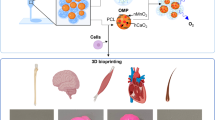
Abstract
Efforts to find sustainable and eco-friendly ways to conduct chemical reactions have led to the mimicking of nature. In this study, a new silica polymerization protein that can produce silica in an environmentally friendly manner was developed using cathepsin L-like protein (AqCtL) from Amphimedon queenslandica with a 61% sequence identity to that of silicatein-alpha, which is a natural biosilicifying enzyme. To stabilize the protein structure, heterologously expressed AqCtL in Escherichia coli was mutated into AqCtLSN by changing the amino acid residues responsible for protease cleavage. The insoluble form of AqCtLSN was reconstituted into a soluble protein through the refolding process, displaying silica-condensing activity from silicic acid. AqCtLSN self-assembled, aggregated, and attached to a support in the PBS buffer without losing silica deposition activity. These properties were applied to fabricate a silica-hybrid material using a gelatin-tyramine-alginate cross-linked hydrogel as a scaffold. FT-IR analysis revealed that a silica hybrid material was produced owing to the in situ silicification by AqCtLSN immobilized on the hydrogel. The surface of biosilica mediated by AqCtLSN demonstrated an increase in cell proliferation, alkaline phosphatase activity, and calcium mineral precipitation in the osteogenesis of MC3T3 E1 cells compared to those without biosilica. In conclusion, AqCtLSN, recombinantly expressed in E. coli, is a novel biosilica-forming protein that can be used to produce composites for biomedical applications, especially bone regeneration.
Similar content being viewed by others
References
L. A. Estroff, Chem. Rev., 108, 4329 (2008).
K. Shimizu, J. Cha, G. D. Stucky and D. E. Morse, Proc. Natl. Acad. Sci. USA, 95, 6234 (1998).
M. J. Olszta, D. J. Odom, E. P. Douglas and L. B. Gower, Connect. Tissue Res., 44 Suppl 1, 326 (2003).
H. P. Wiesmann, U. Meyer, U. Plate and H. J. Hohling, Int. Rev. Cytol., 242, 121 (2005).
L. B. Gower, Chem. Rev., 108, 4551 (2008).
H. M. Moura and M. M. Unterlass, Biomimetics (Basel), 5, 29 (2020).
F. Nudelman and N. A. Sommerdijk, Angew. Chem. Int. Ed. Engl., 51, 6582 (2012).
M. B. Dickerson, K. H. Sandhage and R. R. Naik, Chem. Rev., 108, 4935 (2008).
C. F. Böhm, J. Harris, P. I. Schodder and S. E. Wolf, Materials, 12, 2117 (2019).
N. Kroger, R. Deutzmann and M. Sumper, Science, 286, 1129 (1999).
M. A. A. Abdelhamid and S. P. Pack, Acta Biomater., 120, 38 (2021).
S. M. Bulatovic, Handbook of flotation reagents: Chemistry, theory and practice, in Beneficiation of silica sand, Elsevier, Canada (2015).
S. Jafari, H. Derakhshankhah, L. Alaei, A. Fattahi, B. S. Varnamkhasti and A. A. Saboury, Biomed. Pharmacother., 109, 1100 (2019).
Y. Wang, Y. Huang, H. Bai, G. Wang, X. Hu, S. Kumar and R. Min, Biosensors, 11, 472 (2021).
S. Kumari, K. H. Min, B. K. Kanth, E. K. Jang and S. P. Pack, Biotechnol. Bioprocess Eng., 25, 758 (2020).
H. Li, X. Chen, D. Shen, F. Wu, R. Pleixats and J. Pan, Nanoscale, 13, 15998 (2021).
C. J. Brinker and G. W. Scherer, Sol-gel science: The physics and chemistry of sol-gel processing, Academic Press, Boston, London (1990).
M. B. Dickerson, R. R. Naik, P. M. Sarosi, G. Agarwal, M. O. Stone K. H. Sandhage, J. Nanosci. Nanotechnol., 5, 63 (2005).
M. J. Limo, A. Sola-Rabada, E. Boix, V. Thota, Z. C. Westcott, V. Puddu and C. C. Perry, Chem. Rev., 118, 11118 (2018).
S. Görlich, A. J. Samuel, R. J. Best, R. Seidel, J. Vacelet, F. K. Leonarski, T. Tomizaki, B. Rellinghaus, D. Pohl and I. Zlotnikov, Proc. Natl. Acad. Sci. USA, 117, 31088 (2020).
H. C. Schröder, X. Wang, A. Manfrin, S.-H. Yu, V. A. Grebenjuk, M. Korzhev, M. Wiens, U. Schlossmacher and W. E. G. Müller, J. Biol. Chem., 287, 22196 (2012).
M.-R. Ki, E.-K. Jang and S. P. Pack, Process Biochem., 49, 95 (2014).
A. Krasko, B. Lorenz, R. Batel, H. C. Schroder, I. M. Muller and W. E. Muller, Eur. J. Biochem., 267, 4878 (2000).
W. E. Muller, A. Krasko, G. Le Pennec, R. Steffen, M. Wiens, M. S. Ammar, I. M. Muller and H. C. Schroder, Prog. Mol. Subcell. Biol., 33, 195 (2003).
M. Pozzolini, L. Sturla, C. Cerrano, G. Bavestrello, L. Camardella, A. M. Parodi, F. Raheli, U. Benatti, W. E. Muller and M. Giovine, Mar. Biotechnol. (NY), 6, 594 (2004).
X. Wang, H. C. Schröder and W. E. G. Müller, Trends. Biotechnol., 32, 441 (2014).
S. Y. Tabatabaei Dakhili, S. A. Caslin, A. S. Faponle, P. Quayle, S. P. de Visser and L. S. Wong, Proc. Natl. Acad. Sci. USA, 114, E5285 (2017).
M. R. Ki, K. B. Yeo and S. P. Pack, Bioprocess Biosyst. Eng., 36, 643 (2012).
H. Oguri, K. Nakashima, K. Godigamuwa, J. Okamoto, Y. Takeda, F. Okazaki, M. Sakono and S. Kawasaki, J. Biosci. Bioeng., 133, 222 (2022).
M. Fairhead, K. A. Johnson, T. Kowatz, S. A. McMahon, L. G. Carter, M. Oke, H. Liu, J. H. Naismith and C. F. van der Walle, Chem. Commun. (Camb), 15, 1765 (2008).
H. Kirschke, A. J. Barrett and N. D. Rawlings, Lysosomal cysteine proteases, 2nd Edn., Oxford University Press, Oxford, New York (1998).
W. E. Muller, A. Boreiko, X. Wang, S. I. Belikov, M. Wiens, V. A. Grebenjuk, U. Schlossmacher and H. C. Schroder, Gene, 395, 62 (2007).
N. V. Povarova, N. A. Barinov, M. S. Baranov, N. M. Markina, A. M. Varizhuk, G. E. Pozmogova, D. V. Klinov, V. B. Kozhemyako and K. A. Lukyanov, Sci. Rep., 8, 16759 (2018).
D. G. Kamenev, Y. N. Shkryl, G. N. Veremeichik, V. A. Golotin, N. N. Naryshkina, Y. O. Timofeeva, S. N. Kovalchuk, I. V. Semiletova and V. P. Bulgakov, J. Nanosci. Nanotechnol., 15, 10046 (2015).
C. L. Fisher and G. K. Pei, BioTechniques, 23, 570 (1997).
T. A. Elkhooly, W. E. G. Müller, X. Wang, W. Tremel, S. Isbert and M. Wiens, Bioinspir. Biomim., 9, 044001 (2014).
M. R. Ki, S. H. Kim, T. K. M. Nguyen, R. G. Son, S. H. Jun and S. P. Pack, ACS Biomater. Sci. Eng., (2022). DOI: https://doi.org/10.1021/acsbiomaterials.1c01095.
D. K. Lee, M.-R. Ki, E. H. Kim, C.-J. Park, J. J. Ryu, H. S. Jang, S. P. Pack, Y. K. Jo and S. H. Jun, Biomater. Res., 25, 13 (2021).
R. Andre, M. N. Tahir, T. Link, F. D. Jochum, U. Kolb, P. Theato, R. Berger, M. Wiens, H. C. Schroder, W. E. Muller and W. Tremel, Langmuir, 27, 5464 (2011).
E.-j. Cheon, S.-H. Kim, D.-K. Lee, Y.-K. Jo, M.-R. Ki, C.-J. Park, H.-S. Jang, J.-S. Ahn, S.-P. Pack and S.-H. Jun, Biotechnol. Bioprocess Eng., 26, 923, (2021).
L. N. Niu, K. Jiao, Y. P. Qi, S. Nikonov, C. K. Yiu, D. D. Arola, S. Q. Gong, A. El-Marakby, M. R. Carrilho, M. W. Hamrick, K. M. Hargreaves, A. Diogenes, J. H. Chen, D. H. Pashley and F. R. Tay, FASEB J., 26, 4517 (2012).
Z. Lipinszki, V. Vernyik, N. Farago, T. Sari, L. G. Puskas, F. R. Blattner, G. Posfai and Z. Gyorfy, ACS Synth. Biol., 7, 2656 (2018).
R. Menard, E. Carmona, S. Takebe, E. Dufour, C. Plouffe, P. Mason and J. S. Mort, J. Biol. Chem., 273, 4478 (1998).
J. D. Bendtsen, H. Nielsen, G. von Heijne and S. Brunak, J. Mol. Biol., 340, 783 (2004).
T. Coradin, A. Coupé and J. Livage, Colloids Surf. B Biointerfaces, 29, 189 (2003).
C. J. Oldfield and A. K. Dunker, Annu. Rev. Biochem., 83, 553 (2014).
J. Kozlowska, N. Stachowiak and A. Sionkowska, Polymers, 10, 456 (2018).
B.-P. Jiang, L. Zhang, Y. Zhu, X.-C. Shen, S.-C. Ji, X.-Y. Tan, L. Cheng and H. Liang, J. Mater. Chem. B., 3, 3767 (2015).
G. Singh, H. B. Dizaji, H. Puttuswamy and S. Sharma, Sustainability, 14, 539 (2022).
R. Plowright, N. Dinjaski, S. Zhou, D. J. Belton, D. L. Kaplan and C. C. Perry, RSC Adv., 6, 21776 (2016).
S. Wang, X. Wang, F. G. Draenert, O. Albert, H. C. Schröder, V. Mailänder, G. Mitov and W. E. G. Müller, Bone, 67, 292 (2014).
W. E. G. Müller, H. C. Schröder, Q. Feng, U. Schlossmacher, T. Link and X. Wang, J. Tissue Eng. Regen. Med., 9, E39 (2015).
W. Zhu, X. Gao, X. Zou, W. E. G. Müller, S. Wang, Y. Wang and Y. Liu, J. Biomater Tissue Eng., 8, 258 (2018).
O. Dudik, S. Amorim, J. R. Xavier, H. T. Rapp, T. H. Silva, R. A. Pires and R. L. Reis, Front. Mar. Sci., 8, 637810 (2021).
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (NRF-2021R1A5A8032895, NRF-2021R1A2C2011564). This work was also supported by the National Research Foundation of Korea (NRF) funded by the Korea Ministry of Education (NRF-2021 R1I1A3046565). This work was also supported by Korea Environmental Industry & Technology Institute (KEITI) through Project to develop eco-friendly new materials and processing technology derived from wildlife, funded by Korea Ministry of Environment (MOE) (RE202101398).
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ki, MR., Park, K.S., Abdelhamid, M.A.A. et al. Novel silicatein-like protein for biosilica production from Amphimedon queenslandica and its use in osteogenic composite fabrication. Korean J. Chem. Eng. 40, 419–428 (2023). https://doi.org/10.1007/s11814-022-1314-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-022-1314-x