Abstract
The purpose of the study was to pretreat fermented rooibos biomass with partially purified horseradish peroxidase (HRP) for lignin removal and to convert delignified biomass to soluble sugars through saccharification with a formulated holocellulolytic enzyme cocktail (HEC). HRP enzyme was extracted from the horseradish root tissue and was partially purified by membrane filters and characterised biochemically. HRP enzyme was used to pretreat the fermented rooibos biomass to remove lignin before hydrolysing it with the HEC. Our findings indicated that HRP is versatile because it displayed activity on guaiacol, 8-aminoquinoline, and decolourised methylene blue dye. HRP had a pH optimum of 4.5 and displayed a mesophilic temperature range. The kinetics studies indicated that HRP displayed a higher affinity towards guaiacol (Km= 0.082 mg/mL) followed by 8-aminoquinoline (Km= 0.221 mg/mL). However, the catalytic efficiency revealed that the enzyme hydrolysed guaiacol (63436.48 s− 1. mg/mL) and 8-aminoquinoline (59189.81 s− 1. mg/mL) efficiently. HRP pretreatment of rooibos biomass significantly removed lignin content and increased pores on the surface as visualised with SEM. FTIR validated the SEM results by showing reductions at 3324.81, 1615.16 and 1018.75 cm− 1, corresponding to crystalline cellulose, lignin and holocellulose regions, respectively. HRP pretreated biomass had the lowest crystallinity index of 11.2% compared to 20% of the control. HRP delignified rooibos biomass was hydrolysed effectively by the HEC, which released about 10% yield of soluble sugars compared to 6% of control. We conclude that HRP pretreatment significantly modified the structural and chemical properties of the biomass, making it more accessible to hydrolytic enzymes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Economic activities such as burning fossil fuels (coal, oil and gas) enhance the greenhouse effect leading to global warming. Hence, biofuel production from agricultural residues is one of the preferred fossil fuels because it can decrease carbon monoxide emissions (I.P.C.C 2007; Houghton 2005). Plants can use the emissions of carbon dioxide from the biofuel during photosynthesis resulting in an ecofriendly process known as the carbon cycle (I.P.C.C 2007). The biorefinery sector continues to investigate cheaper methods for producing secondary-generation biofuel and value-added products (VAPs) from agricultural residues, which have the potential to produce simple sugars that can be fermented into ethanol (Olsson and Hahn-Hagerdal 1996; Mafa et al. 2021). However, removing lignin from the biomass via ecofriendly means and using enzyme cocktails to degrade lignocellulose to simple sugars has been a challenge, resulting in a higher price for biofuel than petroleum production (Olsson and Hahn-Hagerdal 1996). These observations prompt more research that probes newer, efficient and environmentally friendly methods to remove lignin from lignocellulose or formations of enzyme cocktails with superior biomass conversion to fermentable sugars (Mafa et al. 2021; Van Dyk and Pletschke 2012).
Agricultural wastes such as rooibos (Aspalathus linearis) residues are carbon-rich biomass that can be used to produce second-generation biofuels without affecting food supply due to their high abundance (Kong et al. 2017; Malhotra and Suman 2021). Rooibos is part of the fynbos vegetation found in the Western Cape and Northern Cape Provinces of South Africa (Joubert and Schulz 2006; Lotter and Maitre 2014). It is one of the major drinks consumed as a ready-made tea (iced tea), or as a hot tea (Joubert and Schulz 2006). The production of iced tea results in tons of rooibos bagasse produced annually, and the manufacturing companies typically dispose of the waste by burning, dumping it into landfills or using it as compost (Kondo et al. 2007). We propose that some of the rooibos residues disposed of annually can be used to produce biofuel and VAPs through enzymatic degradation.
The physiological and biochemical features of the rooibos plant make it an attractive biomass source. Rooibos is a shrub (woody plant) that grows along the ground and can reach 2 m in length (Joubert and Schulz 2006; Lotter and le Maitre 2014; Pengilly et al. 2008) demonstrated that soluble sugars (e.g., xylose, glucose, arabinose and galactose) were easily extracted from rooibos samples by boiling in water. Compared to black tea (Camellia sinensis), rooibos contains lower tannin content, which forms part of lignin in most dicots (Joubert and de Beer 2011). Dossou-Yovo et al. (2021) study showed that rooibos contains about 45% soluble matter, 37% holocellulose, 9% soluble lignin, and 82% of the rooibos biomass is hydrolysable. Other studies argue that the holocellulose constitutes major content (Malhotra and Suman 2021), where cellulose forms 36–61%, and hemicellulose constitutes 13–39% (Olsson and Hahn-Hagerdal 1996; Calderan-Rodrigues et al. 2019).
Cellulose and hemicellulose are hydrolysed by glycoside hydrolases (GHs) and debranching enzymes to produce sugars that can be fermented to produce ethanol or VAP (Van Dyk and Pletschke 2012; Malgas et al. 2019; Mafa et al. 2021). However, the presence of lignin can lead to non-specific substrate-binding of holocellulolytic enzymes or inhibits the enzymatic degradation of cellulose and hemicellulose (Malhotra and Suman 2021; Kong et al. 2017). Lignin is a polymer comprised of randomly linked hydroxycinnamyl alcohols such as p-coumaryl alcohol, coniferyl alcohol, and sinapyl alcohol (Mnich et al. 2020). Due to its protective nature, lignin must be removed before the lignocellulosic biomass is hydrolysed through enzymatic application. Generally, lignin removal results in a higher enzymatic biomass conversion rate with a higher yield of soluble sugars. (Mnich et al. 2020; (Mafa et al. 2020a, b; Van Dyk and Pletschke 2012).
Most of the developed pretreatment methods used to remove lignin include chemicals (particularly acid pretreatments) which are expensive and produce toxic compounds, such as black liquor, that are detrimental to the environment and human health (Kong et al. 2017; Bagewadi et al. 2017). Additionally, they alter the lignin and hemicellulose structure during lignin degradation and produce inhibitory compounds, such as furfural and hydroxymethylfurfural, which impede enzymatic hydrolysis and the fermentation process (Kong et al. 2017; Harmsen et al. 2010). In contrast, the use of alkaline pretreatments, such as sodium hydroxide, is cost-effective compared to acid pretreatment. Alkaline pretreatments lower the degree of polymerisation (DP) and cellulose crystallinity, as a result, it increases the efficacy of the enzymes that hydrolyse the carbohydrates (Mafa et al. 2020a; Hendriks and Zeeman 2009). However, the limitation of alkaline pretreatment is that the method’s efficacy depends on the amount of lignin present in the biomass, and it can also hydrolyse hemicellulose content at higher concentration (Mafa et al. 2020a, b; Hendriks and Zeeman 2009). The biological approach for lignin removal (including the use of ligninase, peroxidase, laccase and carbohydrate esterase enzymes or microbes), is a cheaper alternative that is sustainable, environmentally friendly and effective without the loss of polysaccharides and the production of inhibitory compounds (Giacobbe et al. 2018).
The biological pre-treatments use enzymes produced by microorganisms and plants to remove lignin from the biomass. Enzymes that remove lignin are known as ligninolytic enzymes, which include lignin peroxidase (LiP, EC 1.11.1.14), manganese peroxidase (MnP, EC 1.11.1.13), versatile peroxidase EC 1.11.1.16), and dye decolourising peroxidase (DyP, EC 1.11.1.19) (Malhotra and Suman 2021; Biko et al. 2022). The current study will focus on the use of horseradish peroxidase to delignify rooibos bagasse. The peroxidases (EC 1.11.1.7) require H2O2 as a co-substrate to initiate the reaction for lignin degradation, and in the same reaction, H2O2 is converted to water (Sarika et al. 2015; Rad et al. 2007; Lavery et al. 2010; Liu et al. 2013). Ligninolytic enzymes, such as peroxidase from horseradish roots (Armoracia rusticana), are heme-containing enzymes used in immunohistochemistry, the treatment of waste containing phenolic compounds, removal of colour from waste or the removal of peroxide from industrial waste (Miranda and Cascone 1995; Lavery et al. 2010; Liu et al. 2013; Sarika et al. 2015). The need for cheaper biofuel and other VAPs produced from lignocellulosic material is the driving force behind the search for cheaper and eco-friendly de-lignification of the biomass. The aims of the study are to comprehensively characterise horseradish peroxidase (HRP) and use it to de-lignify rooibos biomass. Secondly, we wanted to formulate a holocellulolytic enzyme cocktail and apply it to the de-lignified rooibos to convert it to soluble sugar.
Materials and methods
Materials
Rooibos (Aspalathus linearis) samples were supplied by rooibos limited (Clanwilliam, Western Cape, South Africa). The horseradish root (Armoracia rusticana) was provided by Mr Barry Newton and Mr John Parr from Yaxham farm situated in Tweespruit, Free State, South Africa. The commercial cellulases and xylanase enzymes such as xylanase from Aspergillus oryza, endoglucanase1 (EG1) from Aspergillus niger, endoglucanase2 (EG2) from Aspergillus sp., β-glucosidase from Aspergillus niger and CBHI from Hypocrea jecorina were all purchased from Sigma (Johannesburg, South Africa). The commercial substrates, such as guaiacol, avicel, 8-aminoquinoline, glucuronoxylan and carboxymethyl cellulose sodium (CMC) were purchased from Sigma (Johannesburg, South Africa), whereas beechwood xylan and wheat arabinoxylan were purchased from Megazyme, (Wicklow, Ireland) and methylene blue dye was supplied by protea laboratories. All the analytical chemicals used in the study were purchased from Sigma (Johannesburg, South Africa), unless stated differently.
Extraction of horseradish peroxidase (HRP)
Horseradish peroxidase (HRP) was extracted from 300 g root tissue of horseradish (Armoracia rusticana). The root was chopped into 5 cm pieces, transferred into electric industrial blender and homogenised into a fine paste after adding 500 mL of 50 mM sodium phosphate buffer at pH 6. The homogenate was transferred into 50 mL falcon tubes and incubated on ice for 30 min. The ice-cool samples were filtered using a cheese cloth. The resulting supernatant was transferred into clean falcon tubes and allowed to sediment for 16 h at 4 °C. After 16 h, samples were centrifuged at 15 000 xg for 30 min, and supernatant was collected in a clean beaker, and then filtered using a 0.45 μm, white 47 mm gridded sterile (Millipore) filter. A volume of 400 mL of the filtrate (HRP) was collected in clean Falcon tubes and stored at 4 °C for further use.
HRP purification analysis
About 50 mL crude HRP was added to a 30 kDa, 20-100mL, 4pk Pierce™ protein concentrator PES (Thermoscientific, United Kingdom) for partial purification. The samples were centrifuged at 4 250 xg for 30 min, the flow-through and the retained samples were collected in 50 mL falcon tube. The protein concentration of the partially purified HRP enzyme was determined using Bradford’s method (Bradford 1976). The enzyme purity was determined with 12% (w/v) sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) according to Laemmli (1970). The proteins were loaded on stacking gel wells and separated for 2 h at 118 V. After separation, the gel was stained overnight using the Coomassie-staining solution (40% methanol, 10% acetic acid, and 0.025% Coomassie Brilliant Blue G250), and destained using a destaining solution (40% methanol and 10% glacial acetic acid).
HRP substrate specificity assay
HRP substrate specific assays were conducted using guaiacol, methylene blue and 8-aminoquinoline. About 1.5 mM of guaiacol substrate was dissolved in 50 mM sodium citrate buffer pH 4.5, and 0.5% (v/v) H2O2 and used to test HRP activity. Firstly, we varied HRP concentrations as follows, 1.6 µg/mL, 4.7 µg/mL, 7.8 µg/mL and 16 µg/mL but kept the concentration of the substrate constant. The kinetic absorbance was measured as the rate of the reaction for 3 min at 470 nm. Once the HRP activity on guaiacol was established, about 0.68 mg/mL of HRP was used to demethylate methylene blue dye by dissolving about 1.5 mM methylene blue dye in 50 mM sodium citrate buffer pH 4.5, which was added to HRP, and 0.5% (v/v) H2O2. The reactions were incubated for 16 h and the absorbance was measured at 666 nm. The third reaction to test the versatility of HRP was performed using 1.5 mM of 8-aminoquinoline dissolved in 50 mM sodium citrate buffer pH 4.5, then added to 4.7 µg/mL of HRP and 0.5% (v/v) H2O2. The reactions were incubated at 37ºC for 20 min and the absorbance was measured at 520 nm. All the reactions were performed in triplicate and absorbance readings were performed on an ultraviolet-visible spectrophotometer (Varian Cary 100 Bio UV-Visible). The extinction coefficient of guaiacol and 8-aminoquinoline were 26.6 mM .cm− 1 and 6.58 mol/L .cm− 1, respectively.
HRP biochemical characterization
Biochemical characterization studies were performed to determine the HRP pH optimum, pH stability and thermostability using guaiacol as a substrate. pH optimum studies were assayed using 1.5 mM guaiacol dissolved in different buffers, i.e., 50 mM sodium citrate buffer for pH 4 to 5.5; 50 mM sodium phosphate buffer for pH 6 to 7.5; 50 mM for Tri-HCL buffer for pH 8 to 9) and 4.7 µg/mL of the HRP and 0.5% (v/v) H2O2. For pH stability studies, the enzyme was incubated for 16 h in different buffers with varying pH, and the reactions were conducted as mentioned above. Similarly, for thermostability assays, the enzyme was incubated at 37, 50 and 70 °C for 1, 5, and 16 h to initiate the reaction as mentioned above. All reactions were conducted in triplicate, and the absorbance was measured at 470 nm.
HRP enzyme kinetics studies
The kinetics studies were conducted using 8-aminoquinoline and guaiacol substrates. HRP kinetic constants were determined using various concentrations (0.1, 0.25, 0.5, 0.75, 1, 1.25, 1.5 mM) of 8-aminoquinoline and guaiacol substrate. Each reaction in 1 mL consisted of the substrate dissolved in 50 mM sodium citrate buffer at pH 4.5, 4.7 µg/mL of the HRP-enzyme and 0.5% (v/v) H2O2. For 8-aminoquinoline, the reactions were incubated for 20 min at 37ºC, and the absorbance readings were taken at 520 nm. For guaiacol, the reactions were measured as the rate of the reaction at 470 nm. The reactions were carried out in triplicate and the readings were taken using a spectrophotometer (Varian Cary 100 Bio UV-Visible). After calculation, the reaction rate was converted to mg/mL for the determination of the Michaelis-Menten constant (KM), Vmax, catalytic constant (kcat) and catalytic efficiency using the Lineweaver-Burk plot and Solver analysis in Microsoft Excel (Version 2016).
Fermented rooibos biomass pretreatment with HRP for lignin removal
About 2.5 g of soluble sugar-free fermented rooibos samples were dissolved in 50 mM sodium citrate buffer pH 4.5. The lignin removal reaction was conducted by adding 0.5 mg/mL HRP-enzyme and 1% (v/v) H2O2 in the tubes containing the rooibos biomass. The reaction period was 24 h, and samples were incubated in the water bath set at 37 °C followed by mixing with vortex every 6 h. Additions of enzyme and H2O2 were performed in triplicate (at 0, 6 and 12 h incubation) in the reaction. There were two reaction controls that were performed in the same conditions as the HRP pretreatment reaction, except that first control contained rooibos biomass and buffer only, and the second control reaction contained rooibos biomass and 1% (v/v) H2O2 only. After the lignin removal reaction, the samples were centrifuged at 4 000 xg for 15 min and the supernatant was removed and frozen at -20℃ for later use, while the rooibos material was dried at 60 °C for 72 h and stored in the airtight container at room temperature for later use.
Rooibos biomass analysis with FTIR
The chemical (functional group) analysis of the untreated or pretreated fermented rooibos biomass samples was analysed with Fourier Transform Infrared (FTIR) Spectroscopy. The FTIR spectra of the fermented rooibos control (untreated), and H2O2 or HRP pretreated samples were measured at room temperature using an UATR-FTIR instrument (Thermo scientific, USA). All FTIR spectra were collected in absorbance mode at a spectrum resolution of 4 cm− 1, with 32 co-added scans per sample over the range of 4000 to 650 cm− 1. Peaks’ functional groups were assigned according to Mafa et al. (2020a, b).
X-ray diffraction (XRD) analysis of rooibos biomass
The XRD technique was used to determine the HRP-pretreatment effect on the crystallinity of the fermented rooibos biomass. Once again, 1% (v/v) H2O2 pretreated biomass and untreated rooibos biomass were used as controls. The Bruker diffractometer (USA) was used to do XRD using Cu Ka radiation at 40 kV and 130 mA at Coupled 2θ/Theta scanning angle and a speed of 0.5°/minute. Sample’s crystallinity index was calculated according to Park et al. (2010).
Determination of rooibos biomass topology with SEM
To determine the effect of HRP-pretreatment on rooibos biomass topology and the controls, completely dry samples were analysed with scanning electron microscopic (SEM). Samples were mounted on aluminium pin stubs using double sided carbon tape and coated with Iridium (± 10 nm) for conductivity using a Leica EM ACE600 coating system. Specimens were imaged at 5 kV using a JSM-7800 F Extreme-resolution Analytical Field Emission SEM (Tokyo, Japan). The samples with different magnification ranging between 100 and 1000 times were used to analyse the samples and photos were taken with a built-in camera to document the results.
The effect of HRP on microcrystalline cellulose
About 2.5 g of Avicel samples were dissolved in 50 mM sodium citrate buffer pH 4.5. The reaction was conducted as described in the lignin removal method to determine the effect of the HRP pretreatment on the microcrystalline cellulose. After 24-hour incubation, each of the sample was mixed by vortexing and placed in a falcon tube stand to determine the sedimentation rate according to Koskela et al. (2022) with modification. Photographs were taken every min for 10 min and after 10 min the distance of the sedimentation was measured in millimetre (mm). After the determination of the sedimentation, the samples were centrifuged at 4 000 x g for 15 min and the supernatant was removed and stored at -20℃ for later use, while the Avicel insoluble material was dried at 60 °C for 72 h and stored in an airtight container at room temperature for later use. The supernatant was used to determine the soluble reducing sugars using DNS method (Miller 1959). The readings were taken at 540 nm on an ultraviolet-visible spectrophotometer (Varian Cary 100 Bio UV-Visible).
Enzyme activity was also conducted using EG1 (from A. niger) and EG2 (from Aspergillus sp.), and β-glucosidase (from A. niger) and HRP pretreated Avicel was used a substrate. Each reaction assays for the cellulase activity consisted of about 1% (w/v) substrate (pretreated Avicel dissolved in 50 mM sodium phosphate buffer pH5) added to 0.143 mg/mL for EG1, 0.33 mg/mL for EG2, and 0.25 mg/mL for β-glucosidase of the enzymes. All the reactions were incubated at 37ºC for about 2 h. After the completion of the reactions, samples were centrifuged at 5 000 x g and total reducing sugars were also detected with the DNS method.
Holocellulolytic enzymes substrate specificity assay
Substrate specific assays were conducted using wheat arabinoxylan (WAX), beechwood xylan (BWX), which represent the hemicellulosic content of lignocellulose; and carboxymethyl cellulose sodium (CMC) represented the amorphous cellulose content, while microcrystalline cellulose was represented by Avicel or filter paper. Each reaction assay for the xylanase activity consisted of 1% (w/v) WAX and BWX dissolved in 50 mM sodium phosphate buffer pH 6 and 0.143 mg/mL of xylanase and EG1, or 0.246 mg/mL of EG2. In the case of cellulase two experimental regimes were used; for the CHBI activity, about 0.024 mg/mL of enzyme was added to 1% (w/v) Avicel or 3 discs of filter paper hydrated in 50 mM sodium phosphate buffer pH6, and endoglucanase activities were assayed using 1% (w/v) CMC dissolved in 50 mM sodium phosphate buffer at pH6. All the reactions were incubated for an hour at 37ºC, except for the reaction where enzymes degraded Avicel and filter paper, the reactions were incubated for about 5 h. All the experiments were conducted in triplicate. The enzyme activity was measured using the DNS method as mentioned in the previous section.
Formulation of the cellulolytic cocktail
The most effective endoglucanase enzyme core set was determined using 1% (w/v) of CMC or 3-discs of filter paper dissolved in 50 mM sodium citrate buffer pH 5 and enzyme-loading combinations (100, 75, 50, 25, 0%) of EG1 and EG2 were kept at constant concentration of 0.143 mg/mL. The reactions contained 1% (w/v) CMC, 0.143 mg/mL enzyme (EG1 and EG2 combinations) and 0.0143 of β-glucosidase, and the reactions were incubated for an hour at 37ºC. After the completion of the reactions, reducing sugars were detected using the DNS method. The most effective EG1:EG2 combination (i.e., 25%:75%) was further used in combination with CBHI to determine the cellulolytic cocktail using 1% (w/v) Avicel or 3-discs of filter paper dissolved in 50 mM sodium citrate buffer pH5. The experimental setup was similar to the one used to develop the most effective EG1:EG2 core sets.
Application of the holocellulolytic enzyme cocktail on the delignified rooibos biomass
The cellulolytic enzyme cocktails (consisting of EG1, EG2, CBHI and β-glucosidase) did not show a superior hydrolytic effect compared to EG1, EG2 and β-glucosidase. As a result, the holocellulolytic enzyme cocktail was formulated by investigating the additive effect of xylanase on the most effective EG1:EG2 enzyme core set. The xylanase additive effect was determined using 1% (w/v) of the de-lignified or control rooibos biomass that was hydrated by 50 mM sodium citrate buffer at pH 5. The best EG1:EG2 (25%:75%) was used at a constant concentration of 0.143 mg/mL and 0.0143 mg/mL β-glucosidase, while the xylanase concentration varied at 0.1, 0.5, 0.75 and 1 mg/mL. The reactions were incubated for 24 h at 37ºC. All experiments were performed in triplicate. After the completion of the reactions, reducing sugars were detected using the DNS method as described in the effect of HRP on microcrystalline cellulose section.
Data analysis
All the data (excluding the SDS-page and sedimentation data) was analysed in Excel and used to generate graphs and tables. One-way ANOVAs was used to calculate the significance differences between treatments where necessary. Solver was used to determine the kinetic constants of the HRP enzyme.
Results
HRP partial purification and substrate specificity
Post extraction of HRP from the root of horseradish plant, the enzyme was partially purified and concentrated. The SDS-PAGE analysis showed intense bands with a molecular weight of 44, 48 and 66 kDa (Fig. 1). These bands were visible in the crude HRP samples, partially pure HRP and the dialyzed samples. It is important to note that after dialyzing the concentrated partially pure HRP samples, a few of the bands between the 48 and 60 kDa and a few bands above 60 kDa disappeared from the gel. These observations indicated that we partially purified the HRP enzyme which was used in the follow-up studies.
Determination of HRP partial purity with SDS-PAGE. Lane 1 (PC) represents the protein standard, lane 2 represents crude protein extracted from the horseradish root, lane 3 represents HRP enzyme partially purified with 30 kDa cut-off filters and Lane 4 shows the profile of a further dialyzed partially pure HRP
Substrate specificity assays were conducted to determine the partially purified HRP enzymes’ activities. The enzymes showed extremely high activity on the guaiacol substrate- as a result, this substrate was used to determine the correct HRP concentration for further analysis. The results showed that 4.7 µg/mL concentration of the HRP was sufficient as it did not convert guaiacol to tetraguaiacol more rapidly as did 16 µg/mL (Fig. 2a). So, a concentration of 4.7 µg/mL was used for downstream analysis and to test the HRP activity on one other substrate (Fig. 2c). The enzyme decolourised the methylene blue dye by significantly reducing the absorbance values (Fig. 2b). Also, the HRP enzyme efficiently converted 8-aminoquinoline to form a quinoline dye (Fig. 2c), and the activity of the HRP enzyme was significantly different from when H2O2 was present alone. The assays showed that HRP is a versatile enzyme because it had activity on all three tested substrates.
Represents the activity of the peroxidase enzyme on, a guaiacol substrate at different enzyme concentrations, b on Methylene Blue dye and c on 8 aminoquinoline. The values represents the means ± standard deviation. The * represents significant differences between enzyme concentrations or treatments detected with ANOVA (p < 0.05)
Biochemical properties and kinetics parameters of the HRP enzyme
Guaiacol is a model substrate for peroxidase and it was used to optimize the HRP enzyme biochemical conditions. HRP displayed a pH optimum at 4.5 (Table 1), however at pH 4 and 5.5 the enzyme displayed more than 80% relative activity indicating that the enzyme favoured acidic conditions but gradually lost activity as the conditions became more basic. For pH stability studies, the enzyme still demonstrated high activity at pH 4.5, while it lost between 30 and 40% of its activity at pH 4, and at pH 5 to 7, and lost more than 50% of its activity in the basic range. Thus pH 4.5 was taken as the optimum pH condition for the HRP-enzyme.
The thermostability studies for HRP was conducted by incubated the enzyme at 37, 50, and 70 °C for 1, 5 and 16 h, respectively. The enzyme had high activity at 37 °C for all three-time points (Table 1), and at 50 °C, it had an activity of more than 80% after 1 h of incubation, but gradually lost activity at 50 °C after 5 h incubation followed by 16 h incubation. At 70 °C the enzyme lost activity at all the time points. Thus, 37 °C was taken as the temperature at which the HRP enzymes showed optimal activity.
Kinetics studies showed the rate at which HRP catalysed guaiacol and 8-aminoquinoline substrate into products, as well as HRP’s affinity towards the substrates. The Km values were 0.082 and 0.221 mg/mL for guaiacol and 8-aminoquinoline substrates, respectively (Table 1). These results indicate that HRP enzyme has a higher affinity toward guaiacol compared to 8-aminoquinoline. However, the enzyme displayed a higher Vmax (1.022 mg/mL.min − 1) when it catalyse 8-amniquinoline, which was 3.5 fold higher than the Vmax (0.404 mg/mL.min − 1) it displayed during the catalysis of guaiacol. kcat values of 5185.45 s− 1 for guaiacol and 13096.86 s− 1 for 8-aminoquinoline were recorded. The HRP displayed a catalytic efficiency value of about 63436.48 s− 1. mg/mL for guaiacol and 59189.81 s− 1. mg/mL for 8-aminoquinoline. The catalytic efficiency values were higher when HRP catalysed guaiacol, compared to when it catalysed 8-aminoquinoline. The results showed that HRP enzyme has a higher affinity for guaiacol compared to 8-aminoquinoline, but the enzymes catalysed guaiacol and 8-aminoquinoline efficiently.
Structural, and physicochemical characterization of HRP-treated rooibos biomass
After confirming the substrate specificity, optimum biochemical properties and kinetics parameters for the HRP enzyme, it was used to delignify the rooibos biomass. SEM topological studies were employed to determine the lignin removal efficacy of the HRP enzyme. The control (the untreated rooibos biomass) had a rough surface with a thick layer of lignin covering cellulose and hemicellulose of fermented rooibos, which was visible under 300x and 800x magnification (Fig. 3). The biomass treated with 1% (v/v) H2O2, had a smoother surface that still contained pieces of the lignin layer visible on the biomass. Also, a few pores appeared on the biomass after H2O2 pre-treatment. Interestingly, the biomass treated with HRP showed a significant removal of lignin layer from the fermented rooibos biomass, such that the fibers (holocellulose) were exposed, suggesting that more than 80% of the lignin layer was removed or catalysed by HRP to phenolic compounds. Lastly, the HRP-treated rooibos biomass was more porous compared to the control and H2O2 pretreatment. Increased, pores on the HRP-treated rooibos surface could grant hydrolytic enzymes more access to the biomass, which could lead to increased soluble sugars production.
Changes in the chemical functional groups in the untreated or pretreated fermented rooibos biomass samples were analysed with FTIR. The FTIR results were comparable to the SEM findings because it was clear from the FTIR analysis that HRP pretreatment had an effect on the structural and chemical features of the rooibos biomass (Fig. 4). The control (untreated) biomass showed a high peak at 3324.81 cm− 1 (red asterisk) which represent the OH groups, inter- and intra-hydrogen bonds found in the microcrystalline cellulose region of the biomass. The rooibos biomass pretreated with H2O2 also had a high peak at same wavenumber as untreated samples, while the HRP pretreated samples showed a significant decrease in the absorbance at 3324.81 cm− 1. The observations suggest untreated and H2O2 pretreated biomass had higher crystallinity compared to HRP-treated biomass. In addition, the control (untreated) biomass showed highest peak at the phenolic content range (1615.16 cm− 1), indicating the substantial presence of lignin in the samples. The second higher peak at phenolic content wavenumber was that of H2O2, followed by HRP pretreated biomass with significantly reduced peak in the phenolic content region compared to the control (yellow asterisk above peak 1615.16 in Fig. 4). Once again, the results indicated that HRP pretreatment had a significant effect by removing the lignin from the biomass. There was a minimal difference between control and H2O2 pretreated biomass at the holocellulose region (1018.75 cm− 1). However, the HRP pretreated biomass had a reduced holocellulose peak at 1018.75 cm− 1 (green asterisk in Fig. 4). The FTIR findings reveals that HRP-enzyme treatment had significantly modified the chemical and structural features of fermented rooibos biomass.
Effects of horseradish peroxidase (HRP) enzyme on the chemical functional groups of fermented rooibos samples using FTIR analysis. The HRP-pretreated rooibos FTIR spectrum was compared to that of the control and H2O2 pretreated biomass. Three regions of interest, OH group region, phenolics/lignin region and glycosidic bonds/holocellulose regions are represented by red, yellow and green asterik, respectively
The SEM and FTIR findings were supported by XRD analysis of the HRP-pretreated rooibos biomass. The untreated biomass displayed a higher crystallinity index of 20.4%, while the H2O2 pretreated biomass had a reduction of 3.2% less crystallinity index than the control (Fig. 5). However, the HRP pretreated biomass showed that the pretreatment had an effect on the microcrystalline cellulose as it led to a reduction of 9.2% in the crystallinity index. Taken together, the SEM, FTIR and XRD analysis demonstrated that the HRP-pretreatment significantly modified structural and chemical features of the rooibos biomass.
The effect of HRP on microcrystalline cellulose
The significant decrease in the crystallinity, lignin and holocellulose content after HRP pretreatment (shown by SEM, FTIR and XRD analysis), suggests that the HRP-enzyme hydrolysed the lignin and holocellulose content. As a result, we further investigated the effects of HRP enzyme on the microcrystalline cellulose using Avicel as a suitable substrate. The reducing sugars produced during the Avicel pretreament was determined using the hydrolysate (supernatant) collected after the 24 h reaction. Figure 6 showed that HRP pretreatment produced the highest levels of reducing sugars content compared to the control and H2O2 pretreatment, which had residual activity. The HRP application produced 6 fold higher total reducing sugars compared to the H2O2 pretreated sample; and 10 fold higher total reducing sugars compared to the control. The residual total reducing sugars detected in the control samples and H2O2 treated samples could be due to auto-hydrolysis of avicel substrate during incubation at 37℃ for 24 h.
The reducing sugars produced by horseradish peroxidase (HRP) during Avicel pretreatment. The untreated and H2O2 pretreatment samples were used to compare the HRP activity on Avicel substrate. The values represents the means ± standard deviation. The * represents significant differences between enzyme concentrations or treatments detected with ANOVA (p < 0.05)
Additionally, sedimentation studies were performed to validate the HRP enzyme activity on the Avicel substrate. Figure 7 demonstrates that the rate of Avicel sedimentation after the HRP-pretreament reaction was significantly slower compared to the control (untreated) and H2O2 pretreated Avicel biomass. Avicel sedimented rapidly in the control leaving a clear liquid on top measuring 1.6 cm and a similar response was observed in the H2O2 Avicel with the clear liquid measuring 1.7 cm after 10 min. Interestingly, the HRP pretreated Avicel had a slow sedimentation rate, resulting in a sedimentation distance that was not significantly different between 3, 5 and 7 min (Fig. 7b to d). Only after 10 min sedimentation distance was about 0.9 cm, suggesting that the HRP-pretreatment had an effect on the structural and physicochemical properties of cellulose.
EG1, EG2 and β-glucosidase hydrolysis assays were used to determine the impact of HRP pretreatment on the structural and chemical properties of Avicel. The EG1 and EG2 showed high activity in the HRP pretreated Avicel compared to control (untreated) and H2O2 pretreated samples (Fig. 8). After HRP pretreatment EG1 showed more than 2 fold activity on Avicel compared to control and H2O2 treated samples, respectively. EG2 displayed about 1.2 fold higher activity when it hydrolysed the HRP-treat samples compared to control and H2O2 treated Avicel. EGs are known to show little-to-no activity on Avicel, due to its crystalline nature, which makes it recalcitrant to enzymatic hydrolysis. However, the findings reveals that HRP-treatment had an effect on reducing the recalcitrance of Avicel, resulting in the increased EGs activities on the modified Avicel. β-glucosidase displayed low activity as expected, because it usually hydrolyse disaccharides or shorted oligosaccharides (with a DP less than 5) into monosaccharides, indicating that the polymers produced during HRP pretreatment had higher degrees of polymerisation (long oligosaccharides).
The effects of HRP-pretreatment on the microcrystalline cellulose substrate (Avicel) measured as a function of endoglucanase-1 (EG1), Endoglucanase-2 (EG2) or β-glucosidase. The values represents the means ± standard deviation.The U represent units, which is equal to µmol.h-1. The * represents significant differences of EGs activity on pretreated Avicel; detected with ANOVA (p < 0.05)
Substrate specificity of holocellulolytic enzymes
Before the formulation of the holocellulolytic enzyme cocktail for hydrolysis of rooibos biomass, we determined the specific activities of commercial EG1, EG2, CBH and xylanase. Figure 9a shows that xylanase enzyme had higher activity on BWX followed by WAX and GX. EG1 enzyme also displayed a higher activity on BWX and had the same amount of activity on WAX and GX. It is important to note that EG1 displayed relatively similar results on all xylan substrates, while EG2 displayed 3 fold lower activity on xylan substrates compared to the xylanase (Fig. 9a). Cellulolytic substrate specificity revealed that CBHI had the overall highest activity on both substrates that represents the microcrystalline cellulose. It showed more than 3 fold higher activity on filter paper and Avicel compared to the activity of EG1, EG2, and xylanase. While CBHI has the overall highest activity on CMC (Fig. 9b). Both endoglucases exhibited the second highest activity on CMC, compared to xylanase.
Substrate specific activity of the cellulases and xylanases on the xylan and cellulose substrates. In a xylanase, endoglucanase-1 (EG1) and endoglucanase-2 (EG2) activity tested on Beechwood-xylan (BWX), wheat arabinoxylan (WAX) and glucuronoxylan (GX) substrates. In b, xylanase, CBHI, EG1 and EG2 activity was tested on Avicel, filter paper and carboxymethyl cellulose sodium (CMC) substrates. The U represent units, which is equal to µmol.h− 1
Endoglucanase binary synergy
The most effective endoglucanase combination was determined by using varying combinations of the EG enzymes. β-glucosidase was applied at 10% (v/v) relative to the total concentration of the EGs. The most effective combination of EGs was 25% EG1 and 75% EG2, as this combination had a significantly (p < 0.05) higher specific activity compared to other combinations (Fig. 10). The next best combination of EGs that had significantly higher activity was 50% EG1 and 50% EG2. The enzyme combination 25% EG1 and 75% EG2 was selected for the formulation of the cellulase cocktail.
Cellulase enzyme cocktail
The most effective cellulase combination was formulated by using varying combinations of the selected endoglucanase enzyme core-set and exoglucanase (CBHI) as shown in Fig. 11. The concentration of the enzymes was kept constant at 0.84 mg/mL and β-glucosidase application concentration was 0.084 mg/mL. The endoglucanase enzyme core set dosed at 100% showed significantly (p < 0.05) higher activity compared to any CBHI and EGs combinations (Fig. 11). The next best combination was a 75% endoglucanase and 25% exoglucanase combination, which showed second significantly (p < 0.05) higher activity (Fig. 11). However, we anticipated that the best activity would be observed where all three cellulase enzymes were combined.
Formulation of the cellulase cocktail composed of two endoglucanases (EG1 and EG2) and exoglucanase (Exo). All experiments were performed in triplicate, the values and error bars represents the means ± SD, respectively. The U represent units, which is equal to µmol.h− 1. The * represents significant differences of EGs and exoglucanase combination on CMC or filter papers detected with ANOVA (p < 0.05)
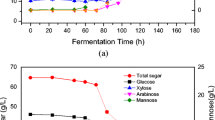
Additive effect of xylanase on the rooibos biomass hydrolysis
The additive effect of xylanase enzyme on enhancing the cellulase cocktail activity was tested using rooibos biomass. The final volume and the concentration of the endoglucanase combination was kept constant at 1 mg/mL, and the substrate loading at 1% (w/v), respectively. The fermented rooibos biomass was treated with peroxidase enzyme to remove the lignin content, while the control substrate was only subjected to buffer without peroxidase. Xylanase enzyme was used by increasing the concentration from 0.1 to 1 mg/mL. Figure 12a shows the combined activity of the selected EG enzyme cocktail and xylanase was significantly higher (p < 0.5) at 0.5 mg/mL xylanase enzyme loading. Thereafter, the activity did not change, indicating that 0.5 mg/mL xylanase enzyme loading was an ideal concentration to have maximum effect on hydrolysis of the rooibos biomass. However, the fermented biomass treated with peroxidase had the overall highest activity, which may give an indication that lignin was indeed removed increasing the efficiency of enzyme hydrolysis. Another indication that the removal of lignin increased the efficiency of the enzymes is displayed in Fig. 12b, where the yield produced in the biomass treated with peroxidase was higher by 10% compared to the 6% in the control. It also indicated that 0.5 mg/mL of xylanase, in addition to the endoglucanase cocktail, is required in order to produce more sugar products.
The additive effect of xylanase on the endoglucanase cocktail activity (a) and reducing sugar yield (b) during fermented rooibos biomass hydrolysis. The values and error bars represents the means ± SD, respectively. The * represents significant differences of holocellulolytic enzyme cocktail activity on fermented rooibos detected with ANOVA (p < 0.05)
Discussion
The partially pure horseradish peroxidase contained bands with molecular weights of 44, 48 and 66 kDa, which were similarly to molecular weight of horseradish peroxidase reported by Paul and Stigbrand (1970). Peroxidase from sycamore maple and peanuts contained bands of 42 and 40–42 kDa molecular weight, respectively (Dean et al. 1994; Chibbar and Van Huystee 1984). Duarte-Vázquez et al. (2007) reported that peroxidases typically sourced from plants have a molecular weight of between 30 and 60 kDa. These studies suggest that the three intense band observed on the SDS-PAGE in our study could be isozymes of horseradish peroxidase. In addition, the partially pure HRP showed versatility as it hydrolysed three tested substrates. The enzyme in the presence of the co-substrate H2O2 converted the model substrate guaiacol to tetra-guaiacol, which is measured as the change in absorbance overtime (Fig. SI1), similar to the HRP enzyme reported by Lavery et al. (2010). The enzyme also acts as a decolourizing agent by removing colour from methylene blue dye. Bholay et al. (2012) proposed that bacterial peroxidase with decolourizing activity showed lignin modifying activity. The 8-aminoquinoline is mainly used as an antimalaria agent (Strother et al. 1981), however, it is similar to the 4-aminoantipyrine-phenol substrate that acts as a hydrogen donor in the presence of hydrogen peroxide and HRP, resulting in the production of the quinone chromogen. Our studies also found 8-aminoquinoline was hydrolysed by HRP enzyme to produce quinoline dye (Fig. SI2), giving an intense pink colour formation (Sarika et al. 2015). We propose that 44, 48 and 66 kDa protein bands on the SDS-PAGE gel could be the HRP-isozymes, which contribute to the versatile nature of this enzyme. We are conducting follow-up studies to identify and verify if this protein band are isozyme of the HRP.
Biochemical studies showed that the HRP pH optimum was 4.5, which is within the pH range of peroxidases reported in literature; for instance, date-palm peroxidases showed an optimum pH at pH 4.5 (Al-Bagmi et al. 2019), while other peroxidases have optimum pH from 4 to 6 (Sarika et al. 2015; Biko et al. 2022). The HRP enzyme is not thermostable, because the enzyme lost activity at 50 and 70℃ within one hour of incubation period and was only stable at 37℃. However, peroxidase from Moringa oleifera L. leaves was stable at 55 °C (Khatun et al. 2012). The Km value represents the affinity or possible degree at which an enzyme tends to bind to a substrate. HRP displayed higher affinity for guaiacol compared to 8-aminoquinoline, but catalytic efficiency was relatively higher in both substrates. These implies that the HRP is effective in producing more products at a faster rate during catalysis of both substrates (Sulyman et al. 2020).
After successfully characterising HRP and confirming its versatility in hydrolysing three substrates, we used it to delignify fermented rooibos biomass. The SEM results showed the efficacy of HRP pretreatment in removing lignin from the fermented rooibos biomass. The HRP pretreatment removed the thick rough layer of lignin, exposing the pores and fibers of the biomass, which generally increase the area accessible to enzymes for hydrolysis. Similar studies on the pretreatment of corn stover with lignin degrading fungi and isolated Trametes hirsuta yj9, showed that the pretreatments removed lignin and increased the biomass conversion to simple sugar (Su et al. 2018; Sun et al. 2011). Liu et al. (2022) also used bactetrial (Raoultella ornithinolytica RS-1) pretreatment to remove the lignin from the the corn stover. It was shown that the RS-1 strain was successful in removing lignin because it secreted lignin-peroxidase, manganese-peroxidase and unclassified peroxidase (Liu et al. 2022). The studies validated our SEM findings, which showed that HRP-pretreatment modified or removed the lignin from rooiboss biomass.
The FTIR results validated the claim that HRP pretreatment removed lignin from the rooibos biomass. Liu et al. (2022) also found that biological pretreatment of the corn stover decreased the peak at 1630 cm− 1, which corresponded to the C = O stretching vibration of aromatic rings in lignin. Our findings also showed a simily trand because the HRP pretreated rooibos biomass had a significant decrease at 1625 cm− 1. At 1718 cm− 1 corresponding to ester bonds (lignin hemicellulose conjugation region) peak slightly increase in the HRP pretreated rooiboss biomass compared to H2O2 control, suggesting that the products of lignin degradation increased. In addition, the pretreatment of rice straw with renewable bioionic liquids showed a decrease in the phenolic content, indicating the removal of lignin (Hou et al. 2013). Additionally, the FTIR analysis show the broad peak between 3000 cm− 1 and 3500 cm− 1 corresponding to OH stretching vibration and intra/inter hydrogen bonds was significantly reduced in the HRP-treated biomass. The region (3000–3500 cm− 1) is generally assigned to microcrystalline cellulose (Mafa et al. 2020a, b), so the decreased peak at this region indicate that HRP treatment modified the microcrystalline cellulose of rooibos biomass. The XRD results confirmed a decrease in the crystallinity of cellulose due to the HRP pretreament. Other studies also used FTIR analysis to show that pretreatment of corn stover with lignin degrading fungi or bacteria had an increased cellulose and hemicellulose content, and XRD analysis displayed a reduction in the crystallinity of cellulose (Su et al. 2018; Lui et al. 2022).
The SEM, FTIR and XRD results indicated that the HRP pre-treatment also hydrolysed the holocellulose content of the rooibos. This observation implies that the HRP enzyme also possesses lytic polysaccharide monooxygenase-like (LPMO-like) activity, which catalyse the holocellulose content (cellulose or hemicellulose) of the biomass (Walton and Davies 2016; Meier et al. 2018). Further analysis were conducted by pretreating microcrystalline model substrate (Avicel) with the HRP enzyme. The results showed that the hydrolysate from the pretreatment contained reducing sugars produced by the HRP enzyme. Additionally, the sedimentation study showed that HRP pretreatment modified the structure of Avicel. We propose that HRP decreased recalcitrance of cellulose towards enzymatic hydrolysis because after HRP-pretreatment the Avicel was effectively hydrolysed by endoglucanases (Fig. SI3). Eijsink et al. (2019) argued that LPMOs requires the presence of a reductant (e.g., H2O2) to catalyse recalcitrant polysaccharides to produce oxidised oligosaccharides. The authors detected both reducing sugars and oxidised sugars, and the presences of reducing sugars was attributed to contamination. Interestingly, manganese peroxidase sourced from Phanerochaete chrysosporium that degraded lignin from the biomass, could also produce high amounts of soluble reducing sugars froms CMC, Avicel and xylan substrates (Min et al. 2022). The manganese peroxidase from Min et al. (2022) and HRP used in our study have similar properties such as, LPMO-like activity, cellulase activity boosting effects and reduced the recalcitrance of Avicel. We are conducting additional studies to establish the LPMO mechanism employed by the HRP enzyme during cellulose hydrolysis.
The HRP-pretreated rooibos biomass was hydrolysed by the holocellulolytic enzyme cocktail. Commercial enzymes used to formulate the enzyme cocktail were analysed with binary synergy assays (for endoglucanases) and ternary synergy assays as described by (Mafa et al. 2021 and Thoresen et al. 2021). Synergy studies of the two endoglucanases were used to formulate the best endoglucanase enzyme core set. The endoglucanase enzymes hydrolyse glycosidic bonds in the amorphous region of cellulose, while the exoglucanase (CBHI) enzyme hydrolyses glycosidic bonds from the reducing-end region of the microcrystalline cellulose (Van Dyk and Pletschke 2012; Zhang and Zhang 2013, Mafa et al. 2021). Thus, CMC was used as the substrate for endoglucanases synergy studies, which demonstrated that the best endoglucanase combination was 25% EG1 and 75% EG2. This EGs combination was used along with CBHI to formulate the cellulase cocktail. The results were not expected because the EGs combination at 100% enzyme dose had higher activity than the combinations of EGs and CBHI on Avicel or filter paper. Thoresen et al. (2021) demonstrated that CBHs and endoglucanases do not always display synergy. As a result, some EG and CBH combinations don’t produce higher concentration of soluble sugars, the phenomena is called anti-synergy (Thoresen et al. 2021; Mafa et al. 2021). Therefore, the best EGs combination were used to convert the HRP de-lignified rooibos biomass.
EGs combination was supplemented with various concentrations of xylanase and the enzyme cocktail was used to hydrolyze the delignified rooibos. The removal of lignin allowed the holocellulolytic enzyme cocktail to convert the rooibos biomass to soluble sugars better than control. In addition, the holocellulolytic enzyme cocktail yielded 10% soluble sugars during the hydrolysis of delignified rooibos biomass which about 2 fold higher compare to the yield of soluble sugars produced from the control. Eijsink et al. (2019) demonstrated that the use of LPMOs together with commercial cellulases enzymes resulted in a 10% yield of glucose produced from the lignocellulose biomass. The manganise peroxidase had boosting effects on the cellulase activity that degraded CMC, such that about 75% of CMC was converted to glucose (Min et al. 2022). In addition, a bacterial strain that secreted three types of peroxidases was used to pretreating the corn stover and thereafter, used commercial cellulase and β-glucose to convert 20% of pretreated corn stover to soluble sugars (Liu et al. 2022). These studies support our observation that the HRP pretreated rooibos biomass was efficiently degraded by the holocellulolytic enzyme cocktail.
Conclusion
Horseradish peroxidase (HRP) is a versatile enzyme with activity on multiple substrates. It also displays LPMO-like activity that produces significant amounts of reducing sugars from Avicel. Additionally, the LPMO-like activity of HRP decreased Avicel’s recalcitrance against endoglucanase enzymes - as a result, endoglucanases showed higher activity on HRP-treated Avicel. Furthermore, SEM analysis showed that HRP pre-treated fermented rooibos biomass removed lignin, increased pores on the biomass surfaces, and there were visible signs that it degraded holocellulose content. To the best of our knowledge, this is the second study reporting on versatile peroxidase that possesses LPMO-like activity and can remove lignin from lignocellulose substrate after Min et al. (2022). We are working on elucidating the LPMO mechanisms employed by HRP to produce reducing sugars from microcrystalline cellulose. The efficacy of HRP pretreatment on rooibos biomass was confirmed by FTIR and XRD analysis, which showed a decreased crystallinity, reduced holocellulose content, and a reduction in lignin from fermented rooibos biomass. Finally, saccharification of HRP pretreatment rooibos biomass with holocellulolytic enzyme cocktail released more reducing sugars and resulted in about a 10% yield compared to the control. Thus, we recommend the use of HRP pretreatment over chemical pretreatment because it removes lignin and reduces the crystallinity of lignocellulosic biomass.
Data availability
Available upon request.
Abbreviations
- BWX:
-
Beechwood xylan
- CBHI:
-
Cellobiohydrolase-I
- CMC:
-
Carboxymethyl cellulose sodium
- DNS:
-
Dinitrosalicylic acid reagent
- DP:
-
Degree of polymerisation
- Dyp:
-
Dye decolourising peroxidase
- EG1:
-
Endoglucanase-1
- EG2:
-
Endoglucanase-2
- EGs:
-
Endoglucanases
- FTIR:
-
Fourier Transform Infrared Spectroscopy
- H2O2 :
-
Hydrogen peroxide
- HRP:
-
Horseradish peroxidase
- I.P.C.C:
-
Intergovernmental Panel on Climate Change
- LiP:
-
lignin peroxidase
- LPMO:
-
Lytic polysaccharide monooxygenase
- MnP:
-
Manganese peroxidase
- SDS-PAGE:
-
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis
- SEM:
-
Scanning electron microscope
- VAPs:
-
Value-added products
- WAX:
-
Wheat arabinoxylan
- XRD:
-
X-ray diffraction
References
Al-Bagmi MS, Khan MS, Ismael MA, Al-Senaidy AM, Bacha AB, Husain FM, Alamery SF (2019) An efficient methodology for the purification of date palm peroxidase: Stability comparison with horseradish peroxidase (HRP). Saudi J Biol Sci 26:301–307. https://doi.org/10.1016/j.sjbs.2018.04.002
Bagewadi ZK, Mulla SI, Ninnekar HZ (2017) Optimization of laccase production and its application in delignification of biomass. IJROWA 6:351–365. https://doi.org/10.1007/s40093-017-0184-4
Bholay AD, Borkhataria BV, Jadhav PU, Palekar KS, Dhalkari MV, Nalawade PM (2012) Bacterial lignin peroxidase: a tool for biobleaching and biodegradation of industrial effluents. Univers J Environ Res Technol 2:58–64
Biko OD, Viljoen-Bloom M, van Zyl WH (2022) Medium optimization for enhanced production of recombinant lignin peroxidase in Pichia pastoris. Biotechnol Lett 45:105–113. https://doi.org/10.1007/s10529-022-03321-3
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Calderan-Rodrigues MJ, Fonseca JG, de Moraes FE, VazSetem L, CarmanhanisBegossi A, Labate CA (2019) Plant cell wall proteomics: A focus on monocot species, Brachypodiumdistachyon, Saccharum spp. and Oryza sativa. Int J Mol Sci 20:1975. https://doi.org/10.3390/ijms20081975
Chibbar RN, van Huystee RB (1984) Characterization of peroxidase in plant cells. Plant Physiol 75:956–958. https://doi.org/10.1104/pp.75.4.956
Dean JFD, Sterjiades R, Eriksson KEL (1994) Purification, characterization of an anionic peroxidase from sycamore maple (Acer pseudoplatanus) cell suspension cultures. Physiol Plant 92:233–240. https://doi.org/10.1111/j.1399-3054.1994.tb05331.x
Dossou-Yovo W, Parent S, Ziadi N, Parent É, Parent L (2021) Tea bag index to assess Carbon Decomposition Rate in Cranberry Agroecosystems. Soil Syst 5:44. https://doi.org/10.3390/soilsystems5030044
Duarte-Vázquez MA, García-Padilla S, García-Almendárez BE, Regalado C (2007) Purification of natural plant peroxidases and their physiological roles. Funct Plant Sci Biotechnol 1:18–31
Eijsink VG, Petrovic D, Forsberg Z, Mekasha S, Røhr ÃK, Várnai A, Bissaro B, Vaaje-Kolstad G (2019) On the functional characterization of lytic polysaccharide monooxygenases (LPMOs). Biotechnol Biofuels 12:1–16. https://doi.org/10.1186/s13068-019-1392-0
Giacobbe S, Pezzella C, Lettera V, Sannia G, Piscitelli A (2018) Laccase pretreatment for agrofood wastes valorization. Bioresour Technol 265:59–65. https://doi.org/10.1016/j.biortech.2018.05.108
Harmsen PF, Huijgen W, Bermudez L, Bakker R (2010) Literature review of physical and chemical pretreatment processes for lignocellulosic biomass.(Report /Wageningen UR, Food & Biobased Research; No. 1184). Wageningen UR - Food & Biobased Research. https://edepot.wur.nl/150289
Hendriks ATWM, Zeeman G (2009) Pretreatments to enhance the digestibility of lignocellulosic biomass. Bioresour Technol 100:10–18. https://doi.org/10.1016/j.biortech.2008.05.027
Hou XD, Li N, Zong MH (2013) Renewable bio ionic liquids-water mixtures‐mediated selective removal of lignin from rice straw: visualization of changes in composition and cell wall structure. Biotechnol Bioeng 110:1895–1902. https://doi.org/10.1002/bit.24862
Houghton J (2005) Global warming. Rep Prog Phys 68:1343–1403. https://doi.org/10.1088/0034-4885/68/6/R02
I.P.C.C. Climate change (2007) The physical science basis. Agenda 6(07):333
Joubert E, Schulz H (2006) Production and quality aspects of rooibos tea and related products. J Appl Bot Food Qual 80:138–144
Khatun S, Ashraduzzaman M, Karim MR, Pervin F, Absar N, Rosma A (2012) Purification and characterization of peroxidase from Moringa oleifera L. leaves. BioResources 7:3237–3251
Kondo M, Hidaka M, Kita K, Yokota H (2007) Ensiled green tea and black tea waste as protein supplement for goats. Options Méditerr Sér 74:165–169
Kong W, Fu X, Wang L, Alhujaily A, Zhang J, Ma F, Zhang X, Yu H (2017) A novel and efficient fungal delignification strategy based on versatile peroxidase for lignocellulose bioconversion. Biotechnol Biofuels 10:1–15. https://doi.org/10.1186/s13068-017-0906-x
Koskela S, Zha L, Wang S, Yan M, Zhou Q (2022) Hemicellulose content affects the properties of cellulose nanofibrils produced from softwood pulp fibres by LPMO. Green Chem 24:7137–7147. https://doi.org/10.1039/d2gc02237k
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lavery CB, MacInnis MC, MacDonald MJ, Williams JB, Spencer CA, Burke AA, Irwin DJ, D’Cunha GB (2010) Purification of peroxidase from horseradish (Armoracia rusticana) roots. J Agric Food Chem 58:8471–8476
Liu G, Han D, Yang S (2022) Combinations of mild chemical and bacterial pretreatment for improving enzymatic saccharification of corn stover. Biotechnol Biotechnol Equip 36:598–608. https://doi.org/10.1080/13102818.2022.2112910
Liu J, Yang B, Chen C (2013) A novel membrane-based process to isolate peroxidase from horseradish roots: optimisation of operating parameters. Bioprocess Biosyst Eng 36:251–257. https://doi.org/10.1007/s00449-012-0781-6
Lotter D, le Maitre D (2014) Modelling the distribution of Aspalathus linearis (Rooibos tea): implications of climate change for livelihoods dependent on both cultivation and harvesting from the wild. Ecol Evol 4:1209–1221. https://doi.org/10.1002/ece3.985
Mafa MS, Malgas S, Bhattacharya A, Rashamuse K, Pletschke BI (2020a) The effects of alkaline pretreatment on agricultural biomasses (corn cob and sweet sorghum bagasse) and their hydrolysis by a termite-derived enzyme cocktail. Agronomy 10:1211. https://doi.org/10.3390/agronomy10081211
Mafa MS, Malgas S, Rashamuse K, Pletschke BI (2020b) Delineating functional properties of a cello-oligosaccharide and β-glucan specific cellobiohydrolase (GH5_38): its synergism with Cel6A and Cel7A for β-(1, 3) -(1, 4)-glucan degradation. Carbohydr Res 495:108081. https://doi.org/10.1016/j.carres.2020.108081
Mafa MS, Pletschke BI, Malgas S (2021) Defining the frontier of synergism between cellulolytic enzymes for improved hydrolysis of lignocellulosic feedstocks. Catalysts 11:1343. https://doi.org/10.3390/catal11111343
Malgas S, Mafa MS, Mkabayi L, Pletschke BI (2019) A mini review of xylanolytic enzymes with regards to their synergistic interactions during hetero-xylan degradation. World J Microbiol Biotechnol 35:1–13. https://doi.org/10.1007/s11274-019-2765-z
Malhotra M, Suman SK (2021) Laccase-mediated delignification and detoxification of lignocellulosic biomass: removing obstacles in energy generation. Environ Sci Pollut Res 28:58929–58944. https://doi.org/10.1007/s11356-021-13283-0
Meier KK, Jones SM, Kaper T, Hansson H, Koetsier MJ, Karkehabadi S, Solomon EI, Sandgren M, Kelemen B (2018) Oxygen activation by Cu LPMOs in recalcitrant carbohydrate polysaccharide conversion to monomer sugars. Chem Rev 118:2593–2635. https://doi.org/10.1021/acs.chemrev.7b00421
Miller LG (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Min K, Kim YH, Kim J, Kim Y, Gong G, Um Y (2022) Effect of manganese peroxidase on the decomposition of cellulosic component: direct cellulolytic activity and synergistic effect with cellulase. Bioresour Technol 343:126138. https://doi.org/10.1016/j.biortech.2021.126138
Miranda MIV, Cascone O (1995) Horseradish peroxidase extraction and purification by aqueous two-phase partition. Appl Biochem Biotechnol 53:147–154. https://doi.org/10.1007/BF02788604
Mnich E, Bjarnholt N, Eudes A, Harholt J, Holland C, Jorgensen B, Larsen FH, Liu Manat R, Meyer AS, Mikkelsen JD (2020) Phenolic cross-links: building and de-constructing. Nat Prod Rep 37:919–961. https://doi.org/10.1039/c9np00028c
Olsson L, Hahn-Hagerdal B (1996) Fermentation of lignocellulosic hydrolysates for ethanol production. Enzyme Microb Technol 18:312–331. https://doi.org/10.1016/0141-0229(95)00157-3
Park S, Baker JO, Himmel ME, Parilla PA, Johnson DK (2010) Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnol Biofuels 3:1–10. https://doi.org/10.1186/1754-6834-3-10
Paul KG, Stigbrand T (1970) Four isoperoxidases from horseradish root. Acta Chem Scand 24:3607–3617
Pengilly M, Joubert E, van Zyl WH, Botha A, Bloom M (2008) Enhancement of rooibos (Aspalathus linearis) aqueous extract and antioxidant yield with fungal enzymes. J Agric Food Chem 56:4047–4053. https://doi.org/10.1021/jf073095y
Rad AM, Ghourchian H, Moosavi-Movahedi AA, Hong J, Nazari K (2007) Spectrophotometric assay for horseradish peroxidase activity based on pyrocatechol–aniline coupling hydrogen donor. Anal Biochem 362:38–43. https://doi.org/10.1016/j.ab.2006.11.035
Sarika D, Kumar PSSA, Arshad S, Sukumaran MK (2015) Purification and evaluation of horseradish peroxidase activity. Int J Curr Microbiol Appl Sci 4:367–375
Strother A, Fraser IM, Allahyari R, Tilton BE (1981) Metabolism of 8-aminoquinoline antimalarial agents. Bull World Health Organ 59:413
Su Y, Yu X, Sun Y, Wang G, Chen H, Chen G (2018) Evaluation of screened lignin-degrading fungi for the biological pretreatment of corn stover. Sci Rep 8:1–11. https://doi.org/10.1038/s41598-018-23626-6
Sulyman AO, Igunnu A, Malomo SO (2020) Isolation, purification, and characterization of cellulase produced by Aspergillus niger cultured on Arachis hypogaea shells. Heliyon 6:e05668. https://doi.org/10.1016/j.heliyon.2020.e05668
Sun FH, Li J, Yuan YX, Yan ZY, Liu XF (2011) Effect of biological pretreatment with Trametes hirsuta yj9 on enzymatic hydrolysis of corn stover. Int Biodeterior Biodegradation 65:931–938. https://doi.org/10.1016/j.ibiod.2011.07.001
Thoresen M, Malgas S, Mafa MS, Pletschke BI (2021) Revisiting the phenomenon of cellulase action: not all endo-and exo-cellulase interactions are synergistic. Catalysts 11:170. https://doi.org/10.3390/catal11020170
Van Dyk JS, Pletschke BI (2012) A review of lignocellulose bioconversion using enzymatic hydrolysis and synergistic cooperation between enzymes—factors affecting enzymes, conversion and synergy. Biotechnol Adv 30:1458–1480. https://doi.org/10.1016/j.biotechadv.2012.03.002
Walton PH, Davies GJ (2016) On the catalytic mechanisms of lytic polysaccharide monooxygenases. Curr Opin Chem Biol 31:195–207. https://doi.org/10.1016/j.cbpa.2016.04.001
Zhang XZ, Zhang YHP (2013) Cellulases: characteristics, sources, production, and applications. In: Yang S-T, El-Enshasy HA, Thongchul N. (eds) Bioprocessing technologies in biorefinery for sustainable production of fuels, chemicals, and polymers, vol 1. p 131–146. https://doi.org/10.1002/9781118642047.ch8
Acknowledgements
This research was supported by University of the Free State, Department of Plant Science and Centre for Graduate Support. This research was also partially supported by the South African National Research Foundation through a Thuthuka grant awarded to Dr. Mpho S Mafa.
Funding
Open access funding provided by University of the Free State. Ms MMM received MSc bursary from University of the Free State Centre for Graduate Support, MSM received the central research fund (CRF) from the Dean of Natural and Agricultural Sciences and NRF-Thuthuka grants.
Author information
Authors and Affiliations
Contributions
MMM and MSM designed the research; MMM conducted the experiments and prepared the original draft of the manuscript;
MSM, MMM, OA, and BIP analysed the data and wrote the paper; and MSM had primary responsibility for the final content.
Corresponding author
Ethics declarations
Consent for publication
All authors have read and approved the final manuscript.
Competing interests
Authors do not have any competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 342 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mohotloane, M.M., Alexander, O., Pletschke, B.I. et al. Horseradish peroxidase delignification of fermented rooibos modifies biomass structural and chemical properties and improves holocellulolytic enzyme cocktail efficacy. Biologia 78, 1943–1959 (2023). https://doi.org/10.1007/s11756-023-01424-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-023-01424-4