Abstract
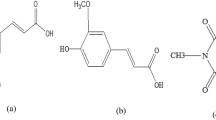
Ferulic acid (4-hydroxy-3-methoxycinnamic acid) is a phytochemical antioxidant that is widely distributed throughout the plant kingdom. Cinnamic acid derivatives are used as biobased ultraviolet (UV) absorbers in sunscreen formulations. Soybean oil ferulate, a biobased UV absorber, was synthesized by reacting soyamide with ferulic acid. The resulting product was characterized by infrared (IR) and nuclear magnetic resonance (NMR) spectroscopy, and gel permeation chromatography (GPC). Spectroscopic studies measured the soyamide-based ferulate maximum absorbance at 327 nm with a molar extinction coefficient of 19,705 L mol−1 cm−1. Soyamide ferulate has potential to be used in coatings.








Similar content being viewed by others
References
Sharma BK, Adhvaryu A, Erhan SZ (2006) Synthesis of hydroxy thio-ether derivatives of vegetable oil. J Agric Food Chem 54:9866–9872
Khot SN, Lascala JJ, Can E, Morye SS, Williams GI, Palmese GR, Kusefoglu SH, Wool RP (2001) Development and application of triglyceride-based polymers and composites. J Appl Polym Sci 82:703–723
Stavarache C, Vinatoru M, Nishimura R, Maeda Y (2005) Fatty acids methyl esters from vegetable oil by means of ultrasonic energy. Ultrason Sonochem 12:367–372
Thames SF, Smith OW, Mendon SK, Quintero C (2004) Vegetable oil macromonomers as copolymerizable hydrophobes in miniemulsion polymerization. Polymer Preprints 2004(45):1110–1111
Dintcheva NT, La Mantia FP (2007) Durability of a starch-based biodegradable polymer. Polym Degrad Stabil 92:630–634
Chang HT, Su YC, Chang ST (2006) Studies on photostability of butyrylated, milled wood lignin using spectroscopic analyses. Polym Degrad Stabil 91:816–822
Muasher M, Sain M (2006) The efficacy of photostabilizers on the color change of wood filled plastic composites. Polym Degrad Stabil 91:1156–1165
Yamashita H, Ohkatsu YA (2003) A new antagonism between hindered amine light stabilizers and acidic compounds including phenolic antioxidant. Polym Degrad Stabil 80:421–426
Ou S, Kwok KC (2004) Ferulic acid: pharmaceutical functions, preparation and applications in foods. J Sci Food Agric 84:1261–1269
Harry-O’kuru RE, Mohamed A, Abbott TP (2005) Synthesis and characterization of tetrahydroxyjojoba wax and ferulates of jojoba oil. Ind Crops Prod 22:125–133
Compton DL, Laszlo JA (2002) Sunscreens from vegetable oils and plant phenols. US Patent 6346236
Compton DL, Kenar JA, Laszlo JA, Felker FC (2007) Starch-encapsulated, soy-based, ultraviolet-absorbing composites with feruloylated monoacyl- and diacylglycerol lipids. Ind Crops Prod 25:17–23
Kemp W (1991) Organic spectroscopy, 3rd edn. ELBS, New Delhi, p 130
Bunzel M, Ralph J, Funk C, Steinhart H (2005) Structural elucidation of new ferulic acid-containing phenolic dimmers and trimers isolated from maize bran. Tetrahedron Lett 46:5845–5850
Masuda T, Yamada K, Maekawa T, Takeda Y, Yamaguchi H (2006) Antioxidant mechanism studies of ferulic acid: identification of oxidative coupling product from methyl ferulate and linoleate. J Agric Food Chem 54:6069–6074
Pan GX, Thomson CI, Leary GJ (2002) UV–visible spectroscopic characteristics of ferulic acid and related compounds. J Wood Chem Technol 22:137–146
Acknowledgments
This material is based upon work supported by the Cooperative State Research, Education, and Extension Service, US Department of Agriculture under Agreement No. 2006-38202-16954.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Rawlins, J.W., Pramanik, M. & Mendon, S.K. Synthesis and Characterization of Soyamide Ferulate. J Am Oil Chem Soc 85, 783–789 (2008). https://doi.org/10.1007/s11746-008-1260-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11746-008-1260-8