Abstract
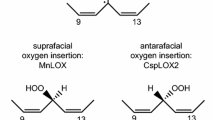
Small catalase-related hemoproteins with a facility to react with fatty acid hydroperoxides were examined for their potential mono-oxygenase activity when activated using iodosylbenzene. The proteins tested were a Fusarium graminearum 41 kD catalase hemoprotein (Fg-cat, gene FGSG_02217), a Pseudomonas fluorescens Pfl01 catalase (37.5 kD, accession number WP_011333788.1), and a Mycobacterium avium ssp. paratuberculosis 33 kD catalase (gene MAP-2744c). 13-Hydroxy-octadecenoic acids (which are normally unreactive) were selected as substrates because these enzymes react specifically with the corresponding 13S-hydroperoxides (Pakhomova et al. 18:2559–2568, 5; Teder et al. 1862:706–715, 14). In the presence of iodosylbenzene Fg-cat converted 13S-hydroxy-fatty acids to two products: the 15,16-double bond of 13S-hydroxy α-linolenic acid was oxidized stereospecifically to the 15S,16R-cis-epoxide or the 13-hydroxyl was oxidized to the 13-ketone. Products were identified by UV, HPLC, LC–MS, NMR and by comparison with authentic standards prepared for this study. The Pfl01-cat displayed similar activity. MAP-2744c oxidized 13S-hydroxy-linoleic acid to the 13-ketone, and epoxidized the double bonds to form the 9,10-epoxy-13-hydroxy, 11,12-epoxy-13-hydroxy, and 9,10-epoxy-13-keto derivatives; equivalent transformations occurred with 9S-hydroxy-linoleic acid as substrate. In parallel incubations in the presence of iodosylbenzene, human catalase displayed no activity towards 13S-hydroxy-linoleic acid, as expected from the highly restricted access to its active site. The results indicated that with suitable transformation to Compound I, monooxygenase activity can be demonstrated by these catalase-related hemoproteins with tyrosine as the proximal heme ligand.










Similar content being viewed by others
Abbreviations
- PhIO:
-
Iodosylbenzene
- cAOS:
-
Catalase-related allene oxide synthase
- COSY:
-
Correlation spectroscopy
- Fg-cat:
-
Fusarium graminearum catalase of the gene FGSG_02217
- H(P)ODE:
-
Hydro(pero)xyoctadecadienoic acid
- H(P)OTE:
-
Hydro(pero)xyoctadecatrienoic acid
- HODE:
-
Hydroxyoctadecadienoic acid
- LOX:
-
Lipoxygenase
- MAP-2744c:
-
Mycobacterium avium ssp. paratuberculosis catalase of gene MAP-2744c
- mCPBA:
-
Meta-chloroperoxybenzoic acid
- Pfl01-cat:
-
Pseudomonas fluorescens Pf01 catalase from the gene, accession number WP_011333788.1
- RP-HPLC:
-
Reversed phase high pressure liquid chromatography
- SP-HPLC:
-
Straight phase high pressure liquid chromatography
References
Mashhadi Z, Newcomer ME, Brash AR (2016) The Thr–His connection on the distal heme of catalase-related hemoproteins: a hallmark of reaction with fatty acid hydroperoxides. ChemBioChem 17:2000–2006
Fita I, Rossman MG (1985) The active center of catalase. J Mol Biol 185:21–37
Putnam CD, Arvai AS, Bourne Y, Tainer JA (2000) Active and inhibited human catalase structures: ligand and NADPH binding and catalytic mechanism. J Mol Biol 296:295–309
Oldham ML, Brash AR, Newcomer ME (2005) The structure of coral allene oxide synthase reveals a catalase adapted for metabolism of a fatty acid hydroperoxide. Proc Natl Acad Sci USA 102:297–302
Pakhomova S, Gao B, Boeglin WE, Brash AR, Newcomer ME (2009) The structure and peroxidase activity of a 33-kDa catalase-related protein from Mycobacterium avium ssp. paratuberculosis. Protein Sci 18:2559–2568
Ortiz de Montellano PR, De Voss JJ (2002) Oxidizing species in the mechanism of cytochrome P450. Nat Prod Rep 19:477–493
Denisov IG, Makris TM, Sligar SG, Schlichting I (2005) Structure and chemistry of cytochrome P450. Chem Rev 105:2253–2277
Ortiz de Montellano P (1992) Catalytic sites of hemoprotein peroxidases. Ann Rev Pharmacol Toxicol 32:89–107
Boeglin WE, Brash AR (2012) Cytochrome P450-type hydroxylation and epoxidation in a tyrosine-liganded hemoprotein, catalase-related allene oxide synthase. J Biol Chem 287:24139–24147
Lichtenberger F, Nastainczyk W, Ullrich V (1976) Cytochrome P450 as an oxene transferase. Biochem Biophys Res Commun 70:939–946
Gelb MH, Toscano WA Jr, Sligar SG (1982) Chemical mechanisms for cytochrome P-450 oxidation: spectral and catalytic properties of a manganese-substituted protein. Proc Natl Acad Sci USA 79:5758–5762
Macdonald TL, Burka LT, Wright ST, Guengerich FP (1982) Mechanisms of hydroxylation by cytochrome P-450: exchange of iron-oxygen intermediates with water. Biochem Biophys Res Commun 104:620–625
Ortiz de Montellano PR (2010) Hydrocarbon hydroxylation by cytochrome P450 enzymes. Chem Rev 110:932–948
Teder T, Boeglin WE, Schneider C, Brash AR (2017) A fungal catalase reacts selectively with the 13S fatty acid hydroperoxide products of the adjacent lipoxygenase gene and exhibits 13S-hydroperoxide-dependent peroxidase activity. Biochim Biophys Acta 1862:706–715
Brash AR, Song W-C (1996) Detection, assay, and isolation of allene oxide synthase. Methods Enzymol 272:250–259
Thomas CP, Boeglin WE, Garcia-Diaz Y, O’Donnell VB, Brash AR (2013) Steric analysis of epoxyalcohol and trihydroxy derivatives of 9-hydroperoxy-linoleic acid from hematin and enzymatic synthesis. Chem Phys Lipids 167–168:21–32
Gao B, Boeglin WE, Brash AR (2008) Role of the conserved distal heme asparagine of coral allene oxide synthase (Asn137) and human catalase (Asn148): mutations affect the rate but not the essential chemistry of the enzymatic transformations. Arch Biochem Biophys 477:285–290
Cui PH, Duke RK, Duke CC (2008) Monoepoxy octadecadienoates and monoepoxy octadecatrienoates 1: NMR spectral characterization. Chem Phys Lipids 152:122–130
Falck JR, Manna S, Jacobson HR, Estabrook RW, Chacos N, Capdevila J (1984) Absolute configuration of epoxyeicosatrienoic acids (EETs) formed during catalytic oxygenation of arachidonic acid by purified rat liver microsomal cytochrome P-450. J Am Chem Soc 106:3334–3336
Gao B, Boeglin WE, Zheng Y, Schneider C, Brash AR (2009) Evidence for an ionic intermediate in the transformation of fatty acid hydroperoxide by a catalase-related allene oxide synthase from the cyanobacterium Acaryochloris marina. J Biol Chem 284:22087–22098
Teder T, Boeglin WE, Brash AR (2014) Lipoxygenase-catalyzed transformation of epoxy fatty acids to hydroxy-endoperoxides: a potential P450 and lipoxygenase interaction. J Lipid Res 55:2587–2596
Silverstein RM, Webster FX, Kiemle DJ (2005) Spectrometric identification of organic compounds. Wiley, New York
Schneider C, Schreier P, Humpf H-U (1997) Exciton-coupled circular dichroism (ECCD) in acyclic hydroxylated dienes: a sensitive method for the direct stereochemical assignment of lipoxygenase products. Chirality 9:563–567
Gonnella NC, Nakanishi K, Martin VS, Sharpless BK (1982) General method for determining absolute configurations of acyclic allylic alcohols. J Am Chem Soc 104:3775–3776
Humpf H-U, Berova N, Nakanishi K, Jarstfer MB, Poulter CD (1995) Allylic and homoallylic exciton coupled CD: a sensitive method for determining the absolute stereochemistry of natural products. J Org Chem 60:3539–3542
Hamberg M (1989) Fatty acid hydroperoxide isomerase in Saprolegnia parasitica: structural studies of epoxy alcohols formed from isomeric hydroperoxyoctadecadienoates. Lipids 24:249–255
Mercier J, Agoh B (1974) Comportement d’hydroperoxydes allyliques a longue chaine en presence de complexes de certains metaux de transition. II. Structure des époxy-alcools formés à partir d’hydroperoxydes d’octadécène-9 oates de méthyle cis et trans en présence d’acétylacétonate de vanadyle. Chem Phys Lipids 12:239–248
Hamberg M, Herman RP, Jacobsson U (1986) Stereochemistry of two epoxy alcohols from Saprolegnia parasitica. Biochim Biophys Acta 879:410–418
Guengerich FP (2003) Cytochrome P450 oxidations in the generation of reactive electrophiles: epoxidation and related reactions. Arch Biochem Biophys 409:59–71
Hofrichter M, Ullrich R (2014) Oxidations catalyzed by fungal peroxygenases. Curr Opin Chem Biol 19:116–125
Blée E, Flenet M, Boachon B, Fauconnier ML (2012) A non-canonical caleosin from Arabidopsis efficiently epoxidized physiological unsaturated fatty acids with complete stereoselectivity. FEBS J 279:3981–3995
Rao SI, Wilks A, Hamberg M, Ortiz de Montellano PR (1994) The lipoxygenase activity of myoglobin. Oxidation of linoleic acid by the ferryl oxygen rather than protein radical. J Biol Chem 269:7210–7216
Hamberg M (1997) Myoglobin-catalyzed bis-allylic hydroxylation and epoxidation of linoleic acid. Arch Biochem Biophys 344:194–199
Bellucci G, Chiappe C, Pucci L, Gervasi PG (1996) The mechanism of oxidation of allylic alcohols to alpha, beta-unsaturated ketones by cytochrome P450. Chem Res Toxicol 9:871–874
Matsunaga T, Tanaka H, Higuchi S, Shibayama K, Kishi N, Watanabe K, Yamamoto I (2001) Oxidation mechanism of 7-hydroxy-delta 8-tetrahydrocannabinol and 8-hydroxy-delta 9-tetrahydrocannabinol to the corresponding ketones by CYP3A11. Drug Metab Dispos 29:1485–1491
Wang Y, Yang C, Wang H, Han K, Shaik S (2007) A new mechanism for ethanol oxidation mediated by cytochrome P450 2E1: bulk polarity of the active site makes a difference. ChemBioChem 8:277–281
Rettie AE, Rettenmeier AW, Howald WN, Baillie TA (1987) Cytochrome P-450-catalyzed formation of D4-VPA, a toxic metabolite of valproic acid. Science 235:890–893
Rettie AE, Sheffels PR, Korzekwa KR, Gonzalez FJ, Philpot RM, Baillie TA (1995) CYP4 isozyme specificity and the relationship between omega-hydroxylation and terminal desaturation of valproic acid. Biochemistry 34:7889–7895
Korzekwa KR, Trager WF, Nagata K, Parkinson A, Gillette JR (1990) Isotope effect studies on the mechanism of the cytochrome P-450IIA1-catalyzed formation of delta 6-testosterone from testosterone. Drug Metab Dispos 18:974–979
Brash AR (2009) Mechanistic aspects of CYP74 allene oxide synthases and related cytochrome P450 enzymes. Phytochemistry 70:1522–1531
Acknowledgements
This work was supported in part by National Institutes of Health Grants GM-074888 and GM-15431 (to A. R. B.) and by the Institutional Research Funding IUT19-9 of the Estonian Ministry of Education and Research and the Estonian Science Foundation Grant 9410 (to Professor Nigulas Samel).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Teder, T., Boeglin, W.E. & Brash, A.R. Oxidation of C18 Hydroxy-Polyunsaturated Fatty Acids to Epoxide or Ketone by Catalase-Related Hemoproteins Activated with Iodosylbenzene. Lipids 52, 587–597 (2017). https://doi.org/10.1007/s11745-017-4271-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-017-4271-0