Abstract
Background
Adherence to positive airway pressure (PAP) therapy is essential for the benefits of therapy to be realised. Telemedicine-based strategies provide a new option for enhanced monitoring and intervention to promote adherence during PAP. This study investigated the impact of telemedicine-based proactive patient management on PAP therapy termination rates versus standard care.
Methods
Observational data were obtained from ResMed Germany Healthcare, a German homecare provider. Patients were undergoing routine homecare using either a standard or proactive management strategy. The proactive strategy used data from AirView™, a cloud-based remote monitoring system, to prompt patient contact and information sharing/education. Patients receiving their first PAP therapy were included and analysed in matched pairs.
Results
In all, 3401 patients were included in each group. In the first year of PAP therapy, overall therapy termination rate was significantly lower (5.4% vs 11.0%; p < 0.001) and time to therapy termination was significantly longer (348 ± 58 vs 337 ± 76 days; p < 0.05) in the proactive versus standard care group. Cox proportional hazard analysis revealed a significantly reduced risk of PAP termination in the proactive versus the standard care group (hazard ratio 0.48, 95% confidence interval 0.4–0.57). Findings were consistent in subanalyses according to gender, type of device and insurance status, and in patients aged ≥40 years. However, in the subgroup of patients aged younger than 40 years, the risk of PAP termination was similar in the proactive and standard groups.
Conclusion
A telemedicine-based proactive management strategy compared with standard care of PAP patients was associated with a lower long-term therapy termination rate.
Zusammenfassung
Hintergrund
Eine ausreichende Therapieadhärenz ist erforderlich, um das therapeutische Potenzial einer Positivdrucktherapie (PAP) auszuschöpfen. Telemedizinbasierte Strategien ermöglichen durch Therapiemonitoring proaktive individuelle und zeitnahe Interventionen, um die PAP-Adhärenz zu steigern. Diese Studie untersuchte den Einfluss der telemedizinbasierten proaktiven Versorgung von Patienten mit Schlafapnoe auf die Abbruchrate der PAP-Therapie.
Methoden
Beobachtungsdaten wurden durch den Deutschen Homecare Provider ResMed Healthcare bereitgestellt. Im Rahmen eines Fall-Kontroll-Designs wurden Patienten, die erstmalig auf eine PAP-Therapie eingestellt wurden, mit PAP-Standardversorgung und proaktiver PAP-Versorgung verglichen. Die proaktive PAP-Versorgung erfolgte mit Hilfe der AirView™-Technologie, einem Cloud-basierten automatisierten Monitoringsystem, das bei Therapieproblemen einen zeitnahen proaktiven Patientenkontakt und Informationsaustausch bzw. eine Patientenschulung einleiten kann.
Ergebnisse
In beide Gruppen wurden jeweils 3401 Patienten eingeschlossen. Innerhalb des ersten Jahres nach der Einstellung auf eine PAP-Therapie war in der Gruppe mit proaktiver PAP-Versorgung im Vergleich zu einer PAP-Standardversorgung die Abbruchrate der PAP-Therapie signifikant niedriger (5,4 % vs. 11,0 %; p < 0,001) und die durchschnittliche Zeit bis zum Therapieabbruch signifikant länger (348 ± 58 vs. 337 ± 76 Tage; p < 0,05). Diese Verbesserung war unabhängig von Geschlecht, Gerätetyp und Art der Krankenversicherung sowie bei Patienten im Alter von >40 Jahren nachweisbar. In der Gruppe der Patienten unter 40 Jahren war kein Vorteil einer proaktiven Strategie nachweisbar.
Schlussfolgerung
Ein telemedizinbasiertes proaktives PAP-Versorgungsmodell ist mit einer signifikant geringeren Abbruchrate als bei einer konventionellen PAP-Versorgung assoziiert.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Continuous positive airway pressure (CPAP) is the most effective treatment for obstructive sleep apnoea (OSA) and is mandated by current guidelines [1]. However, the benefits of treatment cannot be realised if patients do not adhere to prescribed therapy.
A number of different approaches to improve compliance with CPAP therapy have been assessed in clinical trials, including patient education [2–5], peer support [6, 7], behavioural interventions [3, 4], clinical support [8] and choice of equipment [9–12]. In addition, features of the healthcare system and service/treatment delivery can have an impact on adherence [13]. Despite this, health stakeholders have not traditionally focussed on addressing adherence issues, and there are relatively few data on interventions to improve adherence, which in turn could improve patient outcomes [13].
Regular follow-up by clinicians and objective assessment of device data have been suggested to be essential in facilitating good compliance with CPAP therapy for OSA [14]. However, current management of CPAP compliance is reactive. Patients who are experiencing challenges or difficulties during CPAP therapy have to contact their healthcare provider to request assistance. Alternatively, less motivated patients might just stop using therapy altogether if there is a problem, or if they perceive there to be a problem.
Telemedicine is a term used to describe the use of information and communications technology to deliver healthcare at a distance [15]. It also offers the opportunity to provide quality care without dramatically escalating healthcare costs [16]. It is especially suited to the management of chronic diseases that require long-term monitoring to ensure successful therapy [17]. Having remote access to CPAP therapy data creates an opportunity for healthcare professionals to intervene proactively and provide appropriate support to individual patients with the aim of having them continue to use PAP therapy. Remote data are useful for tracking CPAP adherence and can also provide information on mask leak and residual apnoea–hypopnoea index (AHI), but there are few data on how use of these systems impacts on outcomes in OSA patients [18].
This study used data from a large German homecare provider to investigate the impact of a proactive patient management programme supported by remote access to PAP therapy data on therapy termination compared with standard care.
Methods
Study data were obtained from ResMed Germany Healthcare, a German homecare provider. German data protection law allows for the use of such data, if anonymised, for scientific purposes. Therefore, patient informed consent was not necessary in this study.
Subjects
Patients who started PAP therapy for the first time between 1 September 2009 and 30 April 2014 and were being treated with CPAP, automatic continuous positive airway pressure (APAP), bilevel PAP or adaptive servo-ventilation (ASV) devices, administered via a nasal mask, nasal pillows or full face mask interface, were eligible for inclusion in this analysis.
Patients who had started PAP therapy ≥1 year previously were included. Those who were managed using a proactive, telemedicine-based strategy were matched with PAP users managed using standard care. Patients were excluded from the analysis if they had terminated PAP therapy for a reason that was not related to PAP compliance (e. g. their insurer refused to cover PAP therapy, patient death, transfer to ventilation device). The proactive and standard care cohorts were matched for gender, insurance (public versus private), age (10-year bands), first device used (CPAP, APAP, bilevel PAP or ASV), therapy start date, initial interface (nasal mask, nasal pillows or full face mask) and introduction to device and interface (in sleep laboratory, at home by the homecare provider or at the homecare provider’s service centre).
Patient management
Standard care for each patient varied depending on their health insurance, but generally included polygraphy after about 6 months and when clinically necessary thereafter, and or patient contact with questions about device usage. If issues arose during PAP therapy the patient was advised to contact his/her healthcare provider, sleep laboratory and/or treating physician. Depending on their insurer, standard care patients might be contacted at 6 months to provide device usage data.
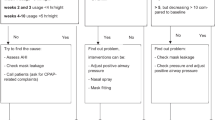
The proactive management strategy was facilitated by data obtained via AirView™, a cloud-based remote monitoring system that connects wirelessly with the PAP therapy devices used by ResMed Germany Healthcare. If AirView™ data showed that compliance during the first 2 weeks of PAP therapy did not meet the required level (average <4 h/day), patients received a telephone call from the homecare provider. For the period from 2 weeks to 6 months, average usage was checked every 14 days and patients were called again each time continued periods of no or low (<4 h/day) usage were identified from AirView™ data. From 6 months of therapy onwards, patients were notified via telephone call or letter if AirView™ data showed that PAP device usage dropped significantly or did not reach the required threshold (based on the insurance-specified usage criteria for reimbursement [approximate average of 4 h/night]). When contacted, patients were proactively provided with detailed information on use of their PAP therapy device and management of side effects (e. g. upper airway dryness, pressure, ulcers).
Assessments
Patient characteristics, therapy termination rates and time to therapy termination were calculated for the standard care and proactive strategy groups based on data extracted from patient records held by ResMed Germany Healthcare, a German homecare provider. Therapy termination was then described as compliance related if it occurred as the result of patient decision or behavior, was related to complaints by the patient, or was related to the PAP interface (i. e. not accepting or tolerating PAP therapy). Terminations that were not compliance related occurred when a patient was lost to follow-up, transferred to a ventilation device or died, or were related to insurance coverage issues or patient transfer to another homecare provider.
Data analysis
Descriptive statistics are shown as absolute and relative frequency or mean ± standard deviation (SD), as appropriate. Numerical differences between groups were analysed using t‑tests because the sample size allows for this approximation. For categorical data, differences in proportions were analysed using Z‑tests.
Matching was used to eliminate the influence of unequal distributions of risk factors on differences in compliance between the proactive care and standard care groups. Propensity score matching was conducted based on the following factors: gender, insurance, age (10-year bands), first device, therapy start date, first mask and therapy set up. Time to therapy termination was analyzed using Cox proportional hazards regression with standard vs. proactive care included as an explanatory factor. The results for marginal times to termination were visualized using Kaplan–Meier plots. A p-value of <0.05 was considered statistically significant. All statistical analyses were performed using IBM SPSS Statistics 22 and R version 3.0.2.
Results
A total of 3401 patients who were managed using the proactive strategy and 3401 matched patients receiving standard care were included in the analysis. There were no significant differences between the matched groups at baseline (Table 1).
PAP termination
The overall therapy termination rate was more than halved by the proactive strategy compared with standard care (5.4% vs 11.0%, respectively) (Table 2). Mean time to therapy termination in the first year was 348 ± 58 days in the proactive strategy group compared with 337 ± 76 days in the standard care group. Cox proportional hazard analysis revealed a significantly reduced risk of PAP termination in the proactive versus the standard care group (hazard ratio 0.48, 95% confidence interval 0.4–0.54) (Fig. 1).
Subanalyses according to potential modifiers
Therapy termination rates with the standard and proactive strategies were similar in males and females (Table 2), and for both genders the proactive strategy significantly reduced the risk of therapy termination compared with standard management. The risk of therapy termination was significantly reduced by the proactive strategy versus standard care in patients aged ≥40–69 years and ≥70 years, but there was no significant difference between the two management strategies in younger patients (age <40 years) (Fig. 2). Therapy termination risk reduction with proactive management was the greatest (63.9%) in patients receiving bilevel PAP or ASV, although the risk of therapy termination did not differ significantly based on the type of device used (Table 2). The overall significant benefit of the proactive strategy compared with standard care on therapy termination was the same in patients whose first device was APAP or CPAP, or bilevel or ASV. The proactive strategy was also effective in reducing the therapy termination rate in patients with either public or private insurance, with a slightly greater beneficial effect in those with public insurance.
Discussion
The results of this study showed that a telemedicine-based proactive strategy for the management of patients receiving PAP therapy reduced the rate of therapy termination over time, irrespective of gender, device used and the type of insurance. The finding that the proactive strategy was effective irrespective of the type of PAP device used might suggest that therapy acceptability (e. g. interface comfort) may play a bigger role than the type of device being used. Interestingly, the positive effects of proactive care were seen in all age groups except those aged <40 years. One possible explanation for this may be that the youngest patients are more comfortable with using information technology to better inform themselves about their condition.
To the best of our knowledge, this study is the first to investigate the impact of a telemedicine-based strategy on long-term continuation of PAP therapy. The main outcome measure in many previous studies has been compliance in terms of hours of device usage per night, and there is a lack of data on therapy termination rates. The majority of these studies have focussed only on CPAP in patients with OSA, used a variety of different telemedicine-based strategies, and reported inconsistent results. In addition, the majority were small, single-centre and/or short-term studies [19–24].
Interface problems and side effects are the most common causes of CPAP intolerance and an important potential benefit of telemedicine-based strategies is early identification of these issues and the implementation of strategies to resolve them in a timely fashion. Other benefits of telemonitoring strategies have been reported, such as reduced labour time for healthcare staff, allowing the same levels of adherence and efficacy to be achieved with fewer staff resources [17, 25]. This is important in the context of a projected increase in the number of patients presenting with sleep-disordered breathing due to the aging population and the growing rates of comorbidities such as obesity and diabetes that will increase demand for already stretched healthcare services. The ability to utilise telemedicine to provide patient-focussed, proactive and intensive management of patients using PAP therapy facilitates the management of large patient populations, as well as significantly improving therapy continuation rates and usage. Telemedicine also appears to be a cost effective strategy for managing CPAP treatment in patients with OSA [16].
Improved patient engagement is another potential advantage of telemedicine strategies, which in turn could contribute to additional gains in terms of PAP adherence and outcomes. The positive impact of patient involvement in therapy was highlighted in a recent study which showed that providing patients with web-based access to their personal PAP usage data increased device usage in the first week of therapy and over 3 months of follow-up compared with usual care [26].
According to the World Health Organisation (WHO), it is important that health systems evolve to meet new challenges [13]. The introduction of new technologies, such as telemedicine, is one way in which healthcare systems and providers can advance patient care. In the case of PAP therapy this could contribute to better adherence and improved patient outcomes. WHO has also recognised that interventions targeted at improving adherence should provide a significant positive return on investment via prevention of adverse health outcomes, and that there are also economic benefits associated with improved quality of life, including reductions in indirect costs and increased productivity [13]. In the context of CPAP therapy for OSA, the importance of increasing adherence has also been recognised by a Cochrane review that emphasised the need for new strategies to promote CPAP compliance [27].
One of the main limitations of this analysis is the lack of specific data on device usage in the standard care group. Thus, we report therapy termination rates rather than device usage hours, which made comparison with existing data more difficult. However, it could be argued that therapy termination is the most important endpoint to assess because once a patient stops therapy getting them to restart is likely to be difficult. In contrast, reduced usage provides a cue for intervention, and there is always the possibility that hours of device use will increase. Another potential limitation is that the ability to accurately determine therapy termination is different in the two groups – it is very obvious when patients in the proactive strategy group stop PAP therapy based on device data received, but therapy termination might be less obvious in the standard care group for whom the requirement for a homecare provider visit may be the only indicator of therapy problems or termination. Interrogation of big data allows inclusion of a very large population of patients treated in routine clinical practice. However, databases created for administrative, rather than scientific, purposes do not include a full set of clinical data. In the current analysis, this means that we had no data on clinical features, insomnia and other factors that might have influenced the outcomes under study. Furthermore, these are observational data and therefore do not provide proof for a causative relationship between the intervention (proactive care) and the outcome (reduced therapy termination). Data from randomised clinical trials are needed to more clearly elucidate the effects of a telemedicine-based proactive management strategy on PAP therapy termination rates. In addition, future research should further investigate the potential of telemonitoring-guided proactive care on additional endpoints, including device usage, resource use and healthcare professional time requirements.
Conclusion
This analysis of a large population of PAP therapy users in Germany showed that the long-term rate of therapy termination was significantly lower in patients managed using a telemedicine-based proactive management strategy compared with standard care. This association was observed in both men and women, across a wide range of patient ages, for all PAP devices and in patients with public or private insurance. Implementation of telemedicine-based strategies has the potential to improve adherence and patient outcomes and may allow more efficient allocation of scarce healthcare resources. Randomized, controlled clinical trials are needed to further assess both therapy termination rates and device usage, which would complement our large data set from routine clinical practice.
References
Epstein LJ, Kristo D, Strollo PJ Jr., Friedman N, Malhotra A, Patil SP et al (2009) Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med 5:263–276
Damjanovic D, Fluck A, Bremer H, Muller-Quernheim J, Idzko M, Sorichter S (2009) Compliance in sleep apnoea therapy: influence of home care support and pressure mode. Eur Respir J 33:804–811
Smith I, Nadig V, Lasserson TJ (2009) Educational, supportive and behavioural interventions to improve usage of continuous positive airway pressure machines for adults with obstructive sleep apnoea. Cochrane Database Syst Rev 2009:CD007736
Smith CE, Dauz E, Clements F, Werkowitch M, Whitman R (2009) Patient education combined in a music and habit-forming intervention for adherence to continuous positive airway (CPAP) prescribed for sleep apnea. Patient Educ Couns 74:184–190
Fuchs FS, Pittarelli A, Hahn EG, Ficker JH (2010) Adherence to continuous positive airway pressure therapy for obstructive sleep apnea: impact of patient education after a longer treatment period. Respiration 80:32–37
Brostrom A, Fridlund B, Ulander M, Sunnergren O, Svanborg E, Nilsen P (2013) A mixed method evaluation of a group-based educational programme for CPAP use in patients with obstructive sleep apnea. J Eval Clin Pract 19:173–184
Parthasarathy S, Wendel C, Haynes PL, Atwood C, Kuna S (2013) A pilot study of CPAP adherence promotion by peer buddies with sleep apnea. J Clin Sleep Med 9:543–550
Hoy CJ, Vennelle M, Kingshott RN, Engleman HM, Douglas NJ (1999) Can intensive support improve continuous positive airway pressure use in patients with the sleep apnea/hypopnea syndrome? Am J Respir Crit Care Med 159:1096–1100
Massie CA, Hart RW (2003) Clinical outcomes related to interface type in patients with obstructive sleep apnea/hypopnea syndrome who are using continuous positive airway pressure. Chest 123:1112–1118
Vennelle M, White S, Riha RL, Mackay TW, Engleman HM, Douglas NJ (2010) Randomized controlled trial of variable-pressure versus fixed-pressure continuous positive airway pressure (CPAP) treatment for patients with obstructive sleep apnea/hypopnea syndrome (OSAHS). Sleep 33:267–271
Wimms AJ, Richards GN, Benjafield AV (2013) Assessment of the impact on compliance of a new CPAP system in obstructive sleep apnea. Sleep Breath 17:69–76
Wiest GH, Harsch IA, Fuchs FS, Kitzbichler S, Bogner K, Brueckl WM et al (2002) Initiation of CPAP therapy for OSA: does prophylactic humidification during CPAP pressure titration improve initial patient acceptance and comfort? Respiration 69:406–412
Sabate E (2003) Adherence to long-term therapies: evidence for action. World Health Organization, Geneva
Bollig SM (2010) Encouraging CPAP adherence: it is everyone’s job. Respir Care 55:1230–1239
Pare G, Jaana M, Sicotte C (2007) Systematic review of home telemonitoring for chronic diseases: the evidence base. J Am Med Inform Assoc 14:269–277
Isetta V, Negrin MA, Monasterio C, Masa JF, Feu N, Alvarez A et al (2015) A Bayesian cost-effectiveness analysis of a telemedicine-based strategy for the management of sleep apnoea: a multicentre randomised controlled trial. Thorax 70:1054–1061
Munafo D, Hevener W, Crocker M, Willes L, Sridasome S, Muhsin M (2016) A telehealth program for CPAP adherence reduces labor and yields similar adherence and efficacy when compared to standard of care. Sleep Breath. doi:10.1007/s11325-015-1298-4
Schwab RJ, Badr SM, Epstein LJ, Gay PC, Gozal D, Kohler M et al (2013) An official American Thoracic Society statement: continuous positive airway pressure adherence tracking systems. The optimal monitoring strategies and outcome measures in adults. Am J Respir Crit Care Med 188:613–620
Fox N, Hirsch-Allen AJ, Goodfellow E, Wenner J, Fleetham J, Ryan CF et al (2012) The impact of a telemedicine monitoring system on positive airway pressure adherence in patients with obstructive sleep apnea: a randomized controlled trial. Sleep 35:477–481
Smith CE, Dauz ER, Clements F, Puno FN, Cook D, Doolittle G et al (2006) Telehealth services to improve nonadherence: a placebo-controlled study. Telemed J E Health 12:289–296
Sparrow D, Aloia M, Demolles DA, Gottlieb DJ (2010) A telemedicine intervention to improve adherence to continuous positive airway pressure: a randomised controlled trial. Thorax 65:1061–1066
Stepnowsky CJ, Palau JJ, Marler MR, Gifford AL (2007) Pilot randomized trial of the effect of wireless telemonitoring on compliance and treatment efficacy in obstructive sleep apnea. J Med Internet Res 9:e14
Taylor Y, Eliasson A, Andrada T, Kristo D, Howard R (2006) The role of telemedicine in CPAP compliance for patients with obstructive sleep apnea syndrome. Sleep Breath 10:132–138
Frasnelli M, Baty F, Niedermann J, Brutsche MH, Schoch OD (2016) Effect of telemetric monitoring in the first 30 days of continuous positive airway pressure adaptation for obstructive sleep apnoea syndrome – a controlled pilot study. J Telemed Telecare 22:209–214
Anttalainen U, Melkko S, Hakko S, Laitinen T, Saaresranta T (2016) Telemonitoring of CPAP therapy may save nursing time. Sleep Breath. doi:10.1007/s11325-016-1337-9
Kuna ST, Shuttleworth D, Chi L, Schutte-Rodin S, Friedman E, Guo H et al (2015) Web-based access to positive airway pressure usage with or without an initial financial incentive improves treatment use in patients with obstructive sleep apnea. Sleep 38:1229–1236
Balk EM, Chung M, Chan JA, Moorthy D, Patel K, Concannon TW et al (2012) Future research needs for treatment of obstructive sleep apnea. Future research needs Paper No. 12. (prepared by the Tufts Evidence-based Practice Center under contract No. 290-2007-10055 I.) AHRQ Publication No. 12-EHC033-EF. Rockville, MD: Agency for Healthcare Research and Quality. https://www.ncbi.nlm.nih.gov/books/NBK84494/ (Created February 2012)
Acknowledgements
Medical writing assistance was provided by Nicola Ryan, independent medical writer, funded by ResMed.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
H. Woehrle was employed by ResMed during the conduct of the study. Dr. Ficker reports personal fees and non-financial support from ResMed, personal fees and non-financial support from Weinmann, outside the submitted work. A. Graml is an employee of ResMed Germany Inc. I. Fietze reports grants and personal fees from ResMed, grants from Philips, grants from Fisher&Paykel, grants from Hoffrichter, grants from Heinen&Löwenstein, grants from Weinmann, outside the submitted work. Dr. Young reports personal fees from Sanofi Genzyme, personal fees from Biomarin, personal fees from UCB Pharma, personal fees from Medice, personal fees from ResMed, personal fees from Heinen&Löwenstein, personal fees from Vanda, grants from Löwensteinstiftung, grants from German Ministry of Education and Science (BMBF), outside the submitted work. Dr. Teschler reports grants, personal fees and non-financial support from ResMed, during the conduct of the study; grants, personal fees and non-financial support from ResMed, outside the submitted work. Dr. Arzt reports grants and personal fees from ResMed, grants and personal fees from Philips Respironics, outside the submitted work.
Study data were obtained from ResMed Germany Healthcare, a German homecare provider. German data protection law allows for the use of such data, if anonymised, for scientific purposes. Therefore, patient informed consent was not necessary in this study.
Rights and permissions
Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Woehrle, H., Ficker, J.H., Graml, A. et al. Telemedicine-based proactive patient management during positive airway pressure therapy. Somnologie 21, 121–127 (2017). https://doi.org/10.1007/s11818-016-0098-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11818-016-0098-9