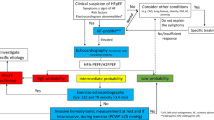
Abstract
Purpose of Review
Cardiac troponin (cTn) is the biomarker of choice for the diagnosis of acute myocardial infarction (MI); use of this biomarker has centered around the 99th percentile upper reference limit (URL) for healthy populations. Recent development and regulatory approval of high-sensitivity cardiac troponin (hs-cTn) assays have required a fresh look at utilization of the 99th percentile URL. This review covers issues regarding the 99th percentile URL and approaches for use of cTn as biomarker for detecting cardiac injury and diagnosis of acute myocardial infarction.
Recent Findings
Development of hs-cTn assays has allowed determination and use of sex-specific 99th percentile URLs for assessing cardiac injury and increased the utility of cTn values below the 99th percentile URL. This improved analytical performance for hs-cTn assays has allowed for development of accelerated diagnostic protocols (ADPs) for rapid assessment and disposition of patients based on serial sampling of cTn for use in acute MI diagnosis as soon as 0–1 h after clinical presentation.
Summary
The 99th percentile URLs of cTn is essential for detecting cardiac injury; however, use of the 99th percentile URLs in the era of hs-cTn results may be modified. ADPs have the potential to substantially decrease the time many patients spend under evaluation for acute MI, thereby potentiating improvement in patient satisfaction, decreased healthcare costs, and reducing the burden on emergency departments.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Boeddinghaus J, Nestelberger T, Twerenbold R, Koechlin L, Meier M, Troester V, et al. High-sensitivity cardiac troponin I assay for early diagnosis of acute myocardial infarction. (in eng), Clin Chem. Jul 2019;65(7):893–904. https://doi.org/10.1373/clinchem.2018.300061.
Wildi K, et al. Misdiagnosis of myocardial infarction related to limitations of the current regulatory approach to define clinical decision values for cardiac troponin. (in eng), Circulation. 2015;131(23):2032–40. https://doi.org/10.1161/circulationaha.114.014129.
Reichlin T, et al. One-hour rule-out and rule-in of acute myocardial infarction using high-sensitivity cardiac troponin T. (in eng), Arch Intern Med. 2012;172(16):1211–8. https://doi.org/10.1001/archinternmed.2012.3698.
Rubini Gimenez M, et al. Rapid rule out of acute myocardial infarction using undetectable levels of high-sensitivity cardiac troponin. (in eng), Int J Cardiol. 2013;168(4):3896–901. https://doi.org/10.1016/j.ijcard.2013.06.049.
Rubini Gimenez M, et al. One-hour rule-in and rule-out of acute myocardial infarction using high-sensitivity cardiac troponin I. (in eng), Am J Med. 2015;128(8):861–870.e4. https://doi.org/10.1016/j.amjmed.2015.01.046.
Twerenbold R, et al. Clinical use of high-sensitivity cardiac troponin in patients with suspected myocardial infarction. (in eng), J Am Coll Cardiol. 2017;70(8):996–1012. https://doi.org/10.1016/j.jacc.2017.07.718.
Chapman AR, Fujisawa T, Lee KK, Andrews JP, Anand A, Sandeman D, et al. Novel high-sensitivity cardiac troponin I assay in patients with suspected acute coronary syndrome. (in eng), Heart. 2019;105(8):616–22. https://doi.org/10.1136/heartjnl-2018-314093.
Christenson RH, et al. Pivotal findings for a high-sensitivity cardiac troponin assay: results of the HIGH-US study. (in eng), Clin Biochem. 2019. https://doi.org/10.1016/j.clinbiochem.2019.10.013.
Sandoval Y, et al. Myocardial infarction risk stratification with a single measurement of high-sensitivity troponin I. (in eng), J Am Coll Cardiol. 2019;74(3):271–82. https://doi.org/10.1016/j.jacc.2019.05.058.
Thygesen K, Alpert JS, Jaffe AS, Chaitman BR, Bax JJ, Morrow DA, et al. Fourth universal definition of myocardial infarction (2018). J Am Coll Cardiol. 2018;72(18):2231–64. https://doi.org/10.1016/j.jacc.2018.08.1038.
Thygesen K, et al. Universal definition of myocardial infarction: Kristian Thygesen, Joseph S. Alpert and Harvey D. White on behalf of the joint ESC/ACCF/AHA/WHF task force for the redefinition of myocardial infarction. Eur Heart J. 2007;28(20):2525–38. https://doi.org/10.1093/eurheartj/ehm355.
Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BR, White HD, et al. Third universal definition of myocardial infarction. (in eng), Eur Heart J. 2012;33(20):2551–67. https://doi.org/10.1093/eurheartj/ehs184.
Myocardial infarction redefined--a consensus document of The Joint European Society of Cardiology/American College of Cardiology Committee for the redefinition of myocardial infarction, (in eng). Eur Heart J. 2000;21(18):1502–13. https://doi.org/10.1053/euhj.2000.2305.
Goodman SG, Steg PG, Eagle KA, Fox KA, López-Sendón J, Montalescot G, et al. The diagnostic and prognostic impact of the redefinition of acute myocardial infarction: lessons from the Global Registry of Acute Coronary Events (GRACE). (in eng), Am Heart J. 2006;151(3):654–60. https://doi.org/10.1016/j.ahj.2005.05.014.
Gerhardt W, Katus H, Ravkilde J, Hamm C, Jørgensen PJ, Peheim E, et al. S-troponin T in suspected ischemic myocardial injury compared with mass and catalytic concentrations of S-creatine kinase isoenzyme MB. (in eng), Clin Chem. 1991;37(8):1405–11.
Apple FS, Collinson PO. Analytical characteristics of high-sensitivity cardiac troponin assays. (in eng), Clin Chem. 2012;58(1):54–61. https://doi.org/10.1373/clinchem.2011.165795.
•• Wu AHB, et al. Clinical laboratory practice recommendations for the use of cardiac troponin in acute coronary syndrome: expert opinion from the Academy of the American Association for Clinical Chemistry and the Task Force on Clinical Applications of Cardiac Bio-Markers of the International Federation of Clinical Chemistry and Laboratory Medicine. (in eng), Clin Chem. 2018;64(4):645–55. https://doi.org/10.1373/clinchem.2017.277186This article provides concensus guidelines prepared by experts in the field for the application of cTnI assays in the clinical setting.
Collinson PO, Saenger AK, Apple FS. High sensitivity, contemporary and point-of-care cardiac troponin assays: educational aids developed by the IFCC Committee on Clinical Application of Cardiac Bio-Markers. (in eng), Clin Chem Lab Med. 2019;57(5):623–32. https://doi.org/10.1515/cclm-2018-1211.
Apple FS, Jaffe AS, Collinson P, Mockel M, Ordonez-Llanos J, Lindahl B, et al. IFCC educational materials on selected analytical and clinical applications of high sensitivity cardiac troponin assays. (in eng), Clin Biochem. 2015;48(4–5):201–3. https://doi.org/10.1016/j.clinbiochem.2014.08.021.
Apple FS, Sandoval Y, Jaffe AS, Ordonez-Llanos J. Cardiac troponin assays: guide to understanding analytical characteristics and their impact on clinical care. (in eng), Clin Chem. 2017;63(1):73–81. https://doi.org/10.1373/clinchem.2016.255109.
Januzzi JL Jr, et al. Recommendations for institutions transitioning to high-sensitivity troponin testing: JACC Scientific Expert Panel. (in eng), J Am Coll Cardiol. 2019;73(9):1059–77. https://doi.org/10.1016/j.jacc.2018.12.046.
Jaffe AS, Apple FS, Morrow DA, Lindahl B, Katus HA. Being rational about (im)precision: a statement from the Biochemistry Subcommittee of the Joint European Society of cardiology/American College of Cardiology Foundation/American Heart Association/World Heart Federation Task Force for the definition of myocardial infarction. (in eng), Clin Chem. 2010;56(6):941–3. https://doi.org/10.1373/clinchem.2010.143958.
Shah ASV, Griffiths M, Lee KK, McAllister DA, Hunter AL, Ferry AV, et al. High sensitivity cardiac troponin and the under-diagnosis of myocardial infarction in women: prospective cohort study. BMJ. 2015;350:g7873. https://doi.org/10.1136/bmj.g7873.
Peacock WF, Baumann BM, Bruton D, Davis TE, Handy B, Jones CW, et al. Efficacy of high-sensitivity troponin T in identifying very-low-risk patients with possible acute coronary syndrome. JAMA Cardiol. 2018;3(2):104–11. https://doi.org/10.1001/jamacardio.2017.4625.
Wu AHB, Apple F, Love SA, Koch D, Myers GL, Christenson RH, et al. Creation of a universal sample bank for determining the 99th percentile for cardiac troponin assays. The Journal of Applied Laboratory Medicine. 2019;1(6):711–9. https://doi.org/10.1373/jalm.2016.022897.
Gore MO, Seliger SL, deFilippi CR, Nambi V, Christenson RH, Hashim IA, et al. Age- and sex-dependent upper reference limits for the high-sensitivity cardiac troponin T assay. J Am Coll Cardiol. 2014;63(14):1441–8. https://doi.org/10.1016/j.jacc.2013.12.032.
Christenson RH, Mullins K, Duh SH. Validation of high-sensitivity performance for a United States Food and Drug Administration cleared cardiac troponin I assay. (in eng), Clin Biochem. 2018;56:4–10. https://doi.org/10.1016/j.clinbiochem.2018.05.004.
Trupp RJ, Albert G, Ziegler A. Sex-specific 99th percentiles derived from the AACC Universal Sample Bank for the Roche Gen 5 cTnT assay: comorbidities and statistical methods influence derivation of reference limits. (in eng) Clin Biochem. 2018;52:173. https://doi.org/10.1016/j.clinbiochem.2017.11.003.
Apple FS, Wu AHB, Sandoval Y, Sexter A, Love SA, Myers G, et al. Sex-specific 99th percentile upper reference limits for high sensitivity cardiac troponin assays derived using a universal sample Bank. Clin Chem. 2020;66:434–44. https://doi.org/10.1093/clinchem/hvz029.
•• Shah ASV, et al. High-sensitivity troponin in the evaluation of patients with suspected acute coronary syndrome: a stepped-wedge, cluster-randomised controlled trial. (in eng) Lancet. 2018;392(10151):919–28. https://doi.org/10.1016/s0140-6736(18)31923-8This study questions the use of the 99th precentile as diagnostic for myocardial infaction and provides evidence suggesting that following the guidelines of the 4th universial definition of myocaridal infaction including the use of the 99th precentile and high-sensitivity cTnI assays did not improve outcomes for patients.
Macrae AR, et al. Assessing the requirement for the 6-hour interval between specimens in the American Heart Association Classification of Myocardial Infarction in Epidemiology and Clinical Research Studies. (in eng), Clin Chem. May 2006;52(5):812–8. https://doi.org/10.1373/clinchem.2005.059550.
Amsterdam EA, et al. AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. (in eng), Circulation. 2014;130(25):2354–94. https://doi.org/10.1161/cir.0000000000000133.
Zhelev Z, et al. Diagnostic accuracy of contemporary and high-sensitivity cardiac troponin assays used in serial testing, versus single-sample testing as a comparator, to triage patients suspected of acute non-ST-segment elevation myocardial infarction: a systematic review protocol. (in eng) BMJ Open. 2019;9(3):e026012. https://doi.org/10.1136/bmjopen-2018-026012.
Cullen LA, Mills NL, Mahler S, Body R. Early rule-out and rule-in strategies for myocardial infarction. (in eng), Clin Chem. 2017;63(1):129–39. https://doi.org/10.1373/clinchem.2016.254730.
• Roffi M, et al. 2015 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: Task Force for the Management of Acute Coronary Syndromes in Patients Presenting without Persistent ST-Segment Elevation of the European Society of Cardiology (ESC). (in eng) Eur Heart J. 2016;37(3):267–315. https://doi.org/10.1093/eurheartj/ehv320This article written by experts in the field supports the use of the 0/1 algorthims and absolute changes in cTnI for rule-in/rule-out of acute myocardial infaction.
Sorensen NA, et al. Diagnostic evaluation of a high-sensitivity troponin I point-of-care assay. (in eng), Clin Chem. 2019;65(12):1592–601. https://doi.org/10.1373/clinchem.2019.307405.
Alpert JS, Jaffe AS. 1-h high-sensitivity troponin rule-out and rule-in approach. Strengths and Potential Weaknesses. 2018;72(6):633–5. https://doi.org/10.1016/j.jacc.2018.05.041.
Nowak RM, et al. Performance of novel high-sensitivity cardiac troponin I assays for 0/1-hour and 0/2- to 3-hour evaluations for acute myocardial infarction: results from the HIGH-US study. (in eng), Ann Emerg Med. 2020. https://doi.org/10.1016/j.annemergmed.2019.12.008.
Caposino PV, Kondratovich MV. Considerations for single-measurement risk-stratification strategies for myocardial infarction using cardiac troponin assays. (in eng) J Am Coll Cardiol. 2019;74(3):283–4. https://doi.org/10.1016/j.jacc.2019.06.014.
Ting HH, et al. Delay from symptom onset to hospital presentation for patients with non-ST-segment elevation myocardial infarction. (in eng) Arch Intern Med. 2010;170(20):1834–41. https://doi.org/10.1001/archinternmed.2010.385.
Montalescot G, Dallongeville J, van Belle E, Rouanet S, Baulac C, Degrandsart A, et al. STEMI and NSTEMI: are they so different? 1 year outcomes in acute myocardial infarction as defined by the ESC/ACC definition (the OPERA registry). (in eng), Eur Heart J. 2007;28(12):1409–17. https://doi.org/10.1093/eurheartj/ehm031.
Twerenbold R, et al. Prospective validation of the 0/1-h algorithm for early diagnosis of myocardial infarction. (in eng) J Am Coll Cardiol. 2018;72(6):620–32. https://doi.org/10.1016/j.jacc.2018.05.040.
Gunsolus IL, Jaffe AS, Sexter A, Schulz K, Ler R, Lindgren B, et al. Sex-specific 99th percentiles derived from the AACC Universal Sample Bank for the Roche Gen 5 cTnT assay: comorbidities and statistical methods influence derivation of reference limits. (in eng), Clin Biochem. 2017;50(18):1073–7. https://doi.org/10.1016/j.clinbiochem.2017.09.009.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Kristin E. Mullins declares no conflict of interest.
Robert H. Christenson reports personal fees from Roche Diagnostics, Siemens Healthineers, Becton Dickinson, Beckman-Coulter, Quidel Diagnostics, and Sphingotech.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Cardiac Biomarkers
Rights and permissions
About this article
Cite this article
Mullins, K.E., Christenson, R.H. Optimal Detection of Acute Myocardial Injury and Infarction with Cardiac Troponin: Beyond the 99th Percentile, into the High-Sensitivity Era. Curr Cardiol Rep 22, 101 (2020). https://doi.org/10.1007/s11886-020-01350-w
Published:
DOI: https://doi.org/10.1007/s11886-020-01350-w