Abstract
Backgrounds and aims
The American Diabetes Association/European Association for the Study of Diabetes recently recommend the preferential use of continuous glucose monitoring(CGM) over self-monitoring of blood glucose for the management of type 1 diabetes (T1DM). For most adults with T1DM, the recommended target time in range is > 70% with < 4% time below range. In Ireland, CGM use has become increasingly popular since 2021. We aimed to audit adult CGM use and analyse CGM metrics in our cohort of adults with diabetes attending a tertiary diabetes centre.
Methods
People with diabetes who were using DEXCOM G6 CGM devices, and sharing their data with the healthcare team on the DEXCOM CLARITY for healthcare professionals platform were included in the audit. Clinical information, glycated haemoglobin (HbA1c) and CGM metrics were gathered retrospectively from medical records and the DEXCOM CLARITY platform.
Results
Data were available for 119 CGM users, 96.9% with T1DM, median age 36 years (IQR = 20) and median diabetes duration 17 years (IQR = 20). Fifty-three per cent of the cohort was male. Mean time in range was 56.2% (SD = 19.2) and mean time below range was 2.3% (SD = 2.6). Mean HbA1c in CGM users was 56.7 mmol/mol (SD = 13.1). This represented a decrease of 6.7 mmol/mol compared to the last HbA1c measurements available pre-commencement of CGM (p ≤ 0.0001, CI 4.4–8.9). The percentage of people in this cohort with a HbA1c < 53 mmol/mol was 40.6% (n = 39/96), compared to 17.5% (n = 18/103) pre-commencement of CGM.
Conclusions
Our study highlights the challenges in optimising the use of CGM. Our team aims to focus on providing additional education to CGM users, more frequent touch-base virtual reviews and increasing access to hybrid closed-loop insulin pump therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Improvements in sensor accuracy, ease of use and expanding reimbursement strategies have seen increased adoption of continuous glucose monitoring (CGM) systems for people with type 1 diabetes mellitus (T1DM) in recent years [1, 2].
Randomised trials have demonstrated significant clinical benefit of CGM use in people with diabetes regardless of insulin delivery method including improvements in glycated haemoglobin (HbA1c), less hypoglycaemic events and better diabetes specific quality of life measures (i.e. diabetes distress, hypoglycaemic confidence) [3,4,5,6,7]. There is also evidence to suggest that CGM can be used successfully in people with poorly controlled type 2 diabetes mellitus (T2DM), not on insulin therapy, to facilitate improved lifestyle choices and medication adherence [8, 9]. CGM was defined as the standard for glucose monitoring over capillary blood glucose monitoring for most adults with type 1 diabetes in the 2021 consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD) [10].
Standardisation of CGM data presentation in the form of the Ambulatory Glucose Profile (AGP) has been developed to assist clinicians and people with diabetes identify glucose patterns to guide clinical decision-making [11]. In 2019, the Advanced Technologies and Treatments for Diabetes (ATTD) consensus published recommendations on clinical targets for CGM metrics including time in range, time below range and time above range [2].
The aim of this audit was to evaluate current CGM use in Beaumont Hospital; we aimed to assess the clinical characteristics of people using DEXCOM CGM in this tertiary diabetes centre, the impact of initiation of DEXCOM CGM on HbA1c and to identify what proportion of CGM users are achieving glycaemic targets.
Methods
People with diabetes who were prescribed DEXCOM CGM and who were sharing their data on the DEXCOM CLARITY platform in a single tertiary centre were identified. This service cares for approximately six thousand people with T2DM and approximately one thousand three hundred people with T1DM. People do not have formal training as part of initiation of the technology. Educational opportunities arise with health care staff at clinic appointments as well as diabetes day centre visits.
Data on age, gender, diabetes type and duration, and duration of CGM use were obtained from the CELLMA electronic medical records software. Data on CGM metrics were collected from the DEXCOM CLARITY Web-based platform. Data collection was undertaken in November and December 2021.
In keeping with international consensus on CGM reporting guidelines, we recorded time in range (% of glucose readings 3.9–10 mmol/L), time above range (% of glucose readings > 10.0 mmol/L), time below range (% of glucose readings < 3.9 mmol/L), glucose variability and percentage time CGM active. These glycaemic metrics were collected and analysed for the first 14 days of CGM use and the 14 days prior to data collection (November/December 2021).
We also recorded the most recent HbA1c prior to the commencement of CGM and the most recent available HbA1c, whilst using CGM from time of data collection (November/December 2021).
Descriptive statistics for demographic and clinical data are reported as number and percentage. Discrete variables are reported as median, interquartile range and range. Continuous variables are reported as mean, standard deviation and range. A paired t-test was used with a p-value of < 0.05 considered statistically significant. Associations between continuous variables including age, baseline HbA1c and change in HbA1c were measured by linear regression. Statistical analysis was performed using Prism GraphPad 9.0.
Results
One hundred and forty-eight people were registered on the DEXCOM CLARITY for health care professionals’ platform. One hundred and nineteen people were sharing data that was available for review and included in this audit (Table 1). The median age of this cohort was 36 years (IQR = 20). Fifty-three per cent were male (n = 63) with a median duration of diabetes of 17 years (IQR = 18). The median duration of CGM use was 12 months (IQR = 9). The mean pre-CGM HbA1c was 64.3 mmol/mol (SD = 14.4). The majority of patients included 97.5% (n = 116) who had type 1 diabetes, whilst the remaining had type 2 diabetes. Demographic data is outlined in Table 1.
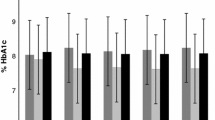
Across the study population, mean HbA1c improved from 64.3 mmol/mol (SD = 14.4) to 57.6 mmol/mol (SD = 13.1), a decrease of 6.7 mmol/mol (p ≤ 0.0001, CI 4.4 – 8.9). This change in HbA1c from pre-CGM to follow-up is displayed in Fig. 1. The percentage of CGM users with a HbA1c < 53 mmol/mol increased from 17.5% (n = 18/103) to 40.6% (n = 39/96).
When looking at the change in CGM metrics from onset of CGM to follow-up, we found no statistically significant change in average glucose (9.5 mmol/L ± 1.8 mmol/L to 9.8 mmol/L ± 2.1 mmol/L, p = 0.09); percentage time in range (59.2% ± 18.04% to 56.2% ± 19.2%, p = 0.089); percentage time above range (38.5% ± 18.8% to 41.5% ± 20.6%, p = 0.1); percentage time below range (2.3% ± 2.8% to 2.3% ± 2.6%, p = 0.88); glucose variability (3.3 SD mmol/L to 3.4 SD mmol/L, p = 0.15); or percentage time CGM active (93.3% ± 9.8% to 92.5% ± 12.3%, p = 0.5). Table 2 outlines HbA1c and CGM metrics at onset of CGM and follow-up.
Change in HbA1c was positively associated with baseline HbA1c as reflected in a larger reduction in HbA1c achieved with higher baseline HbA1c (Pearson r = − 0.48, CI − 0.63 to 0.3, p ≤ 0.0001). We found no significant correlation between age or duration of CGM monitoring and change in HbA1c.
Discussion
This audit demonstrates that despite good engagement with the technology, as evidenced by an excellent percentage time that CGM is active, the majority of CGM users (79%) were not meeting the targets set by the CGM International Vonsensus report in regard to time in range (target > 70% of readings between 3.9 and 10.0 mmol/L), and time above range (< 25% of readings > 10.0 mmol/L) [2]. However, 80% of CGM users were meeting the target time below range (< 4% of readings < 3.9 mmol/L) [2]. This is consistent with data from a UK centre study on the impact of flash glucose monitoring (FGM) and real-time continuous glucose monitoring (RT-CGM) on glycaemic outcomes in 789 adults with type 1 diabetes. In their cohort, 23% of FGM users and 32% of RT-CGM users achieved > 70% of time in range and 70% of RT-CGM and 58% of FGM users spent less than 4% time below range [12]. This data therefore highlights the challenges in optimising the use and maximising the efficacy of CGM devices and other diabetes technologies in people with T1DM.
Continuous glucose monitoring overwhelmingly has been shown to contribute to improvements in diabetes-specific quality of life measures and overall treatment satisfaction [3, 5, 7]. As these technologies are invasive and provide large volumes of data to people with diabetes and health care providers however, they also have the potential to impact negatively on patient treatment satisfaction as well as health care resources [13]. There is also suggestion that technology in isolation does not improve glycaemic control. A study of over 22,000 people with T1DM carried out in the USA demonstrated no improvement in overall HbA1c over a 5-year period despite an increase in CGM and pump use [14].
A number of interventions have been shown to improve the efficacy of diabetes technology and mitigate these risks including diabetes education and training. A randomised control trial in 2019 demonstrated a significant reduction in HbA1c, and improved time in range and diabetes distress scores with a structured education programme for people using flash sensor-based glucose monitoring [15]. For those who choose systems with active alarms and alerts, personalising these settings can prevent alarm fatigue whilst giving people specific instructions on how to respond to alarms can reduce the time spent in hypo- or hyper- glycaemia [16, 17]. It is also important to monitor for skin irritation and poor sensor adhesion as these factors may limit persistent device use and these issues can be successfully managed with topical barrier creams and CGM patches respectively [16].
The integration of CGM with pump technology in the form of sensor-augmented pumps with predictive low glucose suspend function and hybrid closed loop systems has also demonstrated significant benefits including reduction of hypoglycaemic events, improved TIR and improved HbA1c [18,19,20,21]. Improving access to those systems, for those likely to gain the most benefit, should be a priority for health care professionals going forward.
In this audit, CGM use was associated with a statistically significant reduction in HbA1c of 6.7 mmol/mol. This is consistent with other studies. Data from over 10,000 FreeStyle Libre (FSL) flash glucose monitoring device users in the UK demonstrated a − 5.2 mmol/mol change in HbA1c after 7.5 (IQR 3.4–7.8) months of follow-up as well as improved hypoglycaemia awareness and a reduction in hospital admissions [22]. Similarly, the GOLD trial of 161 people with type 1 diabetes showed a 0.43% improvement in HbA1c during CGM compared with self-monitoring of blood glucose [5]. Of course, the retrospective nature of this audit means we cannot exclude the possibility that other changes to our cohorts care or behaviour may have impacted on the change in glycaemic control. The COVID-19 pandemic may have impacted our findings as the study was carried out at the end of 2021. Indeed, an Irish observational study carried out during the first COVID lockdown demonstrated a significant reduction in HbA1c post lockdown [23]. Lifestyle and behavioural changes as well as people attempting to improve glycaemic control due to fears of a more severe disease should they be infected with COVID-19 may be implicated.
Interestingly, in our cohort, there was no statically significant change in CGM metrics at initiation of CGM when compared to follow-up. This suggests that the benefits of CGM begin at initiation of the technology. This is mirrored in a recent meta-analysis investigating the optimal duration of a run-in period for initiating unblinded real-time CGM in a randomised trial. This concluded that improvements in CGM metrics occurred rapidly, with maximal effect achieved within 1–2 weeks [24]. This lack of change in CGM metrics over time however could also reflect the variable duration of CGM use in our population or our relatively small sample size.
Our cohort had a mean baseline HbA1c of 64.3 mmol/L, which is slightly lower than the nationally reported average HbA1c for people with T1DM in Ireland [26]. Previous CGM studies typically have a higher baseline HbA1c [4, 5, 13, 25]. The participants in the DIAMOND trial had a mean baseline HbA1c of 8.6% [SD, 0.6%]; however, the improvement in HbA1c in their randomised control trial was similar to what we have reported in this audit [4]. We demonstrated a positive association between baseline HbA1c and reduction in HbA1c in our study. It is well established that a higher HbA1c corresponds with an increased mortality and an increased risk of diabetes-related complications [27,28,29,30]; hence, expanding interventions to improve glycaemic control in this cohort is hugely important. Recently published ADA and EASD consensus guidelines recommending CGM as the preferred method for glucose monitoring in type 1 diabetes as well as recent NICE guidance recommending FGM or RT-CGM in adults with type 1 diabetes as well as type 2 diabetes under certain conditions should expand the availability of these technologies [10, 31, 32].
Our audit has some limitations; firstly, the timeline to availability of laboratory HbA1c measurements varied during the review. The median time from HbA1c collection to follow-up was 2 months ± 4 months and from collection to CGM initiation was 3 months ± 4 months. As our study timeline overlapped with the COVID-19 pandemic, we assume this contributed to limited patient contact with services. There was also significant variation in the duration of CGM monitoring in our population with a median duration of 12 months (± 9 months) with 12.6% of patients (n = 15/119) using CGM for 3 months of less.
In conclusion, this audit demonstrates that although a statistically significant improvement in glycaemic control occurs with CGM use, the majority of CGM users attending our services have suboptimal time in range highlighting the challenges in optimising the use of CGM in people with T1DM. Our team aims to focus on providing additional education to CGM users, more frequent touch-base virtual reviews and increasing access to hybrid closed-loop insulin pump therapy.
Availability of data and materials
Data supporting this study is stored locally and can be made available on request.
References
Olczuk D, Priefer R (2018) A history of continuous glucose monitors (CGMs) in self-monitoring of diabetes mellitus. Diabetes Metab Syndr 12(2):181–187
Battelino T, Danne T, Bergenstal RM et al (2019) Clinical targets for continuous glucose monitoring data interpretation: recommendations from the international consensus on time in range. Diabetes Care 42(8):1593–1603
Polonsky WH, Hessler D, Ruedy KJ, Beck RW (2017) The impact of continuous glucose monitoring on markers of quality of life in adults with type 1 diabetes: further findings from the DIAMOND Randomized Clinical Trial. Diabetes Care 40(6):736–741
Beck RW, Riddlesworth T, Ruedy K et al (2017) Effect of continuous glucose monitoring on glycemic control in adults with type 1 diabetes using insulin injections: The DIAMOND Randomized Clinical Trial. JAMA 317(4):371–378
Lind M, Polonsky W, Hirsch IB et al (2017) Continuous glucose monitoring vs conventional therapy for glycemic control in adults with type 1 diabetes treated with multiple daily insulin injections: The GOLD Randomized Clinical Trial. JAMA 317(4):379–387
Beck RW, Riddlesworth TD, Ruedy K et al (2017) Continuous glucose monitoring versus usual care in patients with type 2 diabetes receiving multiple daily insulin injections: a randomized trial. Ann Intern Med 167(6):365–374
Heinemann L, Freckmann G, Ehrmann D et al (2018) Real-time continuous glucose monitoring in adults with type 1 diabetes and impaired hypoglycaemia awareness or severe hypoglycaemia treated with multiple daily insulin injections (HypoDE): a multicentre, randomised controlled trial. Lancet 391(10128):1367–1377
Cox DJ, Banton T, Moncrief M et al (2020) Minimizing glucose excursions (GEM) with continuous glucose monitoring in type 2 diabetes: a randomized clinical trial. J Endocr Soc 1;4(11)
Price DA, Deng Q, Kipnes M, Beck SE (2021) Episodic real-time CGM use in adults with type 2 diabetes: results of a pilot randomized controlled trial. Diabetes Ther 12(7):2089–2099
Holt RIG, DeVries JH, Hess-Fischl A et al (2021) The management of type 1 diabetes in adults. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 64(12):2609–2652
Bergenstal RM, Ahmann AJ, Bailey T et al (2013) Recommendations for standardizing glucose reporting and analysis to optimize clinical decision making in diabetes: the Ambulatory Glucose Profile (AGP). Diabetes Technol Ther 15(3):198–211
Lee K, Gunasinghe S, Chapman A et al (2021) Real-world outcomes of glucose sensor use in type 1 diabetes—findings from a Large UK Centre. Biosensors 11(11):457
Gandhi GY, Kovalaske M, Kudva Y et al (2011) Efficacy of continuous glucose monitoring in improving glycemic control and reducing hypoglycemia: a systematic review and meta-analysis of randomized trials. J Diabetes Sci Technol 5(4):952
Foster NC, Beck RW, Miller KM et al (2019) State of type 1 diabetes management and outcomes from the T1D exchange in 2016–2018. Diabetes Technol Ther 21(2):66
Hermanns N, Ehrmann D, Schipfer M et al (2019) The impact of a structured education and treatment programme (FLASH) for people with diabetes using a flash sensor-based glucose monitoring system: results of a randomized controlled trial. Diabetes Res Clin Pract 1(150):111–121
Grunberger G, Sherr J, Allende M et al (2021) American Association of Clinical Endocrinology Clinical Practice Guideline: the use of advanced technology in the management of persons with diabetes mellitus. Endocr Pract 27(6):505–537
Lawton J, Blackburn M, Allen J et al (2018) Patients’ and caregivers’ experiences of using continuous glucose monitoring to support diabetes self-management: qualitative study. BMC Endocr Disord 20;18(1)
Brown SA, Kovatchev BP, Raghinaru D et al (2019) Six-month randomized, multicenter trial of closed-loop control in type 1 diabetes. N Engl J Med 381(18):1707–1717
Ly TT, Nicholas JA, Retterath A et al (2013) Effect of sensor-augmented insulin pump therapy and automated insulin suspension vs standard insulin pump therapy on hypoglycemia in patients with type 1 diabetes: a randomized clinical trial. JAMA 310(12):1240–1247
Bergenstal RM, Klonoff DC, Garg SK et al (2013) Threshold-based insulin-pump interruption for reduction of hypoglycemia. N Engl J Med 369(3):224–232
Isganaitis E, Raghinaru D, Ambler-Osborn L et al (2021) Closed-loop insulin therapy improves glycemic control in adolescents and young adults: outcomes from the International Diabetes Closed-Loop Trial. Diabetes Technol Ther 23(5):342
Deshmukh H, Wilmot EG, Gregory R et al (2020) Effect of flash glucose monitoring on glycemic control, hypoglycemia, diabetes-related distress, and resource utilization in the Association of British Clinical Diabetologists (ABCD) Nationwide Audit. Diabetes Care 43(9):2153–2160
Lockhart M, Green D, Smith D (2022) The impact of COVID-19 lockdown on glycaemic control in young adults with type 1 diabetes mellitus. Ir J Med Sci
Raghinaru D, Calhoun P, Bergenstal RM, Beck RW (2022) The optimal duration of a run-in period to initiate continuous glucose monitoring for randomised trial. Diabetes Technol Ther 24(12):868–872
Yoo JH, Kim JH (2020) Time in range from continuous glucose monitoring: a novel metric for glycemic control. Diabetes Metab J 44(6):828–839. https://doi.org/10.4093/dmj.2020.0257
Prigge R, McKnight JA, Wild SH et al (2022) International comparison of glycaemic control in people with type 1 diabetes: an update and extension. Diabet Med 39:e14766
Nathan DM, Cleary PA, Backlund JC et al (2005) Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med 353(25):2643–2653
Lind M, Odén A, Fahlén M, Eliasson B (2009) The true value of HbA1c as a predictor of diabetic complications: simulations of HbA1c variables. PLoS One 11:4(2)
Lind M, Svensson A-M, Kosiborod M et al (2014) Glycemic control and excess mortality in type 1 diabetes. N Engl J Med 371(21):1972–1982
Ahlén E, Pivodic A, Wedel H et al (2016) Glycemic control, renal complications, and current smoking in relation to excess risk of mortality in persons with type 1 diabetes. J Diabetes Sci Technol 10(5):1006–1014
National Institute for Health and Care Excellence (2015) Type 1 diabetes in adults: diagnosis and management. [London]: NICE; [updated 2022 Aug]
National Institute for Health and Care Excellence (2015) Type 2 diabetes in adults: management . [London]: NICE; [updated 2022 June]
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Aoife Courtney, Diarmuid Smith and Hannah Forde. The first draft of the manuscript was written by Aoife Courtney and all the authors commented on previous versions of the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional audit committee and with the 1964 Helsinki declaration and its later amendments. Patient consent was not required. As this was an audit of current practice compared to recently published standards, no ethical approval was sought.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Courtney, A., Smith, D. & Forde, H. Real-world outcomes of continuous glucose monitoring in adults with diabetes mellitus attending an Irish tertiary hospital. Ir J Med Sci 192, 2763–2768 (2023). https://doi.org/10.1007/s11845-023-03322-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-023-03322-9