, Ivers N, et al. Screening for atrial fibrillation in the older population: a randomized clinical trial. JAMA Cardiology. 2021;6(5):558–67. https://doi.org/10.1001/jamacardio.2021.0038.
Stehlik J, Schmalfuss C, Bozkurt B, Nativi-Nicolau J, Wohlfahrt P, Wegerich S, et al. Continuous wearable monitoring analytics predict heart failure hospitalization. Circ Heart Fail. 2020;13(3):e006513. https://doi.org/10.1161/CIRCHEARTFAILURE.119.006513First study of a wearable sensor providing comparable sensitivity and specificty to invasive sensors for heart failure hospitalisation
Singhal A, Cowie MR. The role of wearables in heart failure. Curr Heart Fail Rep. 2020;17(4):125–32. https://doi.org/10.1007/s11897-020-00467-x.
DeVore AD, Wosik J, Hernandez AF. The future of wearables in heart failure patients. JACC Heart Fail. Nov 2019;7(11):922–932. https://doi.org/10.1016/j.jchf.2019.08.008Recent high quality review of wearable technology in heart failure
Feehan LM, Geldman J, Sayre EC, Park C, Ezzat AM, Yoo JY, et al. Accuracy of fitbit devices: systematic review and narrative syntheses of quantitative data. JMIR Mhealth Uhealth. 2018;6(8): e10527. https://doi.org/10.2196/10527.
Cadmus-Bertram L, Gangnon R, Wirkus EJ, Thraen-Borowski KM, Gorzelitz-Liebhauser J. The accuracy of heart rate monitoring by some wrist-worn activity trackers. Ann Intern Med. 2017;166(8):610–2. https://doi.org/10.7326/l16-0353.
Moayedi Y, Abdulmajeed R, Duero Posada J, Foroutan F, Alba AC, Cafazzo J, et al. Assessing the use of wrist-worn devices in patients with heart failure: feasibility study. JMIR Cardio. 2017;1(2):e8. https://doi.org/10.2196/cardio.8301.
Dörr M, Nohturfft V, Brasier N, Bosshard E, Djurdjevic A, Gross S, et al. The WATCH AF Trial: SmartWATCHes for detection of atrial fibrillation. JACC Clin Electrophysiol. 2019;5(2):199–208. https://doi.org/10.1016/j.jacep.2018.10.006.
Carlisle MA, Fudim M, DeVore AD, Piccini JP. Heart failure and atrial fibrillation, like fire and fury. JACC: Heart Failure. 2019;7(6):447–56. https://doi.org/10.1016/j.jchf.2019.03.005.
National Institute for Health and Care Excellence. KardiaMobile for detecting atrial fibrillation: medical technologies guidance [MTG64]. NICE; 2022. https://www.nice.org.uk/guidance/mtg64. Accessed 11 April 2022.
Downey C, Ng S, Jayne D, Wong D. Reliability of a wearable wireless patch for continuous remote monitoring of vital signs in patients recovering from major surgery: a clinical validation study from the TRaCINg trial. BMJ Open. 2019;9(8):e031150. https://doi.org/10.1136/bmjopen-2019-031150.
Abraham WT, Anker S, Burkhoff D, Cleland J, Gorodeski E, Jaarsma T, et al. Primary results of the sensible medical innovations lung fluid status monitor allows reducing readmission rate of heart failure patients (smile) trial. J Card Fail. 2019;25(11):938. https://doi.org/10.1016/j.cardfail.2019.11.007.
Mullens W, Sharif F, Dupont M, Rothman AMK, Wijns W. Digital health care solution for proactive heart failure management with the Cordella Heart Failure System: results of the SIRONA first-in-human study. Eur J Heart Fail. 2020;22(10):1912–9. https://doi.org/10.1002/ejhf.1870.
Konstam MA, Udelson JE, Butler J, Klein HU, Parker JD, Teerlink JR, et al. Impact of autonomic regulation therapy in patients with heart failure. Circ Heart Fail. 2019;12(11): e005879. https://doi.org/10.1161/CIRCHEARTFAILURE.119.005879.
Premchand RK, Sharma K, Mittal S, Monteiro R, Dixit S, Libbus I, et al. Autonomic regulation therapy via left or right cervical vagus nerve stimulation in patients with chronic heart failure: results of the ANTHEM-HF trial. J Card Fail. 2014;20(11):808–16. https://doi.org/10.1016/j.cardfail.2014.08.009.
Wheeler CM, Patel S, Waldman CE, Panchal J, Sidhu RS, Krol M, et al. Abstract: hearing the heart, validation of a novel digital health earbud technology to measure cardiac time intervals through infrasonic hemodynography. Circulation. 2021;144(Suppl_1):A11669–A11669. https://doi.org/10.1161/circ.144.suppl_1.11669.
Kosevic D, Wiedemann D, Vukovic P, Ristic V, Riebandt J, Radak U, et al. Cardio-microcurrent device for chronic heart failure: first-in-human clinical study. ESC Heart Fail. 2021;8(2):962–70. https://doi.org/10.1002/ehf2.13242.
Lubitz SA, Faranesh AZ, Atlas SJ, McManus DD, Singer DE, Pagoto S, et al. Rationale and design of a large population study to validate software for the assessment of atrial fibrillation from data acquired by a consumer tracker or smartwatch: The Fitbit heart study. Am Heart J. 2021;238:16–26. https://doi.org/10.1016/j.ahj.2021.04.003.
Amir O, Abraham WT, Azzam ZS, Berger G, Anker SD, Pinney SP, et al. Remote speech analysis in the evaluation of hospitalized patients with acute decompensated heart failure. JACC: Heart Failure. 2022;10(1):41–9. https://doi.org/10.1016/j.jchf.2021.08.008.
Cleland JGF, Young R, Jorbenadze A, Shaburishvili T, Demyanchuk V, Buriak R, et al. A first in human multi-center, open label, prospective study to evaluate safety, usability and performance of the VisONE system for heart failure with a reduced left ventricular ejection fraction. J Card Fail. 2020;26(10, Supplement):S64. https://doi.org/10.1016/j.cardfail.2020.09.188.
Ivey-Miranda JB, Wetterling F, Gaul R, Sheridan S, Asher JL, Rao VS, et al. Changes in inferior vena cava area represent a more sensitive metric than changes in filling pressures during experimental manipulation of intravascular volume and tone. Eur J Heart Fail. Nov 27 2021; https://doi.org/10.1002/ejhf.2395
Amir O, Anker SD, Gork I, Abraham WT, Pinney SP, Burkhoff D, et al. Feasibility of remote speech analysis in evaluation of dynamic fluid overload in heart failure patients undergoing haemodialysis treatment. ESC Heart Fail. 2021;8(4):2467–72. https://doi.org/10.1002/ehf2.13367.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The salary of KCCM is funded by a fellowship from Abbott. CEA has chaired the Steering Committee of the MEMS-HF study and is co-chairing the steering committee of the PASSPORT-HF study. She further declares grant support, personal fees and/or non-financial support from Abbott, Astra-Zeneca, Boehringer Ingelheim, Medtronic, Novartis, ResMed, Thermo Fisher and Vifor, all outside of the submitted work, and acknowledges non-financial support from the University Hospital Würzburg, non-financial support from Comprehensive Heart Failure Centre Würzburg and grant support from the German Ministry for Education and Research (BMBF). CEA is the Editor-in-Chief of Current Heart Failure Reports. MRC chairs the steering committee of the COAST study and was a member of the MANAGE-HF, DOT-HF, HOME-HF, SENSE-HF, REM-HF steering committees. MRC has provided consultancy advice to AstraZeneca, Novartis, Bayer, Roche diagnostics, Sevier, Fire1foundry, Abbott, Medtronic and Boston Scientific. His hospital receives research grants for a fellowship — currently held by KCCM.
Human and Animal Rights and Informed Consent.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Digital Medicine in Heart Failure
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
McBeath, K.C.C., Angermann, C.E. & Cowie, M.R. Digital Technologies to Support Better Outcome and Experience of Care in Patients with Heart Failure. Curr Heart Fail Rep 19, 75–108 (2022). https://doi.org/10.1007/s11897-022-00548-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11897-022-00548-z