Abstract
Bioaerosols play a significant role in indoor air quality (IAQ) as they can be the cause of several health problems, including acute allergies and infectious diseases. This study aimed to characterize and compare the microbial air quality of air-conditioned (AC) and naturally ventilated (NV) office rooms in the Upper Silesia region of Poland. The bacterial samples were collected during the late spring season. Culturable bacteria were deposited on the nutrient media on Petri dishes to investigate the viable-culturable count (VCC) of bacteria and bacterial community structure using a Biolog GEN III system. In total, 12 species of bacteria were identified, with the most isolated Macrococcus equipercicus, Micrococcus luteus D, Staphylococcus xylosus (indoor), and Bacillus species (outdoor). The indoor mean concentrations of bacterial aerosol ranged from 102 to 103 CFU m−3, below Polish proposals for threshold limit standards in office buildings. The indoor-to-outdoor (I/O) ratios indicated that studied air pollutants in the office rooms originated from the indoor air. These results, together with community composition of bacteria, indicate that most of the bacteria present in the studied office building were relatively fresh and of human origin. Multi-antibiotic resistance (MAR) tests showed that the most antibiotic-resistant features were present in Macrococcus species. The office building exposure dose (OBED) and mass median aerodynamic diameter (MMAD) of bacteria aerosol were estimated. The highest value of OBED over the study period was obtained for staff working in offices with natural ventilation (141 CFU kg−1), in contrast to the value for staff working in offices equipped with air conditioning (about 100 CFU kg−1). The MMAD of viable airborne bacteria was higher in AC offices (2.4 μm) than in NV offices (2.2 μm).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
People spend more than an average of around 87% of the day indoors (Lee and Chang 2000; Tringe et al. 2008; Ashmore and Dimitroulopoulou 2009; Wichmann et al. 2010). Indoor air quality (IAQ) is an important environmental health matter that needs to be assessed because people inhale 6–10 l of air per minute, which amounts to 15,000 l per day (Wood et al. 2002). According to studies conducted in the last 20 years by the United States Environmental Protection Agency, indoor air is often more polluted than outdoor air, and indoor air pollution has been ranked among the top five risks to public health (Kotzias 2005; Gawrońska and Bakera 2015). The health effects associated with poor air quality contribute to heart and lung diseases (Shin et al. 2015). Moreover, air pollution in 2012 was the cause of 3.7 million premature deaths (Błaszczyk et al. 2017).
Bioaerosol particles are one of the pollutants that can cause a reduction in IAQ in office buildings. They account for 5–34% of indoor air pollution (Gizaw et al. 2016). A bioaerosol is a colloidal suspension formed by liquid droplets and particles of solid matter in the air. These particles suspended in the air may consist of bacteria, fungi, viruses, fragments of any or all of these or their metabolic products (e.g., mycotoxins), endotoxins (part of the outer membrane of the cell wall of Gram-negative bacteria), plant pollen, and fragments of plant tissues. Bioaerosols can become a serious risk to the health of the population, mainly because airborne bacteria and fungi can cause infectious diseases, as well as allergic and toxic effects. Human skin and oral and nasal cavities harbor trillions of microorganisms that may accumulate in offices. Microbes from the soil or from plants can also be vectored by office workers or can be carried on dust particles from the outdoor air (Di Giulio et al. 2010; Hewitt et al. 2012; Nazaroff 2016).
The time-activity studies show that adults spend 18–25% of their time at work (Klepeis et al. 2001; Brasche and Bischof 2005). In fact, an air-conditioning (AC) system is practically the only technical solution used both to improve the air quality and to provide employees with proper working conditions. The outdoor air, which is delivered into the indoor through the AC system, should be free from the pollutants, and the system ensures an ideal temperature and humidity (Gołofit-Szymczak and Górny 2010). AC systems can help remove or dilute more than 80% of air pollutants from atmosphere, but they can also provide favorable breeding grounds for many biological agents (Law et al. 2001). Insufficient maintenance of AC systems can often lead to unintentional pollution of indoor air (Gołofit-Szymczak and Górny 2010).
During the two decades, many studies (Pastuszka et al. 2000; Pastuszka et al. 2005; Bernstein et al. 2008; Aydogdu et al. 2010; Nasir and Colbeck 2010; Pegas et al. 2010; Salleh et al. 2011; Dumała and Dudzińska 2013; Moon et al. 2014; Karottki et al. 2015) carried out in a variety of indoor environments showed a great variation in total concentration of air pollutants. Given the potential of airborne bioaerosols to cause disease, information on the total concentration of bioaerosol particles is insufficient. In practice, information relating to the particle size distribution is critical (Latif et al. 2014). Another important aspect is the dose of inhaled air pollutants. The dose is the amount of the pollutant that actually crosses one of the body’s boundaries and reaches the target tissue. Differences in processes of the ventilation in humans mean that identical exposure may not result in an identical dose. Personal exposure levels and individual susceptibilities are highly variable, and this explains why an inhalation dose has not been established to characterize workers’ real risk from the exposure to bioaerosols resulting from work activities (Watson et al. 1988).
Our initial findings indicated that the role of indoor emission sources in offices was significant and suggested that efficient and regularly maintained air-conditioning systems can ensure a proper hygienic air quality in office buildings. The study includes five aspects. Our primary goal was to evaluate the impact of ventilation design (natural or mechanical) on microbial IAQ. Therefore, we tried to find: (a) the viable-culturable count (VCC) of airborne bacteria in office rooms, (b) the size distributions and mass median aerodynamic diameter (MMAD) of bacterial aerosol, (c) the difference between the bacterial community structures in two types of offices air-conditioned or naturally ventilated. Secondary goals of our study were to calculate the dose of inhaled bacterial aerosol in the office building (OBED) and to test the antibiotic resistance of airborne bacteria (MAR). The results can be used to inform exposure assessment, risk assessment, and risk management activities related to environmental health and to provide data for a better understanding of IAQ in the urban areas of developing countries.
Material and methods
Characteristic of sampling site and office building
The studied area was located in a busy urban region of southern Poland (Silesia) (Fig. 1). Culturable bacterial samples were taken simultaneously in indoor and outdoor areas of Gliwice (50°17′37.1″ N, 18°40′54.9″ E) during the late spring season (from 7 May to 7 June 2017), when the average outdoor air temperature was about 24 °C and the indoor temperature was 21 °C. The air was sampled in a detached office building with seven floors. The usable area of this building is more than 14,000 m2. The offices were occupied by one or two employees per office; there is typical office equipment, computers and printers. Daily cleaning inside the offices occurs in the morning when staff members are not present in the rooms. A short description of the office building and details of its basic environmental parameters are presented in Table 1.
The study was conducted in six offices (three with air-conditioning and three with natural ventilation), each with a volume of 40 m3. In office rooms with AC, the air-conditioning system used refrigerant/Freon R-410A with a multi-split variable refrigerant flow (VRF) system. This system consists of one outdoor unit and 16 indoor units, interconnected by refrigerant pipelines. The multi-split VRF system can be operated in space cooling mode or heating mode by reversing a four-way valve located in the outdoor unit.
Each measurement was conducted between 7:00 a.m. and 8:00 a.m. at the beginning of the working day (to determine the indoor background) and at noon (after 5–6 hours (h) of work). During background sampling in the air-conditioned offices, the AC system was in operation. The air samples were taken in the center of each office room or at a height of about 1.5 m above the ground for the outdoor samples, to simulate aspiration from the human breathing zone.
Downstream analysis was performed as described in “Sampling and analytical methods,” “Microbiological analyses,” “Multi-antibiotic resistance test,” and “Statistical analysis.”
Sampling and analytical methods
Viable-culturable bioaerosol samples were collected using a six-stage Andersen cascade impactor (Thermo Fisher Scientific, Waltham, MA, USA) with aerodynamic diameter cut points of 0.65, 1.1, 2.1, 3.3, 4.7, and > 7.0 μm. The pump ensured a constant flow rate (28.3 L min−1) through the impactor. Collection time was 10 min, calculated following Nevalainen et al. (Nevalainen et al. 1992; Nevalainen et al. 1993).
A Petri dish containing specific nutrient media was placed on all the impactor stages. Impactors were disinfected of spraying 70% ethyl alcohol and wiping. Trypticase soy agar (TSA) was used with actidione for culturing bacteria. The Petri dishes were incubated for 48 h at 36 ± 1 °C. Enumeration of bacteria was conducted according to the Polish standard, and the concentration of bacteria aerosol was measured in CFU (colony forming units)/m−3 of air.
Microbiological analyses
All the strains used in the experiment were screened on a Biolog GEN III (Biolog, Hayward, CA, USA) microtiter plate, containing 71 different carbon sources and 23 chemical sensitivity assays. Strains were grown on TSA plates, and then each pure colony was transferred into inoculating fluid. The bacterial suspension was adjusted to 95% transmittance using a Biolog turbidimeter. Then, 150 μl of the suspension was dispensed into each well of the Biolog GEN III microplate. Inoculated plates were monitored via Biolog’s OmniLog software for 48 h at 37 °C with measurements taken every 15 min.
Multi-antibiotic resistance (MAR) test
Cultivated bacteria, previously identified using the Biolog GEN III technique, were also tested for multi-antibiotic resistance (MAR). In this experiment, isolated strains of the cultivated bacteria were taken. Overnight cultures of the bacterial isolates were diluted to 1 McFarland unit (3 × 108 CFU/ml). Subsequently, 100 μl of bacterial inoculum was spread over the surface of a Mueller-Hinton agar plate (Oxoid, USA). Antimicrobial susceptibility testing discs (Oxoid, USA) were saturated and placed on the inoculated Mueller-Hinton agar plates. The 36 different antibiotics and their concentrations were chosen to include the most common species in the literature on antibiotic resistance. Various groups, e.g., aminocoumarins, carbapenems, cephalosporins, glycopeptides, macrolides, penicillins, quinolones/fluoroquinolones, and tetracyclines, were tested. Each antibiotic test was performed three times. Specific doses of the antibiotics are shown in Table 2.
Petri dishes containing bacteria were incubated at 37 °C for 24 h. After incubation, the zones of inhibited growth were measured and placed on a three-stage scale in order to assess the bacterial resistance to the antibiotics: diameter of growth inhibition < 15 mm—bacterial resistance to antibiotic (R), diameter of growth inhibition between 16 and 25 mm—intermediate bacterial resistance to antibiotic (I), or diameter of growth inhibition > 25 mm—bacterial sensitivity to antibiotic (S).
Laboratory quality control (QC) was practiced during the studies to minimize the risk of error. A detailed description of QC can be found in Brągoszewska et al. 2016a, Brągoszewska et al. 2016b, and Brągoszewska et al. 2018.
Statistical analysis
Statistical analyses were performed using Statistica 12 (StatSoft Inc., Tulsa, USA) software. As the limit criterion of statistical significance assumed p value < 0.05. The concentration values were presented as mean values and standard deviations. Firstly, the distributions of assessed variables were checked with the Shapiro-Wilk test. The data were not normally distributed, and therefore a non-parametric method was employed. The Mann-Whitney U test was applied to assess office differences (AC or natural ventilation).
Results and discussion
Viable-culturable count (VCC) of bacterial aerosol
Exposure to bioaerosols has become a significant public health concern. However, no international standard is available specifying acceptable maximum bioaerosol levels in indoor environments (Mirhoseini et al. 2016). The first observation is that the mean average concentration of the bacterial aerosol in the indoor and outdoor environments differed significantly (Table 3). The level of viable-culturable count (VCC) of bacteria observed in the outdoor (325 CFU m−3) was even four times lower than the level recorded in indoor (from 540 to 1360 CFU m−3). These results indicate that the human activities are usually predominant source of indoor bacteria and agree well with the literature data. The second observation is that the mean average concentration of the bacterial aerosol in the naturally ventilated office rooms was about 1.5 times higher (1360 CFU m−3) than mean concentration of bacteria obtained in air-conditioned office rooms (540 CFU m−3), which indicates that the type of ventilation can be a very important element in shaping of microbiological IAQ. Therefore, in the studied office building, the AC system should be turned on several hours before work and shut down after employees have left.
Similar studies were conducted in four office buildings in Warsaw, Poland, and the results showed that the concentration of bacteria in the office rooms air was 500 CFU m−3 (Gołofit-Szymczak and Górny 2010). Results obtained in office buildings in Gliwice are analogous to the data found in studies carried out in air-conditioned office buildings in Hong Kong, where the mean concentration of airborne bacteria was 580 CFU m−3, with a maximum value of 2300 CFU m−3 (Mui et al. 2008). A significantly lower concentration level of bacterial aerosol was found in Turin, Italy, during the summer, where the average concentration of bacterial aerosol was 198 CFU m−3 and the maximum concentration was 368 CFU m−3 (Bonetta et al. 2010).
On the global scale, there is a lack of standards/guidelines for microbiological IAQ. The main reason for this is the limited well-documented dose-response relationships between the exposure to specific biological pollutants and adverse health effects caused by their exact dose. However, in Poland, proposals for regulatory levels of bacterial aerosols in residences and public buildings have been presented by Górny and Dutkiewicz (2002). A broad view of these proposals for public service buildings can be summarized as follows: 5000 CFU m−3 for mesophilic bacteria. The concentration levels of culturable bacteria obtained in our study were below this proposed standard.
The indoor-to-outdoor ratio (I/O) calculated for all indoor and outdoor bacteria concentrations was 2.97 for offices with AC and 4.19 for office with NV (Table 3). Since I/O ratio was greater than 1, it can clearly be concluded that the major sources of bacterial aerosol are internal sources and ventilation plays an important role in improving the IAQ (Srikanth et al. 2008; Bragoszewska et al. 2018). These results agree well with the literature data (Srikanth et al. 2008; Menteşe et al. 2009; Abosede Sarah 2017).
The Mann-Whitney U test confirmed significant differences between bacterial aerosol concentration in offices with natural ventilation and offices with AC, with p values < 0.05.
Particle size distribution and mass median aerodynamic diameter (MMAD) of airborne bacteria
The size distribution of a bacterial aerosol is a powerful tool for understanding how building design, occupancy, and operation affect human exposure to airborne particles (Nazaroff and Cass 1989). Modeling the dynamics of the airborne bacteria depends upon the community structure of bacteria, the age of the spores, and differences in spore aggregation rates, as well as the type of particles they are associated with, such as mist or particulate matter (Brągoszewska et al. 2017).
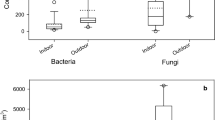
Figure 2 shows an example of the mean size distribution of bacterial aerosols in the outdoor air during our research (late spring season). Bacterial aerosols in the atmosphere reach a maximum concentration of bacteria in the air at diameters > 3.3 μm. Our previous measurements showed that in the atmospheric air (especially in spring and summer seasons), there is an interplay of two opposing mechanisms, i.e., the rapid growth of bacterial particles and a relatively higher mortality of fine particles, which are normally isolated bacterial cells. This results in the observed seasonal size distribution, characterized by a low share of fine particles (< 3.3 μm) and a relatively large share of coarse particles (> 3.3 μm) (Brągoszewska et al. 2017).
Figure 3 shows that small particles, less than 3.3 μm, are more prevalent in indoor air after 5–6 h of work, which means that the bacteria were present in the indoor air mainly as single cells. Results obtained for the indoor background indicate that peak of size distribution was shifted into larger particles, i.e., in the size > 3.3 μm.
The size distributions of bacterial aerosol in the studied office building may vary depending on the type of ventilation and human occupancy. Obtained results indicate that the air-conditioning was more effective in the elimination of respirable fraction of bacterial aerosol (< 3.3 μm) than natural ventilation. Trying to explain this phenomenon, it should be noted that the coarse fraction of bacteria particles remain airborne only for a short time and are removed from the air by gravity and resuspended again when disturbed mechanically. Therefore, ventilation is not a crucial factor in the removal of these particles. However, the behavior of fine particles is different, e.g., particle with aerodynamic diameter of 1 μm does not settle down during 2 h (Nevalainen et al. 1992).
A population of particles can be defined in terms of the mass carried in each particle size range. A measure called the mass median aerodynamic diameter (MMAD) essentially divides the distribution of the mass in half. Particles ≤ 3 μm are thought to have the highest probability of entering the lower airways of the average adult during oral inhalation (Council NR, 2006; Dolovich and Dhand 2011). The parameters of the mass distribution and mass median aerodynamic diameter (MMAD) are given by the following equation:
where
- MMAD:
-
is the mass median aerodynamic diameter,
- n i :
-
is the fraction in stage i, and
- d i :
-
is the cutoff diameter of stage i (Hinds 1999).
The maximum value of MMAD (2.8 μm) was found in the outdoor air. The minimum MMAD (2.2 μm) was found in the offices with natural ventilation (NV). Aerodynamic size diameter may vary depending on the ventilation and air velocity in the vicinity of microbial growth in the structures (Meklin et al. 2002) (Table 4).
Office building exposure dose
The US EPA (Environmental Protection Agency) encourages researchers to assess the risks of environmental exposure for all susceptible populations. The OBED has been calculated on the basis of the EPA’s Exposure Factors Handbook (U.S. EPA 2011) and other publications (Ott et al. 2006; Johnson-Restrepo and Kannan 2009). Total concentrations were used in the calculations of the inhaled dose of airborne bacteria. Although particles with an aerodynamic diameter of > 10 μm cannot usually be inhaled, their total contribution in suspended particles (< 100 μm) is not significant. The OBED is given by the following equation:
where
- OBED:
-
is the office building exposure dose for the indoor environment in CFU kg−1,
- C :
-
is the bacterial aerosol concentration in CFU m−3,
- IEF:
-
is the indoor exposure fraction—the hours spent per day in the office taking into account diverse activity patterns (on average, 8 h),
- IR:
-
is the inhalation rate coefficient characteristic of the selected activity levels, in m3/day (U.S. EPA 2011), and
- BW:
-
is the mean body weight in kg.
A staff time-budget survey was used to acquire information about staff members’ activities during the day. A summary of the information obtained from this questionnaire presented in Tables 5 and 6 shows the calculated results of the inhaled doses of culturable airborne bacteria.
It can be seen that a higher dose (OBED) of bacterial aerosol is inhaled by staff in offices with natural ventilation. However, the bacterial dose inhaled by staff in the studied office buildings is similar to the dose absorbed by the staff of a nursery school in Gliwice, Poland (70–206 CFU kg−1) (Brągoszewska et al. 2016b). The calculated dose absorbed by the staff of the office building is also comparable to the dose absorbed by adults in homes in Upper Silesia, Poland (175.4 CFU kg−1) (Brągoszewska 2014).
Identification of bacterial aerosol
The most abundant of isolated bacteria in our studies was Gram-positive bacteria. Only in the outdoor air Gram-negative bacteria were detected. In total, 12 species of bacteria were identified, with the most isolated Macrococcus equipercicus, Micrococcus luteus D, Staphylococcus xylosus in the indoor, and Bacillus species in the outdoor air (Table 7). The dominant group of bacteria isolated from atmospheric air is common in soil and water. In addition, microbes of this group can be found among the bacterial flora of the skin and mucous membranes of humans and animals. High variability in the virulence of Gram-positive rods forming endospores is known, as potential opportunistic pathogens can cause disease states only in persons with compromised host resistance (Aydogdu et al. 2010).
Similarly to the results obtained in previously published papers, our results also indicate the dominance of Gram-positive cocci in the indoor air (Pastuszka et al. 2000; Kim and Kim 2007; Stryjakowska-Sekulska et al. 2007; Menteşe et al. 2009; Dumała and Dudzińska 2013). The qualitative analysis of the microflora of the office building in Gliwice confirmed the results obtained by Gołofit-Szymczak and Górny (2010), where the air was analyzed in offices located in Warsaw. In both cases, the dominant species were Micrococcus luteus, Micrococcus spp., and Staphylococcus. Microorganisms isolated in our research belong to the most common bacteria in urban environments (offices, schools, residential buildings, and underground) that can be detected in the air (Fang et al. 2007). We can conclude that the indoor air of the examined office building does not create exposure to bioaerosol that may cause an immediate threat of acute health effects; however, prolonged inhalation of such doses of airborne bacteria may cause some adverse health effects, especially in sensitive individuals (Daisey et al. 2003). The level of microbial pollution inside the studied office building should be classified as safe, though some action to improve the indoor air quality is needed.
Based on the results presented in Table 7, it can be concluded that some microorganisms in the indoor air do not originate directly from the outdoor air bioaerosol. Observed differences can be explained by assuming that microorganisms were brought into the room by humans. In our research, Gemella haemolysans was found to be this type of microorganism. The results published by Eberhard et al. (2017) and Zhou et al. (2016) indicate that Gemella haemolysans belongs to the group of oral microbiota and could be spread by saliva particles in the room during conversations or other activities (Zhou et al. 2016; Eberhard et al. 2017).
Identification of Corynebacterium tuberculostearicum in a room with air-conditioning indicates the presence of people with sinusitis in this type of room (Copeland et al. 2018). These are diseases with unspecified etiology, often preceded by inflammation of the nasal mucosa. These bacteria may be absorbed as a result of mucus drying in rooms where the air is too dry (Gwaltney et al. 1981). This is often the case in air-conditioned rooms. The results obtained may confirm this assumption.
Multi-antibiotic resistance (MAR) test
Results of the antibiotic resistance testing (Table 8) are presented as the inhibition rate of the growth diameter around antimicrobial susceptibility test discs, with mean values for each antibiotic and tested strain (in mm).
The highest antibiotic resistance was for Gram-positive cocci, especially Macrococcus brunensis, isolated from the office with natural ventilation. The most antibiotic-sensitive bacterium was Bacillus cereus, except for the cephalosporins and monobactams. Of the all tested groups of antibiotics, the lowest antimicrobial activity toward the culturable bacteria isolated was observed for monobactams. None of the species were sensitive to these antibiotics. On the other hand, the most effective antibiotic was minocycline, which is a broad-spectrum tetracycline antibiotic commonly used in antibiotic treatment.
The results of antibiotic resistance testing of the isolated strains showed that all strains had resistance to more than 12% of the tested antibiotics. The strains were all resistant to metronidazole, aztreonam, and nalidixic acid. The strains were susceptible to teicoplanin except for Bacillus cereus which was resistant to this antibiotic. Many isolates were also resistant to high levels of nitrofurantoin, neomycin, and ceftazidime (83.3%). We observed multiple antibiotic resistance patterns from eight (Corynebacterium tuberculostearicum) to 35 antibiotics (Bacillus pseudomycoides). Corynebacterium tuberculostearicum is a dust-borne Corynebacterium found in households (Pakarinen et al. 2008) and is reported as one of the main constituents of the skin microbiome (Park et al. 2014). However, the pathogenic potential of C. tuberculostearicum was also found and confirmed (Abreu et al. 2012) as relating to childhood asthma and patients with chronic rhinosinusitis (Ege et al. 2012; Copeland et al. 2018). Bacillus pseudomycoides belongs to the group Bacillus cereus, which consists of six different closely related species (Ankolekar et al. 2009). Organisms in that group are common in samples of food, water, or soil, and some can cause human infections (Bavykin et al. 2008). Bacillus pseudomycoides can be found in dust and is ubiquitous in soil and water (Wilson et al. 2006). In our study, this strain was sensitive only to ciprofloxacin.
The evolution of antibiotic-resistant bacteria is a serious problem in modern medicine. It poses a serious threat to human health. In our studies, we suppose that one of the sources of antibiotic-resistant bacteria may be people residing in an indoor space. Results similar to ours have been reported in a study by Raisi (Raisi et al. 2013), where airborne microbial isolates were characterized by differentiated resistance to various antibiotics.
Summary and conclusions
Viable-culturable count (VCC) of the bacterial aerosol was evaluated in two types of offices air-conditioned (AC) or naturally ventilated (NV) in an office building in Gliwice, Poland. The microbiological IAQ also was made taking into consideration the outdoor concentrations of the bacterial aerosol.
It was found that the VCC of bacterial aerosol in the indoor was four times higher than outdoors and the indoor-to-outdoor ratio (I/O) was 2.97 for offices with AC and 4.19 for office with NV. These results indicate that human activity is a major factor in determining bacterial community structure.
We observed that the mean average concentration of bacterial aerosol in the natural ventilated office rooms was about 1.5 times higher than mean concentration of bacteria obtained in offices equipped with air-conditioning and was below Polish proposals for threshold limit values for bioaerosols in this type of working environment. The obtained results indicate that the type of ventilation is an important element in shaping of IAQ and suggest that efficient and regularly maintained air-conditioning systems can ensure a proper better hygienic air quality in work environment.
The size distribution of bacterial aerosol in NV office rooms is characterized by a larger share of particles less than 3.3 μm compared to results of size distribution obtained in offices with AC. Additionally, the mass median aerodynamic diameter (MMAD) of viable airborne bacteria was lower in natural ventilated offices (2.2 μm) than in offices with AC (2.4 μm).
The office building exposure dose (OBED) of bioaerosols was estimated for the personnel in this building. The highest value of OBED over the study period was obtained for staff working in offices with natural ventilation (141 CFU kg−1) compared to staff working in offices equipped with air conditioning (about 100 CFU kg−1). The problem that is becoming more and more serious is the emergence of multi-antibiotic-resistant bacteria. This issue is most often addressed in the context of nosocomial infections, but office rooms can also be a place where these types of microorganisms begin to appear. Currently, antibiotics are used very often in homes without the need for hospitalization. What is more, adults who use drugs still participate in professional life, so they can also transfer resistant microorganisms to the office environment. Gram-positive cocci were the most frequently occurring bacteria in the indoor, with the most isolated genus: Macrococcus equipercicus, Micrococcus luteus D, and Staphylococcus xylosus. Gram-positive rods were the most frequently isolated bacterial isolates in the outdoor. Multi-antibiotic resistance (MAR) tests showed that among the airborne bacteria isolated, the most antibiotic-resistant features were present in Macrococcus species (especially Macrococcus equipercicus and Macrococcus brunensis).
The results obtained in this study indicate that the studied office exposure to airborne bacteria is generally low and safe. However, it is recommended that providing appropriate indoor air office conditions is an important task in order to ensure high productivity of workers. We believe that the results of the current study point to the need for implementing a strategy to control microbiological IAQ in offices. The development and implementation of guidelines for harmful indoor microbiological air pollutants is also recommended.
References
Abosede Sarah A (2017) Indoor and outdoor concentrations of bioaerosols and meteorological conditions of selected salons in four areas of Ibadan North local government area. Int J Environ Mon Analys 5:83. https://doi.org/10.11648/j.ijema.20170503.13
Abreu NA, Nagalingam NA, Song Y, Roediger FC, Pletcher SD, Goldberg AN, Lynch SV (2012) Sinus microbiome diversity depletion and Corynebacterium tuberculostearicum enrichment mediates rhinosinusitis. Sci Transl Med 4:151ra124. https://doi.org/10.1126/scitranslmed.3003783
Ankolekar C, Rahmati T, Labbé RG (2009) Detection of toxigenic Bacillus cereus and Bacillus thuringiensis spores in U.S. rice. Int J Food Microbiol 128:460–466. https://doi.org/10.1016/j.ijfoodmicro.2008.10.006
Aydogdu H, Asan A, Tatman Otkun M (2010) Indoor and outdoor airborne bacteria in child day-care centers in Edirne City (Turkey), seasonal distribution and influence of meteorological factors. Environ Monit Assess 164:53–66. https://doi.org/10.1007/s10661-009-0874-0
Bavykin SG, Mikhailovich VM, Zakharyev VM, Lysov YP, Kelly JJ, Alferov OS, Gavin IM, Kukhtin AV, Jackman J, Stahl DA, Chandler D, Mirzabekov AD (2008) Discrimination of Bacillus anthracis and closely related microorganisms by analysis of 16S and 23S rRNA with oligonucleotide microarray. Chem Biol Interact 171:212–235. https://doi.org/10.1016/j.cbi.2007.09.002
Bernstein JA, Alexis N, Bacchus H et al (2008) The health effects of nonindustrial indoor air pollution. J Allergy Clin Immunol 121:585–591. https://doi.org/10.1016/j.jaci.2007.10.045
Błaszczyk E, Rogula-Kozłowska W, Klejnowski K, Kubiesa P, Fulara I, Mielżyńska-Švach D (2017) Indoor air quality in urban and rural kindergartens: short-term studies in Silesia, Poland. Air Qual Atmos Health 10:1207–1220. https://doi.org/10.1007/s11869-017-0505-9
Bonetta S, Bonetta S, Mosso S, Sampò S, Carraro E (2010) Assessment of microbiological indoor air quality in an Italian office building equipped with an HVAC system. Environ Monit Assess 161:473–483. https://doi.org/10.1007/s10661-009-0761-8
Brągoszewska E, Mainka A, Pastuszka JS, et al (2018) Assessment of bacterial aerosol in a preschool, primary school and high school in Poland. Atmosphere 9. doi: https://doi.org/10.3390/atmos9030087
Brągoszewska E, Mainka A, Pastuszka JS (2017) Concentration and size distribution of culturable bacteria in ambient air during spring and winter in Gliwice: a typical urban area. Atmosphere. 8. https://doi.org/10.3390/atmos8120239
Brągoszewska E (2014) Bacterial aerosol occuring in the atmospheric air in Gliwice and its share of the total human exposure to the bacteria absorbed by inhalation—PhD Thesis
Brągoszewska E, Mainka A, Pastuszka J (2016a) Bacterial and fungal aerosols in rural nursery schools in Southern Poland. Atmosphere 7:142. https://doi.org/10.3390/atmos7110142
Brągoszewska E, Mainka A, Pastuszka JS (2016b) Bacterial aerosols in an urban nursery school in Gliwice, Poland: a case study. Aerobiologia 32:469–480. https://doi.org/10.1007/s10453-015-9419-x
Brasche S, Bischof W (2005) Daily time spent indoors in German homes - Baseline data for the assessment of indoor exposure of German occupants. Int J Hyg Environ Health 208:247–253. https://doi.org/10.1016/j.ijheh.2005.03.003
Copeland E, Leonard K, Carney R, Kong J, Forer M, Naidoo Y, Oliver BGG, Seymour JR, Woodcock S, Burke CM, Stow NW (2018) Chronic rhinosinusitis: potential role of microbial dysbiosis and recommendations for sampling sites. Front Cell Infect Microbiol 8. https://doi.org/10.3389/fcimb.2018.00057
Council NR (2006) Overcoming challenges to develop countermeasures against aerosolized bioterrorism agents. National Academies Press, Washington, D.C.
Daisey JM, Angell WJ, Apte MG (2003) Indoor air quality, ventilation and health symptoms in schools: an analysis of existing information. Indoor Air 13:53–64. https://doi.org/10.1034/j.1600-0668.2003.00153.x
Di Giulio M, Grande R, Di Campli E et al (2010) Indoor air quality in university environments. Environ Monit Assess 170:509–517. https://doi.org/10.1007/s10661-009-1252-7
Dolovich MB, Dhand R (2011) Aerosol drug delivery: developments in device design and clinical use. Lancet 377:1032–1045
Dumała SM, Dudzińska MR (2013) Microbiological indoor air quality in Polish schools. Ann Set Environ Protect 15:231–244
Eberhard J, Stumpp N, Winkel A, Schrimpf C, Bisdas T, Orzak P, Teebken OE, Haverich A, Stiesch M (2017) Streptococcus mitis and Gemella haemolysans were simultaneously found in atherosclerotic and oral plaques of elderly without periodontitis—a pilot study. Clin Oral Investig 21:447–452. https://doi.org/10.1007/s00784-016-1811-6
Ege MJ, Mayer M, Schwaiger K, Mattes J, Pershagen G, van Hage M, Scheynius A, Bauer J, von Mutius E (2012) Environmental bacteria and childhood asthma. Allergy: European J Allergy Clin Immunol 67:1565–1571. doi: https://doi.org/10.1111/all.12028
Fang Z, Ouyang Z, Zheng H, Wang X, Hu L (2007) Culturable airborne bacteria in outdoor environments in Beijing, China. Microb Ecol 54:487–496. https://doi.org/10.1007/s00248-007-9216-3
Gawrońska H, Bakera B (2015) Phytoremediation of particulate matter from indoor air by Chlorophytum comosum L. plants. Air Qual Atmos Health 8:265–272. https://doi.org/10.1007/s11869-014-0285-4
Gizaw Z, Gebrehiwot M, Yenew C (2016) High bacterial load of indoor air in hospital wards: the case of University of Gondar teaching hospital, Northwest Ethiopia. Multidiscip Respir Med 11:24. https://doi.org/10.1186/s40248-016-0061-4
Gołofit-Szymczak M, Górny RL (2010) Bacterial and fungal aerosols in air-conditioned office buildings in Warsaw, Poland—the winter season. Int J Occup Saf Ergon 16:465–476
Górny RL, Dutkiewicz J (2002) Bacterial and fungal aerosols in indoor environment in Central and Eastern European countries. Ann Agric Environ Med : AAEM 9:17–23
Gwaltney J, Sydnor J, Sande MA (1981) Etiology and antimicrobial treatment of acute sinusitis. Ann Otol Rhinol Laryngol 90:68–71
Hewitt KM, Gerba CP, Maxwell SL, Kelley ST (2012) Office space bacterial abundance and diversity in three metropolitan areas. PLoS One 7:e37849. https://doi.org/10.1371/journal.pone.0037849
Hinds WC (1999) Aerosol technology: properties, behavior, and measurement of airborne particles
Johnson-Restrepo B, Kannan K (2009) An assessment of sources and pathways of human exposure to polybrominated diphenyl ethers in the United States. Chemosphere 76:542–548. https://doi.org/10.1016/j.chemosphere.2009.02.068
Karottki D, Spilak M, Frederiksen M, Jovanovic Andersen Z, Madsen A, Ketzel M, Massling A, Gunnarsen L, Møller P, Loft S (2015) Indoor and outdoor exposure to ultrafine, fine and microbiologically derived particulate matter related to cardiovascular and respiratory effects in a panel of elderly urban citizens. Int J Environ Res Public Health 12:1667–1686. https://doi.org/10.3390/ijerph120201667
Kim KY, Kim CN (2007) Airborne microbiological characteristics in public buildings of Korea. Build Environ 42:2188–2196. https://doi.org/10.1016/j.buildenv.2006.04.013
Klepeis NE, Nelson WC, Ott WR, et al (2001) The National human activity pattern survey (NHAPS): a resource for assessing exposure to environmental pollutants. J Expo Anal Environ Epidemiol 11:231–252. https://doi.org/10.1038/sj.jea.7500165
Kotzias D (2005) Indoor air and human exposure assessment—needs and approaches. Exp Toxicol Pathol 57:5–7. https://doi.org/10.1016/j.etp.2005.05.002
Latif MT, Yong SM, Saad A, Mohamad N, Baharudin NH, Mokhtar MB, Tahir NM (2014) Composition of heavy metals in indoor dust and their possible exposure: a case study of preschool children in Malaysia. Air Qual Atmos Health 7:181–193. https://doi.org/10.1007/s11869-013-0224-9
Law AKY, Chau CK, Chan GYS (2001) Characteristics of bioaerosol profile in office buildings in Hong Kong. Build Environ 36:527–541. https://doi.org/10.1016/S0360-1323(00)00020-2
Lee S, Chang M (2000) Indoor and outdoor air quality investigation at schools in Hong Kong. Chemosphere 41:109–113. https://doi.org/10.1016/S0045-6535(99)00396-3
Meklin T, Reponen T, Toivola M, Koponen V, Husman T, Hyvärinen A, Nevalainen A (2002) Size distributions of airborne microbes in moisture-damaged and reference school buildings of two construction types. Atmos Environ 36:6031–6039. https://doi.org/10.1016/S1352-2310(02)00769-0
Menteşe S, Arisoy M, Rad AY, Güllü G (2009) Bacteria and fungi levels in various indoor and outdoor environments in Ankara, Turkey. Clean - Soil Air Water 37:487–493. https://doi.org/10.1002/clen.200800220
Mirhoseini SH, Nikaeen M, Satoh K, Makimura K (2016) Assessment of airborne particles in indoor environments: applicability of particle counting for prediction of bioaerosol concentrations. Aerosol Air Qual Res 16:1903–1910. https://doi.org/10.4209/aaqr.2015.08.0528
Moon KW, Huh EH, Jeong HC (2014) Seasonal evaluation of bioaerosols from indoor air of residential apartments within the metropolitan area in South Korea. Environ Monit Assess 186:2111–2120. https://doi.org/10.1007/s10661-013-3521-8
Mui KW, Wong LT, Hui PS (2008) Risks of unsatisfactory airborne bacteria level in air-conditioned offices of subtropical climates. Build Environ 43:475–479. https://doi.org/10.1016/j.buildenv.2007.01.012
Nasir ZA, Colbeck I (2010) Assessment of bacterial and fungal aerosol in different residential settings. Water Air Soil Pollut 211:367–377. https://doi.org/10.1007/s11270-009-0306-3
Nazaroff WW (2016) Indoor bioaerosol dynamics. Indoor Air 26:61–78. https://doi.org/10.1111/ina.12174
Nazaroff WW, Cass GR (1989) Mathematical modelling of indoor aerosol dynamics. Environ Sci Technol 23:157–166
Nevalainen A, Pastuszka J, Liebhaber F, Willeke K (1992) Performance of bioaerosol samplers: collection characteristics and sampler design considerations. Atmos Environ Part A 26:531–540. https://doi.org/10.1016/0960-1686(92)90166-I
Nevalainen A, Willeke K, Liebhaber F, et al (1993) Bioaerosol sampling. In: Willeke K, Baron P Van Nostrand Reinhold (eds) Aerosol measurement: principles, techniques and applications., New York, pp 471–492
Ott WR, Steinemann AC, Wallace LA (2006) Exposure analysis, CRC Press, London. CRC Press, London
Pakarinen J, Hyvärinen A, Salkinoja-Salonen M, Laitinen S, Nevalainen A, Mäkelä MJ, Haahtela T, von Hertzen L (2008) Predominance of Gram-positive bacteria in house dust in the low-allergy risk Russian Karelia. Environ Microbiol 10:3317–3325. https://doi.org/10.1111/j.1462-2920.2008.01723.x
Park HK, Han JH, Joung Y, Cho SH, Kim SA, Kim SB (2014) Bacterial diversity in the indoor air of pharmaceutical environment. J Appl Microbiol 116:718–727. https://doi.org/10.1111/jam.12416
Pastuszka JS, Kyaw Tha Paw U, Lis DO, Wlazło A, Ulfig K (2000) Bacterial and fungal aerosol in indoor environment in Upper Silesia, Poland. Atmos Environ 34:3833–3842. https://doi.org/10.1016/S1352-2310(99)00527-0
Pastuszka JS, Marchwińska-Wyrwał E, Wlazło A (2005) Bacterial aerosol in Silesian hospitals: preliminary results. Pol J Environ Stud 14:883–890
Pegas PN, Evtyugina MG, Alves CA, Nunes T, Cerqueira M, Franchi M, Pio C, Almeida SM, Freitas MC (2010) Outdoor/indoor air quality in primary schools in Lisbon: a preliminary study. Quim Nova 33:1145–1149
Raisi L, Aleksandropoulou V, Lazaridis M, Katsivela E (2013) Size distribution of viable, cultivable, airborne microbes and their relationship to particulate matter concentrations and meteorological conditions in a Mediterranean site. Aerobiologia 29:233–248. https://doi.org/10.1007/s10453-012-9276-9
Salleh NM, Kamaruzzaman SN, Sulaiman R, Mahbob NS (2011) Indoor air quality at school: ventilation rates and it impacts towards children—a review. 2nd International Conference on Environmental Science and Technology 6:vol.6, 418–422
Shin S-K, Kim J, Ha S, Oh HS, Chun J, Sohn J, Yi H (2015) Metagenomic insights into the bioaerosols in the indoor and outdoor environments of childcare facilities. PLoS One 10:1–17. https://doi.org/10.1371/journal.pone.0126960
Srikanth P, Sudharsanam S, Steinberg R (2008) Bio-aerosols in indoor environment: composition, health effects and analysis. Indian J Med Microbiol 26:302–312. https://doi.org/10.4103/0255-0857.43555
Stryjakowska-Sekulska M, Piotraszewska-Pajak A, Szyszka A et al (2007) Microbiological quality of indoor air in university rooms. Pol J Environ Stud 16:623–632. https://doi.org/10.12980/APJTB.4.2014C807
Tringe SG, Zhang T, Liu X, et al (2008) The airbone metagenome in an indoor urban environment. PLoS ONE. https://doi.org/10.1371/journal.pone.0001862
Ashmore MR, Dimitroulopoulou C (2009) Personal exposure of children to air pollution. Atmos Environ 43:128–141. https://doi.org/10.1016/j.atmosenv.2008.09.024
U.S. EPA (2011) Exposure factors handbook; EPA, Environmental Protection Agency
Watson AY, Bates RR, Kennedy D (1988) Assessment of human exposure to air pollution: methods, measurements, and models. Air Pollution, the Automobile, and Public Health, National Academy Press, Washington
Wichmann J, Lind T, Nilsson MAM, Bellander T (2010) PM2.5, soot and NO2 indoor-outdoor relationships at homes, pre-schools and schools in Stockholm, Sweden. Atmos Environ 44:4536–4544. https://doi.org/10.1016/j.atmosenv.2010.08.023
Wilson C, Brigmon RL, Knox A, Seaman J, Smith G (2006) Effects of microbial and phosphate amendments on the bioavailability of lead (Pb) in shooting range soil. Bull Environ Contam Toxicol 76:392–399. https://doi.org/10.1007/s00128-006-0934-x
Wood RA, Burchett MD, Orwell RA, et al (2002) Plant/soil capacities to remove harmful substances from polluted indoor air
Zhou Y, Yang J, Zhang L, Zhou X, Cisar JO, Palmer RJ Jr (2016) Differential utilization of basic proline-rich glycoproteins during growth of oral bacteria in saliva. Appl Environ Microbiol 82:5249–5258. https://doi.org/10.1128/AEM.01111-16
Funding
This work was supported by the Faculty of Power and Environmental Engineering, Silesian University of Technology (statutory research).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Brągoszewska, E., Biedroń, I., Kozielska, B. et al. Microbiological indoor air quality in an office building in Gliwice, Poland: analysis of the case study. Air Qual Atmos Health 11, 729–740 (2018). https://doi.org/10.1007/s11869-018-0579-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11869-018-0579-z