Abstract
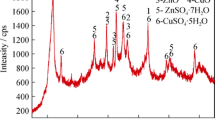
In this work, zinc neutral leaching residue was mechanically activated by ball-milling. The subsequent leaching behavior and kinetics of cadmium extraction in a mixed SO2-H2SO4 system were studied. Changes in the crystalline phase, lattice distortion, particle size and morphology, which were induced by mechanical activation, were also investigated. The activated samples showed different physicochemical characteristics, and cadmium extraction was found to be easier than for the un-activated samples. Under the same conditions, mechanical activation contributed to higher cadmium leaching. The cadmium extraction kinetics at 75–95°C was found to fit the shrinking core model. The raw neutral leaching residue, and the samples activated for 60 min and 120 min had a calculated activation energy of 65.02 kJ/mol, 59.45 kJ/mol and 53.46 kJ/mol, respectively. The leaching residue was characterized by ICP, XRD and SEM analysis. According to XRD analysis, the main phases in the residue were lead sulfate (PbSO4), zinc sulfide (ZnS) and cadmium sulfide (CdS).













Similar content being viewed by others
References
X.W. Wu, Y.H. Liang, T.T.Y. Jin, T.T. Ye, Q.H. Kong, Z.J. Wang, L.J. Lei, I.A. Bergdahl, and G.F. Nordberg, Environ. Res. 108, 233 (2008).
M. Trzcinka-Ochocka, M. Jakubowski, G. Razniewska, T. Halatek, and A. Gazewski, Environ. Res. 95, 143 (2004).
M. Gharabaghi, M. Irannajad, and A.R. Azadmehr, Sep. Purif. Technol. 86, 9 (2012).
M.S. Safarzadeh, D. Moradkhani, and M. Ojaghi-Ilkhchi, J. Hazard. Mater. 163, 880 (2009).
M.S. Safarzadeh, D. Moradkhani, M.O. Ilkhchi, and N.H. Golshan, Sep. Purif. Technol. 58, 367 (2008).
N. Leclerc, E. Meux, and J.M. Lecuire, Hydrometallurgy 70, 175 (2003).
M. Benamor, Z. Bouariche, T. Belaid, and M.T. Draa, Sep. Purif. Technol. 59, 74 (2008).
J. Moghaddam, R. Sarraf-Mamoory, M. Abdollahy, and Y. Yamini, Sep. Purif. Technol. 51, 157 (2006).
M.S. Safarzadeh, M.S. Bafghi, D. Moradkhani, and M.O. Ilkhchi, Miner. Eng. 20, 211 (2007).
Y.J. Zhang, X.H. Li, L.P. Pan, X.Y. Liang, and X.P. Li, Hydrometallurgy 100, 172 (2010).
C. Abbruzzese, Hydrometallurgy 25, 85 (1990).
G.K. Das and J.A.B. de Lange, Hydrometallurgy 105, 264 (2011).
D. Grimanelis, P. Neou-Syngouna, and H. Vazarlis, Hydrometallurgy 31, 139 (1992).
K.B. Hallberg, B.M. Grail, C.A. du Plessis, and D. Barrie Johnson, Miner. Eng. 24, 620 (2011).
L.M. Petrie, Appl. Geochem. 10, 253 (1995).
G. Senanayake, J. Childs, B.D. Akerstrom, and D. Pugaev, Hydrometallurgy 110, 13 (2011).
T.H. Kim, G. Senanayake, J.G. Kang, J.S. Sohn, K.I. Rhee, S.W. Lee, and S.M. Shin, Hydrometallurgy 96, 154 (2009).
M. Hashemzadehfini, J. FiceriovÁ, E. Abkhoshk, and B.K. Shahraki, Trans. Nonferrous Metals Soc. China 21, 2744 (2011).
Z.W. Zhao, S. Long, A.L. Chen, G.S. Huo, H.G. Li, X.J. Jia, and X.Y. Chen, Hydrometallurgy 99, 255 (2009).
T.C. Yuan, Q.Y. Cao, and J. Li, Hydrometallurgy 104, 136 (2010).
K. Tkáčová, V. Šepelák, N. Števulová, and V.V. Boldyrev, J. Solid State Chem. 123, 100 (1996).
P. Baláž, L. Takacs, M. Luxová, E. Godočíková, and J. Ficeriová, Int. J. Miner. Process. 74, S365 (2004).
M.T. Tang, Z.H. Xia, L.S. Qing, Y. Luo, and C.B. Tang, Ming Metal. Eng. 25, 53 (2005).
Acknowledgements
The authors gratefully acknowledge the Key Project of Science and Technology of Hunan province (2012FJ1010), National Natural Science Foundation of China (51474247), Science and Technology Program for Public Wellbeing (2012GS430201) and the National high technology research and development program of China (2011AA061001) for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, C., Min, X., Chai, L. et al. Mechanical Activation-Assisted Reductive Leaching of Cadmium from Zinc Neutral Leaching Residue Using Sulfur Dioxide. JOM 67, 3010–3021 (2015). https://doi.org/10.1007/s11837-015-1623-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-015-1623-1