Abstract
Background
In addition to its direct cytotoxic effects, radiation therapy renders tumor cells more susceptible to T cell-mediated cytotoxicity by modulating cell surface molecules involved in antigen presentation. The purpose of the present study was to determine the benefit of combined 89Sr radiation and dendritic cell (DC) vaccine therapy in bone metastasis patients.
Methods
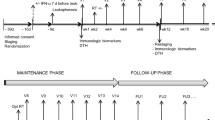
Patients were treated with intravenous 89Sr at a dose of 40 μCi/kg of body weight on the first day after the peripheral blood mononuclear cell collection. Seven days later, patients received DCs once a week for 6 weeks. The first three vaccines were administered by intravenous infusion, and the last three vaccines were administered by 24-point intradermal injection. Clinical response was evaluated by the number of bone metastatic foci demonstrated on bone scintigraphy; cell-mediated cytotoxicity response was evaluated by delayed-type hypersensitivity (DTH) reaction. All treatment-related toxicities including vaccine-induced fever and 89Sr-associated hematological toxicity were carefully monitored.
Results
Twenty-six patients with histologically diagnosed with primary cancers and multiple bone metastases demonstrated on bone scintigraphy were studied. The overall survival rate was 58.3 %. The total positive DTH rate was 50 %. The efficiency rate for pain relief was 60 % (6/10), for quantity of life was 80 %, and for clinic responses was 90 %. Out of 10 cases, the Grade 1 or 2 of hematological depression in 4, erythema in 1, and fever in 7 were observed.
Conclusions
The study has important implications for that combined 89Sr radiation, and DC vaccine therapy can benefit cancer patients with bone metastasis.
Similar content being viewed by others
References
Petrut B, Trinkaus M, Simmons C et al (2008) A primer of bone metastases management in breast cancer patients. Curr Oncol 15:S50–S57. doi:10.3747/co.2008.176
Sartor AO (2011) Progression of metastatic castrate-resistant prostate cancer: impact of therapeutic intervention in the post-docetaxel space. J Hematol Oncol 4:18
Gridley DS, Williams JR, Slater JM (2005) Low-dose/low-dose-rate radiation: a feasible strategy to improve cancer radiotherapy? Cancer Ther 3:105–130
Reddy EK, Robinson RG, Mansfield CM (1986) Strontium 89 for palliation of bone metastases. J Natl Med Assoc 78:27–32
Lewington VJ (1993) Targeted radionuclide therapy for bone metastases. Eur J Nucl Med 20:66–74. doi:10.1007/BF02261248
Kraeber-Bodéré F, Campion L, Rousseau C et al (2000) Treatment of bone metastases of prostate cancer with strontium-89 chloride: efficacy in relation to the degree of bone involvement. Eur J Nucl Med 27:1487–1493. doi:10.1007/s002590000315
Buchali K, Correns HJ, Schuerer M et al (1988) Results of a double blind study of 89-strontium therapy of skeletal metastases of prostatic carcinoma. Eur J Nucl Med 14:349–351
Pons F, Herranz R, Garcia A et al (1997) Strontium-89 for palliation of pain from bone metastases in patients with prostate and breast cancer. Eur J Nucl Med 24:1210–1214. doi:10.1007/s002590050143
Kasalický J, Krajská V (1988) The effect of repeated strontium-89 chloride therapy on bone pain palliation in patients with skeletal cancer metastases. Eur J Nucl Med 25:1362–1367
Soliman H (2010) Developing an effective breast cancer vaccine. Cancer Control 17:183–190
Nguyen-Pham TN, Lee YK, Lee HJ et al (2012) Cellular immunotherapy using dendritic cells against multiple myeloma. Korean J Hematol 47:17–27. doi:10.5045/kjh.2012.47.1.17
Thomas-Kaskel AK, Zeiser R, Jochim R et al (2006) Vaccination of advanced prostate cancer patients with PSCA and PSA peptide-loaded dendritic cells induces DTH responses that correlate with superior overall survival. Int J Cancer 119:2428–2434. doi:10.1002/ijc.22097
Kiessling A, Füssel S, Wehner R et al (2008) Advances in specific immunotherapy for prostate cancer. Eur Urol 53:694–708. doi:10.1016/j.eururo.2007.11.043
Kim CH, Woo SJ, Park JS et al (2007) Enhanced antitumour immunity by combined use of temozolomide and TAT-survivin pulsed dendritic cells in a murine glioma. Immunology 122:615–622. doi:10.1111/j.1365-2567.2007.02680.x
Nguyen-Pham TN, Lee YK, Kim HJ et al (2012) Immunotherapy using dendritic cells against multiple myeloma: how to improve? Clin Dev Immunol 2012:397648. doi:10.1155/2012/397648
Sharp HJ, Wansley EK, Garnett CT et al (2007) Synergistic antitumor activity of immune strategies combined with radiation. Front Biosci 12:4900–4910. doi:10.2741/2436
Tseng CW, Trimble C, Zeng Q et al (2008) Low-dose radiation enhances therapeutic HPV DNA vaccination in tumor-bearing hosts. Cancer Immunol Immunother 58:737–748. doi:10.1007/s00262-008-0596-0
Finkelstein SE, Timmerman R, McBride WH et al (2011) The confluence of stereotactic ablative radiotherapy and tumor immunology. Clin Dev Immunol 2011:439752. doi:10.1155/2011/439752
Persson BRR, Koch CB, Grafström G, et al. (2011) “Abscopal” effect of radiation therapy combined with immune-therapy using IFN-γ gene transfected syngeneic tumor cells, in rats with bilateral implanted N29 tumors. ISRN Immunol 230130. doi:10.5402/2011/230130
Reissfelder C, Timke C, Schmitz-Winnenthal H et al (2011) A randomized controlled trial to investigate the influence of low dose radiotherapy on immune stimulatory effects in liver metastases of colorectal cancer. BMC Cancer 11:419. doi:10.1186/1471-2407-11-419
Liu Y, Zhang W, Zhang B et al (2013) DC vaccine therapy combined concurrently with oral capecitabine in metastatic colorectal cancer patients. Hepatogastroenterology 60:23–27. doi:10.5754/hge12522
Luo J, Sun Y, Wu G et al (1996) Development and assessment of quality of life questionnaire for Chinese cancer patients. J Pract Oncol 11:252–254
Hovey E, Marx G, Kneebone A et al (2009) An Australian clinical perspective: management of hormone refractory (androgen-independent) prostate cancer. Asia Pac J Oncol Hematol 1:77–88
Ma Y, Kepp O, Ghiringhelli F et al (2010) Chemotherapy and radiotherapy: cryptic anticancer vaccines. Semin Immunol 22:113–124. doi:10.1016/j.smim.2010.03.001
Lin CC, Wang TE, Liu CY et al (2008) Potentiation of the immunotherapeutic effect of autologous dendritic cells by pretreating hepatocellular carcinoma with low-dose radiation. Clin Invest Med 31:E150–E159
Chakraborty M, Wansley EK, Carrasquillo JA et al (2008) The use of chelated radionuclide (samarium-153-ethylenediaminetetramethylenephosphonate) to modulate phenotype of tumor cells and enhance T cell-mediated killing. Clin Cancer Res 14:4241–4249. doi:10.1158/1078-0432.CCR-08-0335
Teitz-Tennenbaum S, Li Q, Davis MA et al (2009) Radiotherapy combined with intratumoral dendritic cell vaccination enhances the therapeutic efficacy of adoptive T-cell transfer. J Immunother 32:602–612. doi:10.1097/CJI.0b013e3181a95165
Ridolfi L, Petrini M, Fiammenghi L et al (2010) Unexpected high response rate to traditional therapy after dendritic cell-based vaccine in advanced melanoma: update of clinical outcome and subgroup analysis. Clin Dev Immunol 2010:504979. doi:10.1155/2010/504979
Rotow J, Gameiro SR, Madan RA et al (2010) Vaccines as monotherapy and in combination therapy for prostate cancer. Clin Transl Sci 3:116–122. doi:10.1111/j.1752-8062.2010.00186.x
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, J., Li, J., Fan, Y. et al. Concurrent dendritic cell vaccine and strontium-89 radiation therapy in the management of multiple bone metastases. Ir J Med Sci 184, 457–461 (2015). https://doi.org/10.1007/s11845-014-1145-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-014-1145-9