Abstract
Purpose
The Deonggae coast on Jeju Island is a unique habitat that has not yet been studied from an ecological management or conservation perspective, despite its importance as the northernmost habitat of subtropical plants in Asia and the sole habitat of an endangered fern, Thelypteris interrupta, in Korea. To provide insights into this habitat’s systematic management, we comprehensively characterized its abiotic and biotic components.
Methods
Our study found two distinct plant communities (Persicaria japonica-Thelypteris interrupta and Thelypteris interrupta-Phragmites australis), driven by their respective microhabitats and influenced by soil inundation frequency, water depth, and salinity. We examined the phytosociological similarities and habitat characteristics of these two communities.
Results
Thelypteris interrupta could be distributed in the northernmost part of the Deonggae coast due to its greater heat preference. However, this limits its spread toward polar regions. We suggested minimizing the transformation of wetlands into terrestrial habitats by reducing the introduction of external soils and establishing ecological continuity with neighboring ecosystems while educating visitors about the area’s ecology as two habitat conservation practices.
Conclusion
The Deonggae coast is a natural wetland with a high conservation value as a habitat for endangered wildlife and a refuge for opportunistic species under the influence of climate change.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Korea is a peninsula on the eastern end of the Eurasian continent. Owing to the range of latitudes it stretches across (33–38°), Korea hosts a wide array of climatic zones, including those representing subtropical, temperate, and subarctic climates (Kim 1992; Kong and Paul 1993). This region is characterized by an alternation in the predominance of oceanic and continental climatic systems. For example, the temperate climatic conditions that characterize this island are driven by various warm currents (e.g., Tsushima current, West Korea Coastal current, Yellow Sea current, etc.) (Zhang et al. 1997). In the Korean Peninsula, warm temperatures and the associated subtropical plants are restricted to the southern part of the peninsula and the southern islands; these regions are under the influence of Jeju warm current and the oceanic climate (Choi 2013). Jeju Island, the southernmost island of the Korean Peninsula, has the warmest climate in this region. Its climate its largely shaped by the North Pacific warm current, which facilitates the influx of subtropical plants to the Korean Peninsula.
Due to global climate change, warm temperatures and the subtropical climate normally associated with the southern region of the Korean Peninsula are intensifying. Such changes facilitate the opportunistic spread of plants that prefer warm temperatures beyond their current distributional ranges (Houle 1994; Walck et al. 2011). Temperature and precipitation tend to be the most crucial factors in the distribution of plants (Woodward 1987), as they greatly impact seed germination, growth, and the survival of populations. Therefore, within the context of ongoing global warming, major changes in the current distribution of plants across the are certain (IPCC 2007), especially over the next few decades (Sykes and Prentice 1995; Thuiller et al. 2008).
Thelypteris interrupta is a cosmopolitan species of subtropical ferns that are distributed in tropical and subtropical regions across five continents. The distribution of this species in South Korea has been only confirmed on Jeju Island (International Union for Conservation of Nature 2001; Hemp 2002; Iksan and Aba 2019), which is the northernmost subtropical habitat in east Asia (GBIF 2021). The presence of T. interrupta on Jeju Island (i.e., specifically on the Deonggae coast) was first recorded in 2000. The Deonggae coast is the only known natural habitat of Thelypteris interrupta in Korea (Moon et al. 2002). In response to the rarity of this plant and its small distribution in South Korea, the Ministry of Environment outlined a goal to protect T. interrupta (i.e., by classifying it as an endangered plant) and its habitat (International Union for Conservation of Nature 2001; NIE 2021). However, the ecological dynamics of the habitat of T. interrupta in Korea have not been fully elucidated, to allow for the creation and implementation of robust conservation initiatives.
Ferns have a worldwide distribution due to the great dispersal ability of their small and light spores (Tryon and Tryon 1982; Tryon 1986). However, since ferns are subtropical species, their settlement is affected more by environmental conditions than dispersal ability (Tryon 1972, 1986; Barrington 1993). Specifically, the diversity and abundance of ferns are associated with the availability of water (Kessler et al. 2011). Therefore, climate changes within the context of temperature and precipitation systems may greatly alter the distribution of ferns.
Within the context of South Korea, because T. interrupta is now scattered near the northern boundary of the study area, its sensitivity to environmental disturbances is higher than in its normal habitat in this region. Thus, a thorough examination of the ecological dynamics of the habitat is critical for resolving basic issues in the preservation and management of the habitat of T. interrupta. For example, the study of vascular plant diversity in the habitat of T. interrupta is especially important for a quantitative and qualitative evaluation of plant diversity as a bioindicator of the overall health of the habitat, as well as estimating the environmental impact of the introduction of alien species (Bried et al. 2014; DeBerry et al. 2015; Maginel et al. 2016; Jog et al. 2017).
Therefore, the aim of this study was to characterize the phytosociological characteristics and distribution of the habitat of T. interrupta in Korea. Additionally, we also aimed to yield an accurate assessment of the importance of this habitat within the context of resource provisioning and conservation. Finally, based on our findings, we recommend ecological management measures to ensure proper maintenance of the habitat and its preservation.
Materials and methods
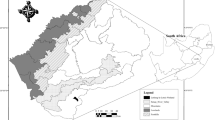
The study area was on the Deonggae coast (33°33ʹ50ʹʹN, 126°45ʹ52ʹʹE) in Gimnyeong-ri, Gujaw-eup, Jeju City, Jeju Special Self-Governing Province (Fig. 1). In terms of the geology of the habitat, it is a quaternary basalt area of the Cenozoic (KIGAM 2022). This area is a rainfall-dependent wetland that is formed in a depression of the lava zone near the coast. Additionally, the Deonggae coast is on the northwestern coast of Jeju Island, which can be directly or indirectly affected by the Kuroshio warm current. Besides the presence of T. interrupta on the coast of the study area, the area is an important phytogeographic site that hosts a wide array of other subtropical plants, namely Hibiscus hamabo, Paliurus ramosissimus, and Crinum asiaticum (Fukui 1977; Choi 2013). The study site had an area of 60 × 40 m with a depression of 4 m. Although the study site had fine-grained soil due to the deposition of basalt bedrock, the soil was not deep, having a depth of only around 15 cm. The average salinity of the study site ranged from 0.19–0.22‰.
Study area. Blue circles show distribution information for Thelypteris interrupta obtained from GBIF (2021). Black box of (A): Jeju-do, Red circle of (B): Study area and (C) is the study site landscape
Vegetation survey
A field vegetation survey was conducted using the traditional Braun-Blanquet method of the Zürich-Montpellier school. This method is based on the phytosociological composition of species (Westhoff and van der Maarel 1978; Kim and Lee 2006). The survey sites for community classification were determined in consideration of the homogeneity of the habitat and the uniformity of the component species. All relevés dominated by T. interrupta (i.e., at least 50% cover in the layer) were selected from the Deonggae coast in Jeju Island. Overall, 12 relevés were obtained. The minimum area was determined according to Dierschke (1994) and relevé sizes average 4 m2 with minimum and maximum values of 3 m2 and 25 m2, respectively. However, a few small plots (2 m2) representing different community types were also included. The survey area was determined to be appropriate to prevent omissions of plant species and vegetation information. The coverage classes of the plant species in the survey area were examined using the combined cover degree of 9 classes: 1: 0.02 ~ 0.1%, 2: 0.1 ~ 2.5%, 3: 2.5 ~ 5%, 4: 5%, 5: 6 ~ 12.5%, 6: 12.5 ~ 25% 7: 26 ~ 50%, 8: 51 ~ 75%, 9: 76 ~ 100% (Van der Maarel 1979). The vegetation survey chart details all the environmental and ecological characteristics associated with the respective communities.
The communities were classified using a hybrid sorting method that simultaneously considers the conventional classification method (Braun-Blanquet 1932; Becking 1957) and numerical syntaxonomy (Mucina and Dale 1989). Regarding the component species, repeated interspecies comparisons were performed to derive the diagnostic group (Chytrý et al. 2002) that reflects vegetation representativeness, and the syntaxa were subsequently recorded on the final classification chart (Table 1). The vegetation communities were defined by sociological species groups including co-occurrence of species (i.e., species under similar ecological conditions that have a similar distribution). For each syntaxon, we quantified the validity of the elucidated vegetation units by calculating the relative net-contribution degree (r-NCD) and percentage net-contribution degree (P-NCD) of the emerging species (Kim and Manyko 1994; Eom 2011). The formulae that were used are:
where ΣCi is a sum of coverage of species i,N is the total number of relevés, ni is the number of relevés with species i, and NCDmax is the maximum NCD value of a certain species for a synthesized vegetation unit.
where NCDtotal is the sum of NCD value of all identified species.
The syntaxa were named in accordance with the international vegetation naming conventions (Theurillat et al. 2020). For each syntaxon, vegetation information such as synecology, syngeography, and syndynamics was described. To characterize and analyze the environmental conditions, the habitat conditions (light, soil moisture, soil nutrients, soil particle, salinity, salt exposure, slope, water depth, etc.) of the location were closely observed and recorded during the field vegetation survey. Salinity was measured using the HI-5522 multi-item water quality meter, and the relative values for wetland points including seawater were compared twice (in May and August).
Mathematical statistical analysis of syntaxa
To analyze factors that shape the distribution of classified vegetation, data of component species were used as input in the software JUICE 7.1 (Tichý 2002). Considering the properties of the values in the vegetation survey chart, the analysis was performed using non-metric multidimensional scaling (NMDS), and the evaluation of significance and correlation analyses were performed using r-NCD.
Survey of vascular flora in the surrounding area
The vascular flora around the habitat of T. interrupta were investigated to determine the status of the habitat (Fig. 2). The scope of the investigation was a radius of 10 m based on T. interrupta samples in the outermost areas, but habitats cut off by coastal roads were excluded. For an accurate identification of species that were not identified in the field, samples with reproductive systems were collected. For some species that could not be physically sampled, their images were captured. Every plant sample was finally given a collection number and coordinates. The evidence samples were preserved in the herbarium of the Temperate and Subtropical Forest Research Institute. The list of vascular plants was created via Engler’s classification system, and the scientific names adhered to the checklist of vascular plants in Korea (KNA 2021).
Results and discussion
Phytosociological characteristics
The plant communities in which T. interrupta occurs were distributed within 200 m from the coast, forming perennial wet herbaceous vegetation. These communities consisted of 13 families, 17 genera, 14 species and 3 variants. Overall, the communities were divided into two main groups, namely, the Persicaria japonica-T. interrupta and the T. interrupta-Phragmites australis communities. This division was made based on the species composition and habitat characteristics (Table 1; Fig. 2).
The P. japonica-T. interrupta community mostly consisted of T. interrupta and P. japonica. Most species in this community were weeds and river-type herbaceous vegetation (Lee 2005), namely, P. japonica (35.39), Humulus scandens (5.90), Juncus decipiens (2.25), Murdannia keisak (0.37), and Rubia cordifolia (0.09). The same trends were observed in the T. interrupta community of the Philippines, Vietnam, Egypt, and South Africa. The formation of a community with weed vegetation on farmlands and papyrus farmlands is a typical example (Kingston et al. 2004; Sieben et al. 2016a, b; Fried et al. 2018). The salinity of the habitat of the P. japonica- T. interrupta community, which is mainly distributed in the periphery of wetlands was 0.19‰, which is close to that of freshwater. This location experiences temporary inundation only during rainfall and maintains a moist soil environment that is not normally submerged in water.
The T. interrupta-P. australis community consists of a two-layered vegetation structure; specifically, it consists of tall grass (with a vegetation height of approximately 1.5 m) and a short grass (with a vegetation height of 0.4 m). The community diagnostic species include P. australis, Carex dimorpholepis, and Lythrum salicaria. Among them, P. australis has the highest r-NCD, and T. interrupta has the second-highest r-NCD. The T. interrupta-P. australis community occurs on gentle slope with a sedimentary layer. Additionally, this location has a stable water level throughout the year, a salinity of 0.22‰, and a soil depth of 50 cm to 1 m. The community is dominated by P. australis; its dominance lies in the fact that it is taller and grows faster than T. interrupta. Similar trends linked to the T. interrupta-P. australis community being associated with tall grass has been reported in India (Kalita 2019; Kalita and Sarma 2019), South Africa (Lötter et al. 2014), and Australia (Sinclair et al. 2012). However, most of these plants (T. interrupta-P. australis community) form communities in marsh plains or wetlands in the background of rivers. Thus, their habitat is greatly differentiated from that of species that grow wild in small wetlands cut off from groundwater in the coastal lava areas of Jeju Island.
In a terrestrialized location around lava rocks in the T. interrupta-P. australis community, species such as Miscanthus sinensis, Lespedeza cuneata, and Imperata cylindrica, which are component species of dry secondary grassland, can be introduced as well. After such species of xerophytic plants have settled, the characteristics of wetland vegetation are greatly weakened by natural succession. We found that terrestrialization progressed as dry herbaceous vegetation and woody plants, such as Rosa multiflora, Ligustrum obtusifolium, and Ulmus parvifolia established.
NMDS analysis for coastal vegetation and T. interrupta communities (P. japonica- T. interrupta community and T. interrupta-P. australis community) on Jeju Island
Phytosociological similarities and habitat characteristics were investigated through the coordinate determination method based on the species composition of the T. interrupta communities and coastal brackish and salt marsh vegetation (200 relevés, 84 species) (Ohba and Sugawara 1979; Lee 2020). This application of this method was achieved as a result of habitat–species composition correspondence (Fig. 3). Overall, we found that the most important environmental factor shaping the distribution of species in the first axis was salt exposure. We also found that the major environmental factors in the second axis were soil particles and water saturation. The T. interrupta community was distributed in a location where the salinity was lower than that of the seven syntaxa that appeared in the salt marsh and brackish zone. More typical coastal vegetations determined by salinity (average salinity: 25.05‰) include Zoysietum sinicae and Limonietum tetragoni. The T. interrupta community was close to the coast, an area in which the influence of salinity was greater. However, it still hosted species that commonly occurred in plant communities inhabiting environments of a lower salinity than that of Phragmitetum australis, Caricetum scabrifoliae, and Phaceluretum latifolii, representing the brackish zone. Compared to the Phragmitetum australis and Caricetum scabrifoliae representing the brackish zone, the T. interrupta-P. australis community showed a coordinate determination characteristic of allocation with lower salinity.
Syntaxa diversity and NMDS analysis of coastal vegetation on Jeju Island (i. Persicaria japonica-Thelypteris interrupta community, ii. Thelypteris interrupta-Phragmites communis community, iii. Phragmitetum australis Savič 1926, iv. Caricetum scabrifoliae Miyawaki et Ohba 1969, v. Phaceluretum latifolii Miyawaki et Okuda 1972, vi. Limonietum tetragoni Miyawaki et Ohba 1969, vii. Artemisietum fukudo Miyawaki et Ohba 1969, viii. Zoysietum sinicae Miyawaki et Ohba 1969, ix. Juncus gracillimus community)
The two plant communities consisting of T. interrupta showed the highest similarity to the P. australis community. In particular, the T. interrupta-P. australis community showed a very high similarity in component species with the P. australis community except for T. interrupta. The Phragmitetum australis is a wet vegetation that is associated with slow flowing fresh and brackish waters. As a tall-grass vegetation, it is dominated by P. australis, and significant differences in the number of emerging species in the community exist depending on the salinity and the frequency of water inundation (Kim 2001; Lee et al. 2003; Lee 2011). The similarity between the T. interrupta- P. australis community and the P. australis community appears to be due to the occurrence of tall-grass vegetation in a low-lying wetland environment. This is due to the fact that the habitat of the T. interrupta-P. australis community is a brackish environment that has been formed by the inflow of irregular seawater, with a water level that is maintained stably from a concave terrain surrounded by lava rocks. In contrast, the P. japonica-T. interrupta community showed a relatively low similarity with the plant community in the brackish zone. This community is at the periphery compared to the T. interrupta-P. australis community, and the water level fluctuates irregularly due to rainfalls or seawater flowing in from large waves. As a result, annual and biennial plants appeared frequently in the location, and the species composition showed low different largely due to these reasons.
The T. interrupta communities (T. interrupta-P. australis community and P. japonica-T. interrupta community) identified in this study showed differences in species composition from the previously reported coastal vegetations on Jeju Island (Kang 2001; Cheong et al. 2013). This indicates that the vegetation by species composition in the rocky coastal habitat of Jeju Island that is restricted to the northernmost parts of this island. Jeju Island has reported approximately 40 new subtropical species over the last 10 years. Some of them still form a community with a low degree of contribution due to the inflow of a small number in warm temperate vegetation. However, some of them, such as T. interrupta, are forming a unique vegetation correlation with a high degree of component species contribution. Thus, it can be predicted that on Jeju Island, vegetation types with subtropical plants as major component species, would increase with the continuous spread of subtropical plants in Korea (Choi 2013).
In general, the geographic factors limiting the distributional expansion of plants toward the poles are more sensitive to climate change because the species in the northernmost part of this island are already inhabiting conditions representing the extreme end of what they can survive (Hampe and Petit 2005). Due to such ecological limits, newly settled species have relatively low plant productivity and population competitiveness under these relatively disadvantageous climatic conditions (Case and Taper 2000), and this will inevitably limit the expansion of T. interrupta. A study has found that the expansion of a population in the periphery of distribution is controlled by low temperature in a temperate region, whereas the equatorial region is affected by the combination of droughts and high temperatures (Hampe and Petit 2005). In this study, the expansion of the T. interrupta community is expected to be limited by the intermittent cold waves. This is because this community is in the transition zone between temperate and subtropical zones. However, the community is expected to also be affected by droughts and high temperature as well because it is distributed in a wetland based on basalt; this area is characterized by high temperatures and droughts. This appears to be a third type that is different from the temperature rise and changes in precipitation patterns due to climate change influencing the expansion or reduction of the distribution of species in contrasting forms in the two limiting regions of temperate and tropical climate conditions (Matías and Jump 2012).
Vascular flora and species distribution
The flora inhabiting the habitat of T. interrupta were found to comprise 175 taxa, including 61 families, 146 genera, 155 species, 19 variants, and 1 variety (Table 2). Among species considered endangered by the Ministry of Environment, there are two species, that is, T. interrupta and Hibiscus hamabo. In terms of plants rare plants and those on the Red List of the Ministry of Environment National, there were 10 species: T. interrupta, Potentilla discolor, Canavalia lineata, Paliurus ramosissimus, Hibiscus hamabo, Vincetoxicum floribundum, Wollastonia dentata, Lilium callosum, and Crinum asiaticum. As for naturalized plants, nine families, 14 genera, 16 species, and these were identified, including Bromus catharticus, Conyza bonariensis, and Silene gallica. Among them, four species were observed as ecosystem disturbance plants designated by the Ministry of Environment: Hypochaeris radicata, Rumex acetosella, Humulus scandens, and Ambrosia artemisiifolia. Furthermore, due to the characteristics of the volcanic rock crevice and salinity, relatively diverse C4 plants of 26 species were identified, including Atriplex gmelinii, Cynodon dactylon, and Cyperus rotundus. The relatively high proportion of C4 plants means that the native area is a habitat where physical/physiological desiccation can easily occur (Pyankov et al. 2010; Wang and Ma 2016). Therefore, these areas are composed of special species with high adaptability to dryness, and it means that evaluation and management considering the characteristics of unique constituent species are necessary.
Habitat characteristics and ecological management measures
In South Korea, the T. interrupta community is distributed locally on the east coast of Jeju Island, which is the northernmost habitat in east Asia. Small-scale brackish wetland vegetation occurs in the wetland of a concave terrain of lava masses on the coast. Based on the topographical features of the wetland, it is a rainfall-dependent wetland that is not directly associated with groundwater. However, seawater irregularly flows in through high waves from the nearby coast. The formation of the T. interrupta community in this type of habitat does not occur anywhere else in the world. It seems that this specific distribution of species was enabled by the combination of various conditions of the micro-environment rather than typical habitat characteristics.
The geology of the current habitat of T. interrupta is largely characterized by quaternary basalt, which is a lithosphere with a very slow rate of soil erosion, and very limited inflow of natural deposits from the surrounding area. In a wetland with shallow soil depth, black bedrock can increase the water temperature via radiant heat, and this can negatively most plants. However, for T. interrupta, which has the characteristics of a thermophilic plant and is a diagnostic species for geothermal habitat (Thorsen et al. 2009), the laval rock environment of Jeju Island that discharges a high radiant heat must have acted as a factor that allowed it to have a competitive advantage in interbreeding with other species. Furthermore, the coastal wetland on Jeju Island has soils of high salinity due to relatively low precipitation in winter. These trends must have played a critical role in reducing the dormant bud frost damage of T. interrupta, a subtropical plant (Mahajan and Tuteja 2005).
Currently, the largest threat to the T. interrupta community is the area expansion of xeromorphic vegetation. We found that the component species of xeromorphic vegetation flowed in at the entity level around the T. interrupta-P. australis community. In some locations, xeromorphic vegetation was dominant, and T. interrupta remained at the entity level. Particularly, during the trail maintenance process, we discovered that the inflow of external soil accelerated the development of xeromorphic vegetation, which caused the loss of the unique characteristics of the wetland as well as the species composition.
Other threats to the preservation of the T. interrupta habitat include the damage of coastal habitats and disturbance of ecological continuity due to various land use practices, including the construction of coastal roads. The development projects of Jeju Island have been concentrated on the coastal areas where the convenience of development and value of utilization are high. Along with the construction of coastal roads, intensive development for housing and commercial buildings are being conducted throughout the island. As of 2021, according to the MOE land cover maps, artificial facilities on the coastal areas of Jeju Island account for 55.47%, including transportation (16.3%), residences (2.61%), public facilities (2.36%), fields (8.05%), and commercial areas (1.85%) (Table 3). Among them, the area of the inland wetland is only 0.29%, and most of this area is occupied by storage facilities. The high-traffic areas including coastal roads, which account for 16.3% and interferes with the continuity of coastal habitats and drives intensive developments.
The third threat includes physical disturbances in the wetlands by visitors. Recently, with the increase in awareness about the T. interrupta habitat on social media, numerous visitors, including amateur photographers, are visiting this region of the island. Currently, it is difficult to monitor disturbances by visitors in the wetland because of the lack of a full-time manager on the wetland. The content of water saturated in soil pores is a critical factor for vegetation maintenance in wetlands (Kozlowski 1999; Kissling et al. 2009; Moor et al. 2017). The increasing effect of trampling due to the increasing number of visitors can be a critical factor accelerating the terrestrialization of wetlands by decreasing soil pores. In particular, the physical disturbance behaviors to the wetlands by some visitors, who are unaware of the value of the habitat, can cause deadly damage to the habitat characteristics.
Therefore, to stably maintain and preserve the T. interrupta habitat, the factors that interfere with the unique hydrological flow of wetlands need to be managed. The artificial introduction of external soils must be strictly avoided, and the artificial structures that interfere with the inflow of seawater by waves need to be relocated. In addition, the stability of the habitat and population needs to be ensured by locating substitute habitats and additional groups that are not cut off by coastal roads. Although there is a dominant community in the habitat, it is also necessary to create an ex-situ conservation center to prevent the unintentional loss of groups in an already restricted and small habitat. In addition, measures to prevent physical damage by people visiting the wetland need to be created (e.g., a fine). The habitats should be clearly defined; for example, by conducting tours only within controlled areas. In particular, a loss of intrinsic topography and influx of surrounding soils may be the most frequent causes of damage by visitors. Hence, management measures to minimize damage to the habitat, such as the installation of exploration decks, need to be implemented.
The coastal areas of Jeju Island are expected to be the initial settlement of numerous new subtropical plants that flow in due to climate change. This implies that the study area is a habitat that can provide an opportunity for securing national biodiversity and expanding bio-sovereignty. Therefore, our findings show that further efforts to preserve the remaining unharmed habitats are warranted, along with additional evaluation of similar locations to preserve biodiversity in the coastal areas of Jeju Island.
This study is a field record of Thelypteris interrupta ‘s the only natural habitat in Korea. Therefore, it was impossible to analyze the typical characteristics of the habitat, the type of vegetation, and the status and dynamics of these species in East Asia. Nevertheless, as one of the northernmost habitats, this study is judged to be meaningful as basic information, and it is necessary to closely observe changes in species and habitats through continuous monitoring studies in future studies.
Data Availability
Not Applicable.
References
Barrington DS (1993) Ecological and historical factors in fern biogeography. J Biogeogr 20(3):275–280. https://doi.org/10.2307/2845635
Becking RW (1957) The Zürich-Montpellier school of phytosociology. Bot Rev 23(7):411–488. https://doi.org/10.1007/BF02872328
Braun-Blanquet J (1932) Plant sociology: the study of plant communities, First edn. McGraw-Hill Book Co., Inc., New York and London, p 439
Bried JT, Jog SK, Dzialowski AR, Davis CA (2014) Potential vegetation criteria for identifying reference-quality wetlands in the south-central United States. Wetlands 34(6):1159–1169. https://doi.org/10.1007/s13157-014-0575-5
Case TJ, Taper ML (2000) Interspecific competition, environmental gradients, gene flow, and the coevolution of species’ borders. Am Nat 155(5):583–605. https://doi.org/10.1086/303351
Cheong JH, Chae JH, Lee SH, Kim BH (2013) Halophyte flora and vegetation of four seashore sites in Jejudo. J Korean Isl 25(3):91–106
Choi BK (2013) Syntaxonomy and syngeography of warm-temperate evergreen broad-leaved forests in Korea. dissertation. Keimyung University, Daegu. [in Korean with English abstract]
Chytrý M, Tichý L, Holt J, Botta-Dukát Z (2002) Determination of diagnostic species with statistical fidelity measures. J Veg Sci 13(1):79–90. https://doi.org/10.1111/j.1654-1103.2002.tb02025.x
DeBerry DA, Chamberlain SJ, Matthews JW (2015) Trends in floristic quality assessment for wetland evaluation. Wetl Sci Pract 32(2):12–22
Dierschke H (1994) Pflanzensoziologie: grundlagen und methoden. Ulmer, Stuttgart, DE
Eom BC (2011) Plant species composition and distributional peculiarity of wild tea (Thea sinensis L.) population in Korean Peninsula. Master’s thesis. Keimyung University, Daegu. [in Korean with English abstract]
Fried O, Kühn I, Schrader J, Sinh Nguyen V, Bergmeier E (2018) Plant diversity and composition of rice field bunds in Southeast Asia. Paddy Water Environ 16(2):359–378. https://doi.org/10.1007/s10333-018-0657-8
Fukui E (ed) (1977) The climate of Japan, vol 8. Elsevier scientific publishing Co., Amsterdam and New York
GBIF (2021) GBIF occurrence download. GBIF.org. Release Date: Accessed 2021-4-8. https://doi.org/10.15468/dl.aztnnq
Hampe A, Petit RJ (2005) Conserving biodiversity under climate change: the rear edge matters. Ecol Lett 8(5):461–467. https://doi.org/10.1111/j.1461-0248.2005.00739.x
Hemp A (2002) Ecology of the pteridophytes on the southern slopes of Mt. Kilimanjaro. I. Altitudinal distribution. Plant Ecol 159(2):211–239. https://doi.org/10.1023/A:1015569125417
Houle G (1994) Spatiotemporal patterns in the components of regeneration of four sympatric tree species, Acer rubrum, A. Saccharum, Betula alleghaniensis, and Fagus grandifolia. J Ecol 82(1):39–53. https://doi.org/10.2307/2261384
Iksan M, Aba LA, Kusrini K (2019) The ability of ferns to accumulate heavy metals (hg, pb, and cd) in the waters of the Gorontalo River. Int J Appl Biol 3(1):36–44. https://doi.org/10.20956/ijab.v3i1.5930
International Union for Conservation of Nature (IUCN) (2001) IUCN Red List categories and criteria, version 3.1, vol International Union for Conservation of Nature and Natural Resources. International Union for Conservation of Nature and Natural Resources Species Survival Commission, Gland and Cambridge, pp –
IPCC (2007) 2007-the physical science basis: working Group I contribution to the fourth assessment report of the IPCC. Climate Change. Cambridge University Press, Cambridge, p 966
Jog SK, Bried JT, Feng X, Dzialowski AR, Papeş M, Davis CA (2017) Can land use indicate wetland floristic quality and taxonomic distinctness? Ecol Indic 78:331–339. https://doi.org/10.1016/j.ecolind.2017.03.033
Kang MS (2001) Studies on the vegetation of the coastal salt marshes around Jeju. Island. Master’s thesis. Cheju National University, Jeju
Kessler M, Kluge J, Hemp A, Ohlemüller R (2011) A global comparative analysis of elevational species richness patterns of ferns. Glob Ecol Biogeogr 20(6):868–880. https://doi.org/10.1111/j.1466-8238.2011.00653.x
KIGAM (2022) Geological map. Release date: accessed 2022-3-25. Korea Institute of Geoscience and Mineral Resources). https://data.kigam.re.kr/
Kim JW (1992) Vegetation of NorthEast Asia – on the syntaxonomy and syngeography of the oak and beech forests. dissertation. Universität Wien, Vienna
Kim YR (2001) Growth dynamics of phragmites communis population and their environment of marsh in the Nakdong River estuary. Master’s thesis. Dongah University, Busan. [in Korean with English abstract]
Kim JW, Lee YK (2006) Classification and assessment of plant communities. World Science, Seoul, p 240
Kim JW, Manyko YI (1994) Syntaxonomical and synchorological characteristics of the cool-temperate mixed forest in the southern Sikhote Alin, Russian Far East. Korean J Environ Ecol 17(3):391–413
Kingston MB, Turnbull JW, Hall PW (2004) Tweed Vegetation Management Strategy, 2004. Ecograph Ecological and Geographic Information Systems Consultants: Accessed 2008-7-16. https://www.tweed.nsw.gov.au. The Tweed Council, Murwillumbah. Appendices, vol 3. Accessed date
Kissling M, Hegetschweiler KT, Rusterholz HP, Baur B (2009) Short-term and long-term effects of human trampling on above-ground vegetation, soil density, soil organic matter and soil microbial processes in suburban beech forests. Appl Soil Ecol 42(3):303–314. https://doi.org/10.1016/j.apsoil.2009.05.008
KNA (2021) Checklist of vascular plants in Korea (native plants). Release Date: Accessed 2021-6-30. http://www.nature.go.kr/kpni/index.do
Kong WS, Paul W (1993) The plant geography of Korea with an emphasis on the Alpine Zones. Springer, Netherlands. eBook ISBN 978-94-011-1624-4
Kozlowski TT (1999) Soil compaction and growth of woody plants. Scand J For Res 14(6):596–619. https://doi.org/10.1080/02827589908540825
Lee YG (2005) Syntaxonomy and synecology of the riparian vegetation, South Korea. dissertation. Keimyung University, Daegu. [in Korean with English abstract]
Lee SH (2011) Distribution of halophyte communities along altitudinal gradient and growth induction of the vegetation in salt marshes. dissertation. Mokwon University, Daejeon. [in Korean with English abstract]
Lee GY (2020) Syntaxonomical and synchorological studies on the coastal vegetation in South Korea. dissertation. Keimyung University, Daegu. [in Korean with English abstract]
Lee SH, Ji KJ, An Y, Ro HM (2003) Soil salinity and vegetation distribution at four tidal reclamation project areas. Korea J Environ Agric 22(2):79–86 [in Korean with English abstract]. https://doi.org/10.5338/KJEA.2003.22.2.079
Lötter MC, Mucina L, Witkowski ETF (2014) Classification of the indigenous forests of Mpumalanga Province, South Africa. S Afr J Bot 90:37–51. https://doi.org/10.1016/j.sajb.2013.09.010
Maginel CJ, Knapp BO, Kabrick JM, Olson EK, Muzika RM (2016) Floristic quality index for woodland ground flora restoration: utility and effectiveness in a fire-managed landscape. Ecol Indic 67:58–67. https://doi.org/10.1016/j.ecolind.2016.02.035
Mahajan S, Tuteja N (2005) Cold, salinity and drought stresses: an overview. Arch Biochem Biophys 444(2):139–158. https://doi.org/10.1016/j.abb.2005.10.018
Matías L, Jump AS (2012) Interactions between growth, demography and biotic interactions in determining species range limits in a warming world: the case of Pinus sylvestris. For Ecol Manag 282:10–22. https://doi.org/10.1016/j.foreco.2012.06.053
Moon MO, Kim CS, Kang YJ, Kim CH, Sun BY (2002) Unrecorded fern species from korean flora: Thelypteris interrupta (Thelypteridaceae) and Dryopteris subexaltata (Dryopteridaceae). Korean J Plant Taxon 32(4):481–489 [in Korean with English abstract]. https://doi.org/10.11110/kjpt.2002.32.4.481
Moor H, Rydin H, Hylander K, Nilsson MB, Lindborg R, Norberg J (2017) Towards a trait-based ecology of wetland vegetation. J Ecol 105(6):1623–1635. https://doi.org/10.1111/1365-2745.12734
Mucina L, Dale MB (1989) Numerical syntaxonomy. Adv Veg Sci. Springer. https://doi.org/10.1007/978-94-009-2432-1. Dordrecht 10
NIE (2021) List of Korean endangered species. Release Date: Accessed 2021-5-25. https://www.nie.re.kr/endangered_species
Ohba T, Sugawara H (1979) Coastal vegetation of Cheju-do (Quelpart is.) S-Korea. J Phytogeogr Taxon 27(1):1–12
Pyankov V, Ziegler H, Akhani H, Deigele C, Luttge U (2010) European plants with C4 photosynthesis: geographical and taxonomic distribution and relations to climate parameters. Bot J Linn Soc 163:283–304. https://doi.org/10.1111/j.1095-83392010.01062.x
Sieben EJJ, Collins NB, Mtshali H, Venter CE (2016a) The vegetation of inland wetlands with salt-tolerant vegetation in South Africa: description, classification and explanatory environmental factors. S Afr J Bot 104:199–207. https://doi.org/10.1016/j.sajb.2015.11.004
Sieben EJJ, Nyambeni T, Mtshali H, Corry FTJ, Venter CE, MacKenzie DR, Matela TE, Pretorius L, Kotze DC (2016b) The herbaceous vegetation of subtropical freshwater wetlands in South Africa: classification, description and explanatory environmental factors. S Afr J Bot 104:158–166. https://doi.org/10.1016/j.sajb.2015.11.005
Sinclair S, Stajsic V, Sutter G (2012) Thelypteris interrupta (Thelypteridaceae): new to Victoria. Muelleria 30(2):183–188
Sykes MT, Prentice IC (1995) Boreal forest futures: modelling the controls on tree species range limits and transient responses to climate change. Water Air Soil Pollut 82(1–2):415–428. https://doi.org/10.1007/BF01182851
Theurillat JP, Wilner W, Fernández-González F, Bültmann H, Čarni A, Gigante D, Mucina L, Weber H (2020) International Code of Phytosociological Nomenclature. 4th edition. Appl. Veg. Sci. https://doi.org/10.1111/avsc.12491
Thorsen MJ, Dickinson KJM, Seddon PJ (2009) Seed dispersal systems in the New Zealand flora. Perspect Plant Ecol Evol Syst 11(4):285–309. https://doi.org/10.1016/j.ppees.2009.06.001
Thuiller W, Albert C, Araújo MB, Berry PM, Cabeza M, Guisan A, Hickler T, Midgley GF, Paterson J, Schurr FM, Sykes MT, Zimmermann NE (2008) Predicting global change impacts on plant species’ distributions: future challenges. Perspect Plant Ecol Evol Syst 9(3–4):137–152. https://doi.org/10.1016/j.ppees.2007.09.004
Tichý L (2002) JUICE, software for vegetation classification. J Veg Sci 13(3):451–453. https://doi.org/10.1111/j.1654-1103.2002.tb02069.x
Tryon R (1972) Endemic areas and geographic speciation in tropical american ferns. Biotropica 4(3):121–131. https://doi.org/10.2307/2989774
Tryon R (1986) The biogeography of species, with special reference to ferns. Bot Rev 52(2):117–156. https://doi.org/10.1007/BF02860999
Tryon RM, Tryon AF (1982) Ferns and allied plants with special reference to tropical America. Springer, New York
van der Maarel E (1979) Transformation of cover-abundance values in phytosociology and its effects on community similarity. Vegetatio 39(2):97–114
Walck JL, Hidayati SN, Dixon KW, Thompson K, Poschlod P (2011) Climate change and plant regeneration from seed. Glob Change Biol 17(6):2145–2161. https://doi.org/10.1111/j.1365-2486.2010.02368.x
Wang R, Ma L (2016) Climate-driven C4 plant distribution in China: divergence in C4 taxa. Sci Rep. https://doi.org/10.1038/srep27977. 6.27977
Woodward FI (1987) Climate and plant distribution. Cambridge University Press, Cambridge
Zhang J, Yasuda Y, Yoshino M (1997) Climate change in post glacial period in monsoon Asia. In: Yoshino M, Domrös M, Douguédroit A, Paszyński J, Nkemdirim LC (eds) Climates and societies—a climatological perspective. The GeoJournal library, vol 36. Springer, Dordrecht, pp 43–60. https://doi.org/10.1007/978-94-017-1055-8_3
Funding
This study was conducted with the support of the “Intensive Survey on National Inland Wetlands (`23), National Institute of Ecology (NIE-A-2023-19), Development of forest water resources management technology for both preventing streamflow reduction and reducing non-point source pollution (FE0401-2021-01-2023).”
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of the study. Material preparation, data collection, and analysis were performed by Jeong-Cheol Lim, Ju-Eun Yang, Gyeong-Yeon Lee and Byoung-Ki Choi. The first draft of the manuscript was written by Jeong-Cheol Lim and Byoung-Ki Choi and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lim, JC., Yang, JE., Lee, GY. et al. An in-depth characterization of the Habitat of Thelypteris interrupta in South Korea. J Coast Conserv 27, 46 (2023). https://doi.org/10.1007/s11852-023-00968-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11852-023-00968-6