Abstract
Citizen science is an increasingly powerful tool to gather large quantities of biological data and to engage the public. The number of citizen science initiatives has rapidly increased in the last 15 years. Invasive alien species such as the harlequin ladybird, Harmonia axyridis (Coleoptera, Coccinellidae), provide a particularly good opportunity for broad-scale use of such initiatives. Harmonia axyridis is parasitized by a fungus, Hesperomyces harmoniae (Ascomycota, Laboulbeniales), that can be found throughout the range of its host. Here we present Beetlehangers.org, a website and data repository where we combine observations of He. harmoniae from literature, online databases, and citizen science initiatives, including new records of both Ha. axyridis and He. harmoniae. Whereas Ha. axyridis is now present in 86 countries across six continents (including seven new country records), the distribution of its parasite He. harmoniae comprises 33 countries in five continents (including two new country records since its description in 2022). We explore spatiotemporal trends of He. harmoniae in light of these records. Finally, we discuss challenges and new opportunities for citizen science in relation to species interactions such as these and provide future perspectives for the website as a home for future Laboulbeniales research and outreach.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Citizen science (CS) is defined as the involvement of non-professionals in voluntarily collecting scientific data (Pocock et al. 2017). Data collection by citizen scientists has become an established approach to engage people with biodiversity, including environmental issues, while generating new knowledge and encouraging public participation in conservation actions (Turrini et al. 2018). While CS is often less structured and contains more noise and bias than research projects with data gathered by professional researchers (Boakes et al. 2010; Probert et al. 2022), the sheer quantity of observations cannot be matched by traditional research approaches (Chandler et al. 2017; Klemann-Junior et al. 2017; Bradter et al. 2018). Large-scale, mass-participation CS projects have grown in number over the last decades and are increasingly being used in biodiversity monitoring to inform conservation, ecology, and management of natural resources (McKinley et al. 2017; Pocock et al. 2017). CS initiatives that span large scales are useful for mapping species distributions and, when covering long time periods, can contribute to studies on climate change and tracking changes in species distributions including those of alien species (Dickinson et al. 2010). Finally, CS projects not only serve to answer research questions, they also increase species literacy and engagement with the natural world among the public (Hooykaas et al. 2022). This, in turn, can build broad-based support for nature conservation and management (Home et al. 2009).
Management of invasive alien species (IAS) greatly benefits from CS projects, since they can cover a much larger geographical area, map ecological interactions of IAS, and identify possible biological control agents (Haelewaters et al. 2017; Encarnação et al. 2021; Groom et al. 2021; Pocock et al. 2024). The harlequin ladybird Harmonia axyridis (Coleoptera, Coccinellidae) is an IAS that was introduced from eastern Asia to North America and Europe as a biocontrol agent of aphids and scale insects and has since spread around the world (Lombaert et al. 2010; Roy et al. 2016; Soares et al. 2022). Due to its negative effects on native insect communities and food production (Koch et al. 2006; Roy et al. 2012; Brown and Roy 2018), it is considered one of the worst pests in Europe (Nentwig et al. 2018). Harmonia axyridis makes a good subject for CS efforts because it is a brightly colored, charismatic, and easily recognized species (Soares et al. 2022). As a result, there is increasing interest to use CS data to monitor it around the world (Stals and Prinsloo 2007; Brown et al. 2008; Steenberg and Harding 2009; Grez et al. 2016; Brown and Roy 2018; Hiller and Haelewaters 2019; Werenkraut et al. 2020; Weyman et al. 2022; Angelidou et al. 2023).
Harmonia axyridis is host to a multitude of natural enemies, including parasites, parasitoids, pathogens, and predators (Ceryngier et al. 2012, 2018; Haelewaters et al. 2017) of which several are poorly known (Roy et al. 2017). Among these, Hesperomyces harmoniae (Ascomycota, Laboulbeniales) is a specialized biotrophic microfungus that is uniquely associated with Ha. axyridis (Haelewaters et al. 2022b). It produces tiny three-dimensional structures, or thalli, that grow externally on the integument of the host. Hesperomyces harmoniae is the most recorded species in Hesperomyces virescens sensu lato, a complex of species that are segregated by host and geographic location (Haelewaters et al. 2018a; Van Caenegem et al. 2023). Although the first records of He. harmoniae within the native range of Ha. axyridis date from the 1930s (Haelewaters et al. 2014), these were previously referred to as He. virescens. Recent results from laboratory experiments point at the potential for the fungus as a biocontrol agent against its invasive host (Haelewaters et al. 2020; Knapp et al. 2022; de Groot and Haelewaters 2022). In different locations in the invasive range of Ha. axyridis, a time lag is observed between the establishment of the ladybird in the wild and the first observation of He. harmoniae (Haelewaters et al. 2017). However, data are limited and would benefit from CS initiatives to increase the number of records in space and time.
In this study, we collated records of He. harmoniae associated with Ha. axyridis from multiple independent sources. The aims were threefold: (1) present an online dataset of global records made available through a newly introduced website Beetlehangers.org, (2) improve our understanding of the spatiotemporal patterns of He. harmoniae, and (3) discuss ways to increase the number of records of He. harmoniae alongside those of Ha. axyridis with the help of citizen scientists.
Materials and methods
Data collection
We define a “record” of He. harmoniae as an observation of one host individual of Ha. axyridis that is infected with He. harmoniae, irrespective of the number of thalli. Records of He. harmoniae were collated from multiple sources. First, a total of 41 CS networks, national organizations, and entomological and mycological societies across Europe were contacted to share unpublished records of He. harmoniae—as part of M.C.’s short-term stay at Ghent University funded by the Alien-CSI European network. Records were retrieved from naturgucker.de (Germany), Projekt LIFE ARTEMIS (Slovenia), National Biodiversity Network (UK), Ukrainian Biodiversity Information Network (Ukraine), and Dutch National Database Flora and Fauna (NDFF, The Netherlands) (Supplementary file S1). Curated records from NDFF were not shown with their exact coordinates, following the conditions of use. They were provided as polygons, of which the centroids were calculated in QGIS 3.32 (QGIS Development Team 2023). The coordinates of the centroids were used in the final Beetlehangers dataset.
Second, records were extracted from published papers. Location descriptions were saved in text format (e.g., “Boston, MA”) and transformed into geographic coordinates using a Python script that leveraged the Geopandas package (Jordahl 2014). The number of He. harmoniae records (infected host individuals) were manually counted per location, when this information was available.
Third, digital records were searched and pooled from three online platforms: iNaturalist (https://www.inaturalist.org/home), Waarnemingen.be (https://waarnemingen.be/), and Flickr (https://www.flickr.com/). Records from iNaturalist were downloaded using their in-house data portals on 11 July 2023. In addition to the collation of previous data from iNaturalist, a dedicated project was initiated, “Beetle Hangers” (https://www.inaturalist.org/projects/beetle-hangers), in tandem with a social media campaign in spring 2022 (Fig. 1; Supplementary file S2), to encourage users to submit observations of He. harmoniae in Europe through this project. A flyer was produced and shared through Facebook and X (then Twitter). To ensure correct identifications of Hesperomyces on iNaturalist given recent taxonomic changes (Haelewaters et al. 2022b), M.D.d.G. and D.H. searched for observations of “Hesperomyces”, checked their suggested identifications, and suggested the appropriate ID towards reaching “Research-Grade” observation status (Iwane 2023). For the final Beetlehangers dataset, all iNaturalist records that were not identified as He. harmoniae were filtered out, even if they belonged in the He. virescens complex.
Representative images used in outreach. Above: illustration by Linshan Feng (Leiden University). Below: Greek version of the visual guide (dubbed “spotting chart”) used for our 2022 social media campaign with an overview of Harmonia axyridis features, images of Hesperomyces harmoniae-infected specimens, and instructions on how to submit observations
Records from Waarnemingen.be were searched on 2 October 2023 using “Hesperomyces harmoniae” as query. The three resulting photo records were manually checked; no validated records of He. harmoniae were retained. Data associated with relevant photo records on Flickr were extracted automatically on 23 September 2022 (i.e., before the formal description of He. harmoniae) following Wu (2019) and with the help of Python’s FlickrAPI package (Ando and Pousson 2022). The search queries used were “Hesperomyces” and “Hesperomyces virescens”. These records along with their geographic coordinates and other metadata were then combined into a single CSV file (Wu 2019) and later compiled within the final Beetlehangers dataset. Records were only pulled from Flickr when geographic coordinates were present, and thus records of He. harmoniae without coordinates are currently not in our dataset. Duplicate records were manually identified and removed in Excel 2210 (Microsoft Corporation, Redmont, Washington).
Data curation
All photographic records received were manually checked for accuracy of species identification. Incorrectly identified records were not retained in the final Beetlehangers dataset. Geographic coordinates (lat, long) were used to assign locations, administrative divisions (states, regions, provinces), two-letter ISO 3166–1 alpha-2 country codes, country names, and continents to all records in the database with the help of the Geopandas and Pycountry packages (Jordahl 2014; Theune 2022). The naming conventions of several countries by Geopandas yielded names with special characters not recognized by Microsoft Excel. These country names were manually fixed. For some records, Geopandas failed to retrieve the necessary location information based on the provided coordinates. Those were manually corrected. Dates of observation were reported for all records (observed_on). However, dates pooled from different sources were not standardized with some following the “little endian” format as mm/dd/yyyy and others the “middle endian” format as dd/mm/yyyy. We employed the datetime package in Python to harmonize the date notations and accurately extract the year for each record. For other date formats, the datetime package was unable to interpret and generated the output “needs year”. These records were manually checked and corrected.
Visualizations
We constructed interactive coordinate maps for the USA and Europe with Google My Maps, showing all collated records of He. harmoniae with Ha. axyridis, each with geographic coordinates, collection date (dd/mm/yyyy), source, and link to original observation when applicable. In addition to these interactive maps, we created static choropleth maps for the world, the United States of America (USA), and Europe with CS records of He. harmoniae, using Google Sheet’s Geochart functionality. We plotted the accumulated number of records of He. harmoniae over time using Excel 2210, including records from the literature (summarized in Haelewaters et al. 2022b) and CS-based observations.
To gain a better understanding of the habitats in which He. harmoniae has been reported thus far, we used the raster layer showing anthropogenic biomes at 5 arc-minute resolution, downloaded from NASA’s Socioeconomic Data and Applications Center (Ellis and Ramankutty 2008a). These anthropogenic biomes are based on population density, land use, and vegetation cover and are organized into six broad groupings: dense settlements, villages, croplands, rangelands, forests, and wildlands (Ellis and Ramankutty 2008b). These data are commonly used for ecological applications and provide global land use information during a specific time frame, useful for studying human–environment interactions. We processed these maps in QGIS 3.32 to calculate the percentage He. harmoniae records in each biome.
Finally, to map the spread of He. harmoniae and Ha. axyridis, both distributions were overlaid using Python’s plotly geo package (Sievert et al. 2021). The global distribution of Ha. axyridis was retrieved from Brown et al. (2011), Orlova-Bienkowskaja et al. (2015), Gorczak et al. (2016), Camacho-Cervantes et al. (2017), Ceryngier and Romanowski (2017), Biranvand et al. (2019), Hiller and Haelewaters (2019), and iNaturalist.
Results
Records of Hesperomyces harmoniae
A total of 4,053 photos were manually checked for accuracy of the identity of the fungus. A total of 3,286 records of He. harmoniae were collected from CS networks, organizations, societies, and online platforms: 2,987 records from iNaturalist, 198 from naturgucker.de, 88 from NDFF, 9 from Flickr, 2 from Ukrainian Biodiversity Information Network, and 1 from Projekt LIFE ARTEMIS and National Biodiversity Network each. In addition, 92 collections of Ha. axyridis infected with He. harmoniae were retrieved from the literature, excluding records covered in the sources above (Garcés and Williams 2004; Riddick and Schaefer 2005; Riddick 2006, 2010; Harwood et al. 2006a, b; Nalepa and Weir 2007; Riddick and Cottrell 2010; Steenberg and Harding 2010; De Kesel 2011; Haelewaters and De Kesel 2011; Cottrell and Riddick 2012; Herz and Kleespiel 2012; Haelewaters et al. 2012a, b; 2014, 2015, 2016, 2017, 2018a, b, 2019, 2022a, b; Ceryngier and Twardowska 2013; Ceryngier et al. 2013; Pfliegler 2014; Raak-van den Berg et al. 2014; Cornejo and González 2015; Gorczak et al. 2016; van Wielink 2017; López-Arroyo et al. 2018; Orlova-Bienkowskaja et al. 2018; Fiedler and Nedvěd 2019; Crous et al. 2021; Knapp et al. 2022). Those collections consisted of one to hundreds of infected ladybirds each, together accounting for 4,559 individual records. All curated records with metadata are available in GitHub: https://github.com/dannyhaelewaters/teamlaboul/tree/main/beetlehangers (in the Data directory).
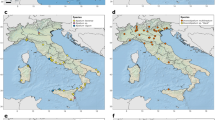
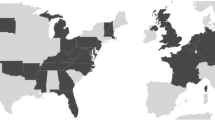
Interactive distribution maps of He. harmoniae in the USA and Europe are available at https://beetlehangers.org/ladybirdproject/ladybird-distributions/. Records are colored by source, and metadata of each record are available by clicking on the icon. Choropleth maps of records of He. harmoniae in the world, the USA and Europe are shown in Figs. 2 and 3. Slovenia and Ukraine represent new country records for He. harmoniae since its formal description by Haelewaters et al. (2022b). Within the USA, new state records are reported from California, Colorado, Delaware, Illinois, Indiana, Iowa, Kansas, Louisiana, Maine, Minnesota, Missouri, South Carolina, Texas, and Washington.
The accumulated number of records of He. harmoniae over time are shown in Fig. 4. When multi-year records in publications were presented in a way that made it impossible to count the number of specimens by year, the first year of observations was used in the plot. Hesperomyces harmoniae was first observed on museum specimens of Ha. axyridis collected in the 1930s in China (Haelewaters et al. 2014) but these records are not shown in the plot. The first field-observed record of He. harmoniae dates from May 2002 (Virginia, USA; Haelewaters et al. 2017). While the number of records retrieved from the literature is higher, the number of CS records is making a steep increase with two thirds of CS observations having been made in the last three years alone (Fig. 4). Using the anthropogenic biome dataset from Ellis and Ramankutty (2008a), we found that He. harmoniae was most recorded in the urban biome (43.2%), followed by the residential rainfed mosaic biome (19.4%), rainfed mosaic villages (13.7%), and dense settlements (10.7%) (Fig. 5). Records of He. harmoniae were very rare in other biomes, ranging from 4.6% in residential irrigated cropland to 0% in cropped & pastoral villages, remote croplands, populated rangelands, and wild forests.
Total cumulative observations of curated Hesperomyces harmoniae records over time, with differentiation between citizen science records (in green) and literature records (in blue). Relevant events are indicated: 1, first record of He. harmoniae in the USA (Virginia); 2, first record of He. harmoniae in Europe (Belgium); 3, launch of iNaturalist; 4, first record of He. harmoniae in Africa (South Africa); 5, first record of He. harmoniae in South America (Ecuador); 6, formal description of He. harmoniae. Color scheme from https://colorbrewer2.org by C.A. Brewer, Department of Geography, Pennsylvania State University
Records of Hesperomyces harmoniae across anthropogenic biomes, colored by group (Ellis and Ramankutty 2008b). Shown is the percentage of records per each biome
Harmonia axyridis is currently present in 86 countries and 6 continents (Fig. 6; Supplementary file S3). Compared to previous literature, this study adds seven countries to the global distribution of Ha. axyridis: El Salvador (North America), Bolivia (South America), Estonia, Finland (Europe), Armenia (Eurasia), and Algeria and Botswana (Africa). Hesperomyces harmoniae is present in 33 countries and 5 continents (Fig. 6). Since its formal description (Haelewaters et al. 2022b), He. harmoniae has been reported from Slovenia and Ukraine. In Africa, He. harmoniae has only been observed in South Africa. It has not been observed in the Middle East, Central Asia, southeastern Asia, Antarctica, and Oceania. Compared with Ha. axyridis, the distribution of He. harmoniae encompasses fewer countries in South America, Europe, Africa, and Asia.
The distributions of both Harmonia axyridis and Hesperomyces harmoniae, by country. Countries in purple have confirmed records of Ha. axyridis. Countries in green have confirmed records of He. harmoniae-infected Ha. axyridis. Countries in grey have no confirmed records of either He. harmoniae or Ha. axyridis. Note that Python’s plotly geo package uses ISO 3166–1 alpha-3 country codes and thus while He. harmoniae is only known from England, the whole United Kingdom (also including North Ireland, Scotland, and Wales) is shown in green. Color scheme from https://colorbrewer2.org by C.A. Brewer, Department of Geography, Pennsylvania State University
Records of Hesperomyces spp.
During curation of records, 133 records were separated; only those of He. harmoniae were retained in the final Beetlehangers dataset. Of the 133 separated records, eight represent new country and island records of He. virescens s.l. These are: Vanuatu on Olla v-nigrum (iNaturalist #113191012, #118881273); Martinique on Cladis nitidula (#148527333); Peru on Cycloneda sanguinea (#55402948) and Psyllobora sp. (#168800632); Haiti on Cycloneda sanguinea (#69453498); Puerto Rico on Cycloneda sanguinea (#179605779); Cuba on Cycloneda sanguinea (#17474775). Additionally, seven new country and island records of Hesperomyces spp. are reported from Australia on Cryptolaemus montrouzieri (iNaturalist #172129139) and Rhyzobius lophanthae (#2648522), Bermuda on Scymnus loewii (#115450130), and New Zealand on Rhyzobius fagus (#1050649, #170578845) and R. forestieri (#159954218, #170581377).
Discussion
Beetlehangers.org, a new resource for Laboulbeniales research
Already in the nineteenth century, Laboulbeniales microfungi were colloquially referred to as “beetle hangers” (Cooke 1892). This is where the name stems from for our website and data archive with records of He. harmoniae from published literature, online databases, and citizen science initiatives: Beetlehangers.org. The online interactive distribution maps of He. harmoniae will be updated when new records are contributed and corrected when an error in data entry is reported to the corresponding author. New records will be quality-screened based on the criteria presented above before being added to the Beetlehangers dataset. With Beetlehangers.org, we hope to (1) promote research on this common ectoparasite of Ha. axyridis (Haelewaters et al. 2018a, 2022b), (2) increase public knowledge and interest in the utility of CS records of parasitic fungi, and (3) continue to increase records including in poorly sampled regions with the help of targeted social media campaigns, such as the one we did in Europe in collaboration with local researchers.
While Beetlehangers.org was initially established in 2018 as a small-scale project for He. harmoniae records, we now intend the website to become a resource of Laboulbeniales knowledge for the general audience as well as an open-access data repository. For instance, in the past few years, Laboulbeniales on bat flies (Diptera, Nycteribiidae and Streblidae) have garnered some attention (e.g., Dogonniuck et al. 2019; Szentiványi et al. 2019; Liu et al. 2020) and a dataset of bat–bat fly–Laboulbeniales interactions is under construction (de Groot et al. 2020). Additionally, a database of all known species of Laboulbeniomycetes along with their holotype location and other geographic records was created (Haelewaters et al. 2024) and will continue to be updated on Beetlehangers.org. Since Laboulbeniales infect a swathe of different arthropod hosts, there are many opportunities for projects focusing on different host groups. We envision Beetlehangers.org to become a home to provide public outreach and share research results of different projects related to Laboulbeniales microfungi.
Records of Hesperomyces harmoniae over time and space
This study aimed at investigating observations of He. harmoniae on a spatiotemporal scale. The usefulness of CS platforms to track species over time and space may be limited due to geospatial biases inherent to CS-based projects (see below). However, they do hold a lot of power for analytical work given the high number of records that cannot be achieved by traditional research (Chandler et al. 2017; Adler et al. 2020). For example, data from CS projects have previously been used for questions related to geographic distributions (van Strien et al. 2013), population trends (Horns et al. 2018), and the spread of invasive species (Brown et al. 2001; Howard et al. 2022). To our knowledge, our Beetlehangers dataset is the first to track an organism alongside its invasive host.
Our dataset shows a steep increase in the number of CS records since 2019 (Fig. 4). First, CS apps and online platforms are becoming more user-friendly and accessible to non-professionals, leading to more records being amassed through them (Boakes et al. 2016; Petersen et al. 2021; Price-Jones et al. 2022). Second, Laboulbeniales research – and Hesperomyces research in particular – has seen a recent surge by D.H. and collaborators, associated with active efforts to perform public outreach efforts and to adopt open science practices. Third, many CS initiatives have involved the public in the recording of ladybirds, and in particular Ha. axyridis since its arrival as an IAS around the world (Grez et al. 2010; Gardiner et al. 2012; Brown and Roy 2018; Werenkraut et al. 2020; Skuhrovec et al. 2021). The combination of these three factors may have resulted in a domino effect, whereby increasingly more people become aware of both the ladybird and its associated fungus and start recording their observations.
We used the complete Beetlehangers dataset to create visualizations of the geospatial distribution of records of He. harmoniae in relation to its host (Fig. 6). As He. harmoniae is an obligate parasite – i.e., it cannot survive without its host – its distribution is expected to fall completely within the distribution of Ha. axyridis. However, He. Harmoniae’s range reaches neither the highest nor the lowest latitudes of the range of Ha. axyridis. These results indicate that the habitable climatic range of the ladybird may be wider than that of the fungus. A recent field-based study showed that climate factors did not have an effect on parasitism of Ha. axyridis by He. harmoniae but this study was limited to data from Central Europe (Haelewaters et al. 2022a). Laboratory experiments did show that there may be an optimal range of humidity and temperature for the fungus to grow (M.D. de Groot and B. Santamaria unpubl. data).
Challenges related to citizen science projects
Hundreds of biodiversity-related CS projects have been initiated during just the last decade (Theobald et al. 2015), many of which focus on IAS (Silvertown 2009; Adriaens et al. 2015; Brown and Roy 2018; Roy et al. 2018; Howard et al. 2022; Price-Jones et al. 2022). Among the general challenges in designing and running CS projects are the management of data quality (data curation), the recruitment and retaining of participants, and taxonomic and geospatial biases. We will discuss some of these biases in light of our work with Laboulbeniales microfungi and He. harmoniae more specifically.
Taxonomic biases
How to overcome taxonomic biases is an important challenge that becomes even more strenuous when dealing with ectoparasitic microfungi (e.g., Gonçalves et al. 2021; Lofgren and Stajich 2021). Public records are heavily skewed by organismal preference, as larger, more charismatic, and more easily identified species represent the majority of observed species in CS platforms (Kosmala et al. 2016; Járic et al. 2020). Our interactions with the 41 CS networks, national organizations, and societies that we had contacted for records of He. harmoniae underlined this taxonomic bias. We only received records from five of these sources (one additional source referred to Angelidou et al. 2023), and the number of records was relatively low: 290 combined records, contributed mostly by naturgucker.de and NDFF, versus 2,987 records from iNaturalist. Future CS projects focusing on Laboulbeniales may benefit from a combination of infographics, a dedicated project website, active social media outreach, instructional videos, and sharing of intermediate and final research results with the participants. Such a broad communication strategy along with an active training component can help to engage and motivate participants (Jordan et al. 2012; Maund et al. 2020). In general, microfungal ectobionts in the order Laboulbeniales represent a group of organisms that need advanced training and equipment for accurate species identification, which makes them harder to use in CS projects. Laboulbeniales can also “fall between two stools”, where entomologists do not record the presence of fungal thalli, and mycologists do not routinely collect Laboulbeniales hosts. For this reason, targeted initiatives such as Beetlehangers.org can increase awareness and appreciation of this understudied fungal group. In the past, projects like the Big Seaweed Search (Brodie et al. 2023) and the FunDiS Northeast Rare Fungi Challenge (Fungal Diversity Survey 2022) have shone a light on neglected taxa with the help of citizen scientists.
Geospatial biases
Observations from citizen scientists are distributionally skewed towards temperate countries and easily accessible localities (Tiago et al. 2017), thereby mirroring the biases from traditional research projects (Rodrigues et al. 2010; Hortal et al. 2015; Meyer et al. 2015; Quandt and Haelewaters 2021). For Laboulbeniales, several species have extremely disjunct distribution patterns that are attributed to a lack of study across large geographical areas (Haelewaters et al. 2024). This Wallacean shortfall (sensu Lomolino 2004) helps to explain the uneven and patchy distribution of He. harmoniae as seen in Fig. 6. Our analysis of He. harmoniae records across anthropogenic biomes suggests that He. harmoniae is mostly present in areas with substantial human populations (Fig. 5). Both these observations are more likely to reflect the distribution of citizen scientists and active researchers rather than the actual species distribution. However, there may also be biological factors involved. Welch et al. (2001) reported that the parasite prevalence of Hesperomyces on Adalia bipunctata was highest in the city center of London and decreased rapidly towards the outskirts. The authors attributed this to the “urban island heat effect”, which shortens winters and may promote aphid growth – increasing survival of ladybirds and thereby providing more opportunities for transmission of ascospores. An alternative explanation may come from the increased number of artificial shelters (attics, basements, windowsills, etc.) in city centers (Adriaens et al. 2008; Roy and Brown 2015) that results in more opportunities for spore transfer among ladybirds as they form dense aggregations in winter (Riddick and Shaefer 2005; Nalepa and Weir 2007).
Interesting records of Hesperomyces virescens sensu lato and Hesperomyces spp.
The genus Hesperomyces has recently received considerable attention. The main contributing reason for this is He. harmoniae being commonly observed as an ectoparasite of the globally invasive Ha. axyridis, and potential biocontrol applications for this fungus (Roy et al. 2011; Haelewaters et al. 2020, 2022a; de Groot and Haelewaters 2022). Hesperomyces harmoniae, however, was only formally described as a species in late 2022 (Haelewaters et al. 2022b). Previously, He. harmoniae was hidden under the name Hesperomyces virescens (Haelewaters et al. 2018a). As the “Beetle Hangers” project in iNaturalist was conceived before this taxonomic decision, the initial focus was He. virescens which, as mentioned above, is a complex of multiple species each with different hosts. As a consequence, various records of He. virescens sensu lato (s.l.), i.e., observations of the fungus of which the host was not Ha. axyridis, were recorded alongside He. harmoniae. Some turned out to be new records for the country or island in which they were found. Even though they do not represent species records in the narrow sense, these mark the first time that a member of Hesperomyces was reported in these localities, including Cuba, Haiti, Martinique, Puerto Rico (North America), Peru (South America), and Vanuatu (Oceania).
Hesperomyces on Clavis nitidula from Martinique in the Caribbean Sea represents the first record of Hesperomyces with this host genus. Clavis is part of the Chilocorini tribe, as are the genera Chilocorus (Ch. stigma is the host of He. virescens sensu stricto; Thaxter 1891) and Parexochomus (host to He. parexochomi; Crous et al. 2021). A few contributed records of Hesperomyces were associated with hosts in the tribes Coccidulini (Cryptolaemus montrouzieri) and Scymnini (Rhyzobius spp.). The only Coccidulini representative found with Hesperomyces thus far is Azya orbigera from Panama, host to an undescribed species of Hesperomyces. Thus, the records of Hesperomyces sp. on Rhyzobius fagus and R. forestieri from New Zealand and R. lophanthae from Australia are the first ones of Hesperomyces associated with this host genus. To date, Scymnini hosts have been found to be associated with He. coccinelloides and He. papuanus. Hesperomyces papuanus was described on a Cryptolaemus affinis from Papua New Guinea (Majewski and Sugiyama 1985). An integrative taxonomy approach is needed to confirm whether this is an actual species. Since photographic records cannot be identified with confidence to species level, the above records remain unidentified until more material is observed and collected for taxonomic investigation.
Towards increased engagement of citizen scientists for parasitology research
We extracted records of He. harmoniae from online databases, among other sources, and used these to identify anthropogenic biomes of high He. harmoniae abundance; our study is an example of how the public can contribute to parasitology research. Likewise, Doherty et al. (2021) pointed out that internet data from non-professionals were reliable to chart the spatial occurrence, temporal trends, and host usage patterns of an easily observed parasite. To date, most CS projects have focused on single-species observations – participants are asked to make observations of a single organism and upload photographs to an app or online platform. However, to gain a more complete picture of species interactions and ecological networks, projects should shift towards multi-species observations. Groom et al. (2021) refer to projects collecting data on species interactions through the public as “next-level” CS and point out that their success depends on eliciting an emotional connection in people, not overcomplicating project aims, sharing results, and designing research questions that are relevant to the participants’ daily lives. To increase the resolution of our Beetlehangers dataset, we propose to incorporate observations of Hesperomyces into existing CS apps and programs for ladybirds, such as the UK Ladybird Survey (Brown and Roy 2018) and the European Ladybirds smartphone app (Skuhrovec et al. 2021).
Data availability
The data that support the findings of this study are available in the Supplementary Information section of this article. All curated records of He. harmoniae with metadata are available in GitHub (https://github.com/dannyhaelewaters/teamlaboul/tree/main/beetlehangers/Data).
References
Adler FR, Green AM, Şekercioğlu CH (2020) Citizen science in ecology: a place for humans in nature. Ann NY Acad Sci 1469(1):52–64. https://doi.org/10.1111/nyas.14340
Adriaens T, Martin S, y Gomez G, Maes D, (2008) Invasion history, habitat preferences and phenology of the invasive ladybird Harmonia axyridis in Belgium. Biocontrol 53:69–88. https://doi.org/10.1007/s10526-007-9137-6
Adriaens T, Sutton-Croft M, Owen K, Brosens D, van Valkenburg J, Kilbey D, Groom Q, Ehmig C, Thürkow F, Van Hende P, Schneider K (2015) Trying to engage the crowd in recording invasive alien species in Europe: experiences from two smartphone applications in northwest Europe. Manag Biol Invasions 6(2):215–225. https://doi.org/10.3391/mbi.2015.6.2.12
Ando K, Pousson E (2022) Package ‘FlickrAPI’. Comprehensive R Archive Network (CRAN). https://cran.r-project.org/web/packages/FlickrAPI/FlickrAPI.pdf. Accessed 29 Sep 2023
Angelidou I, Demetriou J, Christou M, Koutsoukos E, Kazilas C, Georgiades P, Kalaentzis K, Κontodimas DC, Groom Q, Roy HE, Martinou AF (2023) Establishment and spread of the invasive ladybird Harmonia axyridis (Coleoptera: Coccinellidae) in Greece: based on contributions from citizen scientists. Biol Invasions 25:889–900. https://doi.org/10.1007/s10530-022-02955-8
Biranvand A, Nedvěd O, Tomaszewska W, Al Ansi AN, Fekrat L, Haghghadam ZM, Khormizi MZ, Noorinahad S, Şenal D, Shakarami J, Haelewaters D (2019) The genus Harmonia (Coleoptera, Coccinellidae) in the Middle East. Acta Entomol Mus Natl Pragae 59(1):163–170. https://doi.org/10.2478/aemnp-2019-0014
Boakes EH, McGowan PJK, Fuller RA, Chang-qing D, Clark NE, O’Connor K, Mace GM (2010) Distorted views of biodiversity: spatial and temporal bias in species occurrence data. PLoS Biol 8(6):e1000385. https://doi.org/10.1371/journal.pbio.1000385
Boakes EH, Gliozzo G, Seymour V, Harvey M, Smith C, Roy DB, Haklay M (2016) Patterns of contribution to citizen science biodiversity projects increase understanding of volunteers’ recording behaviour. Sci Rep 6(1):33051. https://doi.org/10.1038/srep33051
Bradter U, Mair L, Jönsson M, Knape J, Singer A, Snäll T (2018) Can opportunistically collected citizen science data fill a data gap for habitat suitability models of less common species? Methods Ecol Evol 9(7):1667–1678. https://doi.org/10.1111/2041-210X.13012
Brodie J, Kunzig S, Agate J, Yesson C, Robinson L (2023) The big seaweed search: evaluating a citizen science project for a difficult to identify group of organisms. Aquat Conserv Mar Freshw Ecosyst 33(1):44–55. https://doi.org/10.1002/aqc.3903
Brown PMJ, Roy HE (2018) Native ladybird decline caused by the invasive harlequin ladybird Harmonia axyridis: evidence from a long-term field study. Insect Conserv Divers 11:230–239. https://doi.org/10.1111/icad.12266
Brown WT, Krasny ME, Schoch N (2001) Volunteer monitoring of non-indigenous invasive plant species in the Adirondack Park, New York, USA. Nat Areas J 21(2):189–196
Brown PMJ, Adriaens T, Bathon H, Cuppen J, Goldarazena A, Hägg T, Kenis M, Klausnitzer BEM, Kovář I, Loomans AJM, Majerus MEN, Nedvěd O, Pedersen J, Rabitsch W, Roy HE, Ternois V, Zakharov IA, Roy DB (2008) Harmonia axyridis in Europe: spread and distribution of a non-native coccinellid. Biocontrol 53(1):5–21. https://doi.org/10.1007/s10526-007-9132-y
Camacho-Cervantes M, Ortega-Iturriaga A, Del-Val E (2017) From effective biocontrol agent to successful invader: the harlequin ladybird (Harmonia axyridis) as an example of good ideas that could go wrong. PeerJ 5:e3296. https://doi.org/10.7717/peerj.3296
Ceryngier P, Romanowski J (2017) Harmonia axyridis (Pallas, 1773) (Coleoptera: Coccinellidae) and its parasite in south-western Bulgaria and Northern Greece. BioInvasions Rec 6(4):307–310. https://doi.org/10.3391/bir.2017.6.4.02
Ceryngier P, Twardowska K (2013) Harmonia axyridis (Coleoptera: Coccinellidae) as a host of the parasitic fungus Hesperomyces virescens (Ascomycota: Laboulbeniales, Laboulbeniaceae): a case report and short review. Eur J Entomol 110(4):549–557. https://doi.org/10.14411/eje.2013.075
Ceryngier P, Roy HE, Poland RL (2012) Natural enemies of ladybird beetles. In: Hodek I, van Emden HF, Honek A (eds) Ecology and behaviour of the ladybird beetles (Coccinellidae). Wiley-Blackwell, Chichester
Ceryngier P, Romanowski J, Szymańska J, Galanciak M, Bardzińska M, Romanowski M (2013) Population of the invasive harlequin ladybird Harmonia axyridis (Coleoptera: Coccinellidae) from Pelješac Peninsula, southern Croatia. Stud Ecol Bioeth 11(4):79–91
Ceryngier P, Nedvěd O, Grez AA, Riddick EW, Roy HE, San Martin G, Steenberg T, Veselý P, Zaviezo T, Zúñiga-Reinoso Á, Haelewaters D (2018) Predators and parasitoids of the harlequin ladybird, Harmonia axyridis, in its native range and invaded areas. Biol Invasions 20(4):1009–1031. https://doi.org/10.1007/s10530-017-1608-9
Chandler M, See L, Copas K, Bonde AMZ, López BC, Danielsen F, Legind JK, Masinde S, Miller- Rushing AJ, Newman G, Rosemartin A, Turak E (2017) Contribution of citizen science towards international biodiversity monitoring. Biol Conserv. https://doi.org/10.1016/j.biocon.2016.09.004
Cooke MC (1892) Vegetable wasps and plant worms: a popular history of entomogenous fungi, or fungi parasitic upon insects. Society for Promoting Christian Knowledge, London. https://doi.org/10.5962/bhl.title.34922
Cornejo X, González G (2015) Contribución al conocimiento de la fauna entomológica de los manglares: Olla roatanensis Vandenberg y Cheilomenes sexmaculata Fabricius, dos nuevos registros de Coleoptera: Coccinellidae para ecuador y perú. Rev Cient Cienc Nat Ambient 8(2):76–80
Cottrell TE, Riddick EW (2012) Limited transmission of the ectoparasitic fungus Hesperomyces virescens between lady beetles. Psyche 2012:814378. https://doi.org/10.1155/2012/814378
Crous PW, Osieck ER, Jurjević Ž, Boers J, van Iperen AL, Starink-Willemse M, Dima B, Balashov S, Bulgakov TS, Johnston PR, Morozova OV, Pinruan U, Sommai S, Alvarado P, Decock CA, Lebel T, McMullan-Fisher S, Moreno G, Shivas RG, Zhao L, Abdollahzadeh J, Abrinbana M, Ageev DV, Akhmetova G, Alexandrova AV, Altés A, Amaral AGG, Angelini C, Arenas F, Asselman P, Badali F, Baghela A, Bañares A, Barreto RW, Baseia IG, Bellanger JM, Berraf-Tebbal A, Biketova AYu, Bukharova NV, Burgess TI, Cabero J, Câmara MPS, Cano-Lira JF, Ceryngier P, Chávez R, Cowan D, de Lima AF et al (2021) Fungal Planet description sheets: 1284–1382. Persoonia 47:178–374. https://doi.org/10.3767/persoonia.2021.47.06
de Groot MD, Haelewaters D (2022) Double infections of the invasive ladybird Harmonia axyridis. Front Ecol Evol 10:756972. https://doi.org/10.3389/fevo.2022.756972
de Groot MD, Dumolein I, Hiller T, Sándor AD, Szentiványi T, Schilthuizen M, Aime MC, Verbeken A, Haelewaters D (2020) On the fly: tritrophic associations of bats, bat flies, and fungi. J Fungi 6(4):361. https://doi.org/10.3390/jof6040361
De Kesel A (2011) Hesperomyces (Laboulbeniales) and coccinellid hosts. Sterbeeckia 30:32–37
Dickinson JL, Zuckerberg B, Bonter DN (2010) Citizen science as an ecological research tool: challenges and benefits. Annu Rev Ecol Evol Syst 41:149–172. https://doi.org/10.1146/annurev-ecolsys-102209-144636
Dogonniuck AE, Squires TJ, Weir A (2019) Studies on Dimorphomyceteae I. New species of Nycteromyces and Dimeromyces (Laboulbeniales) on bat flies (Streblidae). Mycologia 111(1):118–126. https://doi.org/10.1080/00275514.2018.1538279
Doherty JF, Filion A, Bennett J, Bhattarai UR, Chai X, de Angeli DD, Donlon E, Jorge F, Milotic M, Park E, Sabadel AJ, Thomas LJ, Poulin R (2021) The people vs science: can passively crowdsourced internet data shed light on host–parasite interactions? Parasitology 148(11):1313–1319. https://doi.org/10.1017/S0031182021000962
Ellis EC, Ramankutty N (2008a) Anthropogenic Biomes of the World, version 1. NASA Socioeconomic Data and Applications Center, Palisades, New York. https://doi.org/10.7927/H4H12ZXD. Accessed 22 Sep 2023
Ellis EC, Ramankutty N (2008b) Putting people in the map: anthropogenic biomes of the world. Front Ecol Environ 6(8):439–447. https://doi.org/10.1890/070062
Encarnação J, Teodósio MA, Morais P (2021) Citizen science and biological invasions: a review. Front Environ Sci 8:602980. https://doi.org/10.3389/fenvs.2020.602980
Fiedler L, Nedvěd O (2019) Fifty shades of the harlequin ladybird and a sexually transmitted fungus. J Insect Sci 19(6):10. https://doi.org/10.1093/jisesa/iez107
Garcés S, Williams R (2004) First record of Hesperomyces virescens Thaxter (Laboulbeniales: Ascomycetes) on Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae). J Kansas Entomol Soc 77(2):156–158. https://doi.org/10.2317/0304.18.1
Gardiner MM, Allee LL, Brown PMJ, Losey JE, Roy HE, Smyth RR (2012) Lessons from lady beetles: accuracy of monitoring data from US and UK citizen-science programs. Front Ecol Environ 10(9):471–476. https://doi.org/10.1890/110185
Gonçalves SC, Haelewaters D, Furci G, Mueller GM (2021) Include all fungi in biodiversity goals. Science 373(6553):403. https://doi.org/10.1126/science.abk1312
Gorczak M, Tischer M, Pawłowska J, Wrzosek M (2016) First record of Hesperomyces virescens (Laboulbeniales, Ascomycota) on Harmonia axyridis (Coccinellidae, Coleoptera) in Poland. Acta Mycol 51(1):1071. https://doi.org/10.5586/am.1071
Grez A, Zaviezo T, González G, Rothmann S (2010) Harmonia axyridis in Chile: a new threat. Int J Agric Nat Resour 37(3):145–149. https://doi.org/10.4067/S0718-16202010000300013
Grez AA, Zaviezo T, Roy HE, Brown PMJ, Bizama G (2016) Rapid spread of Harmonia axyridis in Chile and its effects on local coccinellid biodiversity. Divers Distrib 22(9):982–994. https://doi.org/10.1111/ddi.12455
Groom Q, Pernat N, Adriaens T, de Groot M, Jelaska SD, Marčiulynienė D, Martinou AF, Skuhrovec J, Tricarico E, Wit EC, Roy HE (2021) Species interactions: next-level citizen science. Ecography 44(12):1781–1789. https://doi.org/10.1111/ecog.05790
Haelewaters D, De Kesel A (2011) Laboulbeniales van De Kaaistoep. In: Cramer T, van Wielink P (eds) Natuurstudie in De Kaaistoep. Verslag 2010, 16e onderzoeksjaar. NV Tilburgsche Waterleiding-Maatschappij. KNNV-afdeling Tilburg & Natuurmuseum Brabant, Tilburg
Haelewaters D, Nuytinck J, De Kesel A (2012a) Laboulbeniales (Fungi, Ascomycota) in Nederland: een introductie. Natuurh Maandbl 101(5):88–93
Haelewaters D, van Wielink P, van Zuijlen JW, Verbeken A, De Kesel A (2012b) New records of Laboulbeniales (Fungi, Ascomycota) for The Netherlands. Entomol Ber 72(3):175–183
Haelewaters D, Comont RF, Zhao SY, Pfster DH (2014) Hesperomyces virescens (Fungi, Ascomycota, Laboulbeniales) attacking Harmonia axyridis (Coleoptera, Coccinellidae) in its native range. Chinese Sci Bull 59(5–6):528–532. https://doi.org/10.1007/s11434-014-0283-9
Haelewaters D, Zhao SY, De Kesel A, Royer IR, Handlin RE, Farrell BD, Pfster DH (2015) Laboulbeniales (Ascomycota) of the Boston Harbor Islands I: species parasitizing Coccinellidae and Staphylinidae. Northeast Nat 22(3):459–477. https://doi.org/10.1656/045.022.0304
Haelewaters D, Minnaar IA, Clusella-Trullas S (2016) First finding of the parasitic fungus Hesperomyces virescens (Laboulbeniales) on native and invasive ladybirds (Coleoptera, Coccinellidae) in South Africa. Parasite 23:5. https://doi.org/10.1051/parasite/2016005
Haelewaters D, Zhao SY, Clusella-Trullas S, Cottrell TE, De Kesel A, Fiedler L, Herz A, Hesketh H, Hui C, Kleespies RG, Losey JE, Minnaar IA, Murray KM, Nedvěd O, Pfliegler WP, Raak-van den Berg CL, Riddick EW, Shapiro-Ilan DI, Smyth RR, Steenberg T, van Wielink PS, Viglášová S, Zhao Z, Ceryngier P, Roy HE (2017) Parasites of Harmonia axyridis: current research and perspectives. Biocontrol 62(3):355–371. https://doi.org/10.1007/s10526-016-9766-8
Haelewaters D, De Kesel A, Pfister DH (2018a) Integrative taxonomy reveals hidden species within a common fungal parasite of ladybirds. Sci Rep 8(1):15966. https://doi.org/10.1038/s41598-018-34319-5
Haelewaters D, Hiller T, Gorczak M, Pfster D (2018b) Influence of elytral color pattern, size, and sex of Harmonia axyridis (Coleoptera, Coccinellidae) on parasite prevalence and intensity of Hesperomyces virescens (Ascomycota, Laboulbeniales). InSects 9(2):67. https://doi.org/10.3390/insects9020067
Haelewaters D, De Kesel A, Gorczak M, Bao K, Gort G, Zhao SY, Pfster DH (2019) Laboulbeniales (Ascomycota) of the Boston Harbor Islands II: species parasitizing Carabidae, and the Laboulbenia fagellata species complex. Northeast Nat 25(Sp 9):110–149. https://doi.org/10.1656/045.025.s906
Haelewaters D, Hiller T, Kemp EA, van Wielink PS, Shapiro-Ilan DI, Aime MC, Nedvěd O, Pfister DH, Cottrell TE (2020) Mortality of native and invasive ladybirds co-infected by ectoparasitic and entomopathogenic fungi. PeerJ 8:e10110. https://doi.org/10.7717/peerj.10110
Haelewaters D, Hiller T, Ceryngier P, Eschen R, Gorczak M, Houston ML, Kisło K, Knapp M, Landeka N, Pfliegler WP, ZachAime PMC, Nedvěd O (2022a) Do biotic and abiotic factors infuence the prevalence of a common parasite of the invasive alien ladybird Harmonia axyridis? Front Ecol Evol 10:773423. https://doi.org/10.3389/fevo.2022.773423
Haelewaters D, Van Caenegem W, De Kesel A (2022b) Hesperomyces harmoniae, a new name for a common ectoparasitic fungus on the invasive alien ladybird Harmonia axyridis. Sydowia 75:53–74. https://doi.org/10.12905/0380.sydowia75-2022-053
Haelewaters D, Matthews TJ, Wayman JP, Cazabonne J, Heyman F, Quandt CA, Martin TE (2024) Biological knowledge shortfalls impede conservation efforts in poorly studied taxa—A case study of Laboulbeniomycetes. J Biogeogr 51(1):29–39. https://doi.org/10.1111/jbi.14725
Harwood JD, Ricci C, Romani R, Obrycki JJ (2006a) Historic prevalence of a laboulbenialean fungus infecting introduced coccinellids in the United States. Antenna 30(2):74–79
Harwood JD, Ricci C, Romani R, Pitz KM, Weir A, Obrycki JJ (2006b) Prevalence and association of the laboulbenialean fungus Hesperomyces virescens (Laboulbeniales: Laboulbeniaceae) on coccinellid hosts (Coleoptera: Coccinellidae) in Kentucky, USA. Eur J Entomol 103(4):799–804. https://doi.org/10.14411/eje.2006.109
Herz A, Kleespiel RG (2012) Occurrence of natural enemies in different populations of the invasive ladybird Harmonia axyridis (Pallas, 1771) (Coleoptera, Coccinellidae) in Germany. Mitt Dtsch Ges Allg Angew Entomol 18:201–206
Hiller T, Haelewaters D (2019) A case of silent invasion: citizen science confirms the presence of Harmonia axyridis (Coleoptera, Coccinellidae) in Central America. PLoS ONE 14(7):e0220082. https://doi.org/10.1371/journal.pone.0220082
Home R, Keller C, Nagel P, Bauer N, Hunziker M (2009) Selection criteria for flagship species by conservation organizations. Environ Conserv 36(2):139–148. https://doi.org/10.1017/S0376892909990051
Hooykaas MJ, Schilthuizen M, Albers CJ, Smeets I (2022) Species identification skills predict in-depth knowledge about species. PLoS ONE 17(4):e0266972. https://doi.org/10.1371/journal.pone.0266972
Horns JJ, Adler FR, Şekercioğlu ÇH (2018) Using opportunistic citizen science data to estimate avian population trends. Biol Conserv 221:151–159. https://doi.org/10.1016/j.biocon.2018.02.027
Hortal J, de Bello F, Diniz-Filho JA, Lewinsohn TM, Lobo JM, Ladle RJ (2015) Seven shortfalls that beset large-scale knowledge of biodiversity. Annu Rev Ecol Evol Syst 46:523–549. https://doi.org/10.1146/annurev-ecolsys-112414-054400
Howard L, van Rees CB, Dahlquist Z, Luikart G, Hand BK (2022) A review of invasive species reporting apps for citizen science and opportunities for innovation. NeoBiota 71:165–188. https://doi.org/10.3897/neobiota.71.79597
Iwane T (2023) What is the data quality assessment and how do observations qualify to become "Research Grade"? iNaturalist. https://www.inaturalist.org/pages/help#quality. Accessed 22 Sep 2023
Jarić I, Courchamp F, Correia RA et al (2020) The role of species charisma in biological invasions. Front Ecol Environ 18:345–353. https://doi.org/10.1002/fee.2195
Jordahl K (2014) GeoPandas: Python tools for geographic data. https://github.com/geopandas/geopandas. Accessed 22 Sep 2023
Jordan RC, Ballard HL, Phillips TB (2012) Key issues and new approaches for evaluating citizen-science learning outcomes. Front Ecol Environ 10(6):307–309. https://doi.org/10.1890/110280
Klemann-Junior L, Villegas Vallejos MA, Scherer-Neto P, Vitule JRS (2017) Traditional scientific data vs. uncoordinated citizen science effort: a review of the current status and comparison of data on avifauna in Southern Brazil. PLoS ONE 12(12):e0188819. https://doi.org/10.1371/journal.pone.0188819
Knapp M, Řeřicha M, Haelewaters D, González E (2022) Fungal ectoparasites increase winter mortality of ladybird hosts despite limited effects on their immune system. Proc R Soc B Biol Sci 289(1971):20212538. https://doi.org/10.1098/rspb.2021.2538
Koch RL, Venette RC, Hutchison WD (2006) Invasions by Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae) in the Western Hemisphere: implications for South America. Neotrop Entomol 35(4):421–434. https://doi.org/10.1590/S1519-566X2006000400001
Kosmala M, Wiggins A, Swanson A, Simmons B (2016) Assessing data quality in citizen science. Front Ecol Environ 14(10):551–560. https://doi.org/10.1002/fee.1436
Liu J, Haelewaters D, Pfliegler WP, Page RA, Dick CW, Aime MC (2020) A new species of Gloeandromyces from Ecuador and Panama revealed by morphology and phylogenetic reconstruction, with a discussion of secondary barcodes in Laboulbeniomycetes taxonomy. Mycologia 112(6):1192–1202. https://doi.org/10.1080/00275514.2020.1781496
Lofgren LA, Stajich JE (2021) Fungal biodiversity and conservation mycology in light of new technology, big data, and changing attitudes. Curr Biol 31(19):R1312–R1325. https://doi.org/10.1016/j.cub.2021.06.083
Lombaert E, Guillemaud T, Cornuet JM, Malausa T, Facon B, Estoup A (2010) Bridgehead effect in the worldwide invasion of the biocontrol harlequin ladybird. PLoS ONE 5(3):e9743. https://doi.org/10.1371/journal.pone.0009743
Lomolino MV (2004) Conservation biogeography. In: Lomolino MV, Heaney LR (eds) Frontiers of biogeography: new directions in the geography of nature. Sinauer, Sunderland
López-Arroyo JI, Díaz-Martínez S, Rodríguez-Guerra R, Peña-Carrillo KI, Sánchez-Peña S (2018) Primer registro de ocurrencia del hongo Hesperomyces virescens Thaxter en especies de Coccinellidae (Coleoptera) en México. Control Biol 5:141–147
Majewski T, Sugiyama K (1985) Studies on the Laboulbeniomycetes of Papua New Guinea. 2. Trans Mycol Soc Japan 26:179–188
Maund PR, Irvine KN, Lawson B, Steadman J, Risely K, Cunningham AA, Davies ZG (2020) What motivates the masses: understanding why people contribute to conservation citizen science projects. Biol Conserv 246:108587. https://doi.org/10.1016/j.biocon.2020.108587
Meyer C, Kreft H, Guralnick R, Jetz W (2015) Global priorities for an effective information basis of biodiversity distributions. Nat Commun 6(1):8221. https://doi.org/10.1038/ncomms9221
Nalepa CA, Weir A (2007) Infection of Harmonia axyridis (Coleoptera: Coccinellidae) by Hesperomyces virescens (Ascomycetes: Laboulbeniales): role of mating status and aggregation behavior. J Invertebr Pathol 94(3):196–203. https://doi.org/10.1016/j.jip.2006.11.002
Nentwig W, Bacher S, Kumschick S, Pyšek P, Vilà M (2018) More than 100 worst alien species in Europe. Biol Invasions 20(6):1611–1621. https://doi.org/10.1007/s10530-017-1651-6
Orlova-Bienkowskaja MJ, Ukrainsky AS, Brown PMJ (2015) Harmonia axyridis (Coleoptera: Coccinellidae) in Asia: a re-examination of the native range and invasion to Southeastern Kazakhstan and Kyrgyzstan. Biol Invasions 17(7):1941–1948. https://doi.org/10.1007/s10530-015-0848-9
Orlova-Bienkowskaja MJ, Spiridonov SE, Butorina NN, Bieńkowski AO (2018) Coinvasion by the ladybird Harmonia axyridis (Coleoptera: Coccinellidae) and its parasites, Hesperomyces virescens (Ascomycota: Laboulbeniales) and Parasitylenchus bifurcatus (Nematoda: Tylenchida, Allantonematidae), in the Caucasus. PLoS ONE 13(11):e0202841. https://doi.org/10.1371/journal.pone.0202841
Petersen TK, Speed JD, Grøtan V, Austrheim G (2021) Species data for understanding biodiversity dynamics: the what, where and when of species occurrence data collection. Ecol Sol Evid 2:e12048. https://doi.org/10.1002/2688-8319.12048
Pfiegler WP (2014) First Hungarian record of the fungus Hesperomyces virescens (Ascomycota: Laboulbeniales), parasitic on the harlequin ladybird (Coccinellidae: Harmonia axyridis). e-Acta Nat Pannon 7:139–142
Pocock MJO, Tweddle JC, Savage J, Robinson LD, Roy HE (2017) The diversity and evolution of ecological and environmental citizen science. PLoS ONE 12(4):e0172579. https://doi.org/10.1371/journal.pone.0172579
Pocock MJO, Adriaens T, Bertolino S, Eschen R, Essl F, Hulme PE, Jeschke JM, Roy HE, Teixeira H, de Groot M (2024) Citizen science is a vital partnership for invasive alien species management and research. iScience 27(1):108623. https://doi.org/10.1016/j.isci.2023.108623
Price-Jones V, Brown PMJ, Adriaens T, Tricarico E, Farrow RA, Cardoso AC, Gervasini E, Groom Q, Reyserhove L, Schade S, Tsinaraki C, Marchante E (2022) Eyes on the aliens: citizen science contributes to research, policy and management of biological invasions in Europe. NeoBiota 78:1–24. https://doi.org/10.3897/neobiota.78.81476
Probert AF, Wegmann D, Volery L, Adriaens T, Bakiu R, Bertolino S, Essl F, Gervasini E, Groom Q, Latombe G, Marisavljevic D, Mumford J, Pergl J, Preda C, Roy HE, Scalera R, Teixeira H, Tricarico E, Vanderhoeven S, Bacher S (2022) Identifying, reducing, and communicating uncertainty in community science: a focus on alien species. Biol Invasions 24(11):3395–3421. https://doi.org/10.1007/s10530-022-02858-8
QGIS Development Team (2023) QGIS Geographic Information System. Open Source Geospatial Foundation Project (OSGeo). http://qgis.osgeo.org. Accessed 22 Sep 2023
Quandt CA, Haelewaters D (2021) Phylogenetic advances in Leotiomycetes, an understudied clade of taxonomically and ecologically diverse fungi. In: Zaragoza Ó, Casadevall A (eds) Encyclopedia of Mycology. Elsevier, Oxford
Raak-van den Berg CL, van Wielink PS, de Jong PW, Gort G, Haelewaters D, Helder J, van Lenteren JC (2014) Invasive alien species under attack: natural enemies of Harmonia axyridis in the Netherlands. Biocontrol 59(2):229–240. https://doi.org/10.1007/s10526-014-9561-3
Riddick EW (2006) Infuence of host gender on infection rate, density and distribution of the parasitic fungus, Hesperomyces virescens, on the multicolored Asian lady beetle. Harmonia Axyridis J Insect Sci 6(1):1–15. https://doi.org/10.1673/031.006.4201
Riddick EW (2010) Ectoparasitic mite and fungus on an invasive lady beetle: parasite coexistence and infuence on host survival. Bull Insectol 63(1):13–20
Riddick EW, Cottrell TE (2010) Is the prevalence and intensity of the ectoparasitic fungus Hesperomyces virescens related to the abundance of entomophagous coccinellids? Bull Insectol 63(1):71–78
Riddick EW, Schaefer PW (2005) Occurrence, density, and distribution of parasitic fungus Hesperomyces virescens (Laboulbeniales: Laboulbeniaceae) on multicolored Asian lady beetle (Coleoptera: Coccinellidae). Ann Entomol Soc Am 98(4):615–624. https://doi.org/10.1603/0013-8746(2005)098[0615:ODADOP]2.0.CO;2
Rodrigues ASL, Gray CL, Crowter BJ, Ewers RM, Stuart SN, Whitten T, Manica A (2010) A global assessment of amphibian taxonomic effort and expertise. Bioscience 60:798–806. https://doi.org/10.1525/bio.2010.60.10.6
Roy HE, Brown PMJ (2015) Ten years of invasion: Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae) in Britain. Ecol Entomol 40(4):336–348. https://doi.org/10.1111/een.12203
Roy HE, Rhule E, Harding S, Lawson Handley LJ, Poland RL, Riddick EW, Steenberg T (2011) Living with the enemy: parasites and pathogens of the ladybird Harmonia axyridis. Biocontrol 56(4):663–679. https://doi.org/10.1007/s10526-011-9387-1
Roy HE, Adriaens T, Isaac NB, Kenis M, San Martin Y, Gomez G, Onkelinx T, Brown PMJ, Hautier L, Poland RL, Roy DB, Comont RF, Eschen R, Frost R, Zindel R, Van Vlaenderen J, Nedved O, Ravn HP, Grégoire JC, de Biseau JC, Maes D (2012) Invasive alien predator causes rapid declines of native European ladybirds. Divers Distrib 18(7):717–725. https://doi.org/10.1111/j.1472-4642.2012.00883.x
Roy HE, Brown PMJ, Adriaens T, Berkvens N, Borges I, Clusella-Trullas S, Comont RF, De Clercq P, Eschen R, Estoup A, Evans EW, Facon B, Gardiner MM, Gil A, Grez AA, Guillemaud T, Haelewaters D, Herz A, Honek A, Howe AG, Hui C, Hutchison WD, Kenis M, Koch RL, Kulfan J, Lawson Handley L, Lombaert E, Loomans A, Losey J, Lukashuk AO, Maes D, Magro A, Murray KM, San Martin G, Martinkova Z, Minnaar I, Nedved O, Orlova-Bienkowskaja MJ, Rabitsch W, Ravn HP, Rondoni G, Rorke SL, Ryndevich SK, Saethre M-G, Sloggett JJ, Soares AO, Stals R, Tinsley MC, Vandereycken A, van Wielink P, Viglášová S, Zach P, Zaviezo T, Zhao Z (2016) The harlequin ladybird, Harmonia axyridis: global perspectives on invasion history and ecology. Biol Invasions 18(4):997–1044. https://doi.org/10.1007/s10530-016-1077-6
Roy HE, Hesketh H, Purse BV, Eilenberg J, Santini A, Scalera R, Stentiford GD, Adriaens T, Bacela-Spychalska K, Bass D, Beckmann KM, Bessell P, Bojko J, Booy O, Cardoso AC, Essl F, Groom Q, Harrower C, Kleespies R, Martinou AF, van Oers MM, Peeler EJ, Pergl J, Rabitsch W, Roques A, Schaffner F, Schindler S, Schmidt BR, Schönrogge K, Smith J, Solarz W, Stewart A, Stroo A, Tricarico E, Turvey KMA, Vannini A, Vilà M, Woodward S, Wynns AA, Dunn AM (2017) Alien pathogens on the horizon: opportunities for predicting their threat to wildlife. Convers Lett 10(4):477–484. https://doi.org/10.1111/conl.12297
Roy HE, Groom Q, Adriaens T, Agnello G, Antic M, Archambeau AS, Bacher S, Bonn A, Brown P, Brundu G, López BC, Cleary M, Cogălniceanu D, de Groot M, De Sousa T, Deidun A, Essl F, Pečnikar ŽF, Gazda A, Gervasini E, Glavendekic MM, Gigot G, Jelaska SD, Jeschke JM, Kaminski D, Karachle PK, Komives T, Lapin K, Lucy F, Marchante E, Marisavljevic D, Marja R, Torrijos LM, Martinou A, Matosevic D, Mifsud CM, Motiejūnaitė J, Ojaveer H, Pasalic N, Pekárik L, Per E, Pergl J, Pesic V, Pocock M, Reino L, Ries C, Rozylowicz L, Schade S, Sigurdsson S, Steinitz O, Stern N, Teofilovski A, Thorsson J, Tomov R, Tricarico E, Trichkova T, Tsiamis K, van Valkenburg J, Vella N, Verbrugge L, Vétek G, Villaverde C, Witzell J, Zenetos A, Cardoso AC (2018) Increasing understanding of alien species through citizen science (Alien-CSI). Res Ideas Outcomes 4:e31412. https://doi.org/10.3897/rio.4.e31412
Sievert C, Parmer C, Hocking T, Chamberlain S, Ram K, Corvellec M, Despouy P (2021) Package ‘plotly’ Create Interactive Web Graphics via ‘plotly.js’. Comprehensive R Archive Network (CRAN). https://cran.biotools.fr/web/packages/plotly/plotly.pdf. Accessed 22 Sep 2023
Silvertown J (2009) A new dawn for citizen science. Trends Ecol Evol 24(9):467–471. https://doi.org/10.1016/j.tree.2009.03.017
Skuhrovec J, Roy HE, Brown PMJ, Kazlauskis K, Inghilesi AF, Soares AO, Adriaens T, Roy DB, Nedvěd O, Zach P, Viglášová S, Kulfan J, Honek A, Martinkova Z (2021) Development of the European ladybirds smartphone application: a tool for citizen science. Front Ecol Evol 9:741854. https://doi.org/10.3389/fevo.2021.741854
Soares AO, Haelewaters D, Ameixa OMCC, Borges I, Brown PMJ, Cardoso P, de Groot MD, Evans EW, Grez AA, Hochkirch A, Holecová M, Honek A, Kulfan J, Lillebø AI, Martinkova Z, Michaud JP, Nedvěd O, Omkar RHE, Saxena S, Shandilya A, Sentis A, Skuhrovec J, Viglášová S, Zach P, Zaviezo T, Losey JE (2022) A roadmap for ladybird conservation and recovery. Conserv Biol 37(1):e13965. https://doi.org/10.1111/cobi.13965
Stals R, Prinsloo G (2007) Discovery of an alien invasive, predatory insect in South Africa: the multicoloured Asian ladybird beetle, Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae): research in action. S Afr J Sci 103(3):123–126
Steenberg T, Harding S (2009) The harlequin ladybird (Harmonia axyridis Pallas) in Denmark: spread and phenology during the initial phase of invasion. Entomol Meddr 77(1):27–39
Steenberg T, Harding S (2010) Entomopathogenic fungi found in field populations of the harlequin ladybird, Harmonia axyridis. Iobc/wprs Bull 58:137–141
Fungal Diversity Survey (2022) The FunDiS Northeast Rare Fungi Challenge. 2022 Species Booklet. https://drive.google.com/file/d/1SEvVYEFQ6Eup1Qkbyeto41D9LgMAYlPF/view. Accessed 26 Sep 2023
Szentiványi T, Christe P, Glaizot O (2019) Bat flies and their microparasites: current knowledge and distribution. Front Vet Sci 6:115. https://doi.org/10.3389/fvets.2019.00115
Thaxter R (1891) Supplementary note on North American Laboulbeniaceae. Proc Am Acad Arts Sci 25:261–270. https://doi.org/10.2307/20020441
Theobald EJ, Ettinger AK, Burgess HK, DeBey LB, Schmidt NR, Froehlich HE, Wagner C, HilleRisLambers J, Tewksbury J, Harsch MA, Parrish JK (2015) Global change and local solutions: tapping the unrealized potential of citizen science for biodiversity research. Biol Conserv 181:236–244. https://doi.org/10.1016/j.biocon.2014.10.021
Theune C (2022) Pycountry 22.3.5. Python Package Index (PyPI). https://pypi.org/project/pycountry/. Accessed 22 Sep 2023
Tiago P, Ceia-Hasse A, Marques TA, Capinha C, Pereira HM (2017) Spatial distribution of citizen science casuistic observations for different taxonomic groups. Sci Rep 7(1):12832. https://doi.org/10.1038/s41598-017-13130-8
Van Caenegem W, Ceryngier P, Romanowski J, Pfister DH, Haelewaters D (2023) Hesperomyces (Fungi, Ascomycota) associated with Hyperaspis ladybirds (Coleoptera, Coccinellidae): rethinking host specificity. Front Fungal Biol 3:1040102. https://doi.org/10.3389/ffunb.2022.1040102
van Wielink PS (2017) Harmonia axyridis (Coleoptera: Coccinellidae): 13 jaar gevolgd met lichtvangsten in De Kaaistoep, Noord-Brabant. Entomol Ber 77(3):97–105
van Strien AJ, van Swaay CAM, Termaat T (2013) Opportunistic citizen science data of animal species produce reliable estimates of distribution trends if analysed with occupancy models. J Appl Ecol 50(6):1450–1458. https://doi.org/10.1111/1365-2664.12158
Welch VL, Sloggett JJ, Webberley KM, Hurst GD (2001) Short-range clinal variation in the prevalence of a sexually transmitted fungus associated with urbanisation. Ecol Entomol 26(5):547–550. https://doi.org/10.1046/j.1365-2311.2001.00352.x
Werenkraut V, Baudino F, Roy HE (2020) Citizen science reveals the distribution of the invasive harlequin ladybird (Harmonia axyridis Pallas) in Argentina. Biol Invasions 22(10):2915–2921. https://doi.org/10.1007/s10530-020-02312-7
Weyman G, Butler F, McKeown S (2022) Enhancing our understanding of ladybirds in Ireland—a case study of the use of citizen science. Front Conserv Sci 3:759113. https://doi.org/10.3389/fcosc.2022.759113
Wu T (2019) Automated web scraping using Flickr API with dynamic dashboards for analysis. Medium. https://wutianhao910.medium.com/automated-web-scraping-using-flickr-api-with-dynamic-dashboards-for-analysis-9df5501d2107. Accessed 22 Sep 2023
Acknowledgements
We thank Fred Y. Pan and Fang Yu for their significant contributions to the project at its early stages, Jasmijn Hillaert for advice with the technical setup of the website, Linshan Feng for her illustrations used in the social media campaign, the numerous citizen scientists who submitted their observations to citizen science data portals, #TeamLaboul for general support, and two anonymous reviewers for insightful comments to a previous version of the manuscript.
Funding
Open access publishing supported by the National Technical Library in Prague. This study was financially supported by Research Foundation – Flanders (Fundamental Research Fellowship 1142722N to M.D.d.G., Senior Postdoctoral Fellowship 1206024N to D.H.), the Czech Science Foundation (Grant 21-06446S), and the Alien-CSI COST Action Network CA17122 funded by the Horizon 2020 Framework Programme of the European Union (Short-Term Scientific Mission to M.C.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Handling Editor: Salmah Yaakop and Heikki Hokkanen.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11829_2023_10037_MOESM1_ESM.xlsx
Supplementary file1 (XLSX 18 kb)—Overview of the 41 citizen science networks, organizations, and societies that were contacted, along with the focal country and whether records were contributed.
11829_2023_10037_MOESM3_ESM.xlsx
Supplementary file3 (XLSX 11 kb)—Table with current country records for both Ha. axyridis and He. harmoniae, with sources.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
de Groot, M.D., Christou, M., Pan, J.Y. et al. Beetlehangers.org: harmonizing host–parasite records of Harmonia axyridis and Hesperomyces harmoniae. Arthropod-Plant Interactions (2024). https://doi.org/10.1007/s11829-023-10037-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11829-023-10037-2