Abstract
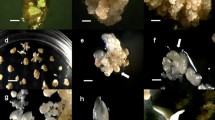
This study describes for the first time in Pinus genus a plant regeneration system via a combined pathway of somatic embryogenesis and organogenesis from immature seeds of radiata pine. Somatic embryos were obtained from embryogenic line 2162 of Pinus radiata D. Don on EDM basal medium containing 60 μM ABA and 6% sucrose. The explants used for organogenesis experiments were either freshly collected somatic embryos or somatic embryos germinated for 1 week. Germination medium was half-strength LP medium, supplemented with 0.2% activated charcoal. Different induction periods and BA concentrations were assayed for shoot induction. After induction treatments, explants were elongated on the same medium used for germination stage. Rooting medium was quarter-strength LP medium supplemented with three different auxin treatments: 1.5 mg L−1 1-naphthalene acetic acid (NAA), 1.5 mg L−1 indole-3-butyric acid (IBA) and 1 mg L−1 IBA with 0.5 mg L−1 NAA (MIX). The effect of the photon flux (120 mmol m−2 s−1 and darkness) in the first week of the explants in the rooting media was also tested. This methodology could offer an alternative to overcome some problems associated with somatic embryogenesis such as the seasonality of embryogenic tissue (ET) initiation or a low embryo production from the ET, a particularly important issue in the case of genetically transformed ETs.




Similar content being viewed by others
References
Aitken-Christie J, Singh AP, Horgan KJ, Thorpe TA (1985) Explant developmental state and shoot formation in Pinus radiata cotyledons. Bot Gaz 146:196–203
Aitken-Christie J, Singh AP, Davies H (1988) Multiplication of meristematic tissue: a new tissue culture system for radiata pine. In: Hanover JW, Keathley DE (eds) Genetic manipulation of woody plants. Plenum, New York, pp 413–432
Alonso P, Moncaleán P, Fernández B, Rodríguez A, Centeno ML, Ordás RJ (2006) An improved micropropagation protocol for stone pine (Pinus pinea L.). Ann For Sci 63:1–7
Álvarez JM, Majada J, Ordás RJ (2009) An improved micropropagation protocol for maritime pine (Pinus pinaster Ait.) isolated cotyledons. Forestry 82:175–184
Bergmann BA, Stomp AM (1992) Influence of taxonomic relatedness and medium composition on meristematic nodule and adventitious shoot formation in nine species. Can J For Res 22:750–755
Biondi S, Thorpe TA (1982) Growth regulator effects, metabolite changes, and respiration during shoot initiation in cultured cotyledon explants of Pinus radiata. Bot Gaz 143:20–25
Brassard N, Brissette L, Lord D, Laliberté S (1996) Elongation, rooting and acclimatization of micropropagated shoots from mature material of hybrid larch. Plant Cell Tissue Organ Cult 44:37–44
Brown MB, Forsythe AB (1974) Robust test for the equality of variance. J Am Stat Assoc 69:364–367
Capuana M, Giannini R (1995) In vitro plantlet regeneration from embryonic explants of Pinus pinea L. In Vitro Cell Dev Biol Plant 31:202–206
Carneros E, Celestino C, Klimaszewska K, Toribio M, Bonga JM (2009) Plant regeneration in Stone pine (Pinus pinea L.) by somatic embryogenesis. Plant Cell Tissue Organ Cult 98:165–178
Choudhury H, Kumaria S, Tandon P (2007) Induction and maturation of somatic embryos from intact megagametophyte explants in Khasi pine (Pinus kesiya Royle ex. Gord.). Curr Sci 95:1433–1438
Cortizo M, de Diego N, Moncaleán P, Ordás RJ (2009) Micropropagation of adult Stone pine (Pinus pinea L.). Trees Struct Funct 23:835–842
Davis JM, Becwar MR (2007) Developments in tree cloning. Developments in fibres and fibre treatment series, PIRA, UK
De Diego N, Montalbán IA, Fernández de Larrinoa E, Moncaleán P (2008) In vitro regeneration of Pinus pinaster adult trees. Can J For Res 38:2607–2615
De Diego N, Montalbán IA, Moncaleán P (2010) In vitro regeneration of adult Pinus sylvestris trees. S Afr J For Res 76:158–162
De Diego N, Montalbán IA, Moncaleán P (2011) Improved micropropagation protocol for maritime pine using zygotic embryos. Scand J For Res (in press)
Fehér A, Pasternak TP, Dudits D (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tissue Organ Cult 74:201–228
Flygh G, Grönroos R, von Arnold S (1993) Induction, rooting, and growth capacity of adventitious shoots of Pinus contorta. Can J For Res 23:1907–1916
Garin E, Isabel N, Plourde A (1998) Screening of large numbers of seed families of Pinus strobus L. for somatic embryogenesis from immature zygotic embryos. Plant Cell Rep 18:37–43
Giri CC, Shyamkumar B, Anjaneyulu C (2004) Progress in tissue culture, genetic transformation and applications of biotechnology to trees: an overview. Trees 18:115–135
Guohua M (1998) Effects of cytokinins and auxins on cassava shoot organogenesis and somatic embryogenesis from somatic embryo explants. Plant Cell Tissue Organ Cult 54:1–7
Gupta PK, Durzan DJ (1986) Somatic polyembryogenesis from callus of mature sugar pine embryos. Bio/Technol 4:643–645
Hargreaves CL, Grace LJ, van der Maas SA, Menzies MI, Kumar S, Holden DG, Foggo MN, Low CB, Dibley MJ (2005) Comparative in vitro and early nursery performance of adventitious shoots from cryopreserved cotyledons and axillary shoots from epicotyls of the same zygotic embryo of control-pollinated Pinus radiata. Can J For Res 35:2629–2641
Jiménez VM (2005) Involvement of plant hormones and plant growth regulators on in vitro somatic embryogenesis. Plant Growth Regul 47:91–110
Khan T, Singh A, Pant RC (2006) Regeneration via somatic embryogenesis and organogenesis in different cultivars of cotton (Gossypium spp.). In Vitro Cell Dev Biol Plant 42:498–501
Kim SW, Oh MJ, Liu JR (2009) Plant regeneration from the root-derived embryonic tissues of Rosa hybrida L. cv. Charming via a combined pathway of somatic embryogenesis and organogenesis. Plant Biotechnol Rep 3:341–345
Klimaszewska K, Park YS, Cyr DR, Overton C, Maceacheron I, Bonga JM (2001) Optimized somatic embryogenesis in Pinus strobus L. In Vitro Cell Dev Biol Plant 37:392–399
Klimaszewska K, Trontin JF, Becwar MR, Devillard C, Park YS, Lelu-Walter MA (2007) Recent progress in somatic embryogenesis of four Pinus spp. Tree For Sci Biotechnol 1:11–25
Lambardi M, Sharma KK, Thorpe TA (1993) Optimization of in vitro bud induction and plantlet formation from mature embryos of aleppo pine (Pinus halepensis Mill.). In Vitro Cell Dev Biol 29:189–199
Lelu-Walter MA, Bernier-Cardou M, Klimaszewska K (2008) Clonal plant production from self- and cross-pollinated seed families of Pinus sylvestris (L.) through somatic embryogenesis. Plant Cell Tissue Organ Cult 92:31–45
MacKay JJ, Beckwar MR, Park YS, Corderro JP, Pullman GS (2006) Genetic control of somatic embryogenesis initiation in loblolly pine and implications for breeding. Tree Genet Genomes 2:1–9
Martinez Pulido C, Harry IS, Thorpe TA (1990) In vitro regeneration of plantlets of Canary Island pine (Pinus canariensis). Can J For Res 20:1200–1211
Martínez Pulido C, Harry IS, Thorpe TA (1992) Optimization of bud induction in cotyledonary explants of Pinus canariensis. Plant Cell Tissue Organ Cult 29:247–255
Martínez Pulido C, Harry IS, Thorpe TA (1994) Effect of various induction treatments on elongation and rooting of adventitious shoots of Canary Island pine (Pinus canariensis). Plant Cell Tissue Organ Cult 39:225–230
Mathur G, Nadgauda R (1999) In vitro plantlet regeneration from mature somatic embryos of Pinus wallichiana A.B. Jacks. Plant Cell Rep 19:74–80
Moncaleán P, Alonso P, Centeno ML, Cortizo M, Rodríguez A, Fernández B, Ordás RJ (2005) Organogenic responses of Pinus pinea cotyledons to hormonal treatments: BA metabolism and cytokinin content. Tree Physiol 25:1–9
Montalbán IA (2011) Desarrollo y optimización de herramientas biotecnológicas para la obtención de material clonal de Pinus radiata D. Don. PhD thesis, University of the Basque Country, Spain, pp 27–57
Montalbán IA, De Diego N, Moncaleán P (2010) Bottlenecks in Pinus radiata somatic embryogenesis: improving maturation and germination. Trees Struct Funct 24:1061–1071
Park YS (2002) Implementation of conifer somatic embryogenesis in clonal forestry: technical requirements and deployment considerations. Ann For Sci 59:651–656
Park YS, Barrett JD, Bonga JM (1998) Application of somatic embryogenesis in high-value clonal forestry: deployment, genetic control, and stability of cryopreserved clones. In Vitro Cell Dev Biol Plant 34:231–239
Prehn D, Serrano C, Mercado A, Stange C, Barrales L, Arce-Johnson P (2003) Regeneration of whole from apical meristems of Pinus radiata. Plant Cell Tissue Organ Cult 73:91–94
Quoirin M, Lepoivre P (1977) Études des milieux adaptés aux cultures in vitro de Prunus. Acta Hortic 78:437–442
Saborio F, Dvorak WS, Donahue JK, Thorpe T (1997) In vitro regeneration of plantlets from mature embryos of Pinus ayacahuite. Tree Physiol 17:787–796
Shapiro SS, Wilk MB (1965) Variance test for normality. Biometrika 52:591–611
Siva R, Rajasekaran C, Mudgal G (2009) Induction of somatic embryogenesis and organogenesis in Oldenlandia umbellata L., a dye-yielding medicinal plant. Plant Cell Tissue Organ Cult 98:205–211
Sriskandarajah S, Lundquist PO (2009) High frequency shoot organogenesis and somatic embryogenesis in juvenile and adult tissues of seabuckthorn (Hippophae rhamnoides L.). Plant Cell Tissue Organ Cult 99:259–268
Stange C, Prehn D, Gebauer M, Arce-Johnson P (1999) Optimization of in vitro culture conditions for Pinus radiata embryos and histological characterization of regenerated shoots. Biol Res 32:19–28
Stasolla C, Thorpe TA (2010) Tissue culture: historical perspectives and applications. In: Kumar A, Sopory SK (eds) Applications of plant biotechnology: in vitro propagation, plant transformations and secondary metabolite production. IK International, India, pp 1–39
Stasolla C, Yeung EC (2003) Recent advances in conifer somatic embryogenesis: improving somatic embryo quality. Plant Cell Tissue Organ Cult 74:15–35
Steward FC, Mapes MO, Smith J (1958) Growth and organized development of cultured cells. I. Growth and division of freely suspended cells. Am J Bot 45:693–703
Stojičić D, Budimir S, Ćulafić L (1999) Micropropagation of Pinus heldreichii. Plant Cell Tissue Organ Cult 59:147–150
Talbert CB, Ritchie GA, Gupta P (1993) Conifer vegetative propagation. In: Ahuja MR, Libby WJ (eds) Clonal forestry I. Genetics and biochemistry. Springer, Berlin, pp 145–181
Tang W, Guo Z (2001) In vitro propagation of loblolly pine via direct somatic organogenesis from mature cotyledons and hypocotyls. Plant Growth Regul 33:25–31
Tang W, Guo Z, Ouyang F (2001) Plant regeneration from embryogenic cultures initiated from mature loblolly pine zygotic embryos. In Vitro Cell Dev Biol Plant 37:558–563
Valdés AE, Centeno ML, Ordás RJ, Fernández B (2001) Relationships between hormonal contents and organogenic response in Pinus pinea L. cotyledons taken from plantlets at different germination periods. Plant Physiol Biochem 39:377–384
Villalobos-Amador E, Rodríguez-Hernández G, Pérez-Molphe-Balch E (2002) Organogenesis and Agrobacterium rhizogenes-induced rooting in Pinus maximartinezii Rzedowsky and P. pinceana Gordon. Plant Cell Rep 20:779–785
von Aderkas P, Label P, Lelu MA (2002) Charcoal affects early development and hormonal concentrations of somatic embryos of hybrid larch. Tree Physiol 22:431–434
Walter C (2004) Genetic engineering in conifer forestry: technical and social considerations. In Vitro Cell Dev Biol Plant 40:434–441
Walter C, Find JI, Grace LJ (1998) Somatic embryogenesis and genetic transformation in Pinus radiata. In: Jain SM, Gupta PK, Newton RJ (eds) Somatic embryogenesis in woody plants, vol 4. Kluwer, The Netherlands, pp 491–504
Webb DT, Flinn BS, Georgis W (1988) Micropropagation of eastern white pine (Pinus strobus L.). Can J For Res 18:1570–1580
Yeung EC, Aitken J, Biondi S, Thorpe TA (1981) Shoot histogenesis in cotyledon explants of radiata pine. Bot Gaz 142:494–501
Yildirim T, Kaya Z, Isik K (2006) Induction of embryogenic tissue and maturation of somatic embryos in Pinus brutia TEN. Plant Cell Tissue Organ Cult 87:67–76
Zdravković-Korać S, Ćalić-Dragosavac D, Uzelac B, Janošević, Budimir S, Vinterhalter B, Vinterhalter D (2008) Secondary somatic embryogenesis versus caulogenesis from somatic embryos of Aesculus carnea Hayne.: developmental stage impact. Plant Cell Tissue Organ Cult 94:225–231
Zimmerman JL (1993) Somatic embryogenesis: a model for early development in higher plants. Plant Cell 5:1411–1423
Acknowledgments
This research was funded by Ministerio de Ciencia y Tecnología-Spain (AGL2005-08214-CO2-02) and Departamento de Agricultura y Pesca-Gobierno Vasco (VEC2004021 and VED2007014), who granted Itziar A. Montalbán a PhD scholarship. Montserrat Mella and Elena Fernández de Larrinoa are thanked for their technical assistance. The authors are grateful to C. Hargreaves and her team from Scion (New Zealand) and K. Klimaszewska from Canadian Forest Service-Laurentian Forestry Center (Canada) for giving Itziar Montalbán the opportunity of learning about SE in their respective laboratories and for enriching discussions during the course of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Montalbán, I.A., De Diego, N., Aguirre Igartua, E. et al. A combined pathway of somatic embryogenesis and organogenesis to regenerate radiata pine plants. Plant Biotechnol Rep 5, 177–186 (2011). https://doi.org/10.1007/s11816-011-0171-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11816-011-0171-6