Abstract
Purpose
Patients with advanced disease are living longer and commonly used patient-reported outcome measures (PROMs) may miss relevant elements of the quality of extended survival. This systematic review examines the measures used to capture aspects of the quality of survival including impact on patients’ everyday lives such as finances, work and family roles.
Methods
Searches were conducted in MEDLINE, EMBASE, CINAHL and PsycINFO restricted to English language articles. Information on study characteristics, instruments and outcomes was systematically extracted and synthesised. A predefined set of criteria was used to rate the quality of studies.
Results
From 2761 potentially relevant articles, 22 met all inclusion criteria, including 10 concerning financial distress, 3 on roles and responsibilities and 9 on multiple aspects of social well-being. Generally, studies were not of high quality; many lacked bias free participant selection, had confounding factors and had not accounted for all participants. High levels of financial distress were reported and were associated with multiple demographic factors such as age and income. There were few reports concerned with impacts on patients’ roles/responsibilities in everyday life although practical and emotional struggles with parenting were identified. Social difficulties were common and associated with multiple factors including being a caregiver. Many studies were single time-point surveys and used non-validated measures. Exceptions were employment of the COST and Social Difficulties Inventory (SDI), validated measures of financial and social distress respectively.
Conclusions
Impact on some important parts of patients’ everyday lives is insufficiently and inconsistently captured. Further PROM development focussing on roles and responsibilities, including work and caring for dependents, is warranted.
Implications for Cancer Survivors
Factors such as finances, employment and responsibility for caring for dependants (e.g. children and elderly relatives) can affect the well-being of cancer survivors. There is a need to ensure that any instruments used to assess patients’ social well-being are broad enough to include these areas so that any difficulties arising can be better understood and appropriately supported.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rapidly emerging novel treatments in oncology, particularly in advanced disease, mean that more patients are living longer [1]; for those who cannot be cured, the goal of therapy is extending survival whilst maintaining or improving quality of life. This changing landscape has implications for the tools both researchers and clinicians have at their disposal to evaluate and improve patient outcomes holistically.
The measurement of a patient’s subjective experience of both the symptoms of disease and treatment-related toxicity within clinical trials has increased [2, 3]. This practice is expanding to routine care and follow-up [4, 5] with the concept of health-related quality of life (HRQoL) subsumed under the broader term of patient-reported outcomes (PROs). Good, well-validated HRQoL instruments have been used in oncology for some time, measuring the impact of disease on functioning and well-being. Many checklists and subscales are available enabling measurement of treatment related side effects as well as symptoms of disease. These are usually designed for use with more generic HRQoL questionnaires revealing the overall impact that disease and treatment may exert on physical, functional, emotional and social well-being. The best validated, generic patient-reported outcome measures (PROMs) used most frequently are the European Organisation for Research and Treatment of Cancer (EORTC) QLQ-C30 [6] and the Functional Assessment of Cancer Therapy (FACT-G) [7] questionnaires. Both have disease-specific cancer modules or subscales, but the FACT has a wider range of treatment-specific subscales.
Although the more frequent use of PROMs is encouraging, there may well be areas of concern impacting the quality of survival that are insufficiently assessed. A previous systematic review by Muzzati and Annunziata [8] showed that some PROMs examine different and limited aspects of patients’ social well-being. For example the subscale of the EORTC QLQ-C30 refers to social and role functioning, the FACT-G to relatives and relationships and the Psychological Screen for Cancer [9] to social support. The authors of this previous systematic review [8] commented that none of the multidimensional HRQoL instruments totally captured the complexity of ‘social impact’. This led them to a systematic search of instruments dedicated solely to the assessment of the social aspects of the cancer experience (or validated in cancer populations) which yielded 27 articles relating to 14 instruments. The social dimensions covered by these tools were predominantly communication (couple/family/caregiver), relationships (with family and friends) and support (from family and friends) and are a reflection of the search terms derived from the social items listed in existing HRQoL measures and a limiting factor of the review. By deriving the search terms using this method, it is not surprising to have identified tools that measure similar issues to those of the social domains of the HRQoL instruments, but potentially covering them in greater depth. Our contention is that the literature on the social concerns and issues faced by cancer patients contains a broader spectrum of social issues than the items on those existing HRQoL instruments. Though Muzzati and Annunziata’s review systematically searched for more depth, it failed to extend its range potentially missing areas of interest. The review did identify one instrument, the Social Difficulties Inventory [10], which included an ‘everyday living’ subscale examining care of dependents, recreation and independence and a ‘money matters’ subscale with items on work, finances and welfare benefits. The social problems that patients with cancer have has been documented for some considerable time with early literature showing that problems can be experienced with employment [11–13], managing daily living [12, 14], finances [15, 16] and insurance [13, 17]. Later work has continued to reveal the social issues that cause significant concern for patients. Both qualitative research [18–20] and pure economic surveys/evaluations [21–25] illustrate that financial worry and hardship are both evident and prevalent. Furthermore, quantitative reports of return to work rates and functional abilities in terms of work performance and obstacles [26–33] and qualitative studies exploring the importance and meaning of work for patients [34, 35] serve to underline the salience of this area. Qualitative studies demonstrate that roles and responsibilities outside of work (i.e. being a parent or carer for others) are also a significant part of patients’ lives and that cancer has consequent interaction and impact on these [36–40]. Wright and colleagues [41] conducted a robust study with patients using focus groups and interviews to describe and categorise cancer patients’ social difficulties. A total of 32 social problems were identified which were able to be categorised under eight headings: (1) managing in the home, (2) health and welfare services, (3) finances, (4) employment, (5) legal matters, (6) relationships, (7) sexuality and body image and (8) recreation. This literature suggests a breadth of social aspects of patients’ lives impacted by cancer that is not traditionally captured alongside treatment toxicity and disease burden, endorsing the need for a further literature search focussed on PROs covering the areas not so well accounted for by the commonly used HRQoL measures.
The current review aims to provide summary and evaluation of PROMs used in studies for reporting the impact of cancer on wider social aspects of patients’ lives, particularly those which may be salient to living with the illness for an extended period, in other words, expanding assessments to look at lifestyle impacts under a conceptual framework which is in the early stages of delineation and referred to as ‘quality of survival’ (defined by four interconnected dimensions: survival, quality of life, side effect management and economic impact management) [42]. This will highlight what has been used, and how and where gaps may still exist which could be addressed with revision or supplementation of currently employed tools. The focus is on the patient with cancer, not caregivers nor relatives (see Shilling et al. [43] for a review of caregiver impact measures).
Methods
Data sources and search
We followed the general principles published by the NHS Centre for Reviews and Dissemination [44] and carried out an electronic search of databases to identify publications using PROs to evaluate the impact of cancer on social aspects of patients’ lives, specifically financial, lifestyle and occupational circumstances, along with roles and responsibilities with dependents (looking after young offspring, a spouse or elderly parents).
Searches were run in MEDLINE (MEDLINE(R) in-process and other non-indexed citations and MEDLINE(R) 1946-present) and EMBASE (1947-present) (both via OvidSP) and CINAHL (1937-present) and PsycINFO (from 1800s–present) (both via EBSCOhost). Terms were modified as appropriate for each database and limited to English language only. A combination of controlled syntax (MeSH) and free-text terms were used. Two groups of terms were generated describing (i) the population and (ii) social aspects of interest. The terms within each group were combined with a Boolean OR command and were searched in combination using a Boolean AND command. Searches were run on 5 March 2015 and an update conducted 8 January 2016 (see Box 1 for the search strategy used for MEDLINE, adapted for other databases). The reference lists of pertinent review articles were also checked for relevant articles.
Box 1: Search strategy used in MEDLINE and adjusted for other databases |
1. exp neoplasm/ |
2. (neoplasm* or cancer).mp. |
3. 1 or 2 |
4. (‘role burden*’ or ‘impact on leisure’ or ‘impact on lifestyle’ or ‘impact on life style’ or ‘role responsibilit*’ or ‘carer responsibilit*’ or ‘childcare role’ or ‘childcare responsibilit*’ or ‘childcare burden*’ or ‘impact on social life’ or ‘social impact’ or ‘family function’).mp. |
5. exp return to work/ |
6. (‘return to work’ or ‘impact on occupation’).mp. |
7. 5 or 6 |
8. (‘out of pocket’ or ‘out-of-pocket’ or ‘financ* burden*’ or ‘financ* impact’ or ‘impact on financ*’).mp. |
9. 4 or 7 or 8 |
10. 3 and 9 |
11. limit 10 to english language |
Study selection criteria
Inclusion criteria were articles in the English language on any quantitative study containing the report of the impact of advanced (or secondary or metastatic or stages III or IV) cancer, as assessed by the use of PROs, on the wider social aspects of adult patients’ lives, that is on financial situation, lifestyle, occupational circumstances, roles and responsibilities with dependents (looking after young offspring or elderly parents) and there was no date restriction.
Excluded were qualitative studies, those with children or caregivers as the sole participants, those involving populations with early cancer (i.e. disease stages I or II) or the very final end-of-life stage; also excluded were studies focussed on the metrics of an instrument, reviews, editorials, letters, opinion, reports published on meeting abstracts and academic theses, any of which an English language version of the PRO had not been developed. Pure monetary costing surveys and functional workability assessment surveys were not included as these give no information on perceived impact on the patient, e.g. recording in a diary a certain amount of money was spent on a hospital parking charge does not establish if, or how, burdensome it was.
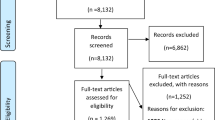
A schema for identification and selection of eligible articles can be seen in Fig. 1. Titles and abstracts were screened independently by two reviewers (SC and VS) against the inclusion and exclusion criteria, and any duplicate papers were recorded and excluded. The full text of potentially relevant papers were retrieved and then further scrutinised independently by both reviewers to identify the final list included in the review. Article selection disagreements were resolved by discussion and a third reviewer (LF) provided adjudication if necessary.
Citation chasing
Backwards citation chasing (one generation) using reference lists of all papers included in the review and forwards citation chasing (one generation) using Science Citation Index Expanded and Social Science Citation Index Expanded via Web of Science were conducted.
Data extraction
For each included paper, the following descriptive data were extracted into a standardised form: first author name, publication year, setting/country, study design and aim, study population (cancer type and demography), number of participants, what social factor/s were assessed and how, reported outcomes and study limitations. Data were extracted and the quality of studies appraised by one reviewer (RS) and checked by a second (SC or VS). Quality appraisal criteria are shown in Box 2 and were developed in accordance with the principles published by the National Health Service Centre for Reviews and Dissemination [44]. Without evidence to inform any differential weighting amongst the criteria, none was applied. A score of one point was awarded where good evidence was provided to meet a criterion, a half point if the evidence provided was weak, and where no evidence was present a zero score was given. Higher total scores represent better quality with articles scoring 0–3.5 designated a +, scores 4–7.5 a ++ and 8–12 a +++.
Box 2: Criteria for assessing the quality of studies |
1. Is the aim of the study clear? |
2. Is the study design/method appropriate? |
3. Are the study eligibility criteria specified? |
4. Is participant selection apparently bias free? |
5. Is there a power calculation for sample size? |
6. Is sample size adequate and response rate satisfactory? (a possible half point could be given for each) |
7. Has a validated measure been used? (i.e. evidence of instrument development, content and construct validity, test-retest reliability, ability to detect change) |
8. Are data analyses appropriate? |
9. Are all study participants accounted for? |
10. Is the study apparently free of confounding factors? |
11. Are the conclusions supported by the results? |
12. Are the results generalizable? |
Results
Articles
The initial search yielded 3115 articles; an additional 6 were identified from hand searches (Fig. 1). After removal of duplicates and initial screening of titles and abstracts, 45 full texts were examined; 12 met the inclusion criteria. A further 8 articles added from the citation searches and 2 from the electronic search update resulted in 22 articles included in the review. The characteristics of the studies are summarised in Table 1, grouped under three broad subheadings dependent upon the focus of investigation: financial distress, roles and responsibilities or multiple aspects of social life. Description of the instruments used and study outcomes is summarised in Table 2.
Most articles comprised single time-point surveys covering a broad array of tumour sites. The majority examined mixed tumour groups, but 7/22 reported on single disease groups. Sample size varied widely, ranging from 26 to over 21,000 patients. Overall, sex representation was clearly specified for all of the studies, but details of participants’ ages were less consistently provided. Most of the studies had been conducted within a time frame of the first 3 years following diagnosis. Some study samples were early disease, some clearly advanced disease and others a mixture or details were not provided within the article; however, no specification of stage was made in the inclusion criteria for the current review.
Quality of publications
Marked against the quality criteria in Box 2, and reported in Table 1, only four studies scored highly (+++) [47, 57, 65, 66]. Some individual criteria warrant specific comment. Only 8 studies were rated as having bias free participant selection, 8 were rated as being apparently free of confounding factors and 11 accounted for all participants in their reporting. Of particular note was the use of diverse, non-validated, study-specific instruments to investigate financial distress; only two of 10 studies used a validated measure.
Financial distress
Table 2 details the 10 articles [45–54] that reported on self-perceived financial distress. Only two used a validated measure. One [47] employed ‘The COST measure’ [68], a recently developed 11-item tool validated in patients with advanced cancer and available via the Functional Assessment of Chronic Illness Therapy (FACIT) suite of questionnaires (see www.facit.org). The second [48] used the ‘Personal Financial Wellness (PFW) Scale’ [69] (formally known as the ‘Incharge Financial Distress/Financial Well-Being Scale’ [70]), which has 8 items, is validated and has been used with cancer patients but without normative data [71]. The remaining eight studies [45, 46, 49–54] used un-validated study-specific tools. The number of questions used in these non-validated measures varied widely, and the content of the items across the measures was diverse and highly individual. Outcomes from the 10 articles show the presence of much self-perceived financial distress amongst the study participants. The findings also demonstrated strategies frequently employed by patients to mitigate their financial burden, including financial reductions to health-related areas and more generally with clothing, food and leisure. Additionally, patients borrowed money, used credit and defaulted on bills. Factors repeatedly associated with greater financial burden were younger age, lower income, poorer health status, more treatment/s, non-white ethnicity and poorer psychological well-being (i.e. greater anxiety and depression).
Roles and responsibilities
Three articles [55–57] investigated the impact on patients’ roles and responsibilities in everyday life. Two [55, 56] used the ‘Parenting Concerns Questionnaire’ (PCQ) [72]. Park and colleagues [56] found that patients had mild to moderate concerns about their cancer affecting their parental role, this was both in terms of performing the usual practical things parents do for their children, but was also about the emotional consequences for the child. Of particular concern was how the child would cope with their death, impact of cancer on the child’s emotions and impact on the co-parent’s emotions. Factors associated with higher levels of concerns about parenting were single marital status, poor functional status, poorer quality of life, psychological distress (anxiety and depression) and lack of social support.
Moore et al. [55] combined the use of the PCQ with another tool, the ‘Parental Efficacy Beliefs Scale’, a non-validated instrument measuring patients’ perceptions of their own ability, and that of their co-parent to perform the parental role. Most (60%) felt they had been extremely well able to perform their parenting responsibilities before diagnosis, dropping to 11% after. Similarly, patients reported less belief in the co-parent after diagnosis (42% dropping to 26%). Factors associated with reported decline in parental efficacy were more frequent clinic visits, poorer quality of life and depression. Higher levels of parenting concerns expressed on the PCQ were accompanied by decline in patients’ belief about themselves and their co-parent being able to parent.
Walsh et al. [57] investigated the impact for women with breast cancer on their roles as wife and mother using the ‘Marital’ and ‘Relationship with Children’ subscales from the CARES instrument [73]. Talking to their spouse/partner about what may happen after their death was problematic for 52.8% of the women, and 37.4% indicated that just talking about cancer with them was difficult. Levels of reported difficulty on the Relationship with Children subscale were not high; however, 19.7% reported problems helping their children talk about their cancer, 16.8% helping their children cope and 12.7% taking care of their children as a result of breast cancer.
Multiple aspects of social life
Nine articles used multidimensional instruments [58–66, N.B. 59 and 60 used the same data set]. Five [58–60, 62, 64] utilised the illness intrusiveness rating scale (IIRS) [74], a validated tool which measures the extent to which an illness and/or treatment affects the patient’s social life. Greater levels of intrusiveness were found to be associated with advanced disease, younger age, lower income, cancer type, recent treatment and poor psychological well-being (depression and hopelessness). Conversely, being older, not having children under 18 years living at home, having early stage disease, fewer symptoms and better psychosocial well-being were all associated with consistently low levels of illness intrusiveness. [64].
The finding that cancer and its treatments commonly impact the social aspects of patients’ lives was evident in three papers [63, 65, 66] that had used the Social Difficulties Inventory (SDI), a validated and psychometrically tested instrument [10, 75–78]. In one article with pooled data from three studies, 45% of participants reported high levels of social difficulty, which was associated more with younger age, those with advanced disease and, to some extent, those from deprived areas [65]. In another study, greater social difficulties were found to be associated with invasive disease (lymph node involvement or distant metastases), undergoing surgery, receiving chemotherapy and lower socioeconomic status, and a reduction in difficulties occurred by 10 months follow-up with the apparent disparity between low and high socioeconomic status groups disappearing [63]. Results from nearly 18,000 patients endorsed previous reports that predictors of social difficulties include younger age, living in a more deprived area, having advanced disease and undergoing surgery or chemotherapy [66]. This study also found additional predictive factors were tumour site, non-white ethnicity, receipt of radiotherapy and being a caregiver. The strongest predictor for social distress was found to be having ≥3 long-term co-morbidities, followed by unemployment and recurrent or non-treatable disease.
Social and financial impacts were also reported using a non-validated questionnaire in a study contrasting rural and urban cancer patients’ experiences [61]. Sixty-four percent of patients experienced at least one financial or social consequence, most frequently this was time-off work (44%), reduced income (31%) and missing family events (23%). City dwellers reported significantly more financial impact, but no similar difference was evident for social impact. Those who were younger, currently working and suffering stress, were found to be at higher risk of experiencing financial or social impacts.
Discussion
Though the studies reviewed here were diverse in terms of study objectives, methods and patient populations, the patient-reported outcomes reported by them repeatedly show cancer and its treatments have significant impact on patients’ finances, roles and responsibilities and on the various facets of their social life.
Moreover, the findings complement reports from published quantitative studies [26–33] that have asked patients to report impact objectively (e.g. by reporting how much money they have spent on an item such as a medication, by indicating the time point of return to work or by indicating ability to perform work tasks) and qualitative studies [34–40] providing first-hand patient accounts of burden. All of this together underlines the far broader nature of the aspects of patients’ social lives that are impacted than can be assessed by relying solely on a social function subscale from one of the validated HRQoL instruments frequently in use, e.g. EORCT-QLQ-C30 [6] or FACT-G [7].
The studies reviewed which have attempted to quantify patients’ self-perceived financial distress have all done so using different, and in the majority of cases [45, 46, 49–54], study-specific measures. This has led to diverse and highly individualised content all scored with incomparable rating scales. This makes summarising findings across studies problematic. However, interest and concern regarding financial burden for patients with cancer have been growing recently [24], driven by the rapid pace of change where expensive novel drugs and living longer with incurable disease are now integral to the picture. In response to this pressure, at least one validated PRO, the COST measure [68], has been developed. To improve the quality of studies conducted and data collected in the future, validated instruments such as this need to be consistently and widely used. This may help researchers and healthcare professionals to better explore, understand and help ameliorate financial distress that patients may experience as a consequence of their cancer and treatments. The current review highlights a paucity of work regarding PROs used to quantify the impacts on patients’ everyday roles and responsibilities in life as a result of cancer and its treatments. Only three studies [55–57] were found, each exploring the impact on the parental role with validated tools. However, data are predominantly from female participants (see Table 1) which should be addressed with future work. The impact on any role/responsibility a patient might have towards other dependants such as elderly parents or an incapacitated spouse/partner was covered with only a single generic item on the SDI, ‘Have you had difficulty with looking after those who depend on you? (e.g. children, dependent adults, pets)’. Yet, evidence suggests that these aspects need to be included in our evaluations; exploratory qualitative analysis has shown that older women with breast cancer who cared for others struggled to balance caregiving responsibilities and their own health needs [36]. In another study, women with early-stage breast cancer and multiple caregiving roles were found to have elevated depressed mood over time [39].
Additionally, there is a survey literature [26–33], not reviewed here, documenting return to work rates and the assessment of work-related functional abilities measured using instruments such as the ‘Work Ability Index’ [79] or the ‘Work Limitations Questionnaire’ [80] in patients with, or treated for, cancer. A few qualitative studies [34, 35, 40] have described the value and meaning of paid work in patients’ lives and barriers to re-joining the workforce. However, the current review found little of patients’ own perceptions of the impact of cancer and its treatments on these aspects of life measured using a comprehensive PRO. The two validated multidimensional instruments identified (IIRS, SDI) have single items asking about impact on work amounting to nothing above that already found in the frequently used, validated, HRQoL instruments (EORCT-QLQ-C30 or FACT-G).
Two validated multidimensional PROs were identified, the IIRS [74] and SDI [10], which capture a broad range of aspects of patients’ everyday lives albeit balanced with brevity. The SDI is of particular relevance since it has been specifically developed for the oncology setting with the aim of being used as a tool to facilitate the integration of the assessment of social well-being into routine care in a way similar to that advocated for psychological well-being [76, 77]. Its purposes are to aid detection and characterisation of social problems and to improve communication between patients and healthcare professionals about this, leading to appropriate service responses, including if necessary onward referral and ultimately better care and enhanced well-being for patients. Other ways that it, and similar instruments, could be used include for the research and development of interventions to help patients manage social impacts like financial burden or family caregiving responsibilities, or perhaps as part of survivorship care plans, as well as for assessing any differential impacts of treatments being compared in clinical trials.
Nevertheless, clearly insufficient data are being gathered with patients to comprehensively understand their problems and needs to enable supporting them living well with their disease, including making decisions about ongoing treatment options. Impacts on the social domain need to be consistently assessed more comprehensively and this has implications for the current HRQoL instruments commonly used. Options for the future include using the SDI in tandem with the existing well-validated quality of life measures and/or generating further assessment modules (i.e. subscales) for the existing instruments.
Limitations
This review was intentionally restricted to identifying studies that reported the use of PRO instruments dedicated solely to the assessment of the financial situation, lifestyle, occupational circumstances, and roles and responsibilities with dependents (looking after young offspring or elderly parents) of adult cancer patients living with advanced disease. In taking this approach, we acknowledge that we have not conducted a mapping and review process of existing social domain subscales of either the general or the more specific health-related quality of life instruments available to use in oncology and that such an undertaking could reveal further useful validated measurement items. One such tool absent from the current review is the Patient-Reported Outcomes Measurement Information System (PROMIS) [81], a product of the National Institutes of Health (NIH) in the USA. This is because existing literature on its social health domain measure (which covers companionship, social isolation, social roles and activities and social support) [82] is about its development and psychometrics, meaning it either did not meet the search terms and/or inclusion criteria or did meet an exclusion criterion. However, it is likely to have significant presence in the future, and an awareness of it is important. It has an item bank approach to its method of development, has been evolved with patients and content experts and has undergone rigorous testing and validation continues to be ongoing. It has a scoring system to norm responses to the general US population with free access and use.
Conclusions
There is a need and opportunity for further work, both in terms of instrument use and development, to ensure that the social areas of patients’ lives are comprehensively assessed. Our tools must keep pace with changes happening in cancer treatment outcomes so that salient social problems are anticipated and/or ameliorated. The COST and SDI, two well-developed and validated tools, could be used to supplement shortfalls in the commonly employed HRQoL instruments. But, there is also scope for developing additional subscales within the existing modular systems to enhance them. In particular, attention needs to be directed towards roles and responsibilities, including work and patients with dependents.
References
John L, Cowey CL. The rapid emergence of novel therapeutics in advanced malignant melanoma. Dermatol Ther (Heidelb). 2015;5(3):151–69.
Wilke RJ, Burke LB, Erickson P. Measuring treatment impact: a review of patient-reported outcomes and other efficacy endpoints in approved product labels. Control Clin Trials. 2004;25:535–52.
Bottomley A, Jones D, Claassens L. Patient-reported outcomes: assessment and current perspectives of the guidelines of the Food and Drug Administration and the reflection paper of the European Medicines Agency. Eur J Cancer. 2009;45:347–53.
Smith AB, Wright EP, Velikova G. Improvements in measuring the health-related quality of life of cancer patients. Expert Rev Pharmacoecon Outcomes Res. 2006;6(1):97–105.
Donaldson M. Using patient-reported outcomes in clinical oncology practice: benefits, challenges and next steps. Expert Rev Pharmacoecon Outcomes Res. 2006;6(1):87–95.
Aaronson NJ, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, Filiberti A, Takeda F. The European Organisation for Research and Treatment of Cancer QLQ- 8 C20: a quality of life instrument for use in international clinical trials of oncology. J Natl Cancer Inst. 1993;85(5):365–76.
Cella DF, Tulsky DS, Gray G, Sarafian B, Linn E, Bonomi A, Silberman M, Yellen SB, Winicour P, Brannon J, Eckberg K, Lloyd S, Purl S, Blendowski C, Goodman M, Barnicle M, Stewart I, McHale M, Bonomi P, Kaplan E, Taylor S, Thomas CR, Harris J. The Functional Assessment of Cancer Therapy scale: development and validation of the general measure. J Clin Oncol. 1993;11:570–9.
Muzzatti B, Annunziata MA. Assessing the social impact of cancer: a review of available tools. Support Care Cancer. 2012;20:2249–57.
Linden W, Yi D, Barroetavena MC, MacKenzie R, Doll R. Development and validation of a psychosocial screening instrument for cancer. Health Qual Life Outcomes. 2005;7:54.
Wright EP, Kiely M, Johnston C, Smith AB, Cull A, Selby PJ. Development and evaluation of an instrument to assess social difficulties in routine oncology practice. Qual Life Res. 2005;14(2):373–86.
Berry DL. Return-to-work experiences of people with cancer. Oncol Nurs Forum. 1993;20:905–11.
Malone M, Harris AL, Luscombe DK. Assessment of the impact of cancer on work, recreation, home management and sleep using a general health status measure. J R Soc Med. 1994;87:386–9.
Van Tulder MW, Aaronson NK, Bruning PF. The quality of life of long term survivors of Hodgkin’s disease. Ann Oncol. 1994;5:153–8.
Stommel M, Given CW, Given BA. The cost of cancer home care to families. Cancer. 1993;71:1867–74.
Berkman BJ, Sampson SE. Psychosocial effects of cancer economics on patients and their families. Cancer. 1993;72:2846–9.
Shultz R, Williamson GM, Knapp JE, Bookwala J, Lave J, Fello M. The psychological, social and economic impact of illness among patients with recurrent cancer. J Psychosoc Oncol. 1995;13:21–45.
Wilson D, Evans J. Non-medical implications of malignant melanoma. Br J Plast Surg. 1993;46:158–9.
Bradley S, Sherwood PR, Donovan HS, Hamilton R, Rosenzweig M, Hricik A, Newberry A, Bender C. I could lose everything: understanding the cost of a brain tumor. J Neuro-Oncol. 2007;85:329–38.
Amir Z, Wilson K, Hennings J, Young A. The meaning of cancer: implications for family finances and consequent impact on lifestyle, activities, roles and relationships. Psycho-Oncology. 2013;21:1167–74.
Timmons A, Gooberman-Hill R, Sharp L. “It’s at a time in your life when you are most vulnerable”: a qualitative exploration of the financial impact of a cancer diagnosis and implications for financial protection in health. PLoS One. 2013;8(11):e77549.
Calhoun EA, Chang C-H, Welshman EE, Fishman DA, Lurain JR, Bennett CL. Evaluating the total costs of chemotherapy-induced toxicity: results from a pilot study with ovarian cancer patients. Oncologist. 2001;6:441–5.
Gorden L, Schuffham P, Hayes S, Newman B. Exploring the economic impact of breast cancers during the 18 months following diagnosis. Psycho-Oncology. 2007;16:1130–9.
McNulty J, Khera N. Financial hardship—an unwanted consequence of cancer treatment. Curr Hematolo Malig Rep. 2015;10(3):205–12.
Meisenberg BR. The financial burden of cancer patients: time to stop averting our eyes. Support Care Cancer. 2015;23:1201–3.
Azzani M, Roslani AC, Su TT. The perceived cancer-related financial hardship among patients and families: a systematic review. Support Care Cancer. 2015;23:889–98.
Feuerstein M, Hansen JA, Calvio LC, Johnson L, Ronquillo JG. Work productivity in brain tumor survivors. JOEM. 2007;49(7):803–11.
Lavigne JE, Griggs JJ, Tu XM, Lerner DJ. Hot flashes, fatigue, treatment exposures and work productivity in breast cancer survivors. J Cancer Surviv. 2008;2:296–302.
de Boer AGEM, Verbeek JHAM, Spelten ER, Uitterhoeve ALJ, Ansink AC, de Reijke TM, Kammeijer M, Sprangers MAG, van Dijk FHJ. Work ability and return-to-work in cancer patients. Br J Cancer. 2008;98:1342–7.
Gudbergsson SB, Fossa SD, Dahl AA. Is cancer survivorship associated with reduced work engagement? A NOCWO study. J Cancer Surviv. 2008;2:159–68.
Gordon LG, Lynch BM, Beesley V, Graves N, McGrath C, O’Rourke P, Webb PM. The Working After Cancer Study (WACS): a population-based study of middle-aged workers diagnosis with colorectal cancer and their return to work experiences. BMC Public Health. 2011;11:604.
de Boer AGEM, Bruinvels DJ, Tytgat KMAJ, Schoorlemmer A, Klinkenbijl JHG, Frings-Dresen MHW. Employment status and work-related problems of gastrointestinal cancer patients at diagnosis: a cross-sectional study. BMJ Open. 2011;2:1–8.
Bains M, Munir F, Yarker J, Bowley D, Thomas A, Armitage N, Steward W. The impact of colorectal cancer and self-efficacy beliefs on work ability and employment status: a longitudinal study. Eur J Cancer Care. 2012;21:634–41.
Braybrook JP, Mimoun S, Zarca D, Elia D, Pinder B, Lloyd AJ, Breheny K, Lomazzi M, Borisch B. Patients’ experiences following breast cancer treatment: an exploratory survey of personal and work experiences of breast cancer patients from three European countries. Eur J Cancer Care. 2015;24:650–61.
Amir Z, Neary D, Luker K. Cancer survivors’ views of work 3 years post diagnosis: a UK perspective. Eur J Oncol Nurs. 2008;12:190–7.
Rasmussen DM, Elverdam B. The meaning of work and working life after cancer: an interview study. Psycho-Oncology. 2008;17:1232–8.
Parrish MM, Adams S. An exploratory qualitative analysis of the emotional impact of breast cancer and caregiving among older women. Care Manag J. 2003;4(4):191–7.
Hirshman KB, Bourjolly JN. How do tangible supports impact the breast cancer experience? Soc Work Health Care. 2005;41(1):17–32.
Buchbinder M, Longhofer J, McCue K. Family routines and rituals when a parent has cancer. Fam Syst Health. 2009;27(3):213–27.
Bailey EH, Perez M, Aft RL, Liu Y, Schootman M, Jeffe DB. Impact of multiple caregiving roles on elevated depressed mood in early-stage breast cancer patients and same-age controls. Breast Cancer Res Treat. 2010;121:709–18.
Mackenzie CR. ‘It is hard for mums to put themselves first’: how mothers diagnosed with breast cancer manage the sociological boundaries between paid work, family and caring for self. Soc Sci Med. 2014;117:96–106.
Wright EP, Kiely MA, Lynch P, Cull A, Selby PJ. Social problems in oncology. Br J Cancer. 2002;87:1099–104.
Fallowfield L, Nadler E, Abernethy AP, Gilloteau I, Greaney M, Gater A, Orsini L, Subar M, Dastani HB, Lyman GH. Quality of survival (QoS) concept framework to assess the quality of prolonged life in advanced melanoma (MEL): principles and application related to treatment with nivolumab (NIVO) [abstract]. In: Proceedings of the 18th ECCO-40th ESMO European Cancer Congress; (2015) Sept 25–29; Vienna, Austria. Abstract number 3331
Shilling V, Matthews L, Jenkins V, Fallowfield L. Patient reported outcome measures for cancer caregivers: a systematic review. Qual Life Res. 2016;25:1859. doi:10.1007/s11136-016-1239-0.
NHS Centre for Reviews and Dissemination. Systematic reviews CRD’s guidance for undertaking reviews in health care. York: Centre for Reviews and Dissemination, University of York ; 2008.www.york.ac.uk/inst/crd/index_guidance.htm (Accessed 14 Nov 2014)
Bennett JA, Brown P, Cameron L, Whitehead LC, Porter D, McPherson KM. Changes in employment and household income during the 24 months following a cancer diagnosis. Support Care Cancer. 2009;17:1057–64.
Delgado-Guay M, Ferrer J, Rieber AG, Rhondali W, Tayjasanant S, Ochoa J, Cantu H, Chisholm G, Williams J, Frisbee-Hume S, Bruera E. Financial distress and its associations with physical and emotional symptoms and quality of life among advanced cancer patients. Oncologist. 2015;20(9):1092–8.
Huntington SF, Weiss BM, Vogal DT, Cohen AD, Garfall AL, Mangan PA, Doshi JA, Stadtmauer EA. Financial toxicity in insured patients with multiple myeloma: a cross-sectional pilot study. Lancet Haematol. 2015;2(10):e408–16.
Meisenberg BR, Varner A, Ellis E, Ebner S, Moxley J, Siegrist E, Weng D. Patient attitudes regarding the cost of illness in cancer care. Oncologist. 2015;20:1199–204.
Meneses K, Azuero A, Hassey L, McNees P, Pisu M. Does economic burden influence quality of life in breast cancer survivors? Gynecol Oncol. 2012;124:437–43.
Pisu M, Kenzik KM, Oster RA, Drentea P, Ashing KT, Fouad M, Martin MY. Economic hardship 1 year after diagnosis: another long-term effect of cancer? Cancer. 2015;121(8):1257–64.
Regenbogen SE, Veenstra CM, Hawley ST, Banerjee M, Ward KC, Kato I, Morris AM. The personal financial burden of complications after colorectal cancer surgery. Cancer. 2014;120(19):3074–81.
Rogers SN, Harvey-Woodworth CN, Hare J, Leong P, Lowe D. Patients’ perception of the financial impact of head and neck cancer and the relationship to health related quality of life. Br J Oral Maxillofac Surg. 2012;50:410–6.
Sharp L, Carsin A-E, Timmons A. Association between cancer-related financial stress and strain and psychological well-being among individuals living with cancer. Psycho-Oncology. 2013;22:745–55.
Zafar SY, Peppercorn JM, Schrag D, Taylor DH, Goetzinger AM, Zhong X, Abernethy AP. The financial toxicity of cancer treatment: a pilot study assessing out-of-pocket expenses and the insured cancer patient’s experience. Oncologist. 2013;18:381–90.
Moore CW, Rauch PK, Baer L, Piri WF, Muriel AC. Parenting changes in adults with cancer. Cancer. 2015;121(19):3551–7.
Park EM, Deal AM, Check DK, Hanson LC, Reeder-Hayes KE, Mayer DK, Yopp JM, Song M-K, Muriel AC, Rosenstein DL. Parenting concerns, quality of life, and psychological distress in patients with advanced cancer. Psycho-Oncology (2015) Aug 17 [Epub ahead of print]
Walsh SR, Manuel JC, Avis NE. The impact of breast cancer on younger women’s relationships with their partner and children. Fam Syst Health. 2005;23(1):80–93.
De Groot JM, Mah K, Fyles A, Winton S, Greenwood S, DePetrillos AD, Devins GM. The psychosocial impact of cervical cancer among affected women and their partners. Int J Gynaecol Cancer. 2005;15:918–25.
Devins GM, Bezjak A, Mah K, Loblaw DA, Gotowiec AP. Context moderates illness-induced lifestyle disruptions across life domains: a test of the illness intrusiveness theoretical framework in six common cancers. Psycho-Oncology. 2006;15:221–33.
Mah K, Bezjak A, Loblaw DA, Gotowiec A, Devins GM. Do ongoing lifestyle disruptions differ across cancer types after the conclusion of cancer treatment? J Cancer Surviv. 2011;5:18–26.
Paul CL, Hall AE, Carey ML, Cameron EC, Clinton-McHarg T. Access to care and impacts of cancer on daily life: do they differ for metropolitan versus regional haematological cancer survivors? J Rural Health. 2013;29:s43–50.
Schimmer AD, Elliott ME, Abbey SE, Raiz L, Keating A, Beanlands HJ, McCay E, Messner HA, Lipton JH, Devins GM. Illness intrusiveness among survivors of autologous blood and marrow transplantation. Cancer. 2001;92(12):3147–54.
Simon AE, Wardle J. Socioeconomic disparities in psychosocial wellbeing in cancer patients. Eur J Cancer. 2008;44:572–8.
Sohl SJ, Levine B, Case D, Danhauer SC, Avis NE. Trajectories of illness intrusiveness domains following a diagnosis of breast cancer. Health Psychol. 2014;33(3):232–41.
Wright P, Smith A, Booth L, Winterbottom A, Kiely M, Velikova G, Selby P. Psychosocial difficulties, deprivation and cancer: three questionnaire studies involving 609 cancer patients. Br J Cancer. 2005;93:622–6.
Wright P, Downing A, Morris EJA, Corner JL, Richards MA, Sebag-Montefiore D, Finan P, Glaser AW. Identifying social distress: a cross-sectional survey of social outcomes 12 to 36 months after colorectal cancer diagnosis. J Clin Oncol. 2015;33(30):3423–30.
Given BA, Given CW, Stommel M. Family and out of pocket costs for women with breast cancer. Cancer Pract. 1994;2(3):187–93.
de Souza JA, Yap BJ, Hlubocky FJ, Wrolewski K, Ratain MJ, Cella D, Daugherty CK. The development of a financial toxicity patient-reported outcome in cancer. Cancer. 2014;120(20):3245–53.
Nielson RB. Assessing financial wellness via computer-assisted telephone interviews. J Financ Couns Plan. 2010;21(2):16–29.
Prawitz AD, Garman ET, Sorhaindo B, O’ Neill B, Kim J, Drentea P. Incharge financial distress/financial well-being scale: development, administration, and score interpretation. Financ Couns Plan. 2006;17(1):34–50.
Bestivina CM, Zullig LL, Rushing C, Chino F, Samsa GP, Altomare I, Tulsky J, Ubel P, Schrag D, Nicolla J, Abernethy A, Peppercorn J, Zafar SY. Patient-oncologist cost communication, financial distress, and medication adherence. J Oncol Pract. 2014;10(3):162–7.
Muriel AC, Moore CW, Baer L, Park ER, Kornblith AB, Pirl W, Prigerson H, Ing J, Rauch PK. Measuring psychosocial distress and parenting concerns among adults with cancer: the Parenting Concerns Questionnaire. Cancer. 2012;118(22):5671–8.
Ganz PA, Schrag CA, Lee JJ, Sim MS. The CARES: a generic measure of health-related quality of life for patients with cancer. Qual Life Res. 1992;1(1):19–29.
Devins GM. Using the illness intrusiveness ratings scale to understand health-related quality of life in chronic disease. J Psychosom Res. 2010;68:591–602.
Smith AB, Wright P, Selby P, Velikova G. Measuring social difficulties in routine patient-centred assessment: a Rasch analysis of the social difficulties inventory. Qual Life Res. 2007;16:823–31.
Wright P, Smith A, Roberts K, Selby P, Velikova G. Screening for social difficulties in cancer patients: clinical utility of the social difficulties inventory. Br J Cancer. 2007;97:1063–70.
Wright P, Marshall L, Smith AB, Velikova G, Selby P. Measurement and interpretation of social distress using the social difficulties inventory (SDI). Eur J Cancer. 2008;44:1529–35.
Wright P, Smith AB, Keding A, Velikova G. The social difficulties inventory (SDI): development of subscales and scoring guidance for staff. Psycho-Oncology. 2011;20:36–43.
Tuomi K, Ilmarinen J, Jahkola A. Work ability index. 2nd revised ed. Helsinki: Finnish Institute of Occupational Health; 1998.
Lerner D, Amick BC, Rogers WH, Malspeis S, Bungay K, Cynn D. The work limitations questionnaire. Med Care. 2001;39:72–85.
Broderick JE, DeWitt EM, Rothrock N, Crane PK, Forrest CB. Advances in patient-reported outcomes: the NIH PROMIS measures. EGEMS (Wash DC). 2013;1(1):1015.
Hahn EA, DeVellis RF, Bode RK, Garcia SF, Castel LD, Eisen SV, Bosworth HB, Heinemann AW, Rothrock N, Cella D. PROMIS Cooperative Group. Measuring social health in the patient-reported outcomes measurement information system (PROMIS): item bank development and testing. Qual Life Res. 2010;19(7):1035–44.
Acknowledgments
This review is part of an investigator initiated study funded by Bristol-Myers Squibb.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This review is part of an investigator initiated study funded by Bristol-Myers Squibb.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
As a review paper, this article does not require ethical approval and does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Catt, S., Starkings, R., Shilling, V. et al. Patient-reported outcome measures of the impact of cancer on patients’ everyday lives: a systematic review. J Cancer Surviv 11, 211–232 (2017). https://doi.org/10.1007/s11764-016-0580-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-016-0580-1