Abstract
A challenging, yet fundamental part of initiating effective control measures against an invasive pest species is developing reliable means of monitoring the pest’s seasonal abundance. Halyomorpha halys, a polyphagous insect pest native to East Asia, has become a major economic threat to agricultural systems following unintentional introductions to North America, South America, and Europe. Research involving the seasonal phenology and monitoring attractant preferences of H. halys from its native range remain scarce. An 11-year collection of H. halys monitoring trap data from black light, incandescent light, and methyl (E, E, Z)-2, 4, 6-decatrienoate (MDT) lured traps from three locations in Kyoto, Japan was analyzed to fill gaps in knowledge relating to the native seasonal abundance and effectiveness of diferent trapping techniques for the pest. Due to a high amount of zero trap counts, a zero-inflated approach was taken to analyze the dataset. Overall, H. halys followed a bell-shaped population trend in Kyoto, with abundance peaking in the mid-summer. The attractant preference of H. halys varied slightly with the season, with black light traps producing to highest mean trap counts. The MDT lure traps generated the lowest mean trap counts, but displayed potential use in the reliable detection of early season H. halys. This work is expected to provide greater insight on H. halys in its native range and ultimately help refine existing management programs in invaded regions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Invasive pest species continually present major economic and environmental challenges to natural and agricultural systems worldwide (Venette and Koch 2009). The estimated costs associated with nonindigenous insect pests such as environmental damages, agricultural losses, and management measures exceeds $14 billion (USD) annually in the United States alone (Pimentel et al. 2000). Following the detection of an invasive species, effective and reliable detection and monitoring methods are an essential component of integrated pest management programs (Rice et al. 2018). Understanding pest populations through field monitoring allows for a more efficient implementation of control measures, including timing and frequency of pesticide applications (Kamiyama et al. 2020). However, invasive pest species monitoring can be difficult due to gaps in knowledge of a pest’s behavior and ecology from its native range (Rice et al. 2018).
Halyomorpha halys (Stål, 1855), is a severe economic pest originating from the subtropical and temperate regions of East Asia, including Japan (Lee et al. 2013). This insect pest has invaded North America (Leskey et al. 2012), South America (Faúndez and Rider 2017), and Europe (Wermelinger et al. 2008). Climate-based species distribution models also identify several currently uninvaded regions as potential sites for the future H. halys establishment (Kriticos et al. 2017). Internationally bound cargo is the primary means of accidental introductions of the pest to foreign areas, as H. halys adults are capable of stowing away and hiding for long periods of time (Kriticos et al. 2017). This pest is highly polyphagous, with hundreds of different hosts spanning from agricultural crops to ornamental and landscape plants, which are accessible for feeding and completing their development (Lee et al. 2013; Rice et al. 2014). The economic impacts imposed by H. halys to agricultural industries in invaded regions have been immense not only due to feeding damage and lower crop yields, but also due to the increased pest management costs (Nielsen and Hamilton 2009; Leskey et al. 2012; Kuhar and Kamminga 2017).
Establishing an effective and dependable monitoring method is imperative for the future control schemes of any invasive pest species (Morrison et al. 2015). However, reliably monitoring and detecting H. halys continues to be particularly challenging, stemming from the pest’s cryptic nocturnal activity and high mobility (Nielsen et al. 2013; Rice et al. 2018; Acebes-Doria et al. 2020). Past studies have focused on determining the most effective H. halys monitoring method by testing pheromone attractants (Leskey et al. 2021), black light traps (Nielsen et al. 2013), sticky traps (Acebes-Doria et al. 2018), sweep netting (Nielsen et al. 2011), and visual sampling (Maistrello et al. 2017). Currently, baiting monitoring traps with H. halys aggregation pheromone (Khrimian et al. 2014), in combination with the aggregation pheromone synergist, methyl (E, E, Z)-2, 4, 6-decatrienoate (MDT) from Plautia stali (Scott, 1874) (Adachi et al. 2007; Weber et al. 2014), is recommended as evidenced from studies conducted in the eastern United States (Leskey et al. 2015; Short et al. 2017) and South Korea (Morrison et al. 2017a). However, the effectiveness of established monitoring and detection techniques may vary with abiotic and biotic factors, emphasizing the importance of investigating different H. halys trapping methods in more regions.
In Japan, H. halys is primarily recognized as a pest of cypress trees and other timber sources, and will opportunistically feed on cultivated fruit crops such as apples as a secondary food source (Tsutsumi 2003). While the pest has been documented in causing injury to cultivated fruit crops in Japan (Funayama 2012), the economic impact of its feeding damage is not widely available and therefore scarce in published literature (Lee et al. 2013). Generally, H. halys persists as an occasional outbreak pest in northern Japan, with damage levels varying between fruit crops (Tsutsumi 2003; Funayama 2012; Lee et al. 2013). In the extensive native range of H. halys, the number of generations per year ranges from one to up to six (Lee et al. 2013), with Japanese H. halys populations typically being bivoltine (Tsutsumi 2003). Light traps and commercial MDT lure traps are common forms of H. halys monitoring attractants in Japan, and the pest’s seasonal abundance changes slightly depending on the monitoring method (Lee et al. 2013; Adachi et al. 2007). From the limited published reports concerning the phenology of H. halys in Japan, light traps reveal peak abundance in the summer (Lee et al. 2013), whereas MDT lure traps suggest highest adult activity in the fall (Funayama 2008). More information about the pest’s phenology in its native range is necessary for refining current management practices.
The Kyoto Prefecture Agriculture, Forestry, and Fisheries Technology Center has produced 11 years of adult H. halys monitoring data from black light, incandescent light, and MDT lured traps from three locations in Kyoto, Japan. The monitoring data was comprised of five-day H. halys trap counts from May through October each year. This information is essential for filling gaps in knowledge pertaining to the seasonal phenology and monitoring methods of this invasive pest in its native range. Using this dataset, we first determined the seasonal population dynamics of H. halys in Japan. Then, we compared the mean adult H. halys trap counts from the black light, incandescent light, and MDT lure monitoring traps. After evaluating over a decade’s worth of H. halys monitoring data from Japan, we present novel results that can supplement current knowledge and contribute to the future management of this invasive pest species.
Materials and methods
Field sites
The H. halys adult monitoring traps were located at three agricultural research stations in Kyotanabe, Kameoka, and Kyotango City, Japan (Fig. 1). Each agricultural facility stationed one black light trap, one incandescent light trap, and one MDT lure trap. The traps were installed to gather H. halys monitoring data, and create simple graphs of adult H. halys outbreaks each year in Kyoto (Kyoto Prefecture Pest Control Center Website 2022https://www.pref.kyoto.jp/byogai/index.html). Kyotanabe is located in the southern portion of the Kyoto Prefecture, Kameoka in the central, and Kyotango in the northern portion. Each research station sparsely cultivated various field crops on a small scale for research purposes. These crops mainly included cucurbits and cereal grains such as rice, and pesticides targeting general pest insects were applied only 1–2 time per year. Monitoring data used for this work was compiled for 11 years from 2010 to 2020, and was provided by the Kyoto agricultural research stations.
Black light and incandescent monitoring traps
Two pest outbreak prediction light traps (model MT-7-N, Ikeda Scientific Co., Ltd., Tokyo, Japan) (Fig. 2) were used to monitor populations of adult H. halys at each of the three trapping locations. Light trap monitoring for H. halys was conducted from May 1 through October 31 at each site from 2010 to 2020. The steel light traps measured 92 cm × 92 cm × 200 cm and weighed 85 kg. At each site, the black light traps were installed with a 20-watt black light, and the incandescent light traps were installed with a 60-watt incandescent light. The black lights and incandescent lights were programmed to automatically turn on by a timer at 6:00 PM and turn off at 6:00 AM every day. The light traps at each site were positioned in open areas near crop plantings, and spaced at least 50 m from each other. Each light trap had a catch basin that automatically rotated every five days, allowing each trap catches to be divided into separate five-day total counts. Upon a collection event, the monitoring trap contents were collected and the total number of adult H. halys were counted and recorded. A total of 36 individual five-day trap counts were recorded per year (six times each month, over the six-month trapping period).
MDT lure monitoring traps
One yellow plastic insect attractor trap (Sankei Chemical Co., Ltd., Kagoshima, Japan) (Fig. 3) was used to monitor adult H. halys populations at the three sites. MDT lure trap monitoring for H. halys took place from May 1 through October 31 at each site from 2010 to 2020. The plastic traps were baited with commercial Plautia stali aggregation pheromone methyl (E, E, Z)-2, 4, 6-decatrienoate (MDT) lures (Senkei Chemical Co., Ltd., Kagoshima, Japan), and water was added to the trap basin to trap specimens. The MDT lures are the most commonly used attractant lure used in Japan for monitoring adult H. halys (Adachi et al. 2007; Lee et al. 2013; Kyoto Prefecture Agriculture, Forestry, and Fisheries Technology Center, personal communication). Traps were hung from a steel pole roughly 1.5 m off the ground in open areas near crop plantings. The MDT lure traps were spaced at least 50 m from light traps. The MDT lure traps were checked every 7–10 days throughout the monitoring period each year, at which time specimens were removed from the trap basin and adult H. halys counted and recorded. Water was replenished in the basin upon every collection event, and MDT lures were replaced every other month per the manufacturer’s recommendation.
Data analysis
Five-day adult H. halys count totals from the black light and incandescent light traps were recorded from 2010 to 2020 for each of the three sites. In the case of the MDT lure traps, the adult H. halys trap count was standardized to every five days to conform with the five-day collection frequency of the light traps. Data from all years and sites were pooled for analysis.
The dataset was analyzed using the RStudio program v.3.4.1 (R Core Team 2019). In order to compare the ability of the different traps throughout the field season, the trapping period each year was divided into three seasons: early summer (May – June), mid-summer (July – August), and fall (September – October). The data did not conform to the assumptions of a normal distribution due to a high amount of zero trap counts. Chi-square analyses were deemed appropriate for the zero-inflated data (Togo and Iwasaki 2013; Johnson et al. 2015), and a chi-square test was run to determine differences in trap counts between the season and trap type factors. Estimated marginal means (Searle et al. 1980), for the trap counts were produced using the emmeans function from the emmeans package. The pairs function was then used to group the means, and a Tukey adjustment function was incorporated in the post-hoc analysis to separate the means of the factors at p < 0.05.
A zero-inflated negative binomial (ZINB) model was generated using the glmmTMB function from the glmmTMB package using the RStudio program to correct for the overdispersion caused by an overabundance of zero counts in the monitoring data. The model implemented a logit-link from the family ‘nbinom2’. This framework allowed us to estimate the relative effectiveness each monitoring trap type (black light, incandescent light, MDT lure) had on attracting H. halys adults, as well as the influence each trap type had on producing a zero count. The ZINB incorporated the factor ‘trap type’ as a random effect, and the factors ‘site’, ‘year’, and the ‘site * year’ interaction as fixed effects. A reduced ZINB model containing only the effect of trap type was also produced. A likelihood ratio test was performed on the full and reduced ZINB models, and the full model was deemed as the best fitting model. Compared to the reduced model, the full model also had lower Akaike information criterion (AIC) and Bayesian Information Criterion (BIC) scores, further evidencing it as the more parsimonious model (Wood 2006).
Results
In total, there were 3049 instances of a recorded zero count from the H. halys monitoring traps out of 3551 total collection events across all trap types, sites, and years. The single high adult H. halys count was 63 for the black light traps, 21 for the incandescent light traps, and 2.1 (standardized for a five-day period) for the MDT lure traps. The total mean five-day adult H. halys count was 1.1 for the black light traps, 0.2 for the incandescent light traps, and 0.05 for the MDT lure traps (Table 1).
Across the entire monitoring period, the black light traps produced the highest five-day mean adult H. halys trap count, the incandescent light traps had the second highest mean count, and the MDT lure traps produced the lowest mean count (χ2 = 46.08, p < 0.001) (Table 1). All three trap types had similar counts in the early summer (χ2 = 4.86, p = 0.09). The black light traps had the highest trap counts in the mid-summer (χ2 = 45.69, p < 0.001), and also in the fall (χ2 = 23.91, p < 0.001). Black light traps produced higher counts in the mid-summer compared to the early summer and fall (χ2 = 61.02, p < 0.001). Similarly, incandescent light traps had higher counts in the mid-summer than the early summer and fall (χ2 = 55.15, p < 0.001). The MDT lure traps had higher trap counts in the early summer than the mid-summer and fall (χ2 = 13.44, p = 0.001).
The ZINB generated coefficient estimates for each trap type in a conditional model and a zero-inflation model. In the conditional model, a positive coefficient estimate indicates higher H. halys abundance in response to trapping method, whereas a negative coefficient estimate indicates lower abundance (Jaffe and Guédot 2019; Kamiyama et al. 2021). In the zero-inflation model, a positive coefficient estimate indicates a higher chance of encountering a zero H. halys count in response to trapping method, whereas a negative coefficient estimate indicates lower chance of encountering a zero count (Brooks et al. 2017). The ZINB conditional model produced a positive coefficient estimate for the black light traps (Z = 5.00, p < 0.001), and negative coefficient estimates for the incandescent light (Z = −2.40, p = 0.002) and MDT lure traps (Z = −5.20, p < 0.001) (Table 2). The ZINB zero-inflation model produced negative coefficient estimates for black light (Z = −7.34, p < 0.001) and incandescent light traps (Z = −7.75, p < 0.001), and a positive coefficient estimate for MDT lure traps (Z = 3.37, p = 0.001).
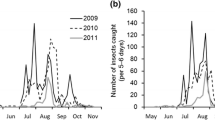
A plot of the mean five-day trap catch data across all years separated by trap type showed that H. halys populations increase from the early summer until they peak in mid-summer, then quickly drop in the fall as measured by the black light traps (Fig. 4). Similarly, the incandescent light traps had increasing trap counts from the early summer through mid-summer, then lower trap counts in the fall. However, the peak trap count as measured by the incandescent light traps was more dampened and drawn out across mid-summer in comparison to the sharper peak produced by the black light traps. The MDT lure traps had relatively low adult H. halys counts throughout the field season, but had highest trap counts in the early summer. Mean five-day trap catch data across all years separated by trap type for each individual site can be viewed in the Online resource: Fig. S1.
Log-transformed mean five-day adult H. halys trap catch across all sites and years for each trap type from May (5) to November (11). A smoothed fit line with standard error band (gray shaded area) is included to help visualize the population trends of H. halys for each trap type. BL = Black light, IL = Incandescent light, MDT = methyl (E, E, Z)-2, 4, 6-decatrienoate lure
Discussion
The pursuit of refining and adopting new monitoring methods remains an important component of current H. halys research (Acebes-Doria et al. 2020; Rondoni et al. 2022). An added caveat to this work is that it is difficult to accurately compare the seasonal abundance of H. halys when measured by different monitoring techniques. For example, light traps with lamps emitting more UV light are more attractive to H. halys than standard incandescent light traps (Endo and Hironaka 2017), and monitoring traps baited with aggregation pheromone lures alone attract less H. halys than traps including a pheromone synergist (Leskey et al. 2015). Our study therefore first establishes a seasonal phenology of adult H. halys in Kyoto, Japan based on adult monitoring using black light, incandescent light, and MDT lure traps, and compares the adult abundance when measured by each respective monitoring method.
Overall, the seasonal phenology of adult H. halys in Kyoto is similar to the previously reported population dynamics of H. halys in Japan (Tsutsumi 2003; Lee et al. 2013), which can be described by our study as low populations in the early summer (May – June), reaching peak activity in the mid-summer (July – August), then numbers dropping in the fall (September – October). However, the population dynamics slightly differed between the trapping methods. The black light and incandescent light trap data revealed H. halys adult numbers are the highest in the mid-summer, which is comparable with previous light trap monitoring studies conducted in Japan showing H. halys captures typically peak in early August (Lee et al. 2013). Black light trapping studies of H. halys in the Mid-Atlantic United States also showed a bell-shaped population trend, with adult activity peaking in the mid-summer (Nielsen et al. 2013). Unlike the light traps, the H. halys trap counts as measured by the P. C. stali aggregation pheromone synergist (MDT) traps in this study were higher in the early summer than the mid-summer and fall. A similar monitoring study done with the MDT lure traps in northern Japan (Akita Prefecture) showed fluctuating H. halys numbers throughout the season, including a peak in activity in mid-May (Funayama 2008). Monitoring research in the eastern United States using the MDT lure together with H. halys aggregation pheromone lures also displayed early season spikes in adult H. halys captures, occurring in late April (Morrison et al. 2017b), and May (Acebes-Doria et al. 2018). This implementation of both H. halys pheromone and MDT lures in combination has resulted in significantly higher adult H. halys captures than MDT lures alone throughout the field season, as shown in monitoring work conducted in South Korea (Morrison et al. 2017a). Unfortunately, aggregation pheromone lures were not included when monitoring for adult H. halys when producing the data set or this work.
The black light traps performed better than the incandescent light traps in capturing adult H. halys, as the black light traps had higher mean trap counts in the mid-summer and fall, as well as the highest overall mean trap count across the entire field season in Kyoto. Black light traps emit large amounts of UV radiation, which has been shown to be more attractive to nocturnal insects than light emitted with higher wavelengths such as incandescent lights (Shimoda and Honda 2013). The increased affinity towards light lower on the wavelength spectrum also applies to H. halys, as evidenced by a field study which demonstrated that applying a UV-absorbing filter to UV light traps reduced their H. halys attractiveness by 75% compared to non-UV filtered traps (Endo and Hironaka 2017). Additionally, black light traps have been determined to be a reliable approach in tracking the spread of H. halys at the landscape level in the United States (Nielsen et al. 2013).
The MDT lure traps produced the lowest mean H. halys trap counts over the course of the entire field season, but had numerically higher trap counts than the black light and incandescent light traps in the early summer when the H. halys field populations were low. There is existing evidence of MDT lure traps being effective in detecting H. halys early in the field season, as MDT lure traps caught H. halys adults earlier than sweep net samples in the eastern United States (Nielsen et al. 2011). Additionally, monitoring work in South Korea reported first adult H. halys capture from MDT lure traps as occurring in May, one month earlier than first captures from light traps (Lee et al. 2002). In the current study, although the MDT lure traps did not produce the mid-summer H. halys abundance peak that both types of light trap did, they were the only trap type to reliably capture H. halys adults in the early summer, indicating MDT lures may hold importance in the early season detection of H. halys.
The preference of H. halys towards different monitoring attractants during different periods of the field season may not only be a result of environmental influence, but also the life cycle of H. halys. Abiotic variables such as temperature, photoperiod, and relative humidity have been determined to affect the behavior of H. halys (Haye et al. 2014; Nielsen et al. 2016; Fisher et al. 2021). After breaking diapause early in the field season, female H. halys require a preoviposition period of about 13 days in which a critical photoperiod is needed for ovarian development (Nielsen et al. 2008; Bergh et al. 2017). Adult H. halys then move in large numbers and aggregate on host crops to feed and replenish carbohydrates lost during diapause (Leskey et al. 2012; Lowenstein and Walton 2018). It is presumed that H. halys become responsive to pheromonal stimuli early in the season (Bergh et al. 2017; Morrison et al. 2017b), and during this period, aggregation pheromone is likely produced in abundance by H. halys to help individuals locate to food resources (Aldrich 1988). This phenomenon in the life cycle of H. halys may explain why the MDT lured traps produced the highest captures in the early summer in Kyoto, compared to later in the mid-summer and fall. Furthermore, when adult H. halys begin to seek out overwintering sites late in the season they become unresponsive to the MDT lure (Weber et al. 2017), rendering the MDT traps impractical during the pre-diapause period. Light traps on the other hand, will act as a consistent source of attraction for H. halys throughout the entire field season. However, the capacity of adult H. halys to fly decreases with decreasing temperature (Lee and Leskey 2015), meaning light traps may be less effective early and late in the season with lower temperatures. A future strategy for maintaining trap effectiveness throughout the field season may be instituting multimodal attractant traps, as research has shown potential in increasing H. halys trap catches by incorporating both light and pheromone attractants in a single trap (Rice et al. 2017; Rondoni et al. 2022).
To summarize, this research describes the seasonal abundance of adult H. halys in Kyoto, and to our knowledge, is the first study that compares the effectiveness of light and MDT lured monitoring traps throughout the field season in Japan. We acknowledge the shortcomings of the 11-year dataset used in this work, such as monitoring in only one prefecture in Japan and overlooking the potential ecological effect of microclimate at each site (Muñoz-Adalia et al. 2022). However, we still believe valuable conclusions can be drawn from this research. Our study demonstrated that black light traps produced the overall highest mean H. halys adult trap counts in Kyoto, and were also the only trap type associated with increased adult H. halys abundance, as evidenced by the ZINB model analysis. Additionally, the MDT lure traps show potential in acting as an early season detection method of adult H. halys. Future research should continue investigating the preferences of H. halys towards different attractants throughout the season, while incorporating a wide range of ecological processes to optimize the monitoring efficiency of this invasive pest.
References
Acebes-Doria AL, Morrison WR III, Short BD, Rice KB, Bush HG, Kuhar TP, Duthie C, Leskey TC (2018) Monitoring and biosurveillance tools for the brown marmorated stink bug, Halyomorpha halys (Stål) (Hemiptera: Pentatomidae). Insects 9:82. https://doi.org/10.3390/insects9030082
Acebes-Doria AL, Agnello AM, Alston DG, Andrews H, Beers EH, Bergh JC, Bessin R, Blaauw BR, Buntin GD, Burkness EC, Chen S, Cottrell TE, Daane KM, Fann LE, Fleischer SJ, Guédot C, Gut LJ, Hamilton GC, Hilton R et al (2020) Season-long monitoring of the brown marmorated stink bug (Hemiptera: Pentatomidae) throughout the United States using commercially available traps and lures. J Econ Entomol 113:159–171. https://doi.org/10.1093/jee/toz240
Adachi I, Uchino K, Mochizuki F (2007) Development of a pyramidal trap for monitoring fruit-piercing stink bugs baited with Plautia crossota stali (Hemiptera: Pentatomidae) aggregation pheromone. Appl Entomol Zool 42:425–431. https://doi.org/10.1303/aez.2007.425
Aldrich JR (1988) Chemical ecology of the Heteroptera. Annu Rev Entomol 33:211–238. https://doi.org/10.1146/annurev.en.33.010188.001235
Bergh JC, Morrison WR III, Joseph SV, Leskey TC (2017) Characterizing spring emergence of adult Halyomorpha halys using experimental overwintering shelters and commercial pheromone traps. Entomol Exp Appl 162:336–345. https://doi.org/10.1111/eea.12539
Brooks ME, Kristensen K, Van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Mächler M, Bolker BM (2017) GlmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400. https://doi.org/10.32614/RJ-2017-066
Endo N, Hironaka M (2017) Differences in the degree of attractiveness of different light sources to stink bugs. Kyushu Plant Prot Res 63:55–61
Faúndez EI, Rider DA (2017) The brown marmorated stink bug Halyomorpha halys (Stål, 1855) (Heteroptera: Pentatomidae) in Chile. Arq Entomol 17:305–307
Fisher JJ, Rijal JP, Zalom FG (2021) Temperature and humidity interact to influence brown marmorated stink bug (Hemiptera: Pentatomidae), survival. Environ Entomol 50:390–398. https://doi.org/10.1093/ee/nvaa146
Funayama K (2008) Seasonal fluctuations and physiological status of Halyomorpha halys (Stål) (Heteroptera: Pentatomidae) adults captured in traps baited with synthetic aggregation pheromone of Plautia crossota stali Scott (Heteroptera: Pentatomidae). Jpn J Appl Entomol Zool 52:69–75. https://doi.org/10.1303/jjaez.2008.69
Funayama K (2012) Control effect on the brown-marmorated stink bug, Halyomorpha halys (Hemiptera: Pentatomidae), by combined spraying of pyrethroid and neonicotinoid insecticides in apple orchards in northern Japan. Jpn J Appl Entomol Zool 47:75–78. https://doi.org/10.1007/s13355-011-0083-5
Haye T, Abdallah S, Gariepy T, Wyniger D (2014) Phenology, life table analysis and temperature requirements of the invasive brown marmorated stink bug, Halyomorpha halys, in Europe. J Pest Sci 87:407–418. https://doi.org/10.1007/s10340-014-0560-z
Jaffe BJ, Guédot C (2019) Vertical and temporal distribution of spotted-wing drosophila (Drosophila suzukii) and pollinators within cultivated raspberries. Pest Manag Sci 75:2188–2194. https://doi.org/10.1002/ps.5343
Johnson WD, Burton JH, Beyl RA, Romer JE (2015) A simple chi-square statistic for testing homogeneity of zero-inflated distributions. Open J Stat 5:483–493. https://doi.org/10.4236/ojs.2015.56050
Kamiyama MT, Bradford BZ, Groves RL, Guédot C (2020) Degree day models to forecast the seasonal phenology of Drosophila suzukii in tart cherry orchards in the Midwest U.S. PLoS One 15:e0227726. https://doi.org/10.1371/journal.pone.0227726
Kamiyama MT, Matsuura K, Yoshimura T, Yang CCS (2021) Improving invasive species management using predictive phenology models: an example from brown marmorated stink bug (Halyomorpha halys) in Japan. Pest Manag Sci 77:5489–5497. https://doi.org/10.1002/ps.6589
Khrimian A, Zhang A, Weber DC, Ho HY, Aldrich JR, Vermillion KE, Siegler MA, Shirali S, Guzman F, Leskey TC (2014) Discovery of the aggregation pheromone of the brown marmorated stink bug (Halyomorpha halys) through the creation of stereoisomeric libraries of 1-bisabolen-3-ols. J Nat Prod 77:1708–1717. https://doi.org/10.1021/np5003753
Kriticos DJ, Kean JM, Phillips CB, Senay SD, Acosta H, Haye T (2017) The potential global distribution of the brown marmorated stink bug, Halyomorpha halys, a critical threat to plant biosecurity. J Pest Sci 90:1033–1043. https://doi.org/10.1007/s10340-017-0869-5
Kuhar TP, Kamminga KL (2017) Review of the chemical control research on Halyomorpha halys in the USA. J Pest Sci 90:1021–1031. https://doi.org/10.1007/s10340-017-0859-7
Kyoto Prefecture Pest Control Center Website (2022) Outbreak situation of brown marmorated stink bug (prediction light). https://www.pref.kyoto.jp/byogai/kusagi.html. Accessed 10 Nov 2020
Lee DH, Leskey T (2015) Flight behavior of foraging and overwintering brown marmorated stink bug, Halyomorpha halys (Hemiptera: Pentatomidae). Bull Entomol Res 105:566–573. https://doi.org/10.1017/S0007485315000462
Lee K-C, Kang C-H, Lee DW, Lee SM, Park C-G, Choo HY (2002) Seasonal occurrence trends of hemipteran bug pests monitored by mercury light and aggregation pheromone traps in sweet persimmon orchards. Korean J Appl Entomol 41:233–238
Lee DH, Short BD, Joseph SV, Bergh JC, Leskey TC (2013) Review of the biology, ecology, and management of Halyomorpha halys (Hemiptera: Pentatomidae) in China, Japan, and the Republic of Korea. Environ Entomol 42:627–641. https://doi.org/10.1603/EN13006
Leskey TC, Short BD, Butler BR, Wright SE (2012) Impact of the invasive brown marmorated stink bug, Halyomorpha halys (Stål), in mid-Atlantic tree fruit orchards in the United States: case studies of commercial management. Psyche 2012:1–14. https://doi.org/10.1155/2012/535062
Leskey TC, Agnello A, Bergh JC, Dively GP, Hamilton GC, Jentsch P, Khrimian A, Krawczyk G, Kuhar TP, Lee D-H, Morrison WR III, Polk DF, Rodriguez-Saona C, Shearer PW, Short BD, Shrewsbury PM, Walgenbach JF, Weber DC, Welty C et al (2015) Attraction of the invasive Halyomorpha halys (Hemiptera: Pentatomidae) to traps baited with semiochemical stimuli across the United States. Environ Entomol 44:746–756. https://doi.org/10.1093/ee/nvv049
Leskey TC, Andrews H, Bády A, Benvenuto L, Bernardinelli I, Blaauw B, Paolo Bortolotti P, Bosco L, Di Bella E, Hamilton G, Kuhar T, Ludwick D, Maistrello L, Malossini G, Nannini R, Nixon LJ, Pasqualini E, Preti M, Short BD et al (2021) Refining pheromone lures for the invasive Halyomorpha halys (Hemiptera: Pentatomidae) through collaborative trials in the USA and Europe. J Econ Entomol 141:1666–1673. https://doi.org/10.1093/jee/toab088
Lowenstein DM, Walton VM (2018) Halyomorpha halys (Hemiptera: Pentatomidae) winter survival, feeding activity, and reproduction rates based on episodic cold shock and winter temperature regimes. J Econ Entomol 111:1210–1218. https://doi.org/10.1093/jee/toy093
Maistrello L, Vaccari G, Caruso S, Costi E, Bortolini S, Macavei L, Foca G, Ulrici A, Bortolotti PP, Nannini R, Casoli L, Fornaciari M, Mazzoli G, Dioli P (2017) Monitoring of the invasive Halyomorpha halys, a new key pest of fruit orchards in northern Italy. J Pest Sci 90:1231–1244. https://doi.org/10.1007/s10340-017-0896-2
Morrison WR III, Cullum JP, Leskey TC (2015) Evaluation of trap designs and deployment strategies for capturing Halyomorpha halys (Hemiptera: Pentatomidae). J Econ Entomol 108:1683–1692. https://doi.org/10.1093/jee/tov159
Morrison WR III, Park CG, Seo BY, Park YL, Kim HG, Rice KB, Lee DH, Leskey TC (2017a) Attraction of the invasive Halyomorpha halys in its native Asian range to traps baited with semiochemical stimuli. J Pest Sci 90:1205–1217. https://doi.org/10.1007/s10340-016-0816-x
Morrison WR III, Acebes-Doria A, Ogburn E, Kuhar TP, Walgenbach JF, Bergh JC, Nottingham L, Dimeglio A, Hipkins P, Leskey TC (2017b) Behavioral response of the brown marmorated stink bug (Hemiptera: Pentatomidae) to semiochemicals deployed inside and outside anthropogenic structures during the overwintering period. J Econ Entomol 110:1002–1009. https://doi.org/10.1093/jee/tox097
Muñoz-Adalia EJ, Ahmed J, Colinas C (2022) Microclimatic conditions drive summer flight phenology of Platypus cylindrus in managed cork oak stands. J Appl Entomol 146:964–974. https://doi.org/10.1111/jen.13025
Nielsen AL, Hamilton GC (2009) Seasonal occurrence and impact of Halyomorpha halys (Hemiptera: Pentatomidae) in tree fruit. J Econ Entomol 102:1133–1140. https://doi.org/10.1603/029.102.0335
Nielsen AL, Hamilton GC, Matadha D (2008) Developmental rate estimation and life table analysis for Halyomorpha halys (Hemiptera: Pentatomidae). Environ Entomol 37:348–355. https://doi.org/10.1093/ee/37.2.348
Nielsen AL, Hamilton GC, Shearer PW (2011) Seasonal phenology and monitoring of the non-native Halyomorpha halys (Hemiptera: Pentatomidae) in soybean. Environ Entomol 40:231–238. https://doi.org/10.1603/EN10187
Nielsen AL, Holmstrom K, Hamilton GC, Cambridge J, Ingerson-Mahar J (2013) Use of black light traps to monitor the abundance, spread, and flight behavior of Halyomorpha halys (Hemiptera: Pentatomidae). J Econ Entomol 106:1495–1502. https://doi.org/10.1603/EC12472
Nielsen AL, Chen S, Fleischer SJ (2016) Coupling developmental, physiology, photoperiod, and temperature to model phenology and dynamics of an invasive Heteropteran, Halyomorpha halys. Front Physiol 7:156. https://doi.org/10.3389/fphys.2016.00165
Pimentel D, Lach L, Zuniga R, Morrison D (2000) Environmental and economic costs of nonindigenous species in the United States. BioScience 50:53–65. https://doi.org/10.1641/0006-3568(2000)050[0053:EAECON]2.3.CO;2
R Core Team (2019) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rice KB, Bergh CJ, Bergmann EJ, Biddinger DJ, Dieckhoff C, Dively G, Fraser H, Gariepy T, Hamilton G, Haye T, Herbert A, Hoelmer K, Hooks CR, Jones A, Krawczyk G, Kuhar T, Martinson H, Mitchell W, Nielson AL et al (2014) Biology, ecology, and management of brown marmorated stink bug (Hemiptera: Pentatomidae). J Integr Pest Manag 5:A1−A13. https://doi.org/10.1603/IPM14002
Rice KB, Cullum JP, Wiman NG, Hilton R, Leskey TC (2017) Halyomorpha halys (Hemiptera: Pentatomidae) response to pyramid traps baited with attractive light and pheromonal stimuli. Fla Entomol 100:449–453. https://doi.org/10.1653/024.100.0207
Rice KB, Morrison WR III, Short BD, Acebes-Doria A, Bergh JC, Leskey TC (2018) Improved trap designs and retention mechanisms for Halyomorpha halys (Hemiptera: Pentatomidae). J Econ Entomol 111:2136–2142. https://doi.org/10.1093/jee/toy185
Rondoni G, Chierici E, Marchetti E, Nasi S, Ferrari R, Conti E (2022) Improved captures of the invasive brown marmorated stink bug, Halyomorpha halys, using a novel multimodal trap. Insects 13:527. https://doi.org/10.3390/insects13060527
Searle SR, Speed FM, Milliken GA (1980) Population marginal means in the linear model: an alternative to least squares means. J Am Stat Assoc 34:216–221. https://doi.org/10.2307/2684063
Shimoda M, Honda K-I (2013) Insect reactions to light and its applications to pest management. Jpn J Appl Entomol Zool 48:413–421. https://doi.org/10.1007/s13355-013-0219-x
Short BD, Khrimian A, Leskey TC (2017) Pheromone-based decision support tools for management of Halyomorpha halys in apple orchards: development of a trap-based treatment threshold. J Pest Sci 90:1191–1204. https://doi.org/10.1007/s10340-016-0812-1
Togo K, Iwasaki M (2013) Group comparisons involving zero-inflated count data in clinical trials. Jpn J Biom 34:53–66
Tsutsumi T (2003) Fruit tree stink bug-interesting ecology and clever prevention. Rural Culture Association, Tokyo
Venette R, Koch R (2009) IPM for invasive species. In: Radcliffe E, Hutchison W, Cancelado R (eds) Integrated pest management: concepts tactics strategies, and case studies. Cambridge University Press, New York
Weber DC, Leskey TC, Walsh GC, Khrimian A (2014) Synergy of aggregation pheromone with methyl (E,E,Z) -2,4,6-decatrienoate in attraction of Halyomorpha halys (Hemiptera: Pentatomidae). J Econ Entomol 107:1061–1068. https://doi.org/10.1603/EC13502
Weber DC, Morrison WR III, Khrimian A, Rice KB, Leskey TC, Rodriguez-Saona C, Nielsen AL, Blaauw BR (2017) Chemical ecology of Halyomorpha halys (Stål): discoveries and applications. J Pest Sci 90:989–1008. https://doi.org/10.3390/insects9030082
Wermelinger B, Wyniger D, Forster B (2008) First records of an invasive bug in Europe: Halyomorpha halys Stal (Heteroptera: Pentatomidae), a new pest on woody ornamentals and fruit trees? Bul Soc Entomol Suisse 81:1–8. https://doi.org/10.5169/seals-402954
Wood SN (2006) Generalized additive models: an introduction with R. Chapman and Hall/CRC, Boca Raton
Acknowledgments
We offer our appreciation and sympathy to the family, friends, and colleges of Dr. Tsuyoshi Yoshimura from Kyoto University, a co-author who passed away during this research. We also express thanks to chief researcher Shinichi Asai and the staff from the Kyoto Prefecture Agriculture, Forestry, and Fisheries Technology Center for providing the monitoring dataset.
Funding
This research was funded by the Japanese Government Monbukagakusho (MEXT) Scholarship and the Virginia Tech New Faculty Start-up Research Fund.
Author information
Authors and Affiliations
Author notes
Tsuyoshi Yoshimura is deceased. This paper is dedicated to his memory.
- Tsuyoshi Yoshimura
Contributions
MTK conceived and designed the research. MTK conducted the research and analyzed the data. MTK and CCSY wrote the manuscript. KM, TH, TY and CCYS secured funding for the research. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 95 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kamiyama, M.T., Matsuura, K., Hata, T. et al. Seasonal abundance and trap comparisons of the invasive brown marmorated stink bug Halyomorpha halys (Hemiptera: Pentatomidae) adults from its native region. Biologia 79, 1341–1349 (2024). https://doi.org/10.1007/s11756-024-01618-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-024-01618-4