Abstract
Purpose
The aim of this review was to summarize the current literature for the effectiveness of activity and nutritional based interventions on health-related quality of life (HRQoL) in older adults living with and beyond cancer (LWBC).
Methods
We conducted systematic structured searches of CINAHL, Embase, Medline, Cochrane CENTRAL databases, and bibliographic review. Two independent researchers selected against inclusion criteria: (1) lifestyle nutrition and/or activity intervention for people with any cancer diagnosis, (2) measured HRQoL, (3) all participants over 60 years of age and (4) randomized controlled trials.
Results
Searches identified 5179 titles; 114 articles had full text review, with 14 studies (participant n = 1660) included. Three had nutrition and activity components, one, nutrition only and ten, activity only. Duration ranged from 7 days to 1 year. Interventions varied from intensive daily prehabilitation to home-based gardening interventions. Studies investigated various HRQoL outcomes including fatigue, general and cancer-specific quality of life (QoL), distress, depression, global side-effect burden and physical functioning. Eight studies reported significant intervention improvements in one or more QoL measure. Seven studies reported using a psychosocial/theoretical framework. There is a gap in tailored nutrition advice.
Conclusions
Among the few studies that targeted older adults with cancer, most were activity-based programmes with half reporting improvements in QoL. Future research should focus on or include tailored nutrition components and consider appropriate behaviour change techniques to maximize potential QoL improvement.
Implications for Cancer Survivors
More research is needed to address the research gap regarding older adults as current recommendations are derived from younger populations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
The proportion of adults aged 65 or older in the United Kingdom (UK) was estimated to be about 18% in 2017, with projections of an increase to around 24% by 2037 [1]. Just under two-thirds of new cancer cases in the UK, on average each year are in people aged 65 and over [2,3,4,5]. Many people post-diagnosis live with multiple adverse side effects that impact both physical and mental health. Cancer treatments are also associated with higher rates of other conditions like cardiovascular disease, type 2 diabetes and subsequent primary cancers [6].
In addition, 1 in 10 people aged 65 years or older is affected by frailty [7]. Frailty is a clinical syndrome characterized by multisystem decline that leads to lower functional reserve, increased vulnerability to dependency and mortality after minor stressor events [8]. Frailty is also associated with adverse outcomes such as increased risk of falls, disability, hospitalization and death [9]. Older adults with cancer are at higher risk of frailty than their younger counterparts. This may limit chemotherapy and other therapeutic options or result in dose reductions and low treatment completion rates.
When coupled with higher rates of sarcopenia (the progressive degeneration of skeletal muscle mass), cachexia (extreme weight loss and muscle wasting due to chronic illness) and nutritional deficiencies (e.g. malnutrition, etc.), cancer and its treatment confer a range of effects which reduce quality of life (QoL) [10]. One recent study found that nearly two-thirds of older people assessed in hospital had at least one tissue loss syndrome (i.e. sarcopenia, frailty, cachexia or malnutrition) [11]. This is concerning as sarcopenia, for example, has been independently associated with 1-year mortality rates in older adults with cancer [12]. Obesity and fat gain have also been identified as a health issue that will become more common among older adults LWBC as the proportion of the general population classified as overweight and obese continues to increase [13, 14]. The American Society of Clinical Oncology (ASCO) has even urged clinicians to intervene and counsel patients, agreeing that obesity is a major concern among people LWBC [15, 16].
Physical activity (PA) benefits people living with or beyond cancer by improving physical function and QoL during and after cancer treatment, and cancer-related outcomes like treatment completion, maintenance of, or faster return to, pre-treatment health, fewer unnecessary healthcare visits and better survival rates [6, 17,18,19,20,21,22]. Improvements are greater in those engaging in PA sooner after a diagnosis [23]. Rehabilitation among people with chronic obstructive pulmonary disease (COPD) is also known to reduce improve function short term [24]. Emerging work indicates that exercise and immune function in the older person are related [25,26,27,28,29,30].
Poor nutritional status is associated with worse overall survival and QoL in patients receiving chemotherapy than those with better nutritional status [31, 32]. A recent review suggests that nutritional interventions, including dietary counselling and a multi-modal approach of exercise and nutrition, may support well-being and patient’s ability to complete treatments; however, further high-quality research is needed [33].
A tailored activity and nutrition intervention, designed to optimize physical function and nutritional status irrespective of treatment plan, started soon after diagnosis may increase the percentage of older people able to complete chemotherapy, and improve QoL and functional ability in those unfit for chemotherapy. Previous work has focused on prehabilitation (e.g. prior to surgery) [34, 35], maintenance during treatment (e.g. alongside chemotherapy) [36] or rehabilitation for cancer survivors post-treatments [37, 38].
Older adults are a growing proportion of the general and cancer populations; yet, they are underrepresented in clinical trials [39, 40]. In fact, a systematic review found that of all RCTs assessed in a 1-year period, only 3% were specifically designed for adults age 65 or older [41]. Additionally, older adults are often excluded based on secondary cancers, co-morbidities and declines in physical function and cognition [42]. The majority of guidance for lifestyle behaviour change in cancer has been derived from early stage breast and prostate cancer populations, a generally younger, fitter, group [43]. As such, recommendations may not be appropriately generalized to older groups of poorer health, for example, adults with lung cancer, the proportion of which being aged 65 or older is 78% [2,3,4,5].
The benefits of exercise in the non-cancer population have widespread acceptance and an extensive evidence base [43], but previous research relating to exercise in cancer patients is less robust and has not been tailored to the older or frail adult. Conversely, programmes developed for older adults have not included people with cancer. The Cancer and Ageing Research Group in Wisconsin observed that “simply extracting results from the larger body of geriatric exercise trials is not sufficient to inform how exercise is prescribed for geriatric oncology patients” [44]. They recommend careful work regarding patient population selection, development of the intervention and choice of outcome measurement to enable rigorous development and testing of programmes prior to rollout in clinical practice. Therefore, we aimed to summarize the current literature regarding activity and nutritional based interventions on health-related quality of life (HRQoL) in older adults with cancer delivered before, during or after active cancer treatments, or as part of best supportive care.
Methods
Study design
The conduct and reporting of this review adhere to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) [45]. A data charting/extraction form was adapted from the Johanna Briggs Institute (JBI) Reviewers’ Manual: Methodology for JBI reviews (2015) [46]. A copy of the final form can be found on our open science framework page (https://osf.io/p23jd/).
Inclusion and exclusion criteria
Studies were included if they met the following a priori eligibility criteria: (1) delivered a lifestyle intervention for nutrition and/or PA to people with any cancer diagnosis, (2) included a measure of HRQoL, (3) participants over 60 years or at least 50% over 60 years with data analyses by age group and (4) randomized controlled trials. Studies were excluded if (1) we could not determine an age range, (2) the intervention was targeting clinicians or carers rather than older adults with cancer, (3) publication language was not in English or (4) findings were conference abstracts only.
Search strategy
Studies were identified through structured searches of all publication years (final update search performed 30 May 2019) in the following electronic databases: Medline via OVID, Embase via OVID, Cochrane Central Register of Controlled Trials (CENTRAL) and Cinahl via EBSCO. The search strategy was developed in consultation with a specialist librarian at the University of Hull and finalized with the aid of an information specialist. MeSH terms in Medline (see supplemental file 1) were developed to search for all key concepts and modified for other databases. Keyword searches restricted to abstract and title were also completed. Boolean logic was used to combine the terms. The original database searches were conducted by a single author (CF) and updated by an information specialist (SG). For the updated search, search filters for RCTs including Cochrane’s Highly Sensitive Search Strategy were included to retrieve randomized controlled trials.
Study selection
All identified articles were uploaded into an EndNote X8 database and duplicates removed. Preliminary screening was undertaken by one author (CF) to remove obvious exclusions (e.g. conference abstracts, etc.) after which two authors (CF and SG) independently screened all articles against eligibility criteria taking title, abstract and full-text into account. Disagreements were discussed and resolved by consensus. Any unresolved items were reviewed by a third author (FS) and their decision stood. If criteria were unclear in the manuscript, corresponding authors were emailed and asked for clarification.
Data extraction
A data extraction form was developed and piloted by the research team to extract data about study details and characteristics (e.g. country, setting, sample characteristics, etc.), intervention details (e.g. group descriptions, intervention components and duration, etc.), QoL outcomes and key findings and messages. The form was independently tested using one article by two authors (CF and SG) and revised following discussion. Data were then extracted using the form by a single reviewer (CF). A second (SG) and third (FS) reviewer randomly selected two articles each (i.e. 25%) and reviewed the data extracted. As there were no discrepancies, data extraction by a second reviewer for the remaining articles was considered unnecessary.
Risk of bias assessment
Two authors (CF and SG) used the Risk of Bias Assessment Tool version 2 available from the Cochrane handbook (2011) to independently assess quality of life outcomes from all included studies. The articles were judged for bias as either low, high or some concerns for the following: (1) selection (random sequence generation and allocation concealment), (2) performance (blinding of participants and study personnel), (3) detection (blinding of outcome assessment), (4) missing outcome data and (5) reporting (selective outcome reporting). The nature of lifestyle behaviour change studies means double-blinding is very difficult but this tool allows fair judgements despite this fact. The authors discussed any differences and reached consensus; therefore, a third party was not necessary.
Outcomes
To describe the nature of studies currently targeting older adults with cancer, we extracted detailed information related to the intervention groups including (1) type of intervention, (2) intervention delivery methods, (3) all components of intervention, (4) study duration and measurement timing and (5) comparator group information. The primary efficacy outcomes of interest for this review were measures related to QoL or HRQoL. The primary outcomes for each study were identified and noted.
Results
Study selection
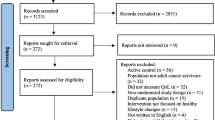
The study selection process is presented in Fig. 1. A total of 6490 records were identified; 5179 remained after de-duplication. After title and abstract screening, 114 articles were identified for full-text review. Of those, 14 studies were deemed eligible and were included in full data extraction for this review.
Study characteristics
This review included 14 papers [47,48,49,50,51,52,53,54,55,56,57,58,59,60]. A further eight related papers were referenced to provide more detail when unable to find information in the primary paper (e.g. protocol papers) [61,62,63,64,65,66,67,68]. One study was a three-arm randomized controlled trial (RCT) [47] with the remaining 13 studies being two-arm RCTs. One study was described as a pilot [51] and seven as feasibility [47, 48, 50, 54, 55, 58, 60] RCTs. Seven studies took place in the USA [49, 50, 55, 56, 59, 60], two in Canada [47, 51] and one each in China [52], Japan [54], Korea [57], Sweden [58] and UK [48]. Five studies recruited patients with prostate cancer only [47, 48, 55, 57, 60], one each recruited bladder [58], lung [52] and breast cancer only [51], with the remaining recruiting a mixed sample of cancer types [49, 50, 53, 54, 56, 59].
Risk of bias assessment
Full results from the assessment can be found in Figs. 2 and 3. Based on assessments from two reviewers, no studies received overall risk of bias judgements of low, ten were judged to have some concerns and four had high risk of bias. Twelve studies were low [47,48,49,50,51, 53, 54, 56,57,58,59,60] and two some concerns [52, 55] for allocation; ten were low [47, 49, 52,53,54, 56,57,58,59,60], two some concerns [48, 50] and two high [51, 55] for intervention deviations; nine rated low [47,48,49,50, 52,53,54, 56, 59], three some concerns [55, 57, 60] and two high [51, 58] for missing data; one ranked low [52] and 13 some concerns [47,48,49,50,51, 53,54,55,56,57,58,59,60] for outcome measurement; and finally, 11 rated low [47,48,49,50,51,52,53,54,55,56,57], two some concerns [58, 59] and one high [60] for selective reporting.
Intervention characteristics
Most interventions focused on PA behaviour only [47, 51,52,53,54,55, 57,58,59,60] and three on both nutrition and activity behaviour [48, 49, 56]. One study reported improving nutrition as its main objective; however, this was a gardening intervention which could also be considered activity [50]. The studies ranged in duration from 7 days to 1 year, six studies [48,49,50, 56, 58] collected follow-up measures beyond post-study though only three studies reported this data in the included articles [48, 49, 58]. Intervention settings included hospital [52,53,54,55, 58], community [57, 59, 60], home [49, 50, 56] or a mixture of settings [47, 48, 51]. Detailed characteristics of the included studies can be found in Table 1.
Activity intervention characteristics
Eight studies included some specific form of aerobic activity [47, 48, 51, 52, 54,55,56, 58], five had traditional-style strength training (e.g. lifting weights or using resistance bands) [47, 48, 56, 58, 60] and five included specific training to build balance and/or functional strength [51, 52, 57,58,59]. Seven interventions included at least one supervised individual session [47, 52,53,54,55, 57, 60], seven had home-based activities [47,48,49,50,51, 53, 56] and four were group-based classes or training sessions [47, 48, 58, 59]. Home-based interventions were delivered using DVD [51], booklets/binders [48,49,50, 53, 56] and four included visits or telephone calls to check-in and determine progress [47, 49, 50, 56]. Six home-based studies included personalized advice on activity [47,48,49, 51, 55, 56].
Nutrition intervention characteristics
One study provided participants with all supplies and guidance on growing their own vegetables at home and had role models in the form of a Master Gardener to teach and assist [50]. One other study provided portion-control tableware to assist eating habits and included tailored nutrition advice as compared to national guidelines [56]. One study had personalized comparisons to general information regarding standard nutrition guidelines [49]. One study had a series of “healthy eating” seminars and a nutrition advice pack [48].
Theoretical intervention characteristics
Three studies explicitly stated they used a theory to develop study materials including social cognitive theory [49, 50, 56], transtheoretical model [49] and social ecological model [50]. Other studies included information regarding habit formation [48], autonomy [48], self-efficacy [49] and action/coping planning [47, 50]. Aside from those studies with group-based session, to increase social support, one study formally included spouses or partners in the intervention [60] and one had a private Facebook group for study participants [50]. Detailed descriptions of the interventions included can be found in Table 2.
Outcomes
Study feasibility was stated as the primary outcome for seven studies [47, 48, 50, 54, 55, 58, 60], of which six were deemed feasible based on recruitment, retention, adherence, and compliance rates [47, 48, 50, 54, 55, 60]. Though attendance and compliance rates were high, one study was deemed not feasible due to the large number of dropouts owing to the more severe illness of bladder cancer patients [58]. Change in activity or diet behaviour was a primary outcome for two studies [48, 49] and five focused on physical [52, 56,57,58] or cognitive functioning [54]. Seven found significant improvements in the primary outcome [48,49,50, 52, 54, 56, 57] while one found no significant group difference [58], though this was a feasibility trial. Further details can be found in Table 3.
To measure QoL, five studies used the Medical Outcomes Study’s Short-Form 36 (SF-36) [50, 56,57,58, 60], five used all or part of the general Functional Assessment of Cancer Therapy (FACT-G) [47, 48, 51, 53, 54] and one used the European Organization for Research and Treatment of Cancer QoL Questionnaire 30 (EORTC-QLQ-30) [52]. Six used cancer-specific measures including the FACT-Prostate [47,48,49, 55], FACT-Breast [49, 51] and the EORTC Lung Cancer supplement [52]. Six studies assessed symptom specific QoL including fatigue [47, 48, 51, 55, 59], anxiety and mood [53] and side effect burden [59]. Six studies reported significant intervention group improvements [48, 55,56,57,58,59] and six reported no significant group difference in one or more QoL measure [48, 49, 51, 52, 54, 60]. Two studies reported significant negative intervention difference in QoL [47, 50]. There was no difference in effectiveness between studies that reported using theory/psychosocial components to guide the interventions versus those reporting no theory. Three studies using theory reported significant group differences in QoL measures [47, 50, 56]; however, two of these were favouring the non-intervention group [47, 50]. Both theory-based and non-theory-based studies had within group improvements in both quality of life and physical outcomes. Select QoL results are described below; full details can be found in Tables 3 and 4.
Quality of life
Participants in a home- or group-based activity programme reported poorer general and cancer-specific QoL than those in a 1:1 personal training group [47]. Participants that received a home-based personalized activity and nutrition intervention reported similar improvements in general QoL to an attention control group receiving general information at study end which was maintained at follow-up [49]. A wait-list control group showed more positive improvement in scores for pain, physical role and overall mental health compared to those receiving a gardening intervention [50]. Both the home-based and group session-based participants in an exercise intervention for women diagnosed with breast cancer improved their overall and breast cancer-specific QoL [51]. No differences were observed between groups on global or lung cancer-specific QoL in those participating in a prehabilitation intervention versus usual care [52]. In a group of mixed cancer patients undergoing chemotherapy, between group differences favouring the intervention were noted among those having poorer social and emotional well-being at baseline measures compared to the wait-list control [53]. No between-group or within-group differences were found for QoL in speed feedback therapy group versus usual care [54]. Those receiving a supervised activity programme had significant between-group improvements in overall and prostate cancer-specific QoL compared to those receiving usual care [55]. While both groups in a home-based activity and nutrition intervention versus wait-list control had declines in overall QoL throughout the study period, the intervention group had significantly smaller declines than the control group [56]. Physical and mental composite scores of the SF-36 returned to preoperative levels in participants in a functional exercise intervention when compared to those in a usual care group [57]. In an exercise study among people with bladder cancer, only the role-physical domain scores improved significantly in the intervention group compared to usual care; all other scores had no differences [58]. No differences were found between exercise and control groups in men with prostate cancer, but among spouses also participating, there was a non-significant increase in partners’ mental health scores [60].
Fatigue and other side effects
Men starting or currently on androgen deprivation therapy (ADT) reported a similar change in fatigue among three groups receiving an activity programme [47]. Participants that received a combination of supervised and home-based activity reported more improvement in fatigue than control groups after the study period which was maintained at follow-up [48]. An activity and nutrition intervention found improvement in depression scores in both the tailored versus non-tailored groups [49].
In a study comparing the same programme either home or group based, there were non-significant improvements in fatigue in both groups [51]. Participants receiving a low- to moderate-intensity home-based activity programme, with poorer anxiety and mood at baseline, had significant improvements compared to the control group [53]. Men with prostate cancer participating in aerobic exercise before radiotherapy reported significantly better fatigue scores than those in usual care [55]. Those participating in a yoga intervention reported significantly lower cancer-related fatigue and global side effect burden than the wait-list control group [59].
Discussion
This review describes the current literature around the nature of PA and nutrition interventions for older adults with cancer. Our initial inclusion age criterion was “aged 70 or older”; however, we had to amend this to 60 or older as we retrieved no studies that met all criteria illustrating the relative paucity of literature relating to older adults. Most available research has targeted relatively young people living with and beyond cancer, limiting the relevance of subsequent clinical guidance to older adults [43, 69,70,71].
We found 14 RCTs relevant to our question; most were feasibility/pilot trials, but 6 were evaluation phase studies. Effects on QoL outcomes were unsurprisingly mixed given that most were not designed to test effectiveness; however, the evaluation phase trials showed positive trends in QoL related to lifestyle interventions [49, 51,52,53, 55, 56]. Trials were globally representative across North America, the Far East and Europe and across all healthcare settings. Most studies were in people with prostate cancer and few included people with advanced disease; even the lung cancer prehabilitation trial included people eligible for radical surgery. Three interventions included both nutrition and PA components, with the vast majority of trials investigating a PA intervention only. Overall, our main findings are that older adults should be considered as a different population, tailoring of interventions increases relevance to the patient and a holistic approach with attention to behavioural self-management strategies with at least some personal contact with a therapist or health professional seems to be necessary.
Compared with the cancer adult population as a whole, older adults have more comorbidities, are at more risk for falls and frailty and current guidelines for behaviour change may not be relevant. However, from studies of pulmonary rehabilitation in non-malignant lung disease, older adults gain as much benefit as younger patients from such interventions although completion rates are lower; those with frailty being twice as likely not to complete [24, 72, 73]. Therefore, tailoring interventions is important. In the behaviour change field, studies that tailor education or interventions to individual participants are more likely to result in meaningful behaviour change [74, 75]. Tailored messages are more personally relevant and are more likely to be read, understood, recalled, higher rated and seen as credible than generalized messages [74, 76]. In this review, studies that tailored programmes to participants’ individual capacity and preference were more likely to lead to change behaviour and in QoL measures [48,49,50, 57]. The more tailored a programme is, the more relevant it will be and is more likely to result in behaviour change [76]. A recent systematic review highlighted the lack of behaviour change technique (BCT) use among thoracic cancer interventions [77]. Though we found mixed results of the effectiveness of theory-based interventions in our review, lifestyle behaviour change programmes that use appropriate BCTs to guide interventions are generally more effective [78].
The subjective nature of QoL may be different for older adults versus younger groups. Values and goals may shift for those in older age and the approach taken for lifestyle behaviour change must reflect this. Research in older adults in the general population highlights the need for more focus on functional fitness and mental well-being to remain independent [44]. The goal in this population is to live as well as possible, for as long as possible. This is reflected in the older adult cancer population where the goal or desired outcome is often functional, not fitness [44]. In this review, the majority of studies found improvements in physical function measures. One study demonstrated fewer days post-surgical recovery and a shorter hospital stay in the intervention group [52], while another study, delivering yoga found reductions in cancer-related fatigue, physical and mental fatigue, and a lower side effect burden [59]. Programmes that are more holistic in nature, focusing on both physical and mental wellness, may be most appropriate, seen as more relevant, and thereby garnering greater engagement.
Interventions with positive QoL outcomes had some form of supervised instruction or training with qualified professionals. Studies that had at least one face-to-face session were more likely to have greater positive changes in QoL measures than those that were home-based only [47, 53, 55, 57, 59]. In some studies, this was only an introductory session. While supervised activity sessions tend to have higher adherence and satisfaction, they are more expensive and resource intensive. However, studies that included telephone professional support also found positive results [49, 56]. The amount of supervision needed to make a lasting difference, or the appropriate “dose” for instigating behaviour change is unknown, and a recommended avenue of study [79].
Most of the study participants were breast or prostate cancer patients during or having completed treatment. While providing important perspectives, this reduces the generalizability of recommendations outside of these groups. In particular, only two studies included people with more severe disease: one with lung cancer [57] and one in bladder cancer [58]. The bladder cancer intervention was deemed not feasible, despite the positive outcomes in the intervention group, due to the low recruitment rate and number of drop-outs highlighting the difficulty in delivering programmes to sicker patients with more comorbidities [80,81,82]. Future research should focus on ways to ensure the most appropriate programmes for these populations by development work with their target populations.
The study among lung cancer patients was also the only prehabilitation study, aimed at providing a programme designed to improve functional outcomes that would therefore reduce post-surgical complications. Participants will have been assessed as fit enough for surgery, thereby not informing clinical practice for most older adults with lung cancer. Further, while potentially very beneficial, prehabilitation studies are difficult to implement given the short time-frames necessary prior to radical cancer treatments [79, 83]. Future research needs to investigate how to deliver interventions in as short a time as possible to have meaningful impact on patient-centred outcomes among those eligible for surgery.
Most studies focused on PA behaviour, highlighting the lack of nutrition interventions in older adults living with and beyond cancer despite nutritional status being a predictor for poor clinical outcomes [84]. Among the included studies, only four had a nutrition component [48,49,50, 56]. The majority of advice was general and focused on comparing current nutritional intake to national guidelines [48, 50, 56]. Only one study that used a nutrition intervention provided more detailed and tailored advice [49]. Diet patterns have been shown to influence QoL in older adult populations [85] but little research is available that tests the differential effects of tailored PA, diet or a combination of both. More research is necessary, particularly randomized controlled trials, to determine the presence and strength of this link among the older adult LWBC population.
Limitations and strengths
Strengths include a broad search method and the use of independent researchers. However, as with any review, important papers may have been missed. As with much of cancer research, most patient groups in this review were either breast or prostate cancer reducing the generalizability of the findings. Over half of the articles included were identified as feasibility studies. Though the majority of studies indicated the interventions were feasible, they were underpowered for effectiveness. Finally, few studies indicated using any theoretical base or specific BCTs. Future research should incorporate appropriate techniques to assist self-management and help encourage higher completion rates for older adults, especially those with frailty and sarcopenia, learning from research in other conditions as relevant [72].
Implications for research
This work identified key gaps in the evidence supporting rehabilitation-based programmes for older adults with cancer, and a paucity of work including nutrition interventions alongside those aiming to improve PA. Development of acceptable and relevant interventions, flexible across the cancer continuum and cancer type and stage are needed. One size is unlikely to fit all. Future research should be underpinned by behaviour change theory and include studies to explore how best to support attendance and completion by those with frailty and sarcopenia. There is likely to be overlap with research in other areas of older adults’ health and rehabilitation but although there is interest in generic rehabilitation programmes, there is little evidence to date to confirm benefits in people with cancer, or in older adults [86, 87].
Conclusions
This review identified very little research that focused on older adults specifically despite the growing proportion of this group. Few studies included a nutritional component. Findings useful to inform the design of activity/nutrition programmes include candidate intervention components, the need to use a holistic and tailored approach with functional goals and some personal professional contact. The tailoring must take into account the older person’s personal goals and be flexible along the cancer continuum depending on current treatment plans. Learning from general older adult populations as well as rehabilitation literature in other disease groups, e.g. chronic obstructive pulmonary disease, will help advance this research.
References
Statistics OfN. Overview of the UK population: November 2018. 2018. https://www.ons.gov.uk/peoplepopulationandcommunity/populationandmigration/populationestimates/articles/overviewoftheukpopulation/november2018. Accessed 23 Aug 2019.
Registry NIC. 2017 Cancer incidence, survival, mortality and prevalence data. 2019. http://www.qub.ac.uk/research-centres/nicr/CancerInformation/official-statistics/. Accessed 23 Aug 2019.
Scotland I. Cancer incidence in Scotland. 2019. https://www.isdscotland.org/Health-Topics/Cancer/Publications/2019-04-30/2019-04-30-Cancer-Incidence-Report.pdf?57526797057. Accessed 23 Aug 2019.
Statistics OfN. Cancer registration statistics, England: 2017. 2019. https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/conditionsanddiseases/bulletins/cancerregistrationstatisticsengland/2017. Accessed 23 Aug 2019.
Unit WCIaS. Cancer incidence in Wales, 2001–2016. 2019. http://www.wcisu.wales.nhs.uk/cancer-incidence-in-wales-1. Accessed 23 Aug 2019.
Li T, Wei S, Shi Y, Pang S, Qin Q, Yin J, et al. The dose-response effect of physical activity on cancer mortality: findings from 71 prospective cohort studies. Br J Sports Med. 2016;50(6):339–45. https://doi.org/10.1136/bjsports-2015-094927.
Collard RM, Boter H, Schoevers RA, Oude Voshaar RC. Prevalence of frailty in community-dwelling older persons: a systematic review. J Am Geriatr Soc. 2012;60(8):1487–92. https://doi.org/10.1111/j.1532-5415.2012.04054.x.
Morley JE, Abbatecola AM, Argiles JM, Baracos V, Bauer J, Bhasin S, et al. Sarcopenia with limited mobility: an international consensus. J Am Med Dir Assoc. 2011;12(6):403–9. https://doi.org/10.1016/j.jamda.2011.04.014.
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381(9868):752–62. https://doi.org/10.1016/s0140-6736(12)62167-9.
Miaskowski C, Wong ML, Cooper BA, Mastick J, Paul SM, Possin K, et al. Distinct physical function profiles in older adults receiving cancer chemotherapy. J Pain Symptom Manag. 2017;54(3):263–72. https://doi.org/10.1016/j.jpainsymman.2017.07.018.
Gingrich A, Volkert D, Kiesswetter E, Thomanek M, Bach S, Sieber CC, et al. Prevalence and overlap of sarcopenia, frailty, cachexia and malnutrition in older medical inpatients. BMC Geriatr. 2019;19(1):120. https://doi.org/10.1186/s12877-019-1115-1.
Otten L, Stobäus N, Franz K, Genton L, Müller-Werdan U, Wirth R, et al. Impact of sarcopenia on 1-year mortality in older patients with cancer. Age Ageing. 2019;48(3):413–8. https://doi.org/10.1093/ageing/afy212.
Lauby-Secretan B, Scoccianti C, Loomis D, Grosse Y, Bianchini F, Straif K. Body fatness and cancer — viewpoint of the IARC Working Group. N Engl J Med. 2016;375(8):794–8. https://doi.org/10.1056/NEJMsr1606602.
Demark-Wahnefried W, Schmitz KH, Alfano CM, Bail JR, Goodwin PJ, Thomson CA, et al. Weight management and physical activity throughout the cancer care continuum. CA Cancer J Clin. 2018;68(1):64–89. https://doi.org/10.3322/caac.21441.
Ligibel JA, Alfano CM, Courneya KS, Demark-Wahnefried W, Burger RA, Chlebowski RT, et al. American Society of Clinical Oncology position statement on obesity and cancer. J Clin Oncol. 2014;32(31):3568–74. https://doi.org/10.1200/JCO.2014.58.4680.
Ligibel JA, Alfano CM, Hershman D, Ballard RM, Bruinooge SS, Courneya KS, et al. Recommendations for obesity clinical trials in cancer survivors: American Society of Clinical Oncology statement. J Clin Oncol. 2015;33(33):3961–7.
Mishra SI, Scherer RW, Snyder C, Geigle P, Gotay C. Are exercise programs effective for improving health-related quality of life among cancer survivors? A systematic review and meta-analysis. Oncol Nurs Forum. 2014;41(6):E326–42. https://doi.org/10.1188/14.ONF.E326-E342.
Stout NL, Baima J, Swisher AK, Winters-Stone KM, Welsh J. A systematic review of exercise systematic reviews in the cancer literature (2005-2017). PM R. 2017;9(9S2):S347–84. https://doi.org/10.1016/j.pmrj.2017.07.074.
Cave J, Paschalis A, Huang CY, West M, Copson E, Jack S, et al. A systematic review of the safety and efficacy of aerobic exercise during cytotoxic chemotherapy treatment. Support Care Cancer. 2018;26(10):3337–51. https://doi.org/10.1007/s00520-018-4295-x.
Abu-Omar K, Rütten A, Burlacu I, Schätzlein V, Messing S, Suhrcke M. The cost-effectiveness of physical activity interventions: a systematic review of reviews. Prev Med Rep. 2017;8:72–8. https://doi.org/10.1016/j.pmedr.2017.08.006.
Kampshoff CS, van Dongen JM, van Mechelen W, Schep G, Vreugdenhil A, Twisk JWR, et al. Long-term effectiveness and cost-effectiveness of high versus low-to-moderate intensity resistance and endurance exercise interventions among cancer survivors. J Cancer Surviv. 2018;12:417–29. https://doi.org/10.1007/s11764-018-0681-0.
Buffart LM, Galvão DA, Brug J, Chinapaw MJM, Newton RU. Evidence-based physical activity guidelines for cancer survivors: current guidelines, knowledge gaps and future research directions. Cancer Treat Rev. 2014;40(2):327–40. https://doi.org/10.1016/j.ctrv.2013.06.007.
Mishra SI, Scherer RW, Snyder C, Geigle P, Gotay C. The effectiveness of exercise interventions for improving health-related quality of life from diagnosis through active cancer treatment. Oncol Nurs Forum. 2015;42(1):E33–53. https://doi.org/10.1188/15.ONF.E33-E53.
Maddocks M, Kon SSC, Canavan JL, Jones SE, Nolan CM, Labey A, et al. Physical frailty and pulmonary rehabilitation in COPD: a prospective cohort study. Thorax. 2016;71(11):988–95. https://doi.org/10.1136/thoraxjnl-2016-208460.
Koelwyn GJ, Wennerberg E, Demaria S, Jones LW. Exercise in regulation of inflammation-immune axis function in cancer initiation and progression. Oncology (Williston Park). 2015;29(12):908–20 922.
Saxton JM, Scott EJ, Daley AJ, Woodroofe MN, Mutrie N, Crank H, et al. Effects of an exercise and hypocaloric healthy eating intervention on indices of psychological health status, hypothalamic-pituitary-adrenal axis regulation and immune function after early-stage breast cancer: a randomised controlled trial. Breast Cancer Res. 2014;16(2):R39. https://doi.org/10.1186/bcr3643.
Kruijsen-Jaarsma M, Revesz D, Bierings MB, Buffart LM, Takken T. Effects of exercise on immune function in patients with cancer: a systematic review. Exerc Immunol Rev. 2013;19:120–43.
Zimmer P, Jäger E, Bloch W, Zopf EM, Baumann FT. Influence of a six month endurance exercise program on the immune function of prostate cancer patients undergoing Antiandrogen- or chemotherapy: design and rationale of the ProImmun study. BMC Cancer. 2013;13(1):272. https://doi.org/10.1186/1471-2407-13-272.
Fairey AS, Courneya KS, Field CJ, Bell GJ, Jones LW, Mackey JR. Randomized controlled trial of exercise and blood immune function in postmenopausal breast cancer survivors. J Appl Physiol. 2005;98(4):1534–40. https://doi.org/10.1152/japplphysiol.00566.2004.
Fairey AS, Courneya KS, Field CJ, Mackey JR. Physical exercise and immune system function in cancer survivors. Cancer. 2002;94(2):539–51. https://doi.org/10.1002/cncr.10244.
Ihara K, Yamaguchi S, Shida Y, Ogata H, Domeki Y, Okamoto K, et al. Poor nutritional status before and during chemotherapy leads to worse prognosis in unresectable advanced or recurrent colorectal cancer. Int Surg. 2015. https://doi.org/10.9738/intsurg-d-15-00079.1.
Hung H-Y, Wu L-M, Chen K-P. Determinants of quality of life in lung cancer patients. J Nurs Scholarsh. 2018;50(3):257–64. https://doi.org/10.1111/jnu.12376.
Kiss N. Nutrition support and dietary interventions for patients with lung cancer: current insights. Lung Cancer. 2016;7:1–9. https://doi.org/10.2147/lctt.s85347.
Steffens D, Beckenkamp PR, Hancock M, Solomon M, Young J. Preoperative exercise halves the postoperative complication rate in patients with lung cancer: a systematic review of the effect of exercise on complications, length of stay and quality of life in patients with cancer. Br J Sports Med. 2018;52:344. https://doi.org/10.1136/bjsports-2017-098032.
Cavalheri V, Granger C. Preoperative exercise training for patients with non-small cell lung cancer. Cochrane Database Syst Rev. 2017;6. https://doi.org/10.1002/14651858.CD012020.pub2.
Mishra SI, Scherer RW, Snyder C, Geigle PM, Berlanstein DR, Topaloglu O. Exercise interventions on health-related quality of life for people with cancer during active treatment. Cochrane Database Syst Rev. 2012;(8):Cd008465. https://doi.org/10.1002/14651858.CD008465.pub2.
Mishra SI, Scherer RW, Geigle PM, Berlanstein DR, Topaloglu O, Gotay CC, et al. Exercise interventions on health-related quality of life for cancer survivors. Cochrane Database Syst Rev. 2012;8. https://doi.org/10.1002/14651858.CD007566.pub2.
Scott DA, Mills M, Black A, Cantwell M, Campbell A, Cardwell CR, et al. Multidimensional rehabilitation programmes for adult cancer survivors. Cochrane Database Syst Rev. 2013;3. https://doi.org/10.1002/14651858.CD007730.pub2.
Gross CP, Herrin J, Wong N, Krumholz HM. Enrolling older persons in cancer trials: the effect of sociodemographic, protocol, and recruitment center characteristics. J Clin Oncol. 2005;23(21):4755–63. https://doi.org/10.1200/JCO.2005.14.365.
Scher KS, Hurria A. Under-representation of older adults in cancer registration trials: known problem, little progress. J Clin Oncol. 2012;30(17):2036–8. https://doi.org/10.1200/JCO.2012.41.6727.
Broekhuizen K, Pothof A, de Craen AJM, Mooijaart SP. Characteristics of randomized controlled trials designed for elderly: a systematic review. PLoS One. 2015;10(5):e0126709. https://doi.org/10.1371/journal.pone.0126709.
Hurria A, Dale W, Mooney M, Rowland JH, Ballman KV, Cohen HJ, et al. Designing therapeutic clinical trials for older and frail adults with cancer: U13 conference recommendations. J Clin Oncol Off J Am Soc Clin Oncol. 2014;32(24):2587–94.
Campbell KL, Winters-Stone KM, Wiskemann J, May AM, Schwartz AL, Courneya KS, et al. Exercise guidelines for cancer survivors: consensus statement from international multidisciplinary roundtable. Med Sci Sports Exerc. 2019;51(11):2375–90. https://doi.org/10.1249/mss.0000000000002116.
Kilari D, Soto-Perez-de-Celis E, Mohile SG, Alibhai SMH, Presley CJ, Wildes TM, et al. Designing exercise clinical trials for older adults with cancer: recommendations from 2015 Cancer and Aging Research Group NCI U13 Meeting. J Geriatr Oncol. 2016;7(4):293–304. https://doi.org/10.1016/j.jgo.2016.04.007.
Moher D, Liberati A, Tetzlaff J, Altman DG, The PG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. https://doi.org/10.1371/journal.pmed.1000097.
Institute TJB. The Joanna Briggs institute reviewers’ manual 2015: methodology for JBI scoping reviews. Adelaide: University of Adelaide; 2015.
Alibhai SMH, Santa Mina D, Ritvo P, Tomlinson G, Sabiston C, Krahn M, et al. A phase II randomized controlled trial of three exercise delivery methods in men with prostate cancer on androgen deprivation therapy. BMC Cancer. 2019;19(1):2. https://doi.org/10.1186/s12885-018-5189-5.
Bourke L, Doll H, Crank H, Daley A, Rosario D, Saxton JM. Lifestyle intervention in men with advanced prostate cancer receiving androgen suppression therapy: a feasibility study. Cancer Epidemiol Biomark Prev. 2011;20(4):647–57. https://doi.org/10.1158/1055-9965.EPI-10-1143.
Demark-Wahnefried W, Clipp EC, Morey MC, Pieper CF, Sloane R, Snyder DC, et al. Lifestyle intervention development study to improve physical function in older adults with cancer: outcomes from Project LEAD. J Clin Oncol Off J Am Soc Clin Oncol. 2006;24(21):3465–73.
Demark-Wahnefried W, Cases MG, Cantor AB, Frugé AD, Smith KP, Locher J, et al. Pilot randomized controlled trial of a home vegetable gardening intervention among older cancer survivors shows feasibility, satisfaction, and promise in improving vegetable and fruit consumption, reassurance of worth, and the trajectory of central adiposity. J Acad Nutr Diet. 2018;118(4):689–704. https://doi.org/10.1016/j.jand.2017.11.001.
Desbiens C, Filion M, Brien MC, Hogue JC, Laflamme C, Lemieux J. Impact of physical activity in group versus individual physical activity on fatigue in patients with breast cancer: a pilot study. Breast. 2017;35:8–13. https://doi.org/10.1016/j.breast.2017.06.001.
Lai Y, Huang J, Yang M, Su J, Liu J, Che G. Seven-day intensive preoperative rehabilitation for elderly patients with lung cancer: a randomized controlled trial. J Surg Res. 2017;209:30–6. https://doi.org/10.1016/j.jss.2016.09.033.
Loh KP, Kleckner IR, Lin PJ, Mohile SG, Canin BE, Flannery MA, et al. Effects of a home-based exercise program on anxiety and mood disturbances in older adults with cancer receiving chemotherapy. J Am Geriatr Soc. 2019;67(5):1005–11. https://doi.org/10.1111/jgs.15951.
Miki E, Kataoka T, Okamura H. Feasibility and efficacy of speed-feedback therapy with a bicycle ergometer on cognitive function in elderly cancer patients in Japan. Psycho-Oncology. 2014;23(8):906–13. https://doi.org/10.1002/pon.3501.
Monga U, Garber SL, Thornby J, Vallbona C, Kerrigan AJ, Monga TN, et al. Exercise prevents fatigue and improves quality of life in prostate cancer patients undergoing radiotherapy. Arch Phys Med Rehabil. 2007;88(11):1416–22.
Morey MC, Snyder DC, Sloane R, Cohen HJ, Peterson B, Hartman TJ, et al. Effects of home-based diet and exercise on functional outcomes among older, overweight long-term cancer survivors: RENEW: a randomized controlled trial. JAMA. 2009;301(18):1883–91. https://doi.org/10.1001/jama.2009.643.
Park SW, Kim TN, Nam JK, Ha HK, Shin DG, Lee W, et al. Recovery of overall exercise ability, quality of life, and continence after 12-week combined exercise intervention in elderly patients who underwent radical prostatectomy: a randomized controlled study. Urology. 2012;80(2):299–305. https://doi.org/10.1016/j.urology.2011.12.060.
Porserud A, Sherif A, Tollback A. The effects of a physical exercise programme after radical cystectomy for urinary bladder cancer. A pilot randomized controlled trial. Clin Rehabil. 2014;28(5):451–9. https://doi.org/10.1177/0269215513506230.
Sprod LK, Fernandez ID, Janelsins MC, Peppone LJ, Atkins JN, Giguere J, et al. Effects of yoga on cancer-related fatigue and global side-effect burden in older cancer survivors. J Geriatr Oncol. 2015;6(1):8–14. https://doi.org/10.1016/j.jgo.2014.09.184.
Winters-Stone KM, Lyons KS, Dobek J, Dieckmann NF, Bennett JA, Nail L, et al. Benefits of partnered strength training for prostate cancer survivors and spouses: results from a randomized controlled trial of the Exercising Together project. J Cancer Surviv: Res Pract. 2016;10(4):633–44. https://doi.org/10.1007/s11764-015-0509-0.
Alibhai SMH, Santa Mina D, Ritvo P, Sabiston C, Krahn M, Tomlinson G, et al. A phase II RCT and economic analysis of three exercise delivery methods in men with prostate cancer on androgen deprivation therapy. BMC Cancer. 2015;15(1):312. https://doi.org/10.1186/s12885-015-1316-8.
Bourke L, Gilbert S, Hooper R, Steed LA, Joshi M, Catto JWF, et al. Lifestyle changes for improving disease-specific quality of life in sedentary men on long-term androgen-deprivation therapy for advanced prostate cancer: a randomised controlled trial. Eur Urol. 2014;65(5):865–72. https://doi.org/10.1016/j.eururo.2013.09.040.
Cases MG, Fruge AD, De Los Santos JF, Locher JL, Cantor AB, Smith KP, et al. Detailed methods of two home-based vegetable gardening intervention trials to improve diet, physical activity, and quality of life in two different populations of cancer survivors. Contemp Clin Trials. 2016;50:201–12. https://doi.org/10.1016/j.cct.2016.08.014.
Demark-Wahnefried W, Morey MC, Clipp EC, Pieper CF, Snyder DC, Sloane R, et al. Leading the Way in Exercise and Diet (Project LEAD): intervening to improve function among older breast and prostate cancer survivors. Control Clin Trials. 2003;24(2):206–23.
Demark-Wahnefried W, Morey MC, Sloane R, Snyder DC, Miller PE, Hartman TJ, et al. Reach out to enhance wellness home-based diet-exercise intervention promotes reproducible and sustainable long-term improvements in health behaviors, body weight, and physical functioning in older, overweight/obese cancer survivors. J Clin Oncol. 2012;30(19):2354–61. https://doi.org/10.1200/JCO.2011.40.0895.
Kim M-S, Park S-W, Chung MK. The effects of 12 Week’s combined exercise intervention on physical function and urinary incontinence after prostatectomy; preliminary study. Korean J Urol Oncol. 2011;9(1):9–16.
Snyder DC, Morey MC, Sloane R, Stull V, Cohen HJ, Peterson B, et al. Reach out to ENhancE Wellness in Older Cancer Survivors (RENEW): design, methods and recruitment challenges of a home-based exercise and diet intervention to improve physical function among long-term survivors of breast, prostate, and colorectal cancer. Psycho-oncology. 2009;18(4):429–39. https://doi.org/10.1002/pon.1491.
Winters-Stone KM, Lyons KS, Nail LM, Beer TM. The Exercising Together project: design and recruitment for a randomized, controlled trial to determine the benefits of partnered strength training for couples coping with prostate cancer. Contemp Clin Trials. 2012;33(2):342–50. https://doi.org/10.1016/j.cct.2011.10.015.
Rowland JH, Bellizzi KM. Cancer survivorship issues: life after treatment and implications for an aging population. J Clin Oncol. 2014;32(24):2662–8. https://doi.org/10.1200/JCO.2014.55.8361.
Sweegers MG, Boyle T, Vallance JK, Chinapaw MJ, Brug J, Aaronson NK, et al. Which cancer survivors are at risk for a physically inactive and sedentary lifestyle? Results from pooled accelerometer data of 1447 cancer survivors. Int J Behav Nutr Phys Act. 2019;16(1):66. https://doi.org/10.1186/s12966-019-0820-7.
Hurria A, Naylor M, Cohen HJ. Improving the quality of cancer care in an aging population: recommendations from an IOM report. JAMA. 2013;310(17):1795–6. https://doi.org/10.1001/jama.2013.280416.
Attwell L, Vassallo M. Response to pulmonary rehabilitation in older people with physical frailty, sarcopenia and chronic lung disease. Geriatrics (Basel, Switzerland). 2017;2(1). https://doi.org/10.3390/geriatrics2010009.
Subramaniam A, McCarthy P, Elshafi M, Bowen B, Henry M. Outcomes of pulmonary rehabilitation for COPD in the elderly: a comparative study. Eur Respir J. 2015;46(suppl 59):PA2216. https://doi.org/10.1183/13993003.congress-2015.PA2216.
Lustria MLA, Noar SM, Cortese J, Van Stee SK, Glueckauf RL, Lee J. A meta-analysis of web-delivered tailored health behavior change interventions. J Health Commun. 2013;18(9):1039–69. https://doi.org/10.1080/10810730.2013.768727.
Krebs P, Prochaska JO, Rossi JS. A meta-analysis of computer-tailored interventions for health behavior change. Prev Med. 2010;51(3–4):214–21.
Noar SM, Benac CN, Harris MS. Does tailoring matter? Meta-analytic review of tailored print health behavior change interventions. Psychol Bull. 2007;133(4):673–93. https://doi.org/10.1037/0033-2909.133.4.673.
Bayly J, Wakefield D, Hepgul N, Wilcock A, Higginson IJ, Maddocks M. Changing health behaviour with rehabilitation in thoracic cancer: a systematic review and synthesis. Psychooncology. 2018;27(7):1675–94. https://doi.org/10.1002/pon.4684.
Rhodes RE, Yao CA. Models accounting for intention-behavior discordance in the physical activity domain: a user’s guide, content overview, and review of current evidence. Int J Behav Nutr Phys Act. 2015;12:9. https://doi.org/10.1186/s12966-015-0168-6.
Grimmett C, Corbett T, Brunet J, Shepherd J, Pinto BM, May CR, et al. Systematic review and meta-analysis of maintenance of physical activity behaviour change in cancer survivors. Int J Behav Nutr Phys Act. 2019;16(1):37. https://doi.org/10.1186/s12966-019-0787-4.
Peddle-McIntyre CJ, Singh F, Thomas R, Newton RU, Galvao DA, Cavalheri V. Exercise training for advanced lung cancer. Cochrane Database Syst Rev. 2019;2:CD012685. https://doi.org/10.1002/14651858.CD012685.pub2.
Titz C, Hummler S, Thomas M, Wiskemann J. Physical exercise in advanced cancer patients undergoing palliative treatment. Exp Rev Qual Life Cancer Care. 2016;1(6):433–42. https://doi.org/10.1080/23809000.2016.1251292.
Munk T, Tolstrup U, Beck AM, Holst M, Rasmussen HH, Hovhannisyan K, et al. Individualised dietary counselling for nutritionally at-risk older patients following discharge from acute hospital to home: a systematic review and meta-analysis. J Hum Nutr Diet. 2016;29(2):196–208.
Levett DZH, Grimmett C. Psychological factors, prehabilitation and surgical outcomes: evidence and future directions. Anaesthesia. 2019;74(S1):36–42. https://doi.org/10.1111/anae.14507.
Presley CJ, Dotan E, Soto-Perez-de-Celis E, Jatoi A, Mohile SG, Won E, et al. Gaps in nutritional research among older adults with cancer. J Geriatr Oncol. 2016;7(4):281–92. https://doi.org/10.1016/j.jgo.2016.04.006.
Govindaraju T, Sahle BW, McCaffrey TA, McNeil JJ, Owen AJ. Dietary patterns and quality of life in older adults: a systematic review. Nutrients. 2018;10(8):971. https://doi.org/10.3390/nu10080971.
Government S. Co-ordinated, integrated and fit for purpose: a delivery framework for adult rehabilitation in Scotland. 2007.
Evans RA, Singh SJ, Collier R, Loke I, Steiner MC, Morgan MDL. Generic, symptom based, exercise rehabilitation; integrating patients with COPD and heart failure. Respir Med. 2010;104(10):1473–81. https://doi.org/10.1016/j.rmed.2010.04.024.
Funding
CF, SG and FS are supported by grants from Yorkshire Cancer Research Foundation.
Author information
Authors and Affiliations
Contributions
CF, FS, ML and MJ created the concept and design of the study. CF and SG created search strategies, performed searches, screened records and extracted data. CF and MJ prepared the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This review does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 28 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Forbes, C.C., Swan, F., Greenley, S.L. et al. Physical activity and nutrition interventions for older adults with cancer: a systematic review. J Cancer Surviv 14, 689–711 (2020). https://doi.org/10.1007/s11764-020-00883-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-020-00883-x