Abstract
Bipartite networks of flowering plants and their visitors (potential pollinators) are increasingly being used in studies of the structure and function of these ecological interactions. Whilst they hold much promise in understanding the ecology of plant–pollinator networks and how this may be altered by environmental perturbations, like land-use change and invasive species, there is no consensus about the scale at which such networks should be constructed and analysed. Ecologists, however, have emphasised that many processes are scale dependent. Here, we compare network- and species-level properties of ecological networks analysed at the level of a site, pooling across sites within a given habitat for each month of surveys, and pooling across all sites and months to create a single network per habitat type. We additionally considered how these three scales of resolution influenced conclusions regarding differences between networks according to two contrasting habitat types (urban bushland remnants and residential gardens) and the influence of honey bee abundance on network properties. We found that most network properties varied markedly depending on the scale of analysis, as did the significance, or lack thereof, of habitat type and honey bee abundance on network properties. We caution against pooling across sites and months as this can create unrealistic links, invalidating conclusions on network structure. In conclusion, consideration of scale of analysis is also important when conducting and interpreting plant–pollinator networks.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Conserving species interactions such as those between plants and pollinating insects is vital for the effective functioning of ecosystems (Tylianakis et al. 2010). The theory and understanding of ecological networks of interacting species can be advanced through analyses of bipartite networks, with practical implications for conservation (Elle et al. 2012). Analysing plant and flower visitor communities using interaction networks has the potential to enhance understandings of ecological patterns and processes (Thébault and Fontaine 2010; Burkle and Alarcón 2011; Vázquez et al. 2009). However, the validity of conclusions from network analyses may be questionable if they are analysed at inappropriate scales. The ability to compare across studies may also be hampered if studies are conducted or analysed at different scales. The question of what the appropriate scale of analysis is for a given system and study objective is an ongoing issue in ecological studies (Hewitt et al. 2001; Peterson et al. 1998; Sutherland et al. 2013). This includes both temporal and spatial aspects. There is presently no agreed upon scale at which to construct ecological networks. However, there is accumulating evidence that scale influences at least some common network indices (Schwarzt et al. 2020).
Bees are integral to many ecosystems due to their roles as pollinators (Ollerton et al. 2011; Garibaldi et al. 2013; Aizen and Feinsinger 2003; Ollerton 2021); however, there are concerns about their conservation (Potts et al. 2016). Pollinators appear to be particularly susceptible to habitat loss (Taki and Kevan 2007), suggesting that anthropogenic processes such as urban development (Güneralp et al. 2013) can lead to a reduction in flower–visitor network integrity (Aizen and Feinsinger 2003). In addition to destruction of natural habitat, introduced competitors may also cause disruptions of plant–pollinator mutualisms (Dohzono and Yokoyama 2010; Prendergast et al. 2021; Prendergast and Ollerton 2021). The topic of honey bee competition with wild pollinators has been controversial (Pyke 1999; Ollerton 2012). Consequently, if conclusions about the impact of honey bees on plant–pollinator networks are affected by the scale at which analyses are carried out, this could alter conclusions about whether honey bees are detrimentally effecting bee–plant mutualisms.
Networks constructed by pooling surveys conducted over an entire season, or by pooling plots separated by kilometres in different habitats, may be unrealistic given the short flight seasons of many wild bees (of the order of a couple of months Batley and Brandley 2014; Prendergast, 2020a, b) and flight ranges that are often less than 500 m (Zurbuchen et al. 2010). Instead, networks and the indices of network structure calculated from them may be better constructed over shorter temporal and spatial scales that coincide with the activity period and flight range of the pollinators.
Here we use a dataset of plant–bee interactions collected monthly across urbanised sites in contrasting habitat types (seven bushland remnants and seven residential gardens) over two years to analyse how the temporal and spatial scale of these networks influences conclusions about network structure. This study assessed how the scale at which networks were constructed influenced network structural properties. We compared site level, monthly (hereafter, site-level) vs. habitat level, monthly (hereafter, habitat-by-month) vs. habitat level, across months (hereafter, habitat-across-months), and finally, networks constructed by pooling across years. This was achieved by constructing bipartite networks from monthly surveys of bee–plant interactions across fourteen sites and calculating network properties. For the site-level analyses the unit of analysis was each survey. For the next, coarser, habitat-by-month scale, we pooled the data from each survey conducted in the same habitat for each month. The coarsest habitat-across-months scale pooled the surveys across all months and sites within a given habitat. By doing this, we specifically assessed how scale altered conclusions regarding the impact of habitat type (remnant vegetation vs. residential gardens) (Prendergast and Ollerton 2021) and the introduced European honey bee Apis mellifera (Prendergast and Ollerton 2022) on network properties of H’ (network generalisation), weighted connectance, NODF (nestedness based on overlap and decreasing fill), bee niche overlap, extinction slope (bee and plant level), and robustness (bee and plant level, functional complementarity, normalised degree, species strength, interaction push–pull, species specificity, and Bluthgen’s d (d’). These properties were chosen due to how they are posited to provide information about the function, structure, completeness, resilience, and vulnerability to disruption, as well as the extent of competitive and specialised interactions of the bee–plant networks (Bascompte and Jordano 2007; Borchardt et al. 2021), and we previously investigated how habitat type and introduced honey bee density influenced these metrics (Prendergast and Ollerton 2021, 2022).
We hypothesised that the level of spatio-temporal resolution at which plant–pollinator networks are constructed will alter the average value of bipartite properties calculated from these networks. More specifically, we predicted that scale would change the significance, or lack thereof, of differences in properties between plant–bee networks in bushland remnant vegetation versus residential gardens and also the influence of honey bee abundance on network properties.
Methods
Plant–bee surveys
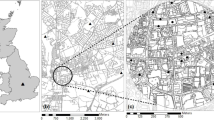
Flower visitation networks were constructed from observations of Australian native bees and the introduced European honey bee visiting flowering plants at fourteen sites in the region of Perth, which is located in the southwest Western Australia biodiversity hotspot (Laurie 2015). Seven of these sites were urban native vegetation remnants and seven were residential gardens (see Prendergast et al. 2022b; Fig. 1). Each site was a minimum of 2 km away from the closest site to ensure independence, since this is beyond the flight range of the majority of bee species (Greenleaf et al. 2007; Hofmann et al. 2020; Smith et al. 2017; Zurbuchen et al. 2010). Sites were surveyed monthly between 10:45 h and 13:45 h over the austral spring/summer from November to February in 2016/2017 (year one) and October to March in 2017/2018 (year two). Surveys were conducted over an approximately 100 × 100-m area of greenspace, encompassing part of the bushland remnant and for residential gardens, the front and backyard, and along road verges (see Prendergast and Ollerton 2021; Prendergast et al. 2022b). A single researcher, KSP, walked haphazardly within the plot observing flowering patches, with a minimum of 5 min spent at each patch, recording the visitations of all native bees and honey bees to flowers. During each survey bee specimens were also collected using an entomological sweep-net for species verification (Prendergast et al. 2020). For further details on the methodology refer to (Prendergast and Ollerton 2021, 2022; Prendergast et al. 2020, 2022b). All flowering plant species were photographed for identification. Australian native flora were identified with reference to Barrett and Tay (2016), using FloraBase, and in consultation with botanists. Weeds were identified using Hussey et al. (1997), and web-based searches and garden community forums were used for identifying other exotic flora. The bushland remnants comprised Kwongan vegetation of the Swan Coastal Plain (Lambers 2014), and residential sites had exotic garden flora and native flora (refer to online datasets in Prendergast 2020b). Each site per habitat had similar vegetation distinct from sites in the alternative habitat type—refer to the NMDS plots in Prendergast and Ollerton (2022).
From this bee–plant visitation data, bipartite networks (hereafter pollination networks) were constructed. All plant taxa were identified and analysed at the level of species (for details refer to online dataset in Prendergast 2020b). As most native bee species cannot be classified to species level by field observation alone (Prendergast et al. 2020), bees were assigned into taxonomic groups based on the lowest level of identification possible in the field: the introduced European honey bee Apis mellifera, Amegilla, Coelyoxis, Euryglossinae, Allodapini, Homalictus, Hylaeinae, Lasioglossum, Leioproctus, Lipotriches, Megachile, Trichocolletes, and Thyreus. These taxonomic groupings were chosen so that phylogenetically based categories of bee taxa that shared similar life-history traits could be used and generalised to other systems. Using this level of taxonomic resolution also provides adequate sample sizes for statistical analyses (e.g. Prendergast and Ollerton 2021, 2022; Prendergast et al. 2021). In addition, it is important to note that it is impossible to identify taxa to species level in the field, as many species within a genus are very similar and diagnostic differences between species are microscopic. Also, due to differences in the catchability of different taxa, we wanted to include the full sample of bees observed visiting flowers, so as to not bias it towards those that were easier to catch, or plant interactions where it is easier to catch bees on particular flowers (Prendergast et al. 2020). We acknowledge that further research is required to determine how taxonomic resolution of taxa in bipartite networks has a bearing on network structure.
Scales of analysis
We first constructed individual flower–visitor networks for each survey at the site scale (N (total number of networks) = 42 for year one, N = 140 for year two). Network metrics were calculated by averaging across all surveys for each habitat type. To assess the effect of scale, we then constructed networks at the habitat scale, pooling all surveys in a given habitat type each month (N = 6 for year one, N = 12 for year two), and then, at the coarsest scale, we constructed whole networks pooling all surveys in a given habitat type across all months (N = 2 for both year one and year two). We also constructed a single bipartite network for each habitat from the data pooled across all surveys over the 2 years.
Network indices
Plant–pollinator networks and associated indices were constructed using the package bipartite (Dormann et al. 2008) in R (version 3.6.2) (R Core Team 2014).
The following network-level indices were calculated for each plant–flower visitor network (see also Prendergast and Ollerton 2021, 2022):
-
Network level
-
H2’: network generalisation
-
Weighted connectance: realised proportion of possible links, weighted by network size
-
NODF (nestedness based on overlap and decreasing fill): the extent to which specialists interact with a subset of species that also interact with generalists
-
Bee niche overlap: mean similarity in interaction patterns between bees visiting flowers
-
Extinction slope (bee and plant level): simulated secondary loss of species with extinctions of species in the other level
-
Robustness ( bee and plant level): “fragility” of a level to losses in the other level
-
Functional complementarity: extent of sharing of interactions between bees
-
For each network, we calculated the following species-level indices:
-
Species level
-
Normalised degree: links per species, scaled by the number of possible partners
-
Species strength: sum of the dependencies for each plants species for a given visitor and is co-determined by the specialisation of other pollinators in the network
-
Interaction push–pull (IPP): asymmetry in dependencies between flower visitors and flowers they visit
-
Species specificity: co-efficient of variability in interactions
-
Bluthgen’s d (d’): a measure of specialisation of a flower visitor taxon in terms of its discrimination from a random sampling of plant partners
-
In the literature about bipartite networks, “species-level” is used to refer to the level of “nodes” as opposed to the entire bipartite network level of analysis (Dormann et al. 2008). In this study, as in many others (e.g. Daniels et al. 2020; Watts et al. 2016; Willcox et al. 2019), the nodes representing the higher-level taxa (here, bees) may not necessarily be represented by single species, but by higher taxonomic groupings, e.g. genus or tribe.
Pollination networks often exhibit modularity, which refers to the division of taxa into modules (compartments), where species within modules share more interactions with each other than they do with species from other modules (Olesen et al. 2007). In bipartite networks, the property of modularity reflects the extent to which nodes (i.e. ‘species’) in a compartment are more likely to be connected to each other than to other nodes of the network (Olesen et al. 2007). Modularity was calculated for each habitat type for the habitat level and whole network level using the computeModules function.
Statistical analysis
Statistical analyses were performed in R (Bates et al. 2015).
Comparison of flower–visitor network metrics and species-level metrics between urban gardens and bushland remnants were made using mixed-effects models (lme4, lmer function) for the site-level networks with site included as a random effect in the models to account for multiple surveys per site and linear models for the habitat-by-month level.
Model fit was checked visually using diagnostic plots (quantile plots) and the data transformed if model assumptions were violated.
The influence of introduced European honey bees on network-level metrics was modelled using linear mixed-effects models (package lme4) and comparing models of network-level metrics as a function of honey bee abundance (ln + 1 transformed due to extreme values). Honey bee abundance was the total number of honey bees observed visiting flowers per survey. When analysed at the coarser scales, as with the bipartite networks, honey bee abundances were summed (pooled) to correspond with the unit of analysis at which the networks were constructed.
The significance of habitat type and honey bee abundance was determined by performing an ANOVA between a model with and without the explanatory variable (Kuznetsova et al. 2017). The variable was considered to contribute to significant variation when the ANOVA produced a value of p ≤ 0.05. The proportion of variance explained by honey bee abundance (R2 values) was calculated from the models using the r.squaredGLMM function from the package MuMIn (Burnham and Anderson 2003).
Due to differences in sample size, comparisons amongst metrics between scales could not be assessed using statistical tests (e.g. in year one, site scale N = 42, habitat by month N = 6, habitat across months N = 1). In addition, statistical tests between habitat types could not be performed at the habitat-across-months scale given the sample size of one per habitat type.
Results
Network summary
For each individual network constructed per survey, network size ranged from 3 to 27 (where network size = bee taxa + plant taxa), with the number of interactions ranging from 10 to 6165 (Prendergast and Ollerton 2021). At the largest scale of analysis, the residential gardens network combining all surveys across both years had a network size of 215 and 25,355 interactions, and the bushland network had a network size of 103, and 42,942 interactions. For raw data on these bee–plant networks, refer to the online data in Prendergast (2020a, b).
Network properties across scales
Average network property values across scales
As scale increased, average H2′ remained fairly similar, within the range of 0.54 to 0.63, as did bee extinction slope, being within the range of 1.61 to 2.03 across scales (Table 1). Changes according to the scale of analysis however could be seen for most other metrics, but how they changed varied according to the metric (Table 1). For weighted connectance, as scale became coarser, weighted connectance was reduced, decreasing by almost 50% for each coarser level (Table 1). For nestedness, networks became more nested as the scale of analysis became coarser (Table 1). Extinction slope of plants also increased as the scale of analysis became coarser, approximately doubling between the levels of analysis (Table 1). In contrast, niche overlap between bees decreased by two-thirds to one half as scale became more coarser (Table 1). Functional complementarity between bee visitors strikingly increased at more coarse scales of analysis, rising in value 3- to fivefold (Table 1). Modularity was comparable between the whole-habitat and habitat-by-month scales of analysis, but did slightly decrease as the scale became finer (Table 1). Modularity could not be calculated from the site-scale networks due to insufficient sample size.
Habitat comparisons between the site- and habitat-level networks
Whether average network-level properties were different between urban bushland remnants and residential gardens depended on the scale of analysis [Table 2, Appendix 1 (Electronic supplementary materials)]. Differences between urban bushland remnants and residential gardens occurred in year two for nestedness only at the coarser scale (habitat by month (p = 0.047), not site scale (p = 0.067)). Extinction slope of bees in year two according to habitat type was significant at the coarser (p = 0.004), but not finer scale (p = 0.440), with the same pattern occurring for robustness (p = 0.593 vs. p = 0.001). Extinction slope of plants between habitat types in year two was significant at the finer (p = 0.001), but not coarser scale (p = 0.335), as was robustness (p = 0.001 vs. p = 0.593).
Niche overlap differed between habitat types in year two at the finer scale (p = 0.011), but not the coarser scale (p = 0.431). Significance (or lack thereof) between habitat types was consistent across scale of analysis in both years for H2’, weighted connectance, and functional complementarity.
Species-level analysis
Average species-level property values across scales
Average species-level properties varied according to scale of analysis, and as with average network properties, trends were heterogeneous according to the particular metric (Fig. 1). Normalised degree decreased as the scale of analysis got coarser, with the greatest change being between the site-level and habitat-by-month levels (Table 3). Average species strength was more than twice as great as the scale of analysis went from site to habitat by month to habitat across months (Table 3). Average interaction push–pull not only increased from site to habitat by month to habitat-across-months scale of analysis, but was on average negative (bees were more dependent on flowers than vice versa) at the finer scales but positive (bees were less dependent on flowers than vice versa) at the coarsest scale of analysis (Table 3). Species specificity index decreased as the scale of analysis got coarser (Table 3). Only Bluthgen’s d’ was similar across scales of analysis, and slight increases/decreases were not consistent between the years (Table 3).
Habitat comparisons between the site- and habitat-level networks
Comparisons of average species-level properties by habitat were similar across the site- and habitat-level networks [Table 4, Appendix 2 (Electronic supplementary materials)], except for species specificity index in year two, which differed between urban bushland remnants at the finer scale (p < 0.001), but not the coarser scale (p = 0.846).
Between and across years
When analyses were performed on networks from the data pooled across years, this did not necessarily represent an average of each separate year. In particular for network-level properties, nestedness and niche overlap were higher than for either year analysed alone, and the much higher values of weighted connectance and functional complementarity obtained in the first year compared to the second were obscured in the pooled year analysis, where the values of these metrics were closer to the second-year values (Table 1). For species-level properties, species strength exceeded the value for year one and year two, and for interaction push–pull, the value was considerably lower than that calculated for either year (Table 2). Comparing the two habitat types, differences or lack thereof between one year and the other were therefore masked when pooling the data from both years (Tables 2, 4).
Association between honey bee abundance and network properties
The scale of analysis affected whether honey bee abundance was associated with network structure [Appendix 3 (Electronic supplementary materials)]. In general the consequence was that significant associations that emerged when analysing the site-scale networks were no longer significant where the unit of analysis was habitat-by-month scale networks. Honey bee abundance was significantly negatively associated with weighted connectance in year one at the site scale (est = -0.014 ± 0.004, X2 = 11.1, p = 0.001) but not habitat-by-month scale (est ≤ − 0.001 ± < 0.001, F = 0.11, p = 0.755). There was a significant positive association between honey bee abundance and functional complementarity at the site scale (est = 0.183 ± 0.09, X2 = 4.11, p = 0.043) but not habitat-by-month scale (est ≤ 0.001 ± < 0.001, F = 0.1, p = 0.345) in year one. Honey bee abundance was significantly negatively associated with H2’ at the site scale (est = − 0.07 ± 0.02, X2 = 13.9, p = 0.020) but not habitat-by-month scale (est ≤ 0.001 ± < 0.001, F = 0.96, p = 0.349) in year two. Niche overlap and honey bee abundance were highly significantly associated at the site scale in year two (est = 0.08 ± 0.02, X2 = 21.5, p < 0.001), but this was non-significant at the habitat-by-month scale (est ≤ 0.001 ± < 0.001, F = 0.1, p = 0.972). The reverse pattern was found for nestedness in year one, which was positively associated with honey bee abundance at the habitat-by-month scale (est = 0.004 ± 0.002, F = 7.09, p = 0.037) but not site scale (est = 0.81 ± 1.97, X2 = 0.29, p = 0.652). Altering the scale of analysis also resulted in changes in the numbers of visits by honey bees relative to native bees such that combining networks led to honey bees comprising a larger proportion of visits [Appendix 4 (Electronic supplementary materials)].
Discussion
Our analyses of the same dataset at different scales of resolution revealed that scale of analysis can significantly alter values of network properties (Fig. 1). The spatio-temporal scale of analysis also can change significance, or lack thereof, of differences in bipartite network properties between habitat types and the influence of honey bees on network properties.
Our previous research used networks constructed per survey (Prendergast and Ollerton 2021, 2022), which we believe is the appropriate level at which to analyse such data. Combining sites within a given habitat type ignores the variation in the presence/absence of particular bees and/or plants between sites. Combining sites across months ignores species’ phenologies. Constructing networks from pooling data from different surveys leads to networks that do not conform to ecological reality. For example, if species were to co-occur in space and/or time, this could alter the behaviour of pollinators, due to facilitation or competition (Ye et al. 2014; Mesgaran et al. 2017). How specialised a species is in their foraging behaviour, especially in terms of bipartite metrics (as opposed to lecty), will depend on what resources are present in a given place and time. If only two plant species are in flower at a site during a survey and there are three flower–visitor taxa, two of the species may use one each and the third bee taxon uses both. If there were twenty plant species, oligoleges would still use one plant species each and a polylectic bee may use none of the original plant taxa, leading to completely different network metrics (Armbruster 2017). Such a scenario, however, will only be possible if the plants and pollinators co-occur in space and time.
We found that average values of network- and species-level properties, as well as the significance or lack thereof of differences between habitats or the influence of honey bees, at times differed between the networks constructed at the site scale, habitat-by-month scale, and habitat-across-month scale. This raises questions about the interpretation of conclusions of previous studies where networks are created by merging data gathered over multiple sites, months, or years. Indeed, plant–flower visitor networks in general are known to display strong temporal dynamics (Olesen et al. 2008; Alarcón et al. 2008; Lázaro et al. 2010; Burkle and Irwin 2009; Trøjelsgaard and Olesen 2016; Prendergast and Ollerton 2021, 2022). Even within a year, plant–flower visitor networks are temporally dynamic, whereby relative species’ abundances and visitation patterns and partners change (Basilio et al. 2006; Biella et al. 2017).
Wider time spans enable a more complete picture of phenological changes to be captured; however, if data are combined into one network, this leads to “forbidden links” (Olesen et al. 2010). For example, some plant species only bloomed in one of the years, but were not in bloom, or were not visited, in the other (see Prendergast 2020a, b). Consequently, when combining the two years of data, this creates “forbidden” links that actually did not occur. Combining such networks also can create improbable interactions. For example, whether a bee visits a plant can be influenced by the other plants concurrently in bloom near it, either acting as “magnets” or competing for pollinators (Mesgaran et al. 2017). The presence or absence of other bees at a particular time and location can also influence the realisation of a bee–plant interaction. This can occur due to competitive exclusion, through recruiting nestmates in the cases of eusocial bees, “eavesdropping” on the attractiveness of a plant based on other visitors, or avoiding plants by detecting predators (Abbott 2006; Lichtenberg et al. 2011; Stout and Goulson 2001; Reinhard and Srinivasan 2009; Roselino et al. 2016; Witjes and Eltz 2007; Yokoi et al. 2007).
Honey bee effects on network structure tended to be significant at site scales, but not at the coarser scales. This may be due to changes in the sample size and therefore degrees of freedom or it may be a reflection of pooling across sites. For example, pooling data across surveys mean there is a greater array of flowering plant species in such a pooled network and indeed we can see that functional complementarity—a mechanism by which species can divide up resources, which can facilitate co-existence (Blüthgen and Klein 2011; Gockele et al. 2014)—attained much higher values at the coarser (pooled) scales. However such a partitioning of resources cannot occur at the site scale if that array of resources is not available in space and time. We also found that honey bee abundance was no longer significantly associated with functional complementarity in year one, nor with niche overlap in year two. Artificial inflations of functional complementarity can also provide misleading interpretations regarding ecosystem functioning (Barry et al. 2019; Fründ et al. 2013; Yachi and Loreau 2007). Native bee species and flowering plant resources often have short activity/bloom periods, especially in seasonally dynamic environments (Cane and Tepedino 2007), and native bees, especially smaller-bodied species, often have short foraging distances (Hofmann et al. 2020). Therefore, assessment of honey bee density on bee–plant networks should be conducted at the finer scales at which competition plays out; pooling across months or sites separated by kilometres creates unrealistic scenarios. If researchers were to assess honey bee impacts at coarser scales they may obscure effects of honey bee competition that occur at biologically relevant scales.
The finding that values change across scales could also give misleading pictures regarding how vulnerable networks may be to perturbations. For example, NODF values were higher at coarser scales of analysis, which may suggest that networks of interacting species are more stable than they actually are (Thébault and Fontaine 2010). In contrast, another study found that nestedness decreased with scale (Schwarzt et al. 2020).
Whilst many network metrics varied according to scale of analysis, others were relatively resilient to change in scales and thus conclusions may be robust at coarser units of analysis. This includes network (H2′) and species (d′) specialisation and plant-level robustness and bee-level extinction slope. The bipartite measures of specialisation are described as being ‘scale invariant’ (Blüthgen et al. 2006) and our results confirm that these metrics can therefore be suitable for comparisons across different networks which may be conducted with different units of analysis. The greater sensitivity of bees than plants to the scale of analysis for robustness warrants further research, but may also have implications for predicting the relative vulnerability of plants/pollinators to extinctions (Astegiano et al. 2015; Memmott et al. 2004; Schleuning et al. 2016). Further research is required to determine if, in addition to spatial and temporal resolution, taxonomic resolution influences network properties. There again is no consensus about what level is appropriate and how this may alter conclusions about plant–pollinator visitation networks.
Our findings on how spatio-temporal scale influences network properties is supported by a recent publication looking at how temporal scale influences networks (Schwarzt et al. 2020). Here, the authors for each site used all observed interactions within the same day, week, month, year, or the total sampling extent, to construct a quantitative interaction matrix, and looked at how different scales affected network properties, including NODF, connectance, and H′2. Like our study, they found a minimal influence of temporal scale on H′2, whereas connectance declined as temporal scale increased. Our study therefore reinforces the findings by Schwarzt et al. (2020) and furthermore shows that temporal scale alters conclusions about habitat and introduced species’ impacts on network structure. Unlike Schwarzt et al. (2020) who only found weak, inconsistent effects of scale on NODF, we found NODF increased with scale. The difference may stem from how Schwarzt et al. (2020) used a number of different datasets which differed therefore in the species pool, spatial scale, and observers.
Our results add to the discussion about the appropriateness of the methods used to assess and analyse plant–pollinator assemblages (Doré et al. 2021; Prendergast and Hogendoorn 2021; Thomson 2021). Previous research has underscored the importance of adequate sampling effort on plant–pollinator networks (Chacoff et al. 2012; Nielsen and Bascompte 2007; Rivera-Hutinel et al. 2012) and here we emphasise the need to consider the spatial and temporal resolution of these networks. Understanding how environmental changes such as conversion of natural habitat to urbanised habitats and introduced species alter pollination network structure holds great potential (Baldock et al. 2019; Elle et al. 2012; Prendergast and Ollerton 2021, 2022; Tylianakis et al. 2010). We have demonstrated however that these need to be analysed at realistic scales based on the ecology and biology of the species concerned to prevent these simply becoming mathematical abstractions with little bearing on reality.
Based on our results, we offer the following guidelines for conducting network analysis and the spatial and temporal scales that are acceptable in evaluating plant–pollinator networks. For spatial scale, sites even separated by a few kilometres can exhibit different community compositions and interaction properties (Prendergast 2021a, b, 2022). This is especially true of different habitats (Prendergast and Ollerton 2021), but sites even within the same habitat should not be combined, e.g. (Prendergast 2021a, b, 2022). This would be especially true in highly heterogeneous landscapes, such as urban areas (Prendergast et al. 2022a, b). Temporal scale may depend on geographical location and phenology of species present—in communities that are highly synchronised with very brief activity seasons or perennially stable seasons, there may be minimal monthly turnover and variation in network structure, which could permit pooling. However in systems where different species have staggered, short activity periods, networks per month or even of finer temporal scales may be necessary, and in such systems pooling surveys between months would be inappropriate. Having said this, plant–pollinator interactions have not been studied in most regions of the planet (Archer et al. 2014) and it is estimated that we have pollinator data for only about 10% of the flowering plant species (Ollerton 2021). Therefore, one circumstance in which pooling might be appropriate is when taking a “meta-network” approach to describe the overall structure of plant–pollinator interactions that have been previously undocumented, in order to stimulate future research on this topic. (Fig. 2).
Summary of how the scale that networks are constructed influences bipartite plant–pollinator network- and species-level properties. For each index, how the scale of analysis influences the calculated value is visualised schematically from red to indicate the highest value, through to orange, then yellow lower, and blue the lowest. A lack of change in colours means that network property’s value was similar across scales. HL higher level, i.e. bee taxa, LL lower level, i.e. plants species visited. ND normalised degree, IPP interaction push–pull, SSI species specificity index, d’ Blüthgen d’
Conclusion
Scale has been increasingly recognised as a major consideration in analysing and interpreting ecological research (Hurlbert and Jetz 2007; Wiens 2001; Schneider 1994). We show here that depending on the spatial–temporal unit that bee–plant networks are constructed, this can alter results and conclusions about network- and species-level properties of pollination networks and effects of habitat and the role of introduced species on pollination network structure.
References
Abbott KR (2006) Bumblebees avoid flowers containing evidence of past predation events. Canadian J Zool 84(9):1240–1247
Aizen M, Feinsinger P (2003) Bees not to be? Responses of insect pollinator faunas and flower pollination to habitat fragmentation. In: Bradshaw GA, Marquet PA (eds) How landscapes change. Ecological studies (analysis and synthesis), vol 162. Springer, Heidelberg, pp 111–129
Alarcón R, Waser NM, Ollerton J (2008) Year-to-year variation in the topology of a plant–pollinator interaction network. Oikos 117:1796–1807
Archer CR, Pirk CWW, Carvalheiro LG, Nicolson SW (2014) Economic and ecological implications of geographic bias in pollinator ecology in the light of pollinator declines. Oikos 123:401–407
Armbruster WS (2017) The specialization continuum in pollination systems: diversity of concepts and implications for ecology, evolution and conservation. Funct Ecol 31:88–100
Astegiano J, Massol F, Vidal MM et al (2015) The Robustness of plant-pollinator assemblages: linking plant interaction patterns and sensitivity to pollinator loss. PLoS ONE 10:e0117243
Baldock KC, Goddard MA, Hicks DM et al (2019) A systems approach reveals urban pollinator hotspots and conservation opportunities. Nat Ecol Evol 3:363
Barrett R, Tay EP (2016) Perth plants: a field guide to the bushland and coastal flora of Kings Park and Bold Park. CSIRO PUBLISHING, Clayton
Barry KE, Mommer L, van Ruijven J et al (2019) The future of complementarity: disentangling causes from consequences. Trends Ecol Evol 34:167–180
Bascompte J, Jordano P (2007) Plant-animal mutualistic networks: the architecture of biodiversity. Ann Rev Ecol Evo Systemat 38:567–593. https://doi.org/10.1146/annurev.ecolsys.38.091206.095818
Basilio AM, Medan D, Torretta JP et al (2006) A year-long plant-pollinator network. Aust Ecol 31:975–983
Bates D, Maechler M, Bolker BM et al (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Batley M, Brandley B (2014) Phenology of the Australian solitary bee species Leioproctus plumosus (Smith) (Hymenoptera: Colletidae). Aust Entomol 41:7–14
Beckett SJ (2016) Improved community detection in weighted bipartite networks. Roy Soc Open Sci 3:140536
Biella P, Ollerton J, Barcella M et al (2017) Network analysis of phenological units to detect important species in plant-pollinator assemblages: can it inform conservation strategies? Community Ecol 18:1–10
Borchardt KE, Morales CL, Aizen MA, Toth AL (2021) Plant–pollinator conservation from the perspective of systems-ecology. Current Op Insect Sci 47:154–161
Burkle LA, Alarcón R (2011) The future of plant–pollinator diversity: understanding interaction networks across time, space, and global change. Am J Bot 98:528–538
Burkle LA, Irwin RE (2009) The importance of interannual variation and bottom–up nitrogen enrichment for plant–pollinator networks. Oikos 118:1816–1829
Burnham KP, Anderson DR (2003) Model selection and multimodel inference: a practical information-theoretic approach. Springer Science & Business Media, New York
Chacoff NP, Vázquez DP, Lomáscolo SB et al (2012) Evaluating sampling completeness in a desert plant–pollinator network. J Anim Ecol 81:190–200
Daniels B, Jedamski J, Ottermanns R, Ross-Nickoll M (2020) A “plan bee” for cities: Pollinator diversity and plant-pollinator interactions in urban green spaces. PLoS ONE 15:e0235492
Dohzono I, Yokoyama J (2010) Impacts of alien bees on native plant-pollinator relationships: a review with special emphasis on plant reproduction. Appl Entomol Zool 45:37–47
Doré M, Fontaine C, Thébault E (2021) Relative effects of anthropogenic pressures, climate, and sampling design on the structure of pollination networks at the global scale. Glob Change Biol 27:1266–1280
Dormann CF, Gruber B, Fründ J (2008) Introducing the bipartite package: analysing ecological networks. Interaction 1:2413793
Elle E, Elwell SL, Gielens GA (2012) The use of pollination networks in conservation. This article is part of a Special issue entitled “Pollination biology research in Canada: perspectives on a mutualism at different scales.” Botany 90:525–534
Fründ J, Dormann CF, Holzschuh A et al (2013) Bee diversity effects on pollination depend on functional complementarity and niche shifts. Ecology 94:2042–2054
Garibaldi LA, Steffan-Dewenter I, Winfree R et al (2013) Wild pollinators enhance fruit set of crops regardless of honey bee abundance. Science 339:1608–1611
Gockele A, Weigelt A, Gessler A et al (2014) Quantifying resource use complementarity in grassland species: a comparison of different nutrient tracers. Pedobiologia 57:251–256
Greenleaf SS, Williams NM, Winfree R et al (2007) Bee foraging ranges and their relationship to body size. Oecologia 153:589–596
Güneralp B, McDonald RI, Fragkias M et al (2013) Urbanization forecasts, effects on land use, biodiversity, and ecosystem services. In: Elmqvist T, Fragkias M, Goodness J et al (eds) Urbanization, biodiversity and ecosystem services: challenges and opportunities. Springer, New York, pp 437–452
Hewitt JE, Thrush SF, Lundquist C (2001) Scale-dependence in ecological systems. eLS. https://doi.org/10.1002/9780470015902.a0021903.pub2
Hofmann MM, Fleischmann A, Renner SS (2020) Foraging distances in six species of solitary bees with body lengths of 6 to 15 mm, inferred from individual tagging, suggest 150 m-rule-of-thumb for flower strip distances. J Hymenopt Res 77:105
Hurlbert AH, Jetz W (2007) Species richness, hotspots, and the scale dependence of range maps in ecology and conservation. P Natl A Sci 104:13384–13389
Hussey B, Keighery G, Cousens R et al (1997) Western weeds: a guide to the weeds of Western Australia. The Plant Protection Society of Western Australia (Inc.), Perth
Kuznetsova A, Brockhoff PB, Christensen RHB (2017) lmerTest package: tests in linear mixed effects models. J Stat Soft 82:26
Lambers H (2014) Plant life on the sandplains in Southwest Australia. UWA Publishing, Crawley, Western Australia
Laurie V (2015) The Southwest: Australia’s biodiversity hotspot. Crawley, UWA Publishing
Lázaro A, Nielsen A, Totland Ø (2010) Factors related to the inter-annual variation in plants’ pollination generalization levels within a community. Oikos 119:825–834
Lichtenberg EM, Hrncir M, Turatti IC, Nieh JC (2011) Olfactory eavesdropping between two competing stingless bee species. Behav Ecol Sociobiol 65:763–774
Memmott J, Waser NM, Price MV (2004) Tolerance of pollination networks to species extinctions. Proc R Soc Lond B Bio 271:2605–2611
Mesgaran MB, Bouhours J, Lewis MA et al (2017) How to be a good neighbour: facilitation and competition between two co-flowering species. J Theor Biol 422:72–83
Nielsen A, Bascompte J (2007) Ecological networks, nestedness and sampling effort. J Ecol 95:1134–1141
Olesen JM, Bascompte J, Dupont YL et al (2007) The modularity of pollination networks. Proc Natl A Sci 104:19891–19896
Olesen JM, Bascompte J, Elberling H et al (2008) Temporal dynamics in a pollination network. Ecology 89:1573–1582
Olesen JM, Bascompte J, Dupont YL et al (2010) Missing and forbidden links in mutualistic networks. Proc R Soc Lond B Bio 278:725–732
Ollerton J (2021) Pollinators & pollination: nature and society. Pelagic Publishing Ltd, Exeter
Ollerton J, Winfree R, Tarrant S (2011) How many flowering plants are pollinated by animals? Oikos 120:321–326
Peterson G, Allen CR, Holling CS (1998) Ecological resilience, biodiversity, and scale. Ecosystems 1:6–18
Potts SG, Imperatriz-Fonseca V, Ngo HT et al (2016) Safeguarding pollinators and their values to human well-being. Nature 540:220–229
Prendergast K (2020a) Species of native bees in the urbanised region of the southwest Western Australian biodiversity hotspot. Curtin University. https://researchdata.edu.au/species-native-bees-biodiversity-hotspot/1462019
Prendergast K (2020b) Plant-pollinator network interaction matrices and flowering plant species composition in urban bushland remnants and residential gardens in the southwest Western Australian biodiversity hotspot. Curtin University. https://doi.org/10.25917/5f3a0aa235fda. https://researchdata.edu.au/plant-pollinator-network-biodiversity-hotspot
Prendergast K (2021a) City of Bayswater native bee surveys November – February 2020/21, City of Bayswater, Western Australia.
Prendergast K (2021b) Kalamunda native bee surveys Spring-Summer 2020/21 at Ledger Hill Reserve, Maida Vale Reserve, and Poison Gully Reserve, Kalamunda, Western Australia
Prendergast K (2022) City of Bayswater Native Bee Surveys October – February 2021/22, City of Bayswater, Western Australia
Prendergast KS, Hogendoorn K (2021) FORUM: methodological shortcomings and lack of taxonomic effort beleaguer Australian bee studies. Aust Ecol 46:880–884
Prendergast K, Ollerton J (2021) Plant-pollinator networks in Australian urban bushland remnants are not structurally equivalent to those in residential gardens. Urban Ecosyst 24:973–987. https://doi.org/10.1007/s11252-020-01089-w
Prendergast KS, Ollerton J (2022) Impacts of the introduced European honey bee on Australian bee-flower network properties in urban bushland remnants and residential gardens. Aust Ecol 47:35–53. https://doi.org/10.1111/aec.13040
Prendergast KS, Menz MHM, Dixon KW et al (2020) The relative performance of sampling methods for native bees: an empirical test and review of the literature. Ecosphere 11:e03076
Prendergast KS, Dixon KW, Bateman PW (2021) Interactions between the introduced European honey bee and native bees in urban areas varies by year, habitat type and native bee guild. Biol J Linn Soc 133:725–743
Prendergast KS, Dixon KW, Bateman PW (2022a) A global review of determinants of native bee assemblages in urbanised landscapes. Insect Conserv Divers 15:385–405
Prendergast KS, Tomlinson S, Dixon KW, Bateman PW, Menz MHM (2022b) Urban native vegetation remnants support more diverse native bee communities than residential gardens in Australia’s southwest biodiversity hotspot. Biol Cons 265:109408
R Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Reinhard J, Srinivasan MV (2009) The role of scents in honey bee foraging and recruitment. In: Jarau S, Hrncir M (eds) Food exploitation by social insects: Ecological, behavioral, and theoretical approaches. CRC Press, Florida, pp 165–182
Rivera-Hutinel A, Bustamante R, Marín V et al (2012) Effects of sampling completeness on the structure of plant–pollinator networks. Ecology 93:1593–1603
Roselino AC, Rodrigues AV, Hrncir M (2016) Stingless bees (Melipona scutellaris) learn to associate footprint cues at food sources with a specific reward context. J Compar Physiol A 202:657–666
Schleuning M, Fründ J, Schweiger O et al (2016) Ecological networks are more sensitive to plant than to animal extinction under climate change. Nat Commun 7:13965
Schneider DC (1994) Quantitative ecology: spatial and temporal scaling. Elsevier, Amsterdam
Schwarzt B, Vázquez DP, CaraDonna PJ et al (2020) Temporal scale-dependence of plant–pollinator networks. Oikos 129:1289–1302
Smith JP, Heard TA, Beekman M, Gloag R (2017) Flight range of the Australian stingless bee Tetragonula carbonaria (Hymenoptera: Apidae). Austral Entomol 56:50–53
Stout JC, Goulson D (2001) The use of conspecific and interspecific scent marks by foraging bumblebees and honey bees. Anim Behav 62:183–189
Sutherland WJ, Freckleton RP, Godfray HCJ et al (2013) Identification of 100 fundamental ecological questions. J Ecol 101:58–67
Taki H, Kevan PG (2007) Does habitat loss affect the communities of plants and insects equally in plant–pollinator interactions? Preliminary findings. Biodivers Conserv 16:3147–3161
Thébault E, Fontaine C (2010) Stability of ecological communities and the architecture of mutualistic and trophic networks. Science 329:853–856
Thomson J (2021) How worthwhile are pollination networks? J Pollinat Ecol 28:i–vi
Trøjelsgaard K, Olesen JM (2016) Ecological networks in motion: micro-and macroscopic variability across scales. Funct Ecol 30:1926–1935
Tylianakis JM, Laliberté E, Nielsen A et al (2010) Conservation of species interaction networks. Biol Conserv 143:2270–2279
Vázquez DP, Blüthgen N, Cagnolo L et al (2009) Uniting pattern and process in plant–animal mutualistic networks: a review. Ann Bot 103:1445–1457
Watts S, Dormann CF, Martín González AM, Ollerton J (2016) The influence of floral traits on specialization and modularity of plant–pollinator networks in a biodiversity hotspot in the Peruvian Andes. Ann Bot 118:415–429
Wiens JA (2001) Understanding the problem of scale in experimental ecology. Scaling relations in experimental ecology. Columbia University Press, Columbia, pp 61–88
Willcox BK, Howlett BG, Robson AJ et al (2019) Evaluating the taxa that provide shared pollination services across multiple crops and regions. Sci Rep 9:1–10
Witjes S, Eltz T (2007) Influence of scent deposits on flower choice: experiments in an artificial flower array with bumblebees. Apidologie 38:12–18
Yachi S, Loreau M (2007) Does complementary resource use enhance ecosystem functioning? A model of light competition in plant communities. Ecol Lett 10:54–62
Ye Z-M, Dai W-K, Jin X-F et al (2014) Competition and facilitation among plants for pollination: can pollinator abundance shift the plant–plant interactions? Plant Ecol 215:3–13
Yokoi T, Goulson D, Fujisaki K (2007) The use of heterospecific scent marks by the sweat bee Halictus aerarius. Naturwissenschaften 94:1021–1024
Zurbuchen A, Landert L, Klaiber J et al (2010) Maximum foraging ranges in solitary bees: only few individuals have the capability to cover long foraging distances. Biol Conserv 143:669–676
Acknowledgements
K. Prendergast would like to acknowledge the assistance of C. Tauss, H. Lambers, and K. Dixon in providing identifications for native flora and thank the property owners and local councils for allowing her to survey the sites.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. Fieldwork by K. Prendergast was conducted during her PhD, which was funded by a Forrest Research Scholarship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Prendergast, K.S., Ollerton, J. Spatial and temporal scale of analysis alter conclusions about the effects of urbanisation on plant–pollinator networks. Arthropod-Plant Interactions 16, 553–565 (2022). https://doi.org/10.1007/s11829-022-09925-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-022-09925-w