Abstract
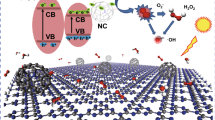
The emission of mercury from anthropogenic activities is a serious concern in both developed and developing countries due to its high toxicity and persistence. Here, carbon nitride nanosheets (CNNS) were attained via a two-step thermal etching method and applied for Hg0 removal. Proper deposition of ZnS can markedly reinforce the Hg0 capture ability of CNNS. 10ZnS/CNNS notably outperforms ZnS and CNNS at 100 °C, which is primarily attributed to surface chemisorbed oxygen species and polysulfide active sites. Besides, Hg0 adsorption and thermal catalytic oxidation pathways are further disclosed using quantum chemistry calculations based on density functional theory (DFT). The calculation results show that the presence of zinc species is beneficial to the decomposition of adsorbed oxygen, which plays a key role in the catalytic oxidation of Hg0, thereby contributing to the enhancement of mercury removal performance.
Similar content being viewed by others
References
C. T. Driscoll, R. P. Mason, H. M. Chan, D. J. Jacob and N. Pirrone, Environ. Sci. Technol., 47, 4967 (2013).
J. H. Pavlish, E. A. Sondreal, M. D. Mann, E. S. Olson, K. C. Galbreath, D. L. Laudal and S. A. Benson, Fuel Process. Technol., 82, 89 (2003).
E. G. Pacyna, J. Pacyna, K. Sundseth, J. Munthe, K. Kindbom, S. Wilson, F. Steenhuisen and P. Maxson, Atmos. Environ., 44, 2487 (2010).
K. C. Galbreath and C. J. Zygarlicke, Fuel Process. Technol., 65–66, 289 (2000).
S. X. Wang, L. Zhang, G. H. Li, Y. Wu, J. M. Hao, N. Pirrone, F. Sprovieri and M. P. Ancora, Atmos. Chem. Phys., 10, 1183 (2010).
H. Q. Yang, Z. H. Xu, M. Fan, A. E. Bland and R. R. Judkins, J. Hazard. Mater., 146, 1 (2007).
J. P. Yang, H. Xu, H. Chen, F. Meng, H. Zu, P. Zhu, Z. Q. Yang, M. Li and H. L. Li, J. Hazard. Mater., 424, 127336 (2022).
A. A. Presto and E. J. Granite, Environ. Sci. Technol., 40, 5601 (2006).
L. Zhao, C. T. Li, X. Zhang, G. Zeng, J. Zhang and Y. Xie, Catal. Sci. Technol., 5, 3459 (2015).
C. Lu, J. Wu and D. J. Liu, Mater. Lett., 227, 308 (2018).
H. Kaur, S. Singh and B. Pal, Korean J. Chem. Eng., 38, 1248 (2021).
J. Zhang, M. Zhang, L. Lin and X. C. Wang, Angew. Chem. Int. Ed., 54, 6297 (2015).
Z. Zhao, Y. Sun and F. Dong, Nanoscale, 7, 15 (2015).
Y. T. Ngo, J. S. Chung and S. H. Hur, Korean J. Chem. Eng., 37, 1589 (2020).
D. J. Liu, Z. Zhang, L. Liu and J. Wu, Surf. Rev. Lett., 27, 2050017 (2020).
Z. Zhang, J. Wu and D. J. Liu, Processes, 7, 279 (2019).
Z. Yang, H. L. Li, S. Feng, P. Li, C. Liao, X. Liu, J. Zhao, J. P. Yang, P. H. Lee and K. Shih, Langmuir, 34, 8739 (2018).
H. L. Li, L. Zhu, J. Wang, L. Li and K. Shih, Environ. Sci. Technol., 50, 9551 (2016).
J. P. Yang, Q. Li, H. Zu, Z. Q. Yang, W. Q. Qu, M. Li and H. L. Li, Environ. Sci. Technol., 54, 16195 (2020).
J. D. Xiao, Q. Han, Y. Xie, J. Yang, Q. Su, Y. Chen and H. Cao, Environ. Sci. Technol., 51, 13380 (2017).
P. Niu, L. Zhang, G. Liu and H. Cheng, Adv. Funct. Mater., 22, 4763 (2012).
W. Liu, H. M. Xu, Y. Liao, Z. Quan, S. Li, S. Zhao, Z. Qu and N. Q. Yan, Fuel, 235, 847 (2019).
D. J. Liu, C. Lu and J. Wu, J. Nanopart. Res., 20, 227 (2018).
S. Liu, L. Chen, X. Mu, M. Xu, J. Yu, G. Yang, X. Luo, H. T. Zhao and T. Wu, Fuel, 254, 115537 (2019).
Y. Wu, W. Xu, Y. Yang, J. Wang and T. Zhu, Catal. Sci. Technol., 8, 297 (2018).
H. Li, Y. Jing, X. Ma, T. Liu, L. Yang, B. Liu, S. Yin, Y. Wei and Y. Wang, RSC Adv., 7, 8688 (2017).
J. A. Bearden, Rev. Mod. Phys., 39, 86 (1967).
H. Mao, J. Han, Y. Fu, Y. Song, C. Yu and X. Dong, Appl. Catal. B-Environ., 102, 417 (2011).
J. Xu, L. Zhang, R. Shi and Y. Zhu, J. Mater. Chem. A, 1, 14766 (2013).
J. Zhang, M. Zhang, G. Zhang and X. C. Wang, ACS Catal., 2, 940 (2012).
J. F. Moulder, W. F. Stickle, P. E. Sobol, K. D. Bomben and J. Chastain, Handbook of X-ray photoelectron spectroscopy: A reference book of standard spectra for identification and interpretation of XPS data, Perkin-Elmer Corporation, Eden Prairie, Minnesota (1992).
J. Li, B. Shen, Z. Hong, B. Lin, B. Gao and Y. Chen, Chem. Commun., 48, 12017 (2012).
C. Wang, D. Wu, H. Wang, Z. Gao, F. Xu and K. Jiang, J. Power Sources, 363, 375 (2017).
G. Dong, L. Yang, F. Wang, L. Zang and C. Wang, ACS Catal., 6, 6511 (2016).
D. J. Liu, W. G. Zhou and J. Wu, Korean J. Chem. Eng., 33, 1837 (2016).
N. D. Hutson, B. C. Attwood and K. G. Scheckel, Environ. Sci. Technol., 41, 1747 (2007).
S. L. Harmer, Miner. Eng., 21, 1005 (2008).
R. S. C. Smart, W. M. Skinner and A. R. Gerson, Surf. Interface Anal., 28, 101 (1999).
W. Liu, H. M. Xu, Y. Liao, Y. Wang, N. Q. Yan and Z. Qu, Environ. Sci. Pollut. Res., 27, 20469 (2020).
D. Jampaiah, S. J. Ippolito, Y. M. Sabri, J. Tardio, P. R. Selvakannan, A. Nafady, B. M. Reddy and S. K. Bhargava, Catal. Sci. Technol., 6, 1792 (2016).
J. Wilcox, E. Sasmaz, A. Kirchofer and S. S. Lee, J. Air Waste Manage. Assoc., 61, 418 (2011).
S. Liu, M. Xu, Y. Chen, J. Yu, C. Pang and T. Wu, Fuel, 305, 121456 (2021).
Acknowledgements
This research is sponsored by National Natural Science Foundation of China (grant no. 52076126), Natural Science Foundation of Shanghai (grant no. 18ZR1416200), and Senior Talent Foundation of Jiangsu University (grant no. 18JDG017).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supporting Information
Additional information as noted in the text. This information is available via the Internet at http://www.springer.com/chemistry/journal/11814.
Supporting Information
11814_2021_1050_MOESM1_ESM.pdf
ZnS-modified carbon nitride nanosheet with enhanced performance of elemental Hg removal: An experimental and density functional theory study
Rights and permissions
About this article
Cite this article
Ling, Y., Wu, J., Yang, L. et al. ZnS-modified carbon nitride nanosheet with enhanced performance of elemental Hg removal: An experimental and density functional theory study. Korean J. Chem. Eng. 39, 1641–1650 (2022). https://doi.org/10.1007/s11814-021-1050-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-021-1050-7