Abstract
Background
Both obesity hypoventilation syndrome (OHS) and opium dependence can negatively affect nocturnal respiratory functionality. However, the effects of a combination of the two are relatively unknown. In this study, the tidal volume (VT) of OHS patients with and without opium dependence was estimated by measuring changes in the circumference of the thorax and the abdomen during sleep.
Materials and methods
This case–control study was performed in Iran, Isfahan. Initially, 20 healthy subjects calibrated the Pneumobelts (Löwenstein Medical Technology GmbH, Hamburg, Germany) by wearing the device and also breathing in and out of a Spirobag. From this, it was possible to estimate the coefficient factors for determining VT based on thoracic and abdominal measures. Subsequently, the study included 55 patients with OHS, who were divided into two groups: opium dependent and non-users. They were then admitted to the sleep lab for a single night of polysomnography (PSG). VT was estimated and then compared between groups.
Results
In total, there were 24 patients in the opium group and 31 in the non-user control group. All PSG variables were analyzed with independent-sample t-tests and an analysis of covariance was used to adjust for potential confounding variables. The following confounding variables were adjusted: sex, smoking, body mass index, and hypertension. VT values were not significantly different between groups (opium: 772 ± 125.4; non-user: 774 ± 95). Average oxygen saturation values were significantly lower in opium-dependent patients (opium: 86.1 ± 5.5; non-user: 89.1 ± 4.1). All other variables from PSG including chest and abdominal circumference were not significantly different between groups.
Conclusion
OHS patients with opium dependency showed similar VT levels when compared to non-users with OHS. Due to the rarity of the sample, it was difficult to gather exactly matched pairs. Therefore, it is possible that with a more closely related sample, we might be able to see a difference in VT between groups.
Zusammenfassung
Hintergrund
Sowohl das Obesitas-Hypoventilations-Syndrom (OHS) als auch die Opiumabhängigkeit können sich negativ auf die nächtliche Atemfunktion auswirken, jedoch sind die Auswirkungen einer Kombination aus beiden Erkrankungen relativ unbekannt. In dieser Studie wurde das Atemzugvolumen (VT) von OHS-Patienten – mit und ohne Opiumabhängigkeit – basierend auf der Messung von Umfangsänderungen des Thorax und des Abdomens während des Schlafs geschätzt.
Material und Methoden
Diese Fall-Kontroll-Studie wurde von Januar 2020 bis Dezember 2020 in Isfahan, Iran, durchgeführt. Zunächst erfolgte die Berechnung der Koeffizienten für die Bestimmung des VT basierend auf Messungen des Thorax- und Abdomenumfangs mittels Pneumobelt (Löwenstein Medical Technology GmbH, Hamburg, Deutschland) während des Ein- und Ausatmens in einen Beutel mit bekannter Menge (Spirobag) bei 20 gesunden Personen. Nach der Bestimmung dieser Faktoren wurden 55 Patienten mit OHS in die Studie eingeschlossen und in 2 Gruppen eingeteilt (opiumabhängig und nicht opiumabhängig), und es erfolgte für eine Nacht eine Polysomnographie (PSG) im Schlaflabor. Das VT wurde geschätzt und dann zwischen den beiden Gruppen verglichen.
Ergebnisse
Insgesamt befanden sich 24 Patienten in der Opiumgruppe und 31 in der Kontrollgruppe ohne Opiumkonsum. Alle PSG-Variablen wurden mit t-Tests für unabhängige Stichproben analysiert, und es wurde eine Kovarianzanalyse durchgeführt, um potenzielle Confounder zu identifizieren. Für die folgenden Confounder wurde adjustiert: Geschlecht, Rauchen, Body Mass Index und Bluthochdruck. Die VT-Werte unterschieden sich nicht signifikant zwischen den Gruppen (Opium: 772 ± 125,4; Nichtkonsumenten: 774 ± 94,5). Die durchschnittlichen SaO2-Werte waren bei opiumabhängigen Patienten signifikant niedriger (Opium: 86,1 ± 5,5; Nichtkonsumenten: 89,1 ± 4,1; p = 0,02). Alle anderen Variablen der PSG, einschließlich des Thorax- und Abdomenumfangs, unterschieden sich nicht signifikant zwischen den Gruppen.
Schlussfolgerung
OHS-Patienten mit Opiumabhängigkeit wiesen im Vergleich zu Nicht-OHS-Konsumenten ähnliche VT-Werte auf. Aufgrund des einmaligen Charakters der Stichprobe war es schwierig, exakt übereinstimmende Paare zu finden. Daher ist es möglich, dass bei einer Stichprobe mit engeren Bezügen ein Unterschied in der VT zwischen den Gruppen feststellbar wäre.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
In this manuscript, we report our findings on nocturnal tidal volume by polysomnography for obesity hypoventilation syndrome patients with and without opium dependence. While there is a vast range of literature reporting tidal volume in obesity hypoventilation syndrome and opium dependence separately, the effect on tidal volume of the combination of the two has received only little attention. Here, we investigated tidal volume indirectly using Pneumobelts instead of intrusive mouthpieces.
Introduction
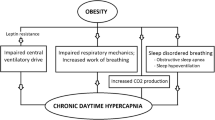
Iran has three times the highest number of opium-dependent individuals compared to the rest of the world, mainly due to sharing a border with Afghanistan—the world’s largest producer of opium [1]. Therefore, opium is relatively accessible to the majority of the nation. The most common method of opium consumption in Iran is by inhalation [2]. An overdose can result in severe respiratory distress, coma, and even death, which means nocturnal respiratory health problems are a real concern in Iran [3]. Previous research has shown the link between opium inhalation and respiratory depression through the reduction of minute ventilation and increased tidal volume (VT) variability. Here, it was shown that opium dependents experienced a change in their respiratory pattern, which lead to respiratory depression [4]. This disturbance is opium’s most profound effect on the respiratory system, which slows and becomes irregular [5]. Another study showed that opium use increased forced expiratory volume (FEV1) and forced vital capacity (FVC), which improved lung functionality [6]. Even though this alleviated some breathing problems, overuse could adversely affect the respiratory system. Consequently, this may follow into the night and influence symptoms of sleep-disordered breathing (SDB) such as apneas and hypopneas [7].
Patients with obesity hypoventilation syndrome (OHS) also suffer from nocturnal respiratory health consequences. For instance, OHS is made up of a combination of hypercapnia (partial pressure of carbon dioxide or PaCo2 level > 45), obesity (body mass index or BMI > 30 kg/m2), and SDB [8]. However, it has been shown that OHS patients have low FVC, FEV1, and VT, and increased respiratory rates [9], which is the opposite in opium-dependent patients, with FEV1 and FVC increasing. This raises the question of what changes to lung functionality occur in patients with a combination of OHS and opium dependence during sleep. Moreover, could opium dependence lower VT currently seen?
One commonly used device to measure lung function is spirometry. It measures the patient’s capability to inhale and exhale air at a given speed or volume [10, 11]. It is an inexpensive and user-friendly method. Another device called a plethysmograph is used to determine other types of lung functions such as functional residual capacity, airway resistance, total lung capacity, and residual volume [12]. Both methods require a facemask or a mouthpiece to breathe into [13]. However, the main drawback is that patients may experience some discomfort when wearing these devices. This can result in unnatural breathing, which leads to inaccurate measurements [14]. Therefore, other more natural methods of measurement are often used to measure lung functions.
Previous research has revealed that the link between upper-body movement (rib cage and abdominal motion) and respiratory mechanisms can predict VT accurately [15, 16]. The proportional sum of rib cage and abdominal motion represents the respiratory volume. The respiratory inductive plethysmograph (RIP, Respitrace) can determine these motion changes by evaluating pulmonary ventilation—the measurement of movement from the chest and abdominal wall [17]. Pneumobelts are RIP devices designed specifically to estimate VT. They contains sensors that respond to chest and abdominal motion. Specifically, they consist of a silicone tube that is blocked at both ends with a rubbery stopper. Stretching of the tubes results in reduction of pressure within the tube, which is proportional to the length change [18]. These belts are placed around the abdomen and rib cage. The change in circumference can estimate VT. In Binks et al.’s research, each patient was asked to complete a few maneuvers in a supine and standing position. During this, the belt circumference changes were recorded [15]. They showed that the relationship between VT, rib cage, and abdominal signals could be calculated with the following equation:
RC and AB are the dimensional changes of the rib cage and abdomen, x and y are the coefficients that describe the relationship between motion and volume change in the rib cage and abdomen [18].
In this study, we estimated the VT of opium-dependent OHS patients by measuring rib cage and abdominal dimension changes during sleep. These results were then compared to OHS patients who do not use opium. We hypothesized that the VT will be lower in opium-dependent OHS patients than in non-users with OHS.
Materials and methods
Participants and study design
This case–control study was performed from January 2020 until December 2020 in Khorshid Hospital, affiliated with Isfahan University of Medical Sciences, Isfahan, Iran. A power calculation of 0.8 with a significance threshold of 5% indicated that it would be suitable to use 30 patients in each group (opium-dependent patients with OHS vs. non-opium users with OHS):
The ∆ represents the statistical power of 0.8 and φ is the ratio of case to control, which is 1. The remaining variables were extracted from a previous study on a similar topic. Specifically, the apnea/hypopnea index (AHI) and the hypopnea index output were extracted and used in this power calculation [2].
Patients were included if they had PaCo2 > 45 whilst awake in an upright position, BMI ≥30 Kg/m2, and were not suspected of having other causes of hypoventilation such as hypothyroidism and neuromuscular disease. These patients had already been diagnosed with OHS and, thus, pulmonary function testing was not needed to confirm this. Patients with congestive heart failure, severe chronic obstructive pulmonary disease, interstitial lung disease, and incomplete documents were excluded from this study. Therefore, both groups contained patients with OHS and the patients in the case group had an opium dependence with a mean consumption of 0.8 g per day. A cohort study of 50,000 Iranians from 2012 indicated that the range of opium consumption was 0.2–1–2 g per day, with a median of 0.6 g per day [19]. This means our current sample is within range of the standard opium-dependent population in Iran. The Research Committee of Isfahan University of Medical Sciences approved the study protocol, and the Ethics Committee confirmed it.
Rib cage and abdominal circumference coefficients
Twenty healthy people without any history of hypoventilation, pulmonary disease, and opium dependence performed breathing exercises whilst awake during the day. This was to estimate the rib cage and abdominal circumference coefficient factors.
Two devices were used to measure breathing. First, two transducer coils (Pneumobelts) from the polysomnography device (Löwenstein Medical Technology GmbH, Hamburg, Germany) were wrapped around the rib cage and abdomen. The respiratory volume could be determined through the expansion and contraction of these areas during breathing. Specifically, one transducer coil is placed around the rib cage, just under the armpits, and the other around the abdomen, with its upper edge below the lowest rib. Secondly, each person was instructed to breathe into the Spirobag in order to measure VT. A Spirobag is a bag that holds 800 cc when fully expanded and it was calibrated so that a 1.0-volt signal equals a 1.0-litre volume. Therefore, the VT was 800 cc due to the Spirobag being 800 cc. This volume remained fixed throughout usage [20]. See Fig. 1 below of a healthy subject using a Pneumobelt and the Spirobag during calibration.
The following breathing maneuvers were performed: five deep breaths in the supine position followed by a three-minute recovery time, then five deep breaths in the standing position. After this, the chest and abdomen frequency changes were evaluated. The relationship between rib cage, abdominal signals, and VT can be modelled as follows:
In the supine position, breathing occurs more through abdominal expansion whilst in the standing position, breathing occurs through chest expansion. Therefore, we determined the changes in both positions. For estimating the x and y calibration factor, the following equations were used:
Uppercase letters represent volumes obtained from a standing patient and lowercase letters represent those from a patient in a supine position [15]. After estimating the x and y calibration factor for each of the 20 healthy participants, the mean range of x and y was calculated.
Procedure and variable assessments
After the coefficients were calculated, both groups of OHS patients (opium-dependent and non-opium users) were admitted to the sleep lab for one night of polysomnography. Here, they received the Pneumobelt without the Spirobag. Demographic data, neck circumference, STOP-Bang (snoring history, tired during the day, observed stop breathing while sleeping, high blood pressure, BMI more than 35 kg/m2, age more than 50 years, neck circumference more than 40 cm, and sex), Epworth Sleepiness Scale (ESS), and the Bilevel positive airway pressure or continuous positive airway pressure treatment data were collected from the patients. Polysomnography was evaluated in two ways: first, the dimensional changes of the rib cage and abdomen were determined according to our previously mentioned equation. This presented us with an estimated VT. Secondly, other polysomnographic data such as average and minimum oxygen saturation and apnea–hypopnea index (AHI) were evaluated in both groups.
Statistical analysis
Continuous and categorical data were reported as mean ± standard deviation (SD) and frequency (percentage). The normality of continuous variables was checked using the Kolmogorov–Smirnov test and the Q‑Q plot. Categorical data were compared between groups using the chi-squared test and independent-samples t-test for normally distributed continuous data. We compared polysomnographic variables with independent-samples t-test as well as analysis of covariance (ANCOVA) for adjusting potential confounding variables. P ≤ 0.05 was considered statistically significant. All statistical analyses were performed using SPSS statistics for Windows v.16 (SPSS Inc., Chicago, IL, USA).
Results
The calibration of the 800 cc Spirobags in 20 healthy people displayed x = 5.33, y = 0.84.
Sixty-one patients entered the study and six were removed from the final analysis due to incomplete documents. This left 24 patients in the case group and 31 in the control group. Demographic and basic clinical data of patients were analyzed in both groups and are shown in Table 1. In the opium group, 91.7% of patients were male, and 45.2% were male in the control group (P < 0.001). BMI and neck circumference were significantly higher in the control group, which meant that patients in the control group were more obese than the case group (P < 0.001). Furthermore, hypertension was more prevalent in the control group (P = 0.002). STOP-Bang and ESS questionnaire scores were not significantly different between groups (P = 0.12, 0.08, respectively).
The comparison of polysomnographic variables between the two groups can be seen in Table 2. After controlling for covariates, VT, chest circumference, and abdominal circumference values were not significantly different between groups. However, the average saturation was significant (P = 0.05). This suggests that opium-dependent patients have a lower oxygen saturation during sleep than the controls. All other polysomnographic data including AHI and minimum oxygen saturation were not significantly different between groups.
Discussion
Our study produced non-significant results when comparing the chest and abdominal circumferences between OHS opium-dependent and non-using patients. This means that the average VT during sleep was not different between groups, which goes against what we hypothesized. Even though the results were not significant, it is important to note that the estimated VT during sleep was still quite low for both groups if we consider that VT is based on predicted body weight and guided by height [21]. This suggests that there is an increased risk of developing SDB in our total sample due to the association between low VT and hypoventilation [9]. Therefore, our study complements previous research well, showing that patients with OHS with or without opium dependence should be investigated for SDB.
Additionally, it was found that oxygen saturation was lower in those with OHS and opium dependence. Previous research has already shown that OHS can reduce oxygen saturation levels and VT during sleep when compared to healthy participants [22]. Considering this and our results, patients with OHS and opium dependence may be at a higher risk of developing nocturnal respiratory health problems related to low oxygen saturation such as hypoxemia. This is when a patient does not obtain enough oxygen in their blood and a typical symptom is shortness of breath throughout the night, which can prevent them from obtaining adequate sleep [23].
In contrast to Fadaii et al.’s study, our results showed the opposite effect on VT during sleep. They reported that opium-dependent patients had larger VT when measured with spirometry in comparison to patients who only smoked cigarettes. In addition, FVC and FEV1 were significantly higher in patients who smoked opium and cigarettes compared to patients who only smoked cigarettes [6]. However, these results are not directly comparable due to our total sample having OHS, and out of the non-opium users, only 16.1% smoked cigarettes. It could be argued that since there were very few smokers in the control condition, their VT may be better than in the case group. Nevertheless, there were no differences between the groups, which could mean that smoking cigarettes or opium has less of a moderating role on breathing patterns as weight increases. This would need to be confirmed in future research.
The main strength of our study was the method we used to estimate VT during sleep. We found body plethysmography to be an excellent measure of VT as it determined pulmonary ventilation through the chest and the abdominal wall [12, 24, 25]. Using this method, we produced an accurate and preliminary insight into the similarities and differences in lung functionality for opium-dependent and non-using patients with OHS. This validates previous research using plethysmography as a measure for VT and provides evidence for future studies to use the same protocol when measuring VT without a mouthpiece [16].
The main limitations of our study came from the sample: first, we had a limited sample size due to excluding persons with missing information. This meant that the opium-dependent OHS group’s total sample size was lower than the estimated power calculation of 30. Therefore, we should interpret these findings with caution. Potentially, the results could change with more patients in the OHS with opium dependence group. Secondly, hypoventilation was studied, but no CO2 values were provided. Thirdly, the heterogeneity of both patient groups regarding BMI and comorbidities was large. The control group had a significantly higher BMI than the case group, which could negatively influence lung volume. For instance, lower FVC, FEV1, and VT, and higher respiratory rates are a consequence of OHS and this could be greater in those who have a higher BMI [9]. Then perhaps the opium also lowers the VT—as previous literature suggests—which could explain why the VT in both groups was similar [5]. In addition, the control group contained significantly more female patients and patients with hypertension, which could further affect the results. Therefore, if this study were to be replicated, the groups should be matched for sex, BMI, and other health conditions to cancel out any potential moderators. This proved to be difficult in this study due to the uniqueness of the sample. Finally, while overadjustment is a concern that should be considered in any statistical analysis, we are confident that this study’s approach to covariate selection and model validation was comprehensive and appropriate, and therefore, the risk of overadjustment is not a major concern in the context of this research.
Conclusion
Overall, OHS patients with opium dependency showed similar VT, chest circumference, and abdominal circumference scores when compared to non-users with OHS. Due to the rarity of the sample, it was difficult to gather exact matched pairs. Therefore, it is possible that with a more closely related sample we might be able to see a difference in VT between groups. Furthermore, it is also important to note that the results produced from this study should be interpreted with caution due to the total sample size being lower than the estimated power calculation. Therefore, with more power, we could have potentially seen a difference. We suggest that future studies use larger and matched populations to rule out any possible moderators.
Main novel aspects
-
Todal volume was similar between groups.
-
Obesity hypoventilation syndrome patients with opium dependency displayed lower levels of average O2 saturation values to the non-user control group.
-
Regardless of opium use, all other polysomnography variables were similar between groups.
References
Soroosh D, Neamatshahi M, Zarmehri B, Nakhaee S, Mehrpour O (2019) Drug-induced prolonged corrected QT interval in patients with methadone and opium overdose. Subst Abuse Treat Prev Policy 14(1):1–5. https://doi.org/10.1186/s13011-019-0196-3
Rostami F, Amra B (2010) The effect of opium on polysomnographic findings in patients with sleep apnea. Tanaffos 9(3):33–36
Radke JB, Owen KP, Sutter ME, Ford JB, Albertson TE (2014) The effects of opioids on the lung. Clinic Rev Allerg Immunol 46:54–64. https://doi.org/10.1007/s12016-013-8373-z
Bouillon T, Bruhn J, Roepcke H, Hoeft A (2003) Opioid-induced respiratory depression is associated with increased tidal volume variability. Eur J Anaesthesiol 20(2):127–133. https://doi.org/10.1017/S0265021503000243
Pattinson KT (2008) Opioids and the control of respiration. Br J Anaesth 100(6):747–758. https://doi.org/10.1093/bja/aen094
Fadaii A, Sohrabpour H, Taherkhanchi B, Moghdaam HRS, Bagheri B (2015) Effects of opium inhalation on pulmonary volumes in smoking patients. Arch Adv Biosci. https://doi.org/10.22037/jps.v6i1.8238
Yamanaka T, Sadikot RT (2013) Opioid effect on lungs. Respirology 18(2):255–262. https://doi.org/10.1111/j.1440-1843.2012.02307.x
Masa JF, Pépin JL, Borel JC, Mokhlesi B, Murphy PB, Sánchez-Quiroga MÁ (2019) Obesity hypoventilation syndrome. Eur Respir Rev. https://doi.org/10.1183/16000617.0097-2018
Littleton SW (2012) Impact of obesity on respiratory function. Respirology 17(1):43–49. https://doi.org/10.1111/j.1440-1843.2011.02096.x
Miller M (2005) ATS/ERS task force: standardisation of spirometry. Eur Respir J 26:319–338. https://doi.org/10.1183/09031936.05.00034805
Criée CP, Baur X, Berdel D, Bösch D, Gappa M, Haidl P, Husemann K, Jörres RA, Kabitz HJ, Kardos P, Köhler D (2015) Leitlinie zur spirometrie leitlinie der deutschen atemwegsliga, der deutschen gesellschaft für pneumologie und beatmungsmedizin und der deutschen gesellschaft für arbeitsmedizin und umweltmedizin zur spirometrie. Pneumologie 69(3):147–164
Criée CP, Sorichter S, Smith HJ, Kardos P, Merget R, Heise D, Berdel D, Köhler D, Magnussen H, Marek W, Mitfessel H (2011) Body plethysmography—its principles and clinical use. Respir Med 105(7):959–971. https://doi.org/10.1016/j.rmed.2011.02.006
Laufer B, Krüger-Ziolek S, Docherty PD, Höflinger F, Reindl L, Möller K (2019) Tidal volume via circumferences of the upper body: a pilot study. In: 2019 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) 3559–3562 https://doi.org/10.1016/10.1109/EMBC.2019.8857688
Perez WI, Tobin MJ (1985) Separation of factors responsible for change in breathing pattern induced by instrumentation. J Appl Physiol 59(5):1515–1520. https://doi.org/10.1152/jappl.1985.59.5.1515
Binks AP, Banzett RB, Duvivier C (2006) An inexpensive, MRI compatible device to measure tidal volume from chest-wall circumference. Physiol Meas 28(2):149. https://doi.org/10.1088/0967-3334/28/2/004
Chadha TS, Watson H, Birch S, Jenouri GA, Schneider AW, Cohn MA, Sackner MA (1982) Validation of respiratory inductive plethysmography using different calibration procedures. Am Rev Respir Dis 125(6):644–649. https://doi.org/10.1164/arrd.1982.125.6.644
Sackner MA, Watson HE, Belsito AS, Feinerman DR, Suarez MA, Gonzalez GE, Bizousky FR, Krieger BR (1989) Calibration of respiratory inductive plethysmograph during natural breathing. J Appl Physiol 66(1):410–420. https://doi.org/10.1152/jappl.1989.66.1.410
Konno K, Mead J (1967) Measurement of the separate volume changes of rib cage and abdomen during breathing. J Appl Physiol 22(3):407–422. https://doi.org/10.1152/jappl.1967.22.3.407
Khademi H, Malekzadeh R, Pourshams A, Jafari E, Salahi R, Semnani S, Abaie B, Islami F, Nasseri-Moghaddam S, Etemadi A, Byrnes G (2012) Opium use and mortality in Golestan Cohort Study: prospective cohort study of 50 000 adults in Iran. BMJ. https://doi.org/10.1136/bmj.e2502
Reljin N, Reyes BA, Chon KH (2015) Tidal volume estimation using the blanket fractal dimension of the tracheal sounds acquired by smartphone. Sensors 15(5):9773–9790. https://doi.org/10.3390/s150509773
Alexandre AR, Rocha FR, Landeiro L, Mota P, Jones J, Gomes JA (2021) Impact of height estimation on tidal volume calculation for protective ventilation—A prospective observational study. Crit Care Explor 3(5):e422. https://doi.org/10.1097/CCE.0000000000000422
Macavei VM, Spurling KJ, Loft J, Makker HK (2013) Diagnostic predictors of obesity-hypoventilation syndrome in patients suspected of having sleep disordered breathing. J Clin Sleep Med 9(9):879–884. https://doi.org/10.5664/jcsm.2986
American Academy of Sleep Medicine. International classification of sleep disorders (2005) Diagnostic and coding manual 51–5.
Goldman M, Smith H, Ulmer W (2005) Whole-body plethysmography. Eur Respir Monogr 31:15
Stocks J, Godfrey S, Beardsmore C, Bar-Yishay E, Castile R (2001) Plethysmographic measurements of lung volume and airway resistance. Eur Respir J 17(2):302–312. https://doi.org/10.1183/09031936.01.17203020
Funding
This research did not receive any grant funding from public agencies, or commercial, or not-for-profit sectors.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
B. Amra, B. Behvandi, F. Soltaninejad, M. Salmasi, A. Feizi, M. Glos, I. Fietze, T. Penzel, and M. Salanitro declare that they have no competing interests. All authors confirm that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Additional informed consent was obtained from all individual participants from whom identifying information is included in this article.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.

Scan QR code & read article online
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Amra, B., Behvandi, B., Soltaninejad, F. et al. Tidal volume measurement in patients with obesity hypoventilation syndrome and opium dependence. Somnologie 27, 265–271 (2023). https://doi.org/10.1007/s11818-023-00434-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11818-023-00434-3