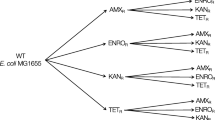
Abstract
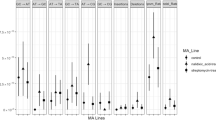
Antibiotic resistance is a major problem for public health worldwide. Since bacteria may rapidly respond to antibiotics and acquire resistance, it is important to understand how the bacterial genome changes under antibiotic treatments. With the advent of high-throughput approaches, experimental limitations in characterizing the roles of new mutations and the selection related to the evolution of antibiotic resistance have been eliminated. Here, to explore the mutation profiles of E. coli populations exposed to sub-inhibitory and then increasing concentrations of the antibiotics norfloxacin and colistin, an evolution experiment was combined with whole-population genomic sequencing. The results showed that, while the occurrence of genome-wide mutations is dramatically higher in the populations that were only treated with norfloxacin, there is a remarkable decrease in the colistin-treated populations compared to norfloxacin. It suggests that antibiotics not only elevate the genome-wide mutation rate but also magnify the rate of occurrence of advantageous mutations that increase the rate of adaptation. This work provides further evidence to support the idea that antibiotics can act indirectly as bacterial mutagens, and also that sequential antibiotic treatments may contribute to acquiring antibiotic resistance by improving the adaptation rate in bacteria in a relatively short time.

Similar content being viewed by others
Data availability
Raw sequence data reported in this study has been deposited in NCBI SRA (Bioproject No.: PRJNA727091).
Abbreviations
- DNM:
-
de novo mutation
- MDR:
-
Multi-drug resistant
- EE:
-
Experimental evolution
- MIC:
-
Minimum inhibitory concentration
- QRDR:
-
Quinolone resistance determining region
- MW U test:
-
Mann-Whitney U test
- LPS:
-
Lipopolysaccharides
References
Andersson DI et al (2020) Antibiotic resistance: turning evolutionary principles into clinical reality. FEMS Microbiol Rev 4:171–188. https://doi.org/10.1093/femsre/fuaa001
Andrade FF, Silva D, Rodriguez A, Pina-Vaz C (2020) Colistin update on its mechanism of action and resistance, present and future challenges. Microorganisms 8:1716. https://doi.org/10.3390/microorganisms8111716
Andrews JM (2001) Determination of minimum inhibitory concentrations. J Antimicrob Chemother 48:5–16. https://doi.org/10.1093/jac/48.suppl_1.5
Aulin LBS et al (2021) Distinct evolution of colistin resistance associated with experimental resistance evolution models in Klebsiella pneumoniae. J Antimicrob Chemother 76:533–535. https://doi.org/10.1093/jac/dkaa450
Aydın M et al (2018) Rapid emergence of colistin resistance and its impact on fatality among healthcare-associated infections. J Hosp Infect 98:260–263. https://doi.org/10.1016/j.jhin.2017.11.014
Bhatnagar K, Wong A (2019) The mutational landscape of quinolone resistance in Escherichia coli. PLoS One 14:e0224650. https://doi.org/10.1371/journal.pone.0224650
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinfomatics 30:2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Bourrel AS, Poirel L, Gea R (2019) Colistin resistance in Parisian inpatient faecal Escherichia coli as the result of two distinct evolutionary pathways. J Antimicrob Chemother 74:1521–1530. https://doi.org/10.1093/jac/dkz090
Buchmeier N, Bossie S, Chen CY, Fang FC, Guiney DG, Libby SJ (1997) SlyA, a transcriptional regulator of Salmonella typhimurium, is required for resistance to oxidative stress and is expressed in the intracellular environment of macrophages. Infect Immun 65:3725–3730. https://doi.org/10.1128/iai.65.9.3725-3730.1997
Cycon M, Mrozik A, Piotrowska-Seget Z (2019) Antibiotics in the soil environment — degradation and their impact on microbial activity and diversity. Front Microbiol 10:338. https://doi.org/10.3389/fmicb.2019.00338
Deatherage DE, Barrick JE (2014) Identification of mutations in laboratory-evolved microbes from next-generation sequencing data using breseq. Methods Mol Biol 1151:165–188. https://doi.org/10.1007/978-1-4939-0554-6_12
Díaz-Cañestro M, Periañez L, Xea M (2018) Ceftolozane/tazobactam for the treatment of multidrug resistant Pseudomonas aeruginosa: experience from the Balearic Islands. Eur J Clin Microbiol Infect Dis 37:2191–2200. https://doi.org/10.1007/s10096-018-3361-0
Dolan KT, Duguid EM, He C (2011) Crystal structures of SlyA protein, a master virulence regulator of Salmonella, in free and DNA-bound states. J Biol Chem 286:22178–22185. https://doi.org/10.1074/jbc.M111.245258
European Medicines Agency (2019) Categorisation of Antibiotics in the European Union. Answer to the Request from the European Commission for Updating the Scientific Advice on the Impact on Public Health and Animal Health of the Use of Antibiotics in Animals. https://www.ema.europa.eu/en/documents/report/categorisation-antibiotics-european-union-answer-request-european-commission-updating-scientific_en.pdf. Accessed 7 May 2021
Everett MJ, Jin YF, Ricci V, Piddock LJ (1996) Contributions of individual mechanisms to fluoroquinolone ressitance in 36 Escherichia coli strains isolated from humans and animals. Antimicrob Agents Chemother 40:2380–2386. https://doi.org/10.1128/AAC.40.10.2380
Fair RJ, Tor Y (2014) Antibiotics and bacterial resistance in the 21st century. Perspect Med Chem 6:25–64. https://doi.org/10.4137/PMC.S14459
Halaby T et al (2016) Genomic characterization of colistin heteroresistance in Klebsiella pneumoniae during a nosocomial outbreak. Antimicrob Agents Chemother 60:6837–6843. https://doi.org/10.1128/AAC.01344-16
Hjort K, Juren P, Toro JC, Hoffner S, Andersson DI, Sandegren L (2020) Dynamics of extensive drug resistance evolution of mycobacterium tuberculosis in a single patient during 9 years of disease and treatment. J Infect Dis:jiaa625. https://doi.org/10.1093/infdis/jiaa625
Hooper DC, Jacoby GA (2016) Topoisomerase inhibitors: fluoroquinolone mechanisms of action and resistance. Cold Spring Harb Perspect Med 6:a025320. https://doi.org/10.1101/cshperspect.a025320
Islam A et al (2017) Colistin resistant Escherichia coli carrying mcr-1 in urban sludge samples: Dhaka. Bangladesh Gut Pathog 9:77. https://doi.org/10.1186/s13099-017-0227-4
Isler B et al (2019) Antibiotic overconsumption and resistance in Turkey. Clin Microbiol Infect 25:651–653. https://doi.org/10.1016/j.cmi.2019.02.024
Janssen AB, Bartholomew TL, Marciszewska NP, Bonten MJM, Willems RJL, Bengoechea JA, van Schaik W (2020) Nonclonal emergence of colistin resistance associated with mutations in the BasRS two-component system in Escherichia coli bloodstream isolates. mSphere 5:e00143–e00120. https://doi.org/10.1128/mSphere.00143-20
Jayol A, Poirel L, Brink A, Villegas M-V, Yilmaz M, Nordmann P (2014) Resistance to colistin associated with a single amino acid change in protein PmrB among Klebsiella pneumoniae isolates of worldwide origin. Antimicrob Agents Chemother 58:4762–4766. https://doi.org/10.1128/AAC.00084-14
Jechalke S, Heuer H, Siemens J, Amelung W, Smalla K (2014) Fate and effects of veterinary antibiotics in soil. Trends Microbiol 22:536–545. https://doi.org/10.1016/j.tim.2014.05.005
Klein EY et al (2018) Global increase and geographic convergence in antibiotic consumption between 2000 and 2015. Proc Natl Acad Sci U S A 115:E3463–E3470. https://doi.org/10.1073/pnas.1717295115
Kleine C-E, Schlabe S, Hischebeth GTR ,al. e (2017) Successful therapy of a multidrug-resistant extended-spectrum β-lactamase–producing and fluoroquinolone-resistant Salmonella enterica subspecies enterica serovar Typhi infection using combination therapy of meropenem and fosfomycin. Clin Infect Dis 65:1754–1756. https://doi.org/10.1093/cid/cix652
Komp Lindgreen P, Karlsson A, Hughes D (2003) Mutation rate and evolution of fluoroquinolone resistance in Escherichia coli isolates from patients with urinary tract infection. Antimicrob Agents Chemother 47:3222–3232. https://doi.org/10.1128/aac.47.10.3222-3232.2003
Lazarus B, Paterson DL, Mollinger JL, Rogers BA (2015) Do human extraintestinal Escherichia coli infections resistant to expanded-spectrum cephalosporins originate from food-producing animals? A systematic review. Clin Infect Dis 60:439–452. https://doi.org/10.1093/cid/ciu785
Lee H, Popodi E, Tang H, Foster PL (2012) Rate and molecular spectrum of spontaneous mutations in the bacterium Escherichia coli as determined by whole-genome sequencing. Proc Natl Acad Sci U S A 109:E2774–E2783. https://doi.org/10.1073/pnas.1210309109
Levin BR, Rozen DE (2006) Non-inherited antibiotic resistance. Nat Rev Microbiol 4:556–562. https://doi.org/10.1038/nrmicro1445
Libby SJ et al (1994) A cytolysin encoded by Salmonella is reqired for survival within macrophages. Proc Natl Acad Sci U S A 91:489–493. https://doi.org/10.1073/pnas.91.2.489
Liu Y-Y et al (2016) Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis 16:161–168. https://doi.org/10.1016/S1473-3099(15)00424-7
Lobanovska M, Pilla G (2017) Penicillin’s discovery and antibiotic resistance: lessons for the future? Yale J Biol Med 90:135–145
Lofton H, Pranting M, Thulin E, Andersson DI (2013) Mechanisms and fitness costs of resistance to antimicrobial peptides LL-37, CNY100HL and wheat germ histones. PLoS One 8:e68875. https://doi.org/10.1371/journal.pone.0068875
Long H et al (2016) Antibiotics treatment enhances the genome-wide mutation of target cells. Proc Natl Acad Sci U S A 113:2498–2505. https://doi.org/10.1073/pnas.160120811
Nation RL, Li J (2009) Colistin in the 21st century. Curr Opin Infect Dis 22:535–543. https://doi.org/10.1097/QCO.0b013e328332e672
Ozdemirel HO, Ulusal D, Kucukyildirim Celik S (2021) Streptomycin and nalidixic acid elevate the spontaneous genome-wide mutation rate in Escherichia coli. Genetica 149:73–80. https://doi.org/10.1007/s10709-021-00114-w
Pereira A, Silva L, Meisel L, Pena A (2015) Fluoroquinolones and tetracycline antibiotics in a Portuguese aquaculture system and aquatic surroundings: occurence and environmental impact. J Toxicol Environ Health A 78:959–975. https://doi.org/10.1080/15287394.2015.1036185
Pribis JP et al (2019) Gamblers: an antibiotic-induced evolvable cell subpopulation differentiated by reactive-oxygen-induced general stress response. Mol Cell 74:785–800. https://doi.org/10.1016/j.molcel.2019.02.037
Rodrigues CHM, Pires DEV, Ascher DB (2021) DynaMut2: assessing changes in stability and flexibility upon single and multiple point missense mutations. Protein Sci 30:60–69. https://doi.org/10.1002/pro.3942
Roemhild R, Schulenburg H (2019) Evolutionary ecology meets the antibiotic crisis: can we control pathogen adaptation through sequential therapy? Evol Med Public Health 2019:37–45. https://doi.org/10.1093/emph/eoz008
Russo A, Bassetti M, Gea C (2019) Bloodstream infections caused by carbapenem-resistant Acinetobacter baumannii: clinical features, therapy and outcome from a multicenter study. J Inf Secur 79:130–138. https://doi.org/10.1016/j.jinf.2019.05.017
Santos-Lopez A, Marshall C, Scribner M, Synder D, Cooper V (2019) Evolutionary pathways to antibiotic resistance era dependent upon environmental structure and bacterial lifestyle. Elife 8:e47612. https://doi.org/10.7554/eLife.47612
Shen LL et al. (1989) Mechanism of inhibition of DNA gyrase by quinolone antibacterials: a cooperative drug--DNA binding model. Biochemistry 28:3886–3894. https://doi.org/10.1021/bi00435a039
Sievert DM et al (2013) Antimicrobial-resistant pathogens associated with healthcare-associated infections: summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2009-2010. Infect Control Hosp Epidemiol 34:1–14. https://doi.org/10.1086/668770
Thorvaldsdottir H, Robinson JT, Mesirov JP (2013) Integrated genomics viewer (IGV): high-performance genomics data visualization and exploration. Brief Bioinform 14:178–192. https://doi.org/10.1093/bib/bbs017
Will WR, Fang FC (2020) The evolution of MarR family transcription factors as counter-silencers in regulatory networks. Curr Opin Microbiol 55:1–8. https://doi.org/10.1016/j.mib.2020.01.002
Yang QE, MacLean C, Aea P (2020) Compensatory mutations modulate the competitiveness and dynamics of plasmid-mediated colistin resistance in Escherichia coli clones. ISME J 14:861–865. https://doi.org/10.1038/s41396-019-0578-6
Yu X, Susa M, Kanbbe C, Schmid RD, Bachmann TT (2004) Development and validation of a diagnostic DNA microarray to detect quinolone-resistant Escherichia coli among clinical isolates. J Clin Microbiol 42. https://doi.org/10.1128/JCM.42.9.4083-4091.2004
Zafer MM, El-Mahallawy HA, Aea A (2019) Emergence of colistin resistance in multidrug-resistant Klebsiella pneumoniae and Escherichia coli strains isolated from cancer patients. Ann Clin Microbiol Antimicrob 18. https://doi.org/10.1186/s12941-019-0339-4
Zurek L, Ghosh A (2014) Insects represent a link between food animal farms and the urban environment for antibiotic resistance traits. Appl Environ Microbiol 80:3562–3567. https://doi.org/10.1128/AEM.00600-14
Acknowledgments
We thank Ozgur Ozdemirel for technical help. This work is supported by the grant from Hacettepe University Research Fund (FHD-2017-13143).
Code availability
Not applicable.
Funding
This work is supported by the grant from Hacettepe University Research Fund (FHD-2017-13143).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest.
Ethics approval
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kucukyildirim, S. Whole-population genomic sequencing reveals the mutational profiles of the antibiotic-treated Escherichia coli populations. Biologia 77, 525–531 (2022). https://doi.org/10.1007/s11756-021-00959-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-021-00959-8