Abstract
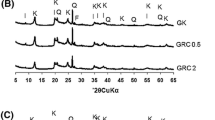
The pore size distribution and specific surface area of the attapulgite was a crucial parameter for the uptake of pigments of oil. Bleaching of the soybean oil with three attapulgites with different pore size distribution, which were assigned a, b, and c, respectively was investigated. The specific surface area and the pore size distribution of the attapulgites were characterized. The Freundlich isotherm analysis was used to evaluate the sorption capacity of the three attapulgite. Sample b gave the highest surface area and sample c the lowest. Sample b exhibited a wider pore distribution (8–65 Å) whereas samples a and c had more micropores smaller than 15 Å. Sample a, in contrast to samples b and c, was characterized by some larger pores (100–170 Å). The sorption capacity followed the sequence: attapulgite sample c > attapulgite sample a > attapulgite sample b. The sorption capacity was decided by the pore size distribution. The more pores with a distribution range 8–32 Å (i.e., close to the diameter of the pigments), the more pigments removed. The attapulgite sample c, which had most pores (8–32 Å) was the best.




Similar content being viewed by others
References
Park EY, Ming H (2004) Oxidation of rapeseed oil in waste activated bleaching earth and its effect on riboflavin production in culture of ashbya gossypii. J Biosci Bioeng 97(1):59–64
Kaynak G, Ersoz M, Kara H (2004) Investigation of the properties of oil at the bleaching unit of an oil refinery. J Colloid Interface Sci 280:131–138
Gülsah KE, Laçin O (2006) Statistical modelling of acid activation on cotton oil bleaching by Turkish bentonite. J Food Eng 75:137–141
Rossi M, Gianazza M, Alamprese C (2003) The role of bleaching clays and synthetic silica in palm oil physical refining. Food Chem 82:291–296
Christidis GE, Scott PW, Dunham AC (1997) Acid activation and bleaching capacity of bentonites from the islands of Milos and Chios, Aegean, Greece. Appl Clay Sci 12:329–347
Akyuz S, Akyuz T (2005) Study on the interaction of nicotinamide with sepiolite, loughlinite and palygorskite by IR spectroscopy. J Mol Struct 744–747:47–52
Potgieter JH, Potgieter VSS, Kalibantong PD (2006) Heavy metals removal from solution by palygorskite clay. Miner Eng 19:463–470
Huang J, Wang X, Jin Q (2007) Removal of phenol from aqueous solution by adsorption onto OTMAC-modified attapulgite. J Environ Manage 84:229–236
Perderiset M, Baillif P, Jaurand MC (1988) Chemical analysis and photoelectron spectroscopy of the adsorption of macromolecules on the surface of attapulgite. J Colloid Interface Sci 121:381–391
Liu P, Guo J (2006) Polyacrylamide grafted attapulgite (PAM-ATP) via surface-initiated atom transfer radical polymerization (SI-ATRP) for removal of Hg(II) ion and dyes. Colloids Surf A 282–283:498–503
Sanchez MMJ, Rodriguez CMS, Andrades MS (2006) Efficiency of different clay minerals modified with a cationic surfactant in the adsorption of pesticides: influence of clay type and pesticide hydrophobicity. Appl Clay Sci 31:216–228
Boki K, Hidehito M, Naohito K (1994) Bleaching rapeseed and soybean oils with synthetic adsorbents and attapulgites. J Am Oil Chem Soc 71:595–601
Taylor DR (2005) Adsorptive Separation of Oils. In: Fereidoon S (ed) Bailey’s industrial oil and fat products, vol.5: edible oil and fat products: processing technologies, 6th edn. Wiley, New York, pp 267–284
Greyt WD, Kellens M (2005) Bleaching. In: Fereidoon S (ed). Bailey’s industrial oil and fat products, vol.5: edible oil and fat products: processing technologies, 6th edn. Wiley, New York, pp 285–339
Proctor A, Toro-Vazquez JF (1996) The freundlich isotherm in studying adsorption in oil processing. J Am Oil Chem Soc 73:1627–1633
Grégorio C, Harmel N P, Frédéric G (2007) Removal of C.I. basic green 4 (malachite green) from aqueous solutions by adsorption using cyclodextrin-based adsorbent: kinetic and equilibrium studies. Sep Purif Technol 5:97–110
Zhang WM, Chen JL, Pan BC (2006) Synergistic adsorption of phenol from aqueous solution onto polymeric adsorbents. J Hazard Mater 128:123–129
Bayrak Y (2003) Adsorption isotherms in bleaching hazelnut oil. J Am Oil Chem Soc 80(11):1143–1146
Sing KSW, Everett DH, Haul RAW (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl Chem 57:603–619
Dubinin MM, Stoeckli HF (1980) Homogeneous and heterogeneous micropore structure in carbonaceous adsorption. J Colloid Interf Sci 75:34–42
Taylor DR, Jenkins DB, Ungermann CB (1989) Bleaching with alternative layered minerals: a comparison with acid-activated montmorillonite for bleaching soybean oil. J Am Oil Chem Soc 66:334–341
Stout LE, Chamberlain DF, Mcklvey JM (1949) Factors influencing vegetable oil bleaching by adsorption. J Am Oil Chem Soc 26:120–126
Xie S, Zhang S, Wang F (2007) Preparation, structure and thermomechanical properties of nylon-6 nanocomposites with lamella-type and fiber-type sepiolite. Compos Sci Technol. doi: 10.1016/j.compscitech.2007.01.012
Boki K, Moriaki K, Naohito K (1992) Adsorption isotherms of pigments from alkali-refined vegetable oils with clays minerals. J Am Oil Chem Soc 69(4):372–378
Franck D, Crini G, Vebrel J (2006) Removal of organic pollutants from aqueous solutions by adsorbents prepared from an agroalimentary by-product. Bioresour Technol 97:2173–2181
Zhang F, Itoh H (2003) Adsorbents made from waste ashes and post-consumer PET and their potential utilization in wastewater treatment. J Hazard Mater 101:323–337
Sukdeb P, Lee KH, Kim JU (2006) Adsorption of cyanuric acid on activated carbon from aqueous solution: effect of carbon surface modification and thermodynamic characteristics. J Colloid Interface Sci 303:39–48
Acknowledgments
The authors express their gratitude to the NSFC (National Natural Science Foundation of China), for its financial support (Contract NO: 20376028). We also thank the Testing and Analysis Center of South Yangtze University.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Huang, J., Liu, Y., Liu, Y. et al. Effect of Attapulgite Pore Size Distribution on Soybean Oil Bleaching. J Amer Oil Chem Soc 84, 687–692 (2007). https://doi.org/10.1007/s11746-007-1094-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11746-007-1094-9