Abstract
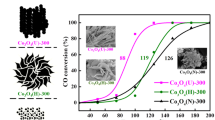
High surface area powders, La0.9Sr0.1CoO3 (LSCO), were prepared by a hard template method with a template of SBA-15. In the preparation process, the solvent was ethanol, the mixture of ethanol and water, and water, respectively. Characterizations were performed to understand the properties of the catalysts. When the solvent is a mixture of ethanol and water, the prepared LSCO has the most easily extracted surface adsorbed oxygen in the H2 atmosphere, exhibiting the best light-off performances for CO oxidation. However, the LSCO prepared in ethanol solvent has the best lattice oxygen mobility, resulting in the best light-off performance for C3H6 oxidation. Moreover, the LSCO prepared in ethanol also shows the best catalytic activity for both CO and C3H6 oxidation under the conditions of simulated diesel exhaust. Furthermore, Pd was impregnated on the LSCO, which was prepared by a sol-gel method and a hard template method, separately; the later one shows higher CO and C3H6 catalytic oxidation activity.
Similar content being viewed by others
References
J. Xu, G. Lu, Y. Guo, Y. Guo and X.-Q. Gong, Appl. Catal. A-Gen., 535, 1 (2017).
C. H. Kim, G. Qi, K. Dahlberg and W. Li, Science, 327, 1624 (2010).
A. Russell and W. S. Epling, Catal. Rev., 53, 337 (2011).
W. Tang, W. Xiao, S. Wang, Z. Ren, J. Ding and P.-X. Gao, Appl. Catal. B-Environ., 226, 585 (2018).
X. Auvray and L. Olsson, Appl. Catal. B-Environ., 168–169, 342 (2015).
D. Pinto and A. Glisenti, Catal. Sci. Technol., 9, 2749 (2019).
A. Glisenti and A. Vittadini, Catalysts, 9, 312 (2019).
H. Ziaei-Azad, A. Khodadadi, P. Esmaeilnejad-Ahranjani and Y. Mortazavi, Appl. Catal. B-Environ., 102, 62 (2011).
P. Li, Q. Yang, H. Zhang, M. Yao, F. Yan and D. Fu, Int. J. Hydrogen Energy, 45, 11802 (2020).
S. Cimino, L. Lisi, S. De Rossi, M. Faticanti and P. Porta, Appl. Catal. B-Environ., 43, 397 (2003).
B. Kucharczyk and W. Tylus, Catal. Today, 90, 121 (2004).
J. Chen, M. Shen, X. Wang, J. Wang, Y. Su and Z. Zhao, Catal. Commun., 37, 105 (2013).
J. Chen, M. Shen, X. Wang, G. Qi, J. Wang and W. Li, Appl. Catal. B-Environ., 134–135, 251 (2013).
J. A. Onrubia, B. Pereda-Ayo, U. De-La-Torre and J. R. González-Velasco, Appl. Catal. B-Environ., 213, 198 (2017).
S. Ponce, M. A. Peña and J. L. G. Fierro, Appl. Catal. B-Environ., 24, 193 (2000).
F. Teng, W. Han, S. Liang, B. Gaugeu, R. Zong and Y. Zhu, J. Catal., 250, 1 (2007).
C. Zhang, W. Hua, C. Wang, Y. Guo, Y. Guo, G. Lu, A. Baylet and A. Giroir-Fendler, Appl. Catal. B-Environ., 134–135, 310 (2013).
N. Mota, M. C. Alvarez-Galván, R. M. Navarro, S. M. Al-Zahrani, A. Goguet, H. Daly, W. Zhang A. Trunschke, R. Schlög and J. L. G. Fierro, Appl. Catal. B-Environ., 113–114, 271 (2012).
P. Xiao, J. Zhu, H. Li, W. Jiang, T. Wang, Y. Zhu, Y. Zhao and J. Li, Chemcatchem, 6, 1774 (2014).
M. V. Landau, L. Titelman, L. Vradman and P. Wilson, Chem. Commun., 5, 594 (2003).
X. Wang, J. Catal., 222, 565 (2004).
P. Li, X. Chen, Y. Li and J. W. Schwank, Catal. Today, 364, 7 (2021).
P. Li, X. Chen, L. Ma, A. Bhat, Y. Li and J. W. Schwank, Catal. Sci. Technol., 9, 1165 (2019).
J. A. Lupescu, J. W. Schwank, K. A. Dahlberg, C. Y. Seo, G. B. Fisher, S. L. Peczonczyk, K. Rhodes, M. J. Jagner and L. P. Haack, Appl. Catal. B-Environ., 183, 343 (2016).
L. Ma, C. Y. Seo, X. Chen, K. Sun and J. W. Schwank, Appl. Catal. B-Environ., 222, 44 (2018).
J. Luo, M. Meng, Y. Zha and L. Guo, J. Phys. Chem. C, 112, 15293 (2008).
L. Ma, C. Y. Seo, X. Chen, K. Sun and J. W. Schwank, Appl. Catal. B-Environ., 222, 44 (2018).
P. Li, X. Chen, Y. Li and J. W. Schwank, Catal. Today, 327, 90 (2018).
P. Li, R. Dong, X. Jiang, S. Zhang, T. Liu, R. Wang, F. Yan and D. Fu, J. Electroanal. Chem., 873, 114513 (2020).
L. Ma, C. Y. Seo, X. Chen, J. Li and J. W. Schwank, Chem. Eng. J., 350, 419 (2018).
M. Aghabararnejad, G. S. Patience and J. Chaouki, Can. J. Chem. Eng., 92, 1903 (2014).
S. Hosokawa, R. Tada, T. Shibano, S. Matsumoto, K. Teramura and T. Tanaka, Catal. Sci. Technol., 6, 7868 (2016).
Acknowledgement
This study was supported by the Fundamental Research Funds for the Central Universities (No. 2020MS124 and 2020MS126) and Natural Science Foundation of Hebei Province (No. B2020502002 and B2020502003)
Author information
Authors and Affiliations
Corresponding author
Additional information
Supporting Information
Additional information as noted in the text. This information is available via the Internet at http://www.springer.com/chemistry/journal/11814.
Supporting Information
Rights and permissions
About this article
Cite this article
Yan, F., Li, P. & Zhang, X. CO and C3H6 oxidation over La0.9Sr0.1CoO3 catalysts: Influence of preparation solvent. Korean J. Chem. Eng. 38, 945–951 (2021). https://doi.org/10.1007/s11814-021-0781-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-021-0781-9