Abstract
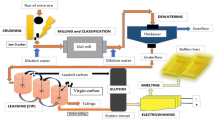
This study investigated the recovery of nickel from tailings (0.2% Ni) obtained in a chromite beneficiation plant using H2SO4 leaching followed by oxalic acid precipitation. The tailings were characterized using mineral liberation analysis. Ni was found as Fe-Ni, Fe-Ni-Co sulfide minerals, which were encapsulated in serpentine, olivine, pyroxene and clinochlore. Liberated fine chromite minerals benefited from magnetic separation followed by Falcon concentrator. A chromite product with a grade > 37% was obtained. The effect of leaching conditions including acid concentration, temperature, time and solid-to-liquid ratio on the leaching behavior of Ni was investigated based on the Taguchi approach. It was possible to extract Ni with a rate of > 98% from the tailings using the following conditions: H2SO4 concentration of 3 M, solid-to-liquid ratio of 1:20 and ambient temperature for 2 h. Finally, Ni was precipitated as an oxalate form with a precipitation rate of 72%. It is believed that implementing additional purification methods like solvent extraction may be essential to obtain a high-purity nickel product from the leach solution of oxalate precipitate. Experimental results indicate that the tailings used in this study are a good alternative as Ni resource in the near future.




Similar content being viewed by others
References
European Commission, Study on the Critical Rwa Materials for the EU (Final Report) (2023).
Energy, USA, Critical Materials Assessment (2023).
T. Schmidt, M. Buchert, and L. Schebek, Resour. Conserv. Recycl. https://doi.org/10.1016/j.resconrec.2016.04.017 (2016).
Z. Xi, Z. Wang, G. Yan, H. Guo, X. Li, Q. Hu, W. Peng, J. Wang, and G. Yan, Hydrometallurgy. https://doi.org/10.1016/j.hydromet.2019.03.007 (2019).
Ş Kaya and Y.A. Topkaya, in Rare earths industry. ed. by I.B.D. Lima, and W.L. Filho (Elsevier, Amsterdam, 2016), p. 171.
J.A. Johnson, B.C. Cashmore, and R.J. Hockridge, Miner. Eng. https://doi.org/10.1016/j.mineng.2005.05.013 (2005).
A.L.A. Santos, E.M.A. Becheleni, P.R.M. Viana, R.M. Papini, F.P.C. Silvas, and S.D.F. Rocha, Min. Metall. Explor. https://doi.org/10.1007/s42461-020-00310-w (2021).
A. Oxley, M.E. Smith, and O. Caceres, Miner. Eng. https://doi.org/10.1016/j.mineng.2015.09.018 (2016).
H.R. Watling, A.D. Elliot, H.M. Fletcher, D.J. Robinson, and D.M. Sully, Aust. J. Earth Sci. https://doi.org/10.1080/08120099.2011.602986 (2011).
V. Bolaños-Benítez, E.D. Van Hullebusch, P.N.L. Lens, C. Quantin, J. Van de Vossenger, S. Subramanian, and Y. Sivry, Minerals. https://doi.org/10.3390/min8060261 (2018).
S. Top, S. Kursunoglu, and Z.T. Ichlas, Can. Metall. Q. https://doi.org/10.1080/00084433.2020.1780560 (2020).
J. MacCarthy, A. Nosrati, W. Skinner, and J. Addai-Mensah, Miner. Eng. https://doi.org/10.1016/j.mineng.2014.12.031 (2015).
S. Kursunoglu and M. Kaya, Int. J. Miner. Process. https://doi.org/10.1016/j.minpro.2016.03.001 (2016).
S. Kursunoglu, Z.T. Ichlas, and M. Kaya, Trans. Nonferrous Metals Soc. China. https://doi.org/10.1016/S1003-6326(18)64808-3 (2018).
P.G. Tzeferis, Min. Metall. Explor. https://doi.org/10.1007/BF03403052 (1994).
D. Pradhan, Mater. Today Proc. https://doi.org/10.1016/j.matpr.2022.06.129 (2022).
D. Pradhan, Lett. Appl. NanoBioSci. https://doi.org/10.33263/LIANBS101.18251832 (2021).
D. Pradhan, D.J. Kim, G.R. Chaudhury, J.S. Sohn, and S.W. Lee, Mater. Trans. https://doi.org/10.2320/matertrans.M2009195 (2010).
D.J. Kim, D. Pradhan, G.R. Chaudhury, J.G. Ahn, and S.W. Lee, Mater. Trans. https://doi.org/10.2320/matertrans.M2009125 (2009).
S. Kursunoglu, Z.T. Ichlas, and M. Kaya, Hydrometallurgy. https://doi.org/10.1016/j.hydromet.2017.01.001 (2017).
J. Borda and R. Torres, S. Afr. J. Chem. Eng. https://doi.org/10.1016/j.sajce.2023.07.001 (2023).
G. Prameswara, F.Y.P. Tyassena, M. Pasaribu, I. Trisnawati, and H.T.B.M. Petrus, Trans. Indian Inst. Metals. https://doi.org/10.1007/s12666-022-02858-1 (2023).
B. Dong, Q. Tian, Z. Xu, X. Guo, Q. Wang, and D. Li, Hydrometallurgy. https://doi.org/10.1016/j.hydromet.2023.106063 (2023).
F. He, B. Ma, Z. Qiu, C. Wang, Y. Chen, and X. Hu, Miner. Eng. https://doi.org/10.1016/j.mineng.2023.108170 (2023).
C. Mang, G. Li, Y. Chen, J. Luo, M. Rao, and T. Jiang, Hydrometallurgy. https://doi.org/10.1016/j.hydromet.2023.106090 (2023).
G.K. Das and J. Li, ACS Omega. https://doi.org/10.1021/acsomega.2c07595 (2023).
K.K. Gupta, S. Haratian, O.V. Mishin, and R. Ambat, npj Mater. Degrad. https://doi.org/10.1038/s41529-023-00393-y (2023).
V. Deniz, Inz. Miner. https://doi.org/10.29227/IM-2019-02-17 (2019).
V. Ross, M. Ramonotsi, and G. Marape, Miner. Eng. https://doi.org/10.1016/j.mineng.2022.107801 (2022).
W. Astuti, F. Nurjaman, F.R. Mufakhir, S. Sumardi, D. Avista, K.C. Wanta, and H.T.B.M. Petrus, Miner. Eng. https://doi.org/10.1016/j.mineng.2022.107982 (2023).
M.F. Gulcan, B.D. Karahan, and O. Keles, J. Appl. Electrochem. https://doi.org/10.1007/s10800-023-01846-7 (2023).
X. Wang, Y. Liu, and B. Liu, J. Hazard. Mater. https://doi.org/10.1016/j.jhazmat.2023.132052 (2023).
M.F. Ye and G.L. Wu, Mineralogical analysis of a chrome ore from South Africa, in Characterization of Minerals, Metals, and Materials 2018. TMS 2018. The Minerals, Metals & Materials Series. ed. by B. Li, et al. (Springer, Cham, 2018). https://doi.org/10.1007/978-3-319-72484-3_65.
M.I. Pownceby, D.A. McCallum, and W.J. Bruckard, Minerals. https://doi.org/10.3390/min13030440 (2023).
B. Lim, R.D. Alorro, M. Aylmore, and D. Grimsey, Miner. Eng. https://doi.org/10.1016/j.mineng.2023.108167 (2023).
S.K. Tripathy, V. Singh, and Y. Ramamurthy, Int. J. Min. Eng. Miner. Process. 3, 101 (2012).
S.K. Tripathy, P.K. Banerjee, and N. Suresh, Int. J. Miner. Metall. Mater. https://doi.org/10.1007/s12613-015-1064-4 (2015).
R.K. Rath, B. Dey, M.K. Mohanta, L.K. Prusty, and R. Singh, Recovery of chromite values from tailings of cob plant using enhanced gravity concentrator. Paper presented at the International Seminar on Mineral Processing Technology 2017, 1–3 February 2017.
M. Gharabaghia, M. Irannajadb, and A.R. Azadmehr, Chem. Eng. Res. Des. https://doi.org/10.1016/j.cherd.2012.11.016 (2013).
S. Kursunoglu and M. Kaya, J. Miner. Process. 150, 1–8 https://doi.org/10.1016/j.minpro.2016.03.001 (2016).
S. Surianti, K.C. Wanta, W. Astuti, F.R. Mufakhir, I. Perdana, and H.T. Petrus, J. Min. Sci. 58(3), 476–485 (2022).
J.A. Allen, J. Phys. Chem. https://doi.org/10.1021/j150508a027 (1953).
Acknowledgement
The study was financially supported by Cukurova University (FBA-2022-14375). The authors thank anonymous reviewers for their suggestion and comments to improve the quality of the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Altiner, M., Ibrahim, A.I.I., Kursunoglu, S. et al. Characterization of Nickel in Chromite Beneficiation Tailings by Mineral Liberation Analysis and Its Recovery by H2SO4 Leaching Followed by Oxalic Acid Precipitation. JOM 76, 1383–1393 (2024). https://doi.org/10.1007/s11837-023-06253-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-023-06253-z