Abstract
While osteotomies are necessary for rotational correction and limb lengthening, angular correction or moderate length inhibition may be achieved by other, less invasive means. Several techniques of epiphysiodesis have evolved, enabling gradual correction of angular correction and/or length equalisation through guided growth. This manuscript comprises a historical and comparative review of those techniques. The 8-plate method of guided growth affords the opportunity to provide a tension band (rather than compression) that expedites angular correction, compared to stapling or transphyseal screws, which rely upon the principle of compression. When applied to each side of a given physis, longitudinal growth is inhibited, in the same fashion as stapling or epiphysiodesis. The physis and periosteum are spared any direct insult, thus making this a reversible process, suitable for use in younger children. The 8-plate is simple to insert and, compared to staples or transphyseal screws, easy to remove.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Orthopaedic surgeons are consulted regarding a wide variety of paediatric problems including limb length discrepancy and torsional and angular deformities of the extremities. While some deformities are physiologic and require only parental reassurance, many may eventually warrant surgical correction. Osteotomy has been the standard choice for many of our surgical interventions; advances in medical imaging and instrumentation have made this relatively safe, improving the outcomes. Nevertheless, immobilisation and deferred weight-bearing are still required during recuperation. Depending upon the aetiology, recurrent deformity may lead to repeat osteotomy.
While osteotomy is necessary for rotational correction and limb lengthening, angular correction may be achieved by other means: several techniques of epiphysiodesis have evolved, enabling gradual correction of angular correction and/or length equalisation through guided growth. This manuscript comprises a historical and comparative review of those techniques.
Methods
Currently there are four available surgical techniques for inhibiting the physis:
-
1.
open epiphysiodesis (Phemister) — permanent;
-
2.
stapling (Blount);
-
3.
transphyseal screw (Metaizeau);
-
4.
8-plate (Stevens).
The history and rationale of our willingness to surgically approach and occasionally instrument the physis is covered in the discussion below. Details regarding the techniques, and applications and results of the first three techniques are available in the literature and will not be reiterated here. The methods and results described in this manuscript are confined to the 8-plate technique, specifically as it applies to the correction of angular deformities and/or for length inhibition.
Patient selection
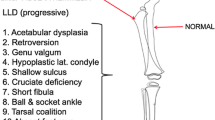
Any child with angular deformity and open physes, excluding physiologic varus and valgus, is a potential candidate for guided growth. Patient selection requires the surgeon to understand the all important differences between physiologic and pathologic deformities [1–4]. The former will resolve without treatment; the latter will progress. When in doubt, follow-up evaluation at 6- month interval(s) may clarify which patients need intervention. The aetiology of the deformity is not critical to the outcome, with the sole exception being an unresectable physeal bar. The general guidelines for length equalisation via epiphysiodesis are well established in the literature, namely a predicted 2–5-cm discrepancy at maturity [5]. For angular deformities of the knee (frontal plane), deviation of the mechanical axis beyond the central 2 quadrants may warrant intervention (Fig. 1).
Mechanical axis zones. If you divide the knee into quadrants, the ideal mechanical axis would bisect the knee (0), with medial zone (−1) or lateral zone (−1) being within physiologic range. With the notable exception of physiologic varus <age 2 and physiologic valgus <age 6, medial or lateral zones 2 or 3 would likely manifest symptoms and gait disturbance and thereby warrant surgical intervention
Pre-operative assessment
The family and general medical history may provide invaluable clues regarding the prognosis and treatment expectations. For example, in hereditary conditions, a parent or family member may have been treated “traditionally” with one or more osteotomies. Naturally this raises the anxiety level on the part of the patient and family. The presence of functional limitations, gait disturbance and pain may influence the timing of intervention. Guided growth is contraindicated for conditions that will self correct. When in doubt about the possible physiologic nature of a given deformity, surgical treatment should be deferred pending a 6-month follow-up evaluation with comparative radiographs; if there is progressive deviation of the mechanical axis, intervention may be warranted. In addition to documenting frontal or sagittal deformities, the clinical evaluation should include gait pattern, strength testing, torsional profile, joint motion and stability, neuromuscular function, and spinal alignment. In unique situations, gait analysis may be useful for comparison before and after correction of malalignment [6].
Medical imaging
A full-length standing radiograph of the legs, with the pelvis levelled (through using an appropriately sized block on the shorter side) and the patellae facing forwards, is most useful for assessing limb lengths and mechanical axis deviation. This study is more helpful than a plain or CT scanogram because it is weightbearing, includes the feet and pelvis, and demonstrates any diaphyseal deformities [7]. This AP radiograph will also reveal concomitant pathology in the hips or ankles; localised views of the pelvis or ankles may be obtained as needed. A lateral view of each extremity may be advisable when sagittal or oblique plane deformities are suspected. For genu valgum deformities, a patellar view is recommended and it may demonstrate a shallow anterior femoral sulcus, subluxation or osteochondral defects.
It may be helpful to determine the bone age if the child is near maturity, as guided growth may not yield sufficient correction if less than 6 months of predicted growth remain [5]. Unless there is history of trauma and a suspected physeal bar, it is unlikely that a CT scan or MRI is warranted.
Surgical planning
The principles of deformity analysis are identical whether applied for guided growth or corrective osteotomies [8, 9]. It is important to decide whether to address the femur, the tibia or both. This is easily accomplished by scrutinising the radiographs. The ideal outcome would be to achieve equal limb lengths and a neutral mechanical axis while preserving a horizontal knee. The best way to anticipate this is to place the radiograph on the view-box so that the knee axis is horizontal and draw a vertical axis at 87° through the middle of the knee. You can readily detect the levels (femoral vs. tibial) and size of deformity (Fig. 2) and determine if hardware on both sides of the knee is needed. If further analysis of the radiograph indicates bilateral or multi-level deformities, these can be addressed at the same sitting.
Rotating the standing AP radiograph so that the knee joint line is horizontal, and drawing an 87° intersecting line through the centre of the knee identifies the source of deformity; the portion of the line proximal to the knee should pass through the centre of the hip joint and that below should pass through the middle of the ankle — where it does not indicate presence of a deformity in that segment
The presence of a concomitant deformity on the lateral film at the same level as observed on the AP view suggests an oblique plane deformity. This would call for an implant at the apex of that deformity in order to accomplish simultaneous coronal and sagittal correction. For example: a flexion and varus deformity might warrant a single anteromedial plate applied to the tibia or femur; fixed equinus may call for an anterior plate on the distal tibia, etc.
Surgical implant
Having had extensive experience with stapling, I noted that some of the most dramatic improvements occurred when staples spread. Unfortunately, this is not a predictable event; sometimes the staple(s) would break or migrate, necessitating premature and unplanned surgical intervention (Fig. 3). This led to the idea of an implant that would facilitate bending (and thus yield to physeal growth) but not be susceptible to migration or breakage; the logical choice was to employ a non-locking plate and screws. It seemed that a flexible implant would be better suited to guide the dynamic physis.
One of the problems with staples has been migration (arrow). A year after proximal tibial stapling to address limb length inequality, this 14-year-old boy has drifted into varus (medial zone 3) because the lateral staples loosened and migrated. He resurfaced with a “bump” on the knee and noticeable genu varum — in addition to a 2.5-cm limb length inequality
Early in the plate series, I tried various plate constructs: one third-tubular, pelvic reconstruction, DC plate with 3.5-mm screws, partially threaded 4.0-mm screws and 4.5-mm screws. I also sought to customise the hardware by selecting and bending small hardware for younger patients and robust hardware for larger teenagers. This led to the design of the precontoured 8-plate (Orthofix Srl, Verona, Italy) in two lengths (12 mm and 16 mm, as measured between the centres of both holes) coupled to standard 4.5-mm fully threaded, cannulated screws in three lengths (16, 24 and 32 mm) (Fig. 4). This serves the full spectrum of patients and diagnoses.
Surgical technique
Tourniquet control is useful for speed and better visualisation. Each incision centred on the physis is 2–3-cm long; the dissection is carried through fascia, between muscles, and leaves the periosteum undisturbed. A needle is inserted through the perichondrial ring to localise the physis (confirmed fluoroscopically) before applying the 8-plate. Because it serves as a tension band, one plate per physis is sufficient (the exception being anterior femur for fixed flexion deformity). Threaded guide pins are inserted with fluoroscopic control through the centres of holes in the plate. The direction of the guide pins are such that they do not encroach on the physis when the screws are finally seated. It is not essential that the guide pins are parallel. It is recommended that you only drill a 3.2-mm starting hole through the cortex; this will give better screw purchase. The 4.5-mm screws are self-tapping and the length (16/24/32 mm) is chosen at the discretion of the surgeon (Fig. 5a–d). Upon removal of the guide pins, each screw should be rechecked and securely tightened to countersink into the plate. Following wound closure, a compression bandage is sufficient; no casts are necessary. Immediate motion and weight-bearing are recommended, with crutches as needed for comfort.
A small incision is centred over the physis. The periosteum is preserved in dissection. The plate is temporarily secured and centred over the physis with a hypodermic needle and followed by threaded pins through the holes of the plate. These are substituted for screws of appropriate length after predrilling of the cortex only
Postoperative management
The surgery is done on an outpatient basis; following discharge, the patient may resume activities as tolerated. For those children who are slow to mobilise, physical therapy may be helpful. Periodic follow-up at 3-month intervals is sufficient to document deformity correction. When the leg(s) is/are straight, follow-up radiographs are taken to document the correction, including neutralisation of the mechanical axis, and plate removal is scheduled accordingly. Due to the vagaries of predicting “rebound” deformity, remove the plate(s) when the mechanical axis is neutral and continue to monitor growth. The process may be repeated as necessary; this is generally preferable to osteotomy.
Results
At the time of writing, I have performed guided growth on approximately 150 patients with over 250 deformities. Approximately 85% of these have been for correction of angular deformities. Compared to my stapling experience, the rate of correction is about 30% faster, averaging 11 months (range 6–26 months) until neutralisation of the mechanical axis (Fig. 6). Early in the series, some of the experimental implants, including one-third tubular plates with 3.5-mm screws and pelvic reconstruction plates with 4.5-mm screws, failed and required revision surgery. Since using the precontoured 8-plate with 4.5-mm cannulated screws, implant migration or failure is rare. Accordingly, the incidence of osteotomy in my practice has declined. Indeed I now consider osteotomy to be a salvage procedure — unless there is an urgent need to correct malrotation or gain significant length.
This 11-year-old girl presented with evolving genu valgum, circumduction gait and progressive symptoms, refractory to nonoperative management. Her mechanical axes, in lateral zone 2, corrected to neutral within 6 months of 8-plate insertion, producing normalisation of her gait and complete resolution of her symptoms. Her plates were removed and growth will be monitored to rule out recurrent valgus. Most corrections can be accomplished within one year (range 6-26 months)
My patients have ranged in age from 19 months to 17 years and in size from 12 to 183 kg. Diagnoses have included idiopathic, metabolic, neuromuscular, genetic, traumatic and developmental. There is no diagnosis that constitutes a contraindication for guided growth — with the exception of physiologic deformities or an unresectable physeal bar. However, I have on occasion combined bar resection with guided growth. Because the approach is practically subcutaneous and the correction is gradual, there have been no vascular or neurological complications and, importantly, no premature physeal arrests.
Given the vagaries of rebound growth, it is not possible to anticipate the likelihood of recurrent deformity. Therefore, I typically remove the plate when the mechanical axis is neutral. Depending upon the age and underlying aetiology of the deformity, rebound growth may still occur. Continued periodic follow-up and parental education are strongly recommended; the parents are easily informed about how to monitor alignment by observing the intercondylar distance (varus) or intermalleolar distance (valgus). If the mechanical axis drifts back out of the physiologic range — usually evident within 12 months of plate removal — guided growth may be repeated.
Discussion
For decades, corrective osteotomies have enjoyed status as the treatment of choice for a variety of paediatric malalignment conditions including idiopathic, metabolic and Blount’s deformities. With careful deformity analysis and meticulous execution, satisfactory results can be achieved [9]. However, while osteotomy is considered “definitive” by some, in fact recurrent deformities are not uncommon. Furthermore, the related costs and potential complications give one pause to reconsider the ostensible standard of care [10, 11].
In 1933, Dr. Dallas Phemister introduced his technique of rotating a rectangle of bone, including a portion of the physis, to produce a bone bridge that would permanently arrest the physis. The advantages of the method include the avoidance of surgical implants and comparatively low cost. Because of the difficulties of predicting growth, the Phemister technique was largely confined to length inhibition in adolescent patients. The same limitations apply to the percutaneous modification of Phemister’s technique [12–14]. A major drawback is the permanent bridging of the physis; the risk of over- or under-correction makes this option less popular than modern instrumented methods.
Haas, who placed a wire loop around a canine distal femoral physis and documented growth inhibition, first demonstrated the resilience of the physis following surgical instrumentation [15, 16]. He noted that growth resumed when the wire broke. Capitalising upon this idea, Dr. Walter Blount introduced his surgical staple in the late 1940s [17, 18]. It featured reinforced shoulders to resist the powerful expansion of the physis and was used in multiples (2 or 3 per physis). Most surgeons reserved this approach for adolescent patients, fearing that permanent physeal closure might ensue [19–22]. Following initial widespread acceptance, this method waned in popularity due, in part, to problems with staple migration or breakage.
I began using staples in younger children, including those with “sick physes” such as observed in rickets and skeletal dysplasia [23]. Often there was only room for a single staple. Despite some surprisingly dramatic improvements, there were still occasions where staple failure resulted in additional but unanticipated surgery comprising either of repeat stapling or osteotomy. For the reasons mentioned above I sought a more reliable implant — one that would be flexible yet secure. The 8-plate was the culmination of this quest.
The application of an extra-periosteal tension band to a given physis has proven to be a versatile solution for a variety of deformities and diagnoses [24, 25]. Contrary to the transphyseal Metaizeau screw method, the physis is not violated [26]. The screws in the 8-plate are free to diverge approximately 30°; that covers 95% of the deformities we are apt to encounter (Figs. 7 and 8). After maximal divergence, the convex, pre-contoured plate may be observed to straighten or even reverse its bend. That is not a problem as long as continued deformity correction is occurring; none of the plates in my series have broken (Fig. 9). On rare occasions a given screw may be retrieved and exchanged percutaneously.
This 15-year-old boy, with documented growth hormone deficiency and comparatively short stature, had received tibial staples a year before this radiograph. It is evident that he has both femoral valgus and residual tibial valgus. Fifteen months post exchange for 8-plates there is notable improvement
Upon closer scrutiny, one can deduce that the fulcrum of the intact staples is at the medial tip of the prongs. This limits the longitudinal growth and slows the process of angular correction. Potential outcomes include staple bending, migration or breakage. The fulcrum of the 8-plates is at (or medial to) the body of the plate. Serving as tension bands, they do not compress the physes, thus permitting more rapid correction. Note the relative straightening of the plates; there is no need to exchange screws unless they loosen or break (rare)
The comparative physiology of the plate vs. staples or percutaneous screws (PETS) is awaiting further study [27]. We have undertaken research in New Zealand rabbits to elucidate the response of the physis to flexible vs. rigid restraint and to better understand the phenomenon of rebound growth following removal. The result of the pilot study was presented at the Paediatric Orthopaedic Society of North America (POSNA) meeting in San Diego in May 2006. It is my belief that a more rapid rate of correction accompanies the use of a flexible implant and that this is probably more physiologic for the physis. Furthermore, it has been suggested that 90% of growth occurs during recumbence; perhaps the plate accommodates this while avoiding stress shielding that would be induced by rigid staples or transphyseal screws.
When considering the costs and risks associated with osteotomies, guided growth presents us with an option of early (or late) intervention with obvious advantages (Fig. 10). Switching to the 8-plate has solved some of the problems encountered with staples, including migration, breakage and difficulty of retrieval. Upon learning the alternatives, the children and their parents enthusiastically accept this method of treatment. The utilisation of healthcare resources and hospital beds is improved accordingly.
This 5-year-old boy presented with previously untreated bilateral Blount’s disease; the deformities are confined to the tibias. He was treated by guided growth on the right and a “Rab” oblique osteotomy of the proximal tibia/fibula on the left. Eighteen months following surgery, his right side is overcorrected (between lateral zones 1 and 2) on the right and undercorrected (medial zone 2) on the left. The 8-plate was removed on the right and a lateral tibial 8-plate inserted on the left
Conclusion
When Dr. Phemister first ventured to operate directly upon the growth plate for deformity correction, a new era of reconstructive orthopaedics was introduced. His technique is still used, albeit with modifications (e.g., percutaneous drilling), when permanent physeal closure is desirable and necessary. The Blount stapling technique, which fell into disfavour in the 1980s, enjoyed a modest resurgence in the 1990s. In Europe and some parts of the USA, the transphyseal screw gained favour as a minimally invasive alternative. However this technique violates the physis unnecessarily, at a calculated risk of premature closure. Hardware retrieval may be problematic and the reversible potential of this technique awaits further study. The 8-plate spares the physis and periosteum, offering perhaps the best solution to date. It is reversible and well tolerated by children of all ages with any diagnosis.
References
Heath CH, Staheli LT (1993) Normal limits of knee angle in children — genu varum and genu valgum. J Pediatr Orthop 13:259–262
Kling TF Jr, Hensinger RN (1983) Angular and torsional deformities of the lower limbs in children. Clin Orthop Relat Res 176:136–147
Levine AM, Drennan JC (1982) Physiological bowing and tibia vara. The metaphyseal-diaphyseal angle in the measurement of bowleg deformities. J Bone Joint Surg Am 64:1158–1163
Salenius P, Vankka E (1975) The development of the tibiofemoral angle in children. J Bone Joint Surg Am 57:259–261
Anderson M, Green WT, Messner MB (1963) Growth and predictions of growth in the lower extremities. J Bone Joint Surg Am 45:1–14
Stevens PM, MacWilliams B, Mohr RA (2004) Gait analysis of stapling for genu valgum. J Pediatr Orthop 24:70–74
Machen MS, Stevens PM (2005) Should full-length standing anteroposterior radiographs replace the scanogram for measurement of limb length discrepancy? J Pediatr Orthop 14:30–37
Dietz FR, Merchant TC (1990) Indications for osteotomy of the tibia in children. J Pediatr Orthop 10:486–490
Paley D, Herzenberg JE, Tetsworth K et al (1994) Deformity planning for frontal and sagittal plane corrective osteotomies. Orthop Clin North Am 25:425–465
Mycoskie P (1981) Complications of osteotomies about the knee in children. Orthopaedics 4:1005–1015
Steel H, Sandrow R, Sullivan P (1971) Complications of tibial osteotomy in children for genu valgum or varum. J Bone Joint Surg Am 53:1629–1635
Bowen JR, Leahey JL, Zhang ZH, MacEwen GD (1985) Partial epiphysiodesis at the knee to correct angular deformity. Clin Orthop Relat Res 198:184–190
Canale S, Russell T, Holcomb R (1986) Percutaneous epiphyseodesis — experimental study and preliminary results. J Pediatr Orthop 6:150
Ogilvie J (1986) Epiphyseodesis evaluation of a new technique. J Pediatr Orthop 6:147
Haas SL (1945) Retardation of bone growth by a wire loop. J Bone Joint Surg 27:25–36
Haas SL (1948) Mechanical retardation of bone growth. J Bone Joint Surg Am 30:506–512
Blount WP, Clarke GR (1949) Control of bone growth by epiphyseal stapling: a preliminary report. J Bone Joint Surg 31:464–478
Blount WP (1971) A mature look at epiphyseal stapling. Clin Orthop Relat Res 77:158–163
Frantz CH (1971) Epiphyseal stapling: a comprehensive review. Clin Orthop Relat Res 77:149–157
Fraser RK, Dickens DR, Cole WG (1995) Medial physeal stapling for primary and secondary genu valgum in late childhood and adolescence. J Bone Joint Surg Br 77:733–735
Stevens PM, Maguire M, Dales MD, Robins AJ (1999) Physeal stapling for idiopathic genu valgum. J Pediatr Orthop 19:645
Zuege RC, Kempken TG, Blount WP (1979) Epiphyseal stapling for angular deformity at the knee. J Bone Joint Surg Am 61:320–329
Mielke CH, Stevens PM (1996) Hemiepiphyseal stapling for knee deformities in children younger than 10 years — a preliminary report. J Pediatr Orthop 16:423–429
Novais E, Stevens PM (2006) Hypophosphatemic rickets — the role of hemiepiphysiodesis. J Pediatr Orthop 26:238–244
Stevens PM, Pease F (2006) Hemiepiphysiodesis for posttraumatic tibial valgus. J Pediatr Orthop 26:385–392
Metaizeau JP, Wong-Chung J, Bertrand H, Pasquier P (1998) Percutaneous epiphysiodesis using transphyseal screws (PETS). J Pediatr Orthop 18:363–369
Aykut US, Yazici M, Kandemir U et al (2005) The effect of temporary hemiepiphyseal stapling on the growth plate: a radiologic and immunohistochemical study in rabbits. J Pediatr Orthop 25:336–341
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Stevens, P.M. Guided growth: 1933 to the present. Strat Traum Limb Recon 1, 29–35 (2006). https://doi.org/10.1007/s11751-006-0003-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11751-006-0003-3